Abstract
Individuals diagnosed with upper gastrointestinal cancers experience a myriad of nutrition impact symptoms (NIS) compromise a person’s ability to adequately meet their nutritional requirements leading to malnutrition, reduced quality of life and poorer survival. Electronic health (eHealth) is a potential strategy for improving the delivery of nutrition interventions by improving early and sustained access to dietitians to address both NIS and malnutrition. This study aimed to explore whether the mode of delivery affected participant disclosure of NIS during a nutrition intervention. Participants in the intervention groups received a nutrition intervention for 18 weeks from a dietitian via telephone or mobile application (app) using behaviour change techniques to assist in goal achievement. Poisson regression determined the proportion of individuals who reported NIS compared between groups. Univariate and multiple regression analyses of demographic variables explored the relationship between demographics and reporting of NIS. The incidence of reporting of NIS was more than 1.8 times higher in the telephone group (n = 38) compared to the mobile group (n = 36). Telephone predicted a higher likelihood of disclosure of self-reported symptoms of fatigue, nausea, and anorexia throughout the intervention period. A trusting therapeutic relationship built on human connection is fundamental and may not be achieved with current models of mobile health technologies.
Background
Individuals diagnosed with upper gastrointestinal cancers (gastric, oesophageal, and pancreatic) experience a myriad of symptoms directly related to their tumour burden and antineoplastic therapies. Many of these nutrition impact symptoms (NIS) including nausea, anorexia, pain, dysphagia, and diarrhoea, compromise a person’s ability to adequately meet their nutritional requirements which confers greater risk of mortality (Baracos, 2006; Hendifar et al., 2019; Khalid et al., 2007; Kubrak et al., 2010; Santos, Mendes, Mansinho, & Santos, 2021). Pancreatic exocrine insufficiency presents an additional challenge for pancreatic cancer patients, where the pancreas is unable to secrete adequate amounts of digestive enzymes (Hendifar et al., 2019), resulting in symptoms of epigastric pain and bloating, nausea, and steatorrhoea (fat-malabsorptive diarrhoea) (Hendifar et al., 2019). NIS contribute to weight loss either at presentation of disease or throughout treatment, which can lead to malnutrition and/or cancer cachexia through reduced nutritional intake and nutrient absorption (Hendifar et al., 2019). A reduction in both quality of life (QoL) and survival result from these debilitating NIS (Lis et al., 2012; Maia et al., 2020; Wheelwright et al., 2013).
Malnutrition at diagnosis is associated with reduced survival (Findlay et al., 2021), as it affects every organ system due to skeletal muscle mass reductions and immune system compromise (Saunders & Smith, 2010). Malnutrition is also an independent predictor of chemotherapy dose reduction (Klute et al., 2016), inhibiting an individual’s ability to receive the full dose of antineoplastic therapy, therefore reducing survival (Dewys et al., 1980; Van Cutsem & Arends, 2005). To improve QoL and survival, the treatment of NIS contributing to malnutrition is key. Appropriate prescription of medication is currently inadequate to control many of the NIS presenting in this cancer population (Andrew et al., 2008), as clinicians are underestimating and undertreating chemotherapy induced nausea and vomiting (Grunberg et al., 2004).
Low levels of malnutrition screening by nursing or support staff such as allied health assistants (Aktas et al., 2017) along with under recognised and/or under documented malnutrition by medical professionals (Aktas et al., 2017), increases the risk that patients undergo antineoplastic treatment without adequate nutrition support. This exacerbates pre-existing malnutrition. Access to dietitians at the critical time from diagnosis to treatment is also poor with one study demonstrating a mean time to first dietitian contact of 70 days (Huggins et al., 2022). A recent study has reported that 43% patients have access to dietitians via a formal nutrition pathway commencing at diagnosis (Deftereos et al., 2023), which is a major improvement from a published 2010 study revealed that only 5% of Australian patients were seen by a dietitian prior to major upper gastrointestinal surgery (Carey et al., 2010).
