Abstract
Recent gene-editing technologies are heralded by proponents as a revolution for developing gene-edited foods (GEFs) while critics demand increased governance and scrutiny of potential societal impacts. Governance of GEFs is different in the United States, where GEFs are entering the market, and Europe, which restricts GEF development. Definitive regulations for governing GEFs are not yet solidified in either region. We identify and compare how English-language media in the United States and Europe portray potential risks, benefits, and regulation of GEFs, and we explore how these portrayals may reflect their regulatory environments. Results show that the regions similarly prioritize benefit frames that emphasize the social and scientific progress GEFs may bring, and few articles express skepticism about potential benefits. Comparing samples across regions exposes differences between Europe and the United States in the risk and benefit portrayals and in governance initiatives. Both regions prioritize policy risks that are counter to their current oversight regimes: the US media sample focused on GEFs being underregulated while the European sample emphasize risks of too much regulation. This may demonstrate the power of media to reflect and even cultivate public opinion and may influence future policy revisions within these distinct regulatory environments.
Recently developed gene-editing technologies like CRISPR have been heralded as a revolution for the science of genetics for being faster, cheaper, and more effective than previous gene editing, genetic modification, and traditional breeding methods (Doudna and Charpentier 2014). Agriculture sees CRISPR as a breakthrough that offers a more granular level of control over the genetic makeup of crops and livestock than previous genome editing tools (Gallo et al. 2018). Proponents note that such tools may allow for wide proliferation of diverse gene-edited foods (GEFs) that could boast several improved features for producers and consumers (Arora and Narula 2017). For instance, the first GEFs, a trans-fat-free oil pressed from gene-edited soybeans entered the commercial marketplace with little media coverage or public concern in March 2019. Yet critics warn that gene editing in this way is an extension to the development of genetically modified food products, which have received increased regulatory scrutiny in various regions of the world and have been the target of concerns about product risks to human and animal health and the environment.
Genetic modification and gene-editing methods fall into the broad category of genetic engineering techniques, yet the nature and complexity of the genetic alterations involved differ. Most genetically modified organisms (GMOs) are
Governmental approaches to regulating GMOs are distinct across different regulatory environments as evidenced by divergent policies and oversight approaches between the United States and Europe. Broadly speaking, the United States is described in policy scholarship as generally utilizing regulation to identify product-based risks of modified food products, demonstrating an adversarial form of adjudication and legalism, and dividing veto powers across Executive, Legislative, and Judicial government branches (Brady and Volden 1998; Kagan 1991). US GMO regulation is centralized at the federal level with GMO regulatory authority distributed among the US Department of Agriculture Animal and Plant Health Inspection Service (USDA-APHIS), Food and Drug Administration (FDA), and Environmental Protection Agency (EPA). The original US governance approach viewed GMOs as not presenting unique risks when compared to their conventionally bred counterparts, so each agency regulated GMOs using existing policies (Galata, Karantininis, and Hess 2014) as described in the Coordinated Framework for Regulation (CFR) of Biotechnology published in 1986 by the Office of Science and Technology Policy in the White House. The first CFR emphasized that biotechnology oversight should be based on scientifically determined risks and data, while also avoiding onerous regulations that could act as barriers to the development of biotechnology innovations (Office of Science and Technology Policy 1986). When determining whether a genetically engineered crop required regulatory oversight, the FDA and EPA assessed the traits of the final plant product and the known risks associated with those traits. FDA allowed GMO producers to self-determine whether a GMO is qualified as Generally Regarded as Safe (GRAS) and then bypass FDA authorization altogether. USDA’s regulations did not specifically target processes or products (Wolt, Wang, and Yang 2016). Instead, it regulated all genetically engineered organisms that could pose a plant pest risk. The agrobacterium used in much genetic modification thought the 1980s and 1990s,
In 2020, the SECURE Rule updated USDA regulatory triggers for genetically engineered crops based on scientific conceptions of risks in the final plant product. A USDA press release explained the SECURE Rule will “bring USDA’s plant biotechnology regulations into the 21st century by removing duplicative and antiquated processes in order to facilitate the development and availability of these technologies through a transparent, consistent, science-based, and risk-proportionate regulatory system” (USDA-APHIS 2020, n.p.n). Under the SECURE Rule, the developers of genetically engineered plants can self-determine whether their product qualifies as exempt from USDA regulations. USDA no longer considers plant DNA modifications through agrobacterium to pose unique risks. An implication is that most genetically engineered crops are expected to be fully exempt from USDA regulation (Wolf forthcoming).
The US approach has led to GMOs gradually dominating US commodity crop production and markets, while the opposite has happened in Europe. By 2013, USDA-APHIS had authorized the release of more than 10,000 GMO plants for field testing (Fernandez-Cornejo et al. 2014). Many farmers have adopted GMO seeds. As of 2020, GMO varieties comprised more than 90 percent of all soybean, cotton, and corn acres in the United States (USDA-ERS 2020). In contrast, only one variety of GMO corn is authorized for cultivation within Europe, and in 2017, only Spain and Portugal planted any GMO corn at all (Rostoks et al. 2019). Most of Europe’s fifty authorized GMO foods and feeds are imported for livestock, rather for human consumption.