Electronic health (eHealth), defined as delivery mechanism of healthcare using information and communication technologies (World Health Organization (WHO), 2016), has been lauded as a potential strategy for improving the delivery of nutrition interventions by improving early and sustained access to health professionals. Our research team undertook a three-arm parallel randomised controlled trial testing the delivery of early and intensive nutrition intervention to improve the quality of life in patients with upper gastrointestinal cancers (Furness et al., 2018; Hanna et al., 2018) to bridge the gap in dietitian access. Participants were randomly assigned to receive the intervention via a synchronous telephone-based method or via an asynchronous mobile application-based method. This study revealed that while earlier commencement of nutrition intervention with the research dietitian was facilitated, there were no differences in quality of life measures between the two intervention groups and control (Huggins et al., 2022). This exploratory study aimed to determine whether the mode of nutrition intervention delivery (telephone or mobile application) affected participant disclosure of NIS during the intervention. It also examined the differences in the type and number of NIS disclosed. It was hypothesised that there would be no difference in disclosure of NIS to the dietitian during the 18-week intervention period.
Methods
Study Design
The primary study in which this exploratory study took place was a three-arm randomised controlled trial exploring two differing modes of early, intensive nutrition intervention (a synchronous (telephone) group and an asynchronous (mobile application) group) compared to a control group in patients with upper gastrointestinal (oesophageal, gastric, and pancreatic) cancers (study protocols published elsewhere) (Furness et al., 2018; Hanna et al., 2018).
Participants and Setting
Four public and private tertiary health services across southeast Melbourne, Victoria, Australia provided the setting for participant recruitment. Recruited participants received the intervention delivered to them at home by the research team in addition to usual nutrition care.
Eligibility
Patients with newly diagnosed (<4 weeks) upper gastrointestinal cancer and who were planned to commence surgical and/or medical (chemotherapy and/or radiotherapy) cancer treatment were eligible for enrolment. Those who had urgent surgical treatment were deemed eligible. Recruitment occurred between April 2017 and July 2019.
Randomisation and Blinding
An independent biostatistician was employed to complete a permuted block randomisation with two group stratification (Malnutrition Screening Tool (MST) score of <3 or ≥3) (Ferguson et al., 1999) performed using computer-generated random numbers (STATA version 18, StataCorp LP, College Station, TX, USA). Researchers involved with conducting recruitment, data collection and analysis were blinded to participant group allocation. Group allocation was revealed to participants by the research dietitian (KF).
Interventions
The 18-week intervention commenced as soon as was feasible after the participant’s consent, recruitment, and baseline data collection.
Participants received either a weekly or fortnightly intervention determined in collaboration between the dietitian and patient. Participants intervention was paused if they were hospitalised as they were deemed to be receiving all necessary care during an admission. If a participant was uncontactable after two attempts for a scheduled session with the dietitian, the session was missed for that week.
The Behaviour Change Technique Taxonomy v1 (BCTTv1) formed the basis for the behaviour change techniques used by the dietitian throughout the delivery of interventions in both groups (Michie et al., 2013). The dietitian provided a person-centred tailored nutrition intervention based on standardised nutrition assessment. The intervention included a food first approach to nutrition support (also commercial oral nutrition support products where appropriate), nutrition impact symptom management (including prompts to administer prescribed medications) and verbal and written education targeting self-management with repeated messaging over the lifespan of the intervention period.
Dietitians use the standardised nutrition care process and a nutrition care plan to document nutrition impact symptoms routinely. We developed a nutrition care plan specifically for this patient population prompting the most common nutrition impact symptoms likely to be reported based on the EviQ treatment protocols for each cancer and treatment type (NSW Government, 2023). All other symptoms were directly reported by patients.
Interventions
Participants’ home phone or mobile telephone was used to deliver the synchronous regular weekly or fortnightly phone contact from the research dietitian. The asynchronous intervention was delivered using a mobile application, myPace (Barnett et al., 2015). If participants did not have a mobile device or internet connectivity, they were provided with a tablet computer (e.g., iPad) and 6 months wireless internet access/connection. The participant and dietitian communicated asynchronously using a messaging function. myPace delivered daily reminders to assist with self-monitoring of weight and completion of scheduled ‘small steps’ (i.e., goals).
Participant Data
Data collected by an independent, blinded researcher (LH) included demographic (age, gender, cancer location, baseline Malnutrition Screening Tool (MST) (Ferguson et al., 1999) and Patient-Generated Subjective Global Assessment Short Form (PG-SGASF) (Abbott et al., 2016) (both total score and symptom summary score (Box 3), where each symptom has its own numerical score), anthropometrical (weight, height, BMI).
The research dietitian (KF) also maintained a set of participant records, collecting data from each week of intervention delivery. All participant reported symptoms, including those ‘other’ than nutrition impact symptoms were recorded e.g., hypertension, body aches.
Data Analysis
Data were analysed (STATA version 18, StataCorp LP, College Station, TX, USA) using Poisson regression to determine the proportion of individuals who reported NIS compared between intervention groups. Univariate logistic regression was used to compare the type and number of NIS reported between groups. Relationships between demographics and NIS were also then explored using backward stepwise regression analysis as described by Hosmer and Lemeshow (Hosmer & Lemeshow, 2000). Demographic data included age, gender, cancer location, baseline PG-SGASF scores. All analyses were presented as an odds ratio.
Results
A total of n = 111 participants were recruited from April 2017 to July 2019. There were n = 37 participants in the control group, n = 36 in the mobile application group and n = 38 in the telephone group. NIS were not recorded in the control group as this group was not contacted outside the planned outcome assessment timepoints.
Demographic Data at Baseline by Intervention Group

Note. *PG-SGASF Summary score = Box 3 of PG-SGASF; PG-SGA Patient Generated Subjective Global Assessment [38]; sd standard deviation; MST Malnutrition Screening Tool [32].
Baseline Reporting of Nutrition Impact Symptoms
Comparison Between Groups of Baseline PG-SGASF Nutrition Impact Score on Individual Symptom Reporting (higher Odds Ratios Indicate telephone group More Likely to Report) Using Poisson Regression Analysis
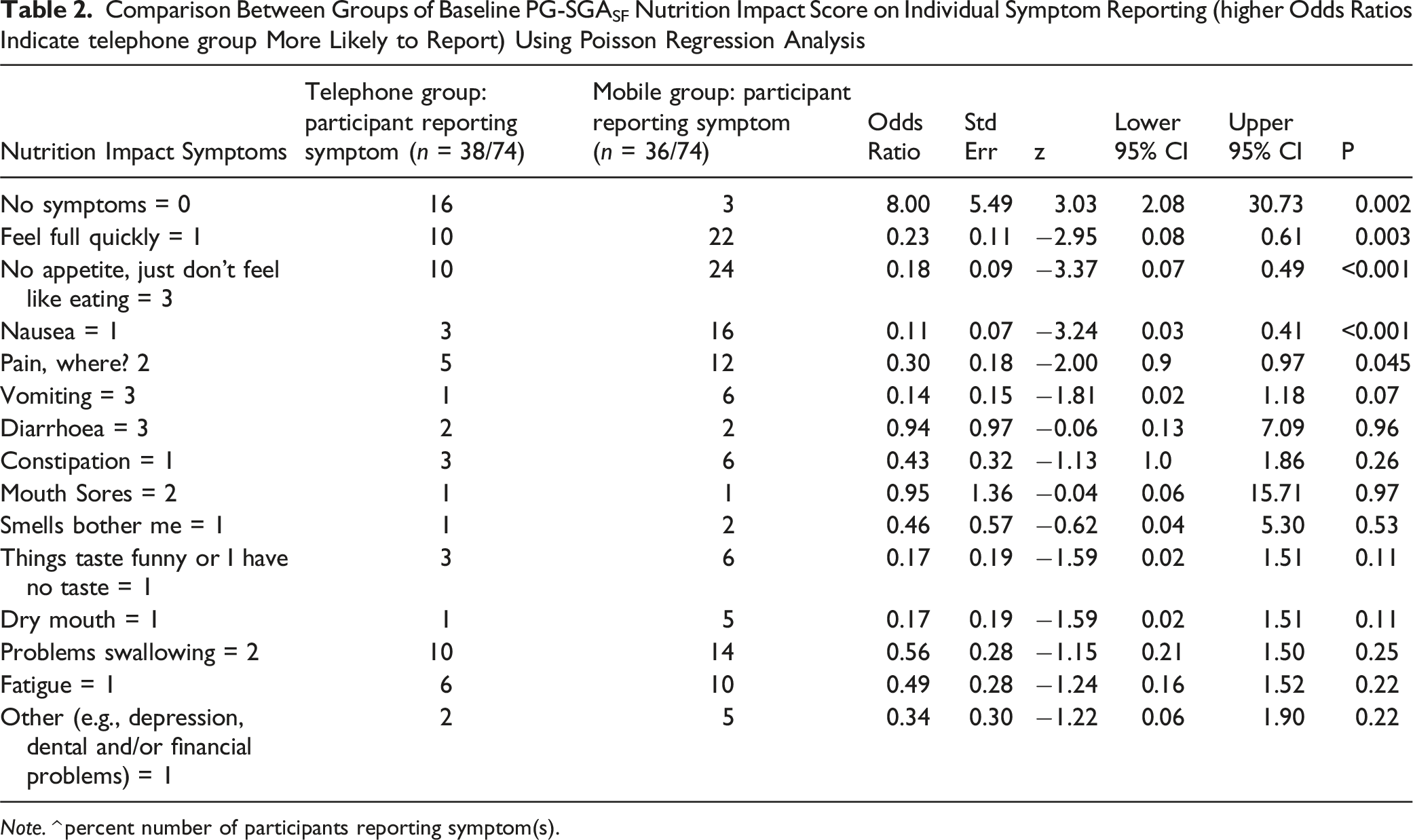
Note. ^ percent number of participants reporting symptom(s).
Nutrition Impact Symptoms Reported during 18-Week Intervention
Difference Between Intervention Groups in Likelihood of Reporting Individual Symptoms (higher Odds Ratios Indicate telephone group More Likely to Report) Using Univariate Analysis
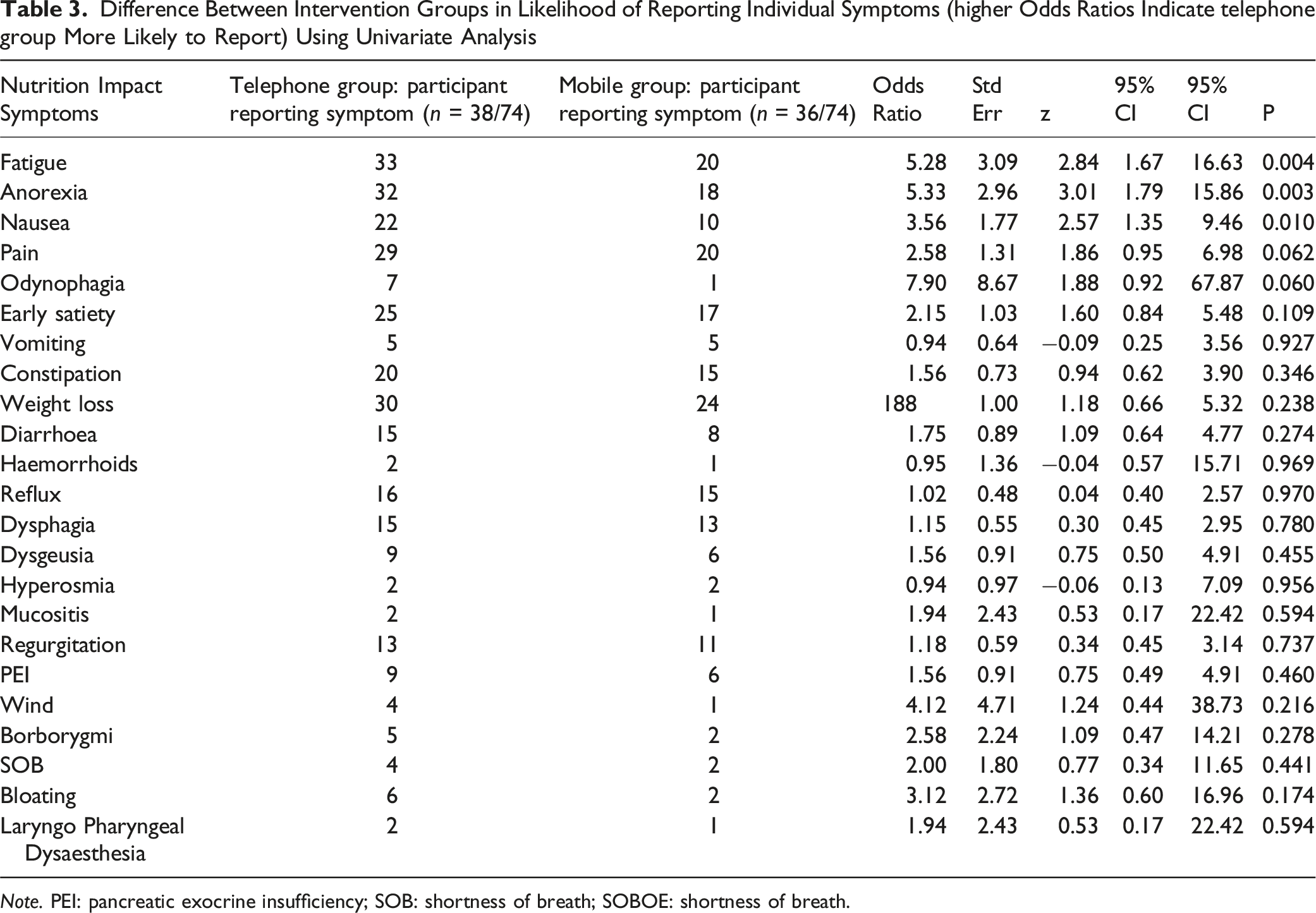
Note. PEI: pancreatic exocrine insufficiency; SOB: shortness of breath; SOBOE: shortness of breath.