Contrary to an open regulatory environment and centralized GMO regulation in the United States, Europe’s GMO regulatory environment is strict, precautionary, closed, and decentralized. Directive 2001/18/EC defines a GMO as the product of genetic engineering processes and outlines the risk assessment and monitoring procedures for GMO developers. The assessment is carried out by the European Food Safety Authority. Later regulations in 2003, 2009, 2015, and 2018 cover GMO issues such as traceability, labeling, food and feed authorization, and transportation. Each Member State also has the right to restrict or prohibit GMO cultivation and sales within its borders due to concerns about impacts to human, animal, or environmental health as well as concerns about socioeconomic impacts (European Parliament 2015).
Precautionary principles are ethics and decision-making tools that are both praised and highly contested (Harremoes et al. 2002). Precautionary policies are proactive approaches to minimizing possible environmental and human harms when uncertainties and risks of large magnitude and irreversibility may be present (Gardiner 2006). Ferrari (2009) has attributed Europe’s precautionary approach to GMO regulations as a product of European cultures and history. Unlike the United States, Europe experienced several food safety crises during the 1980s and 1990s, including the bovine spongiform encephalopathy (BSE) crisis or “mad cow disease.” Food crisis events such as BSE, along with desires for food to be “natural,” contributed to the Europe’s perceptions of risk (Ferrari 2009). The BSE food crisis also negatively impacted public confidence and trust in food regulators, which Kreibohm (2013) suggests has contributed to public distrust of new food technologies such as GMOs. Conversely, critical views on Europe’s precautionary approaches to GMO policy argue that such policies can serve some political, social, and economic interests at the expense of others (Anyshchenko 2019). Aerni (2018, 2019) contends that anti-GMO and pro-precautionary principle narratives serve to conceal vested interests held by different actors. For instance, the precautionary principle is viewed by some as fulfilling protectionist functions beyond managing health and environmental risks (Aerni 2018).
The United States and Europe have also taken divergent routes to regulating GEFs, reflecting their distinct perspectives on potential secondary risks that may arise from use of this novel technology (Cummings, Rosenthal, and Kong, 2021). The SECURE Rule established USDA-APHIS regulatory exemption criteria for many gene-edited crops, allowing those with less-complex edits and traits similar to plant varieties derived from conventional breeding to bypass USDA authorization procedures (Congressional Research Service 2020). This exemption encompasses most gene-edited crops currently in development (Gao 2018; Pirscher et al. 2018).
European regulations focus on potential risks and uncertainties arising from the
However, Gelinsky and Hilbeck (2018) contend that science media and journalists have been “one-sided and biased” in their reporting on the ECJ decision. They assert that proponents’ views are overrepresented and contain unsupported claims, demonstrating journalistic spin on the topic. Gelinsky and Hilbeck (2018) defend the ECJ decision and the precautionary approach to regulating gene editing, arguing that scientific evidence does support rigorous regulations and biosafety research. The European Network of Scientists for Social and Environmental Responsibility published a statement similarly arguing for stringent regulations on gene editing, rejecting proponents’ claims to justify reduced regulations (European Network of Scientists for Social and Environmental Responsibility 2017).
Media Coverage: From GMO to GEF
While media content is rarely the sole foundation of influence, it can play multiple complex roles in attitude and public opinion formation as emphasized across a number of communication theories. For instance, agenda setting theory (McCombs and Shaw 1972) shows there is a strong correlation between the salience of issues reported in mass media and public perceptions. This has been noted by others to be a function of framing effects where the emphasis on certain aspects of an issue can make them more salient for an audience, impacting mental schemas and attitude formation (Scheufele 1999). Issues of science and technology are often portrayed in certain frames that align with expected media logic, such as social progress, conflict, uncertainty, and morality/ethics. By definition, a frame emphasizes certain elements by omitting other possible elements, so the frame chosen for a story will play a role in selecting what an audience member uses to interpret the importance of a topic. These media effects in aggregate can play an influential role in public opinion formation, especially when audiences have few opportunities for direct experience to shape their opinions (McCombs and Valenzuela 2020). As the first to evaluate the media coverage of GEFs within US and European regulatory environments, this study can offer a foundation to help predict how the media influences audiences through varied theoretical lenses.