The top five reported NIS across both intervention groups were weight loss (73%), fatigue (72%), anorexia (68%), pain (66%) and early satiety (57%) (Table 3). Participants in the telephone group were greater than five times more likely to report fatigue (OR 5.3 (SE 3.1), 95% CI 1.7 to 16.6, p = .004) and anorexia (OR 5.3 (SE 3.0), 95% CI 1.8 to 15.9, p = .003), and over three times more likely to report nausea (OR 3.6 (SE 1.8, 95% CI 1.4–9.5), p = .01) compared to the mobile app group over the entire 18 week intervention period. There were no between-group differences in the reporting of remaining 72 NIS.
Difference Between Groups in Likelihood in Reporting Individual Symptoms (higher Odds Ratios Indicate telephone group More Likely to Report) Adjusted for Demographics (age, Gender, Cancer Location, Baseline PG-SGASF Score) Using Backward Elimination Regression Analysis
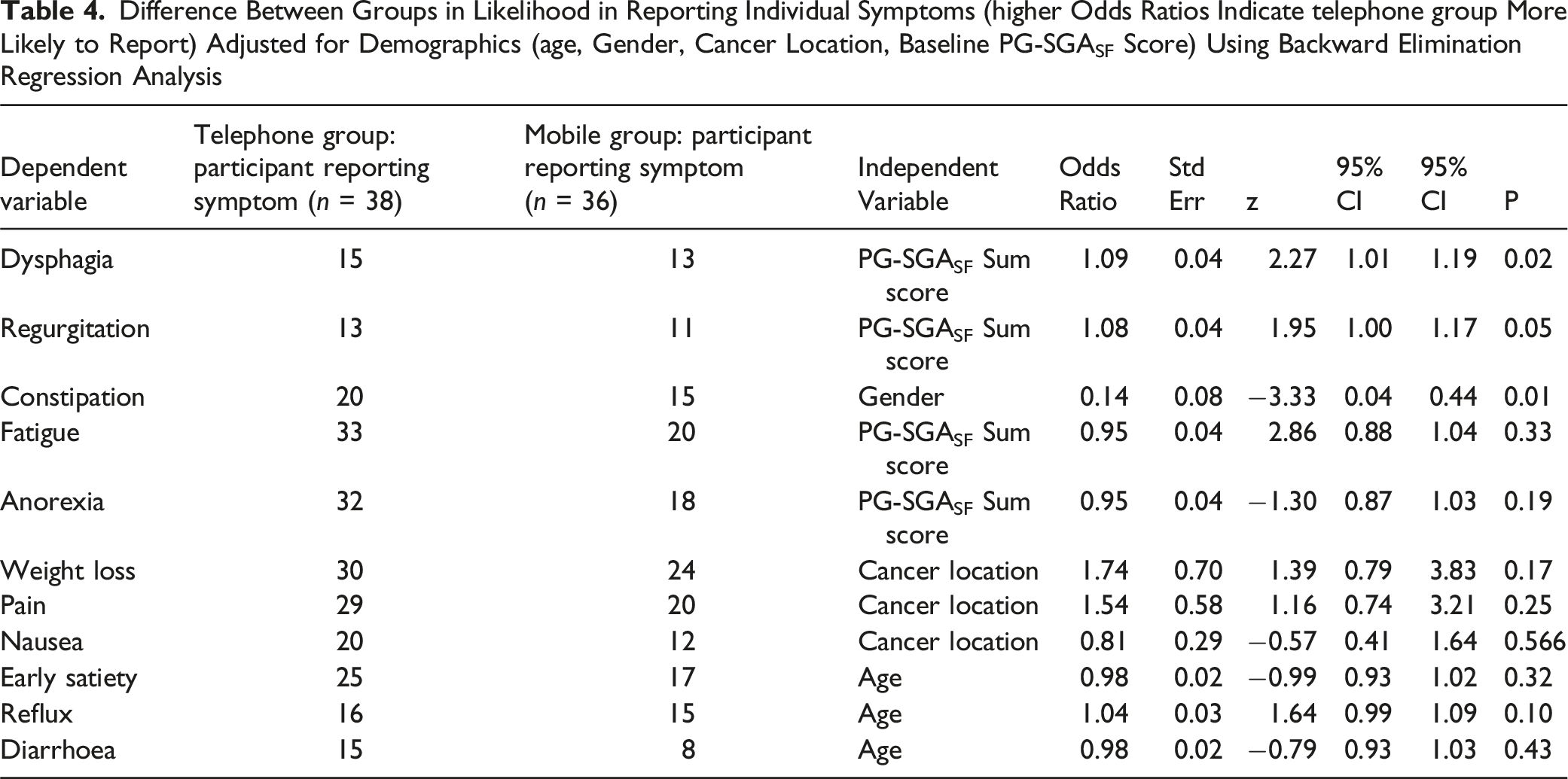
Higher baseline PG-SGASF summary score predicted a very small increase in reporting of both dysphagia and regurgitation in the telephone group. Female gender predicted a small increase in reporting of constipation in the telephone group.
Discussion
Upper gastrointestinal cancer patients randomised to the synchronous, telephone delivery of an early and intensive nutrition intervention disclosed a higher number and type of NIS during the 18-week intervention period, compared to the asynchronous, mobile app group. This finding indicates that use of mobile apps, based on the current technology available in this study, may be a poor substitute for synchronous enquiry led by a health professional in terms of symptoms elicitation.
High symptom and side effect burden, both from tumour load and antineoplastic therapies, are well established, particularly in upper gastrointestinal cancers (Laviano et al., 2003; Van Cutsem & Arends, 2005). Many of these symptoms are both underreported by patients and underrecognised and/or underrated in severity by clinicians (Basch et al., 2006; Di Maio et al., 2015). The reason for underreporting of symptoms by patients is multifaceted, including intentional withholding, believing that there is a certain inevitability of symptoms or that symptoms have been normalised by their medical practitioner, that they may have also been previously dismissed or not heard, and others did not want to burden busy clinicians (Boykoff et al., 2009; Chen et al., 2012; Cleeland, 2007; Frey et al., 2014; Knobf, 2002). The present research adds to this evidence base indicating that reliance on asynchronous mobile app reporting could lead to even further underreporting. Without disclosure, patients may not receive adequate management of their cancer symptoms.
It is important to consider why a telephone-based approach to follow up and symptom elicitation may be superior to an asynchronous online approach. Human connection, where a patient feels respected, is fundamental in developing a trusting therapeutic patient-provider relationship that allows for information sharing, knowledge acquisition and shared decision making (Thórarinsdóttir & Kristjánsson, 2014). Telehealth, particularly with video, allows for this trust to be built and maintained where understanding and empathy can be transferred by voice intonation, expression, and eye contact (Grzybowski et al., 1992; Toh et al., 2016). Asynchronous eHealth interventions are showing benefit in the management of patients remotely (Lin et al., 2005; Nguyen et al., 2021) including reducing healthcare utilisation (Chumbler et al., 2007). However, the therapeutic relationship built through synchronous face to face or voice to voice contact is valued by both clinicians and patients (Furness et al., 2021; Nguyen et al., 2021; Yau et al., 2011) through the manner of questioning and capacity of telephone to have a more conversational style of engagement. Emerging technology that has greater capacity or conversational style discourse may address this limitation with the online approach.
We acknowledge several limitations with this research study. Data collection utilised patient reported outcomes, which may increase the risk that cultural, situational, and environmental factors may impact on whether participants reported their symptoms or not. However, in the context of a randomised controlled trial one would expect this to be theoretically similar between groups and as such should not bias between group comparisons. It is possible that the telephone follow up approach may have elicited a higher number of symptoms as participants may have built a positive relationship with dietitian and therefore were more likely to report obsequious responses. Despite the use of randomisation, there were some differences in symptoms recorded at baseline between the two groups. This may not have biased between group analysis because our data collection wasn’t measuring incident case but rather, we measured the frequency of which symptoms were reported. The small sample size may have made it more difficult to identify differences between groups for the less frequent reported symptoms (Type II statistical error). The results presented are exploratory analyses on results from the overarching randomised controlled trial on primary analyses reported elsewhere (Huggins et al., 2022). Type I error across all these analyses is higher than the individual analysis level alpha of 0.05, consequently these findings should be subjected to further confirmatory research.
The study has highlighted several further future research directions that need more exploration. Understanding is required to determine if the lower levels of engagement and symptom reporting in the mobile app group are specific to this low survival cancer population or are more broadly application to wider groups of patients. With advanced conversational models of automated follow up like chatbots evolving we need to ascertain the acceptability of these models to patients.
Synchronous, telephone communication between the dietitian and patient led to higher eliciting of NIS, compared with the asynchronous mobile app communication mode. The great push to have virtual care approaches integrated into usual care practice requires some caution to ensure we are still providing healthcare that helps patients to build trusting relationships to connect openly and honestly with practitioners.
Supplemental Material
Supplemental Material - Effect of Communication Mode on Disclosure of Nutrition Impact Symptoms During Nutrition Intervention Delivered to People With Upper Gastrointestinal Cancer
Supplemental Material for Effect of Communication Mode on Disclosure of Nutrition Impact Symptoms During Nutrition Intervention Delivered to People With Upper Gastrointestinal Cancer by Kate Furness, Catherine E. Huggins, Lauren Hanna, Daniel Croagh, Mitchell Sarkies, and Terry P. Haines in Evaluation & the Health Professions
Footnotes
Acknowledgments
The authors wish to thank Monash Health for providing support for this project. Professor Helen Truby and Dr Alastair Kwok are acknowledged for their contribution to customising the MyPace application for use in an oncology population.
Author Contributions
Kate Furness was the primary author and drafted the manuscript. Kate Furness completed the data analysis and Terry Haines reviewed for accuracy. All authors (Lauren Hanna, Mitchell Sarkies, Catherine Huggins, Terry Haines, and Daniel Croagh) provided feedback on the draft manuscript. All authors (Lauren Hanna, Mitchell Sarkies, Catherine Huggins, Terry Haines, and Daniel Croagh) reviewed the final manuscript before submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The randomised controlled trial was funded by a Victorian Cancer Agency grant HSR15007.
Ethical Statement
Data Availability Statement
The data that support the findings of this study are not publicly available due to their containing information that could compromise the privacy of research participants but are available from the corresponding author (KF) upon reasonable request
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