GEF messaging has recently entered the food and agriculture media landscape, which had previously been dominated by GMO-focused media messages. Scholars have extensively analyzed media depiction and coverage of GMOs to show what aspects have been prioritized in media messages, how GMOs has been framed, and to better understand how individuals come to hold distinct GMO attitudes (Cook et al. 2006; Gaskell et al. 2004; Heiman and Zilberman 2011; Marks et al. 2007; see McCluskey, Kalaitzandonakes, and Swinnen [2016] for an extensive media literature review). For instance, Vilella-Vila and Costa-Font (2008) analyzed Spain and UK media and found an association between negatively biased news and lack of trust in GMOs. Several studies have focused on media attention cycles, showing fluctuations in GMO media coverage over time (Nisbet and Huge 2006). In some European countries, GMO media coverage increased in the mid- to late-1990s (Bauer and Gaskell 2002; Bonfadelli, Dahinden, and Leonarz 2002; Wheeler et al. 2002) before the EU Parliament passed the GMO-defining Directive 2001/18/EC in 2001. Marks et al. (2007) analyzed mass media coverage of biotechnologies in the United States and UK over a 12-year period and found that US and UK media both framed GMOs as having more risks and hazards than benefits. Similarly, Mintz (2017) examined popular US news media coverage between 2011 and 2013 and found more unfavorable than favorable arguments about GMOs. The analysis also showed more frequent mentions of actors within the biotech industry, US government, food industry, and food processing industry.
From such studies, scholars have advanced research agendas to understand and explain consumer acceptance or rejection of GMOs (Yue et al. 2015). Within the US and European nations, studies show how attitudes and actions toward GMOs intersect with risk and benefit perceptions (Lucht 2015; Rose, Brossard, and Scheufele 2020), risk events (Frewer, Miles, and Marsh 2002), concerns about product safety to human health and the environment, attitudes to naturalness and technology (Rose, Brossard, and Scheufele 2020), media reporting and attitudes toward journalism (Vilella-Vila and Costa-Font 2008), level of GMO scientific knowledge (Ceccoli and Hixon 2012), social movements and nonprofit organization (NGO) influence (Aerni 2018), source credibility (Hunt and Wald 2020; McCluskey and Swinnen 2011), demographic characteristics (Siegrist 2000), and government policies (Zilberman et al. 2013). These studies shows that attitude formation about GMOs is complex and linked to broad social, political, and ethical concerns and values.
Trust or distrust in social actors and organizations involved with GMOs may also influence consumer attitudes (Marques, Critchley, and Walshe 2015; Siegrist, Connor, and Keller 2012). For example, Lang and Hallman (2005) found that US adults reported highest trust in scientists, medical professionals, and universities; moderate trust in NGOs and farmers; and lowest trust in government regulators, media sources, and agribusiness industry. The source of information in media articles may be just as important as what is said.
These are some of the many studies to document contrasting media portrayals of GMOs in the United States and Europe, but there is little to no comparable evaluation of how GEFs have been portrayed in these two jurisdictions. This study conducts a content analysis of how GEFs have been portrayed in the mass media since the introduction of technologies like CRISPR across the United States and Europe. Results provide a granular description of the messaging that serves as the first introduction for most “could-be” consumers of GEFs. These messages are the vital anchors on which potential consumers will base their understanding and form opinions regarding the utility of GEFs including their potential benefits and risks. Messaging for could-be consumers is how most people first learn about how novel food and agriculture products will be governed in coming generations of biotechnological development.
Media coverage regarding GEFs is in its very early phases, so much remains to be known how these novel developments will be portrayed in the media. Do media in the more risk averse and conservative European regulatory environment portray GEFs in a more negative light that in the less regulated US environment? What benefits and risks of GEFs are emphasized by media across these contexts? Answering these questions will provide a foundation for understanding how consumers are being introduced to this novel biotechnology through the mass media and may allow for a formative evaluation of the role of mass media messaging in shaping public opinion and policy development across distinct regulatory environments.
From this premise, the current study asks: How are GEFs portrayed in mass media within US and European regulatory environments?
Our quantitative study, described below, provides a full accounting of indexed major media messages, providing a robust descriptive context for comparing media portrayals across two distinct regulatory environments. Our study provides the first look into this area at a time when GEFs are just beginning to enter US commercial markets, and strong supportive and critical voices are entering the public arena to debate and resolve whether and how GEFs will be produced and used in the future.
Methods
Identification and Collection of Articles
We conducted a quantitative content analysis to answer our research question. We used the LexisNexis Metabase and Nexis-Uni databases to identify and collect English-language articles that discussed GEFs published in US and European local and national media outlets. The European media landscape is diverse in both language and culture, so this English-language sample cannot claim to represent the whole of European coverage. But it does provide a sample of relevant coverage within the more conservative European regulatory environment, which is compared to coverage of GEFs in US mass media.
We searched for articles that included “CRISPR” or variants of “gene edit* or gene-edit* or genome edit* or genome-edit*” anywhere in the text. To refine our results to GEFs rather than gene-editing technologies in general, we also required variants of “food or crop* or agriculture*” to be present in the headline or lead paragraphs.
Because we are interested in understanding the coverage of GEFs in public-facing media, we restricted source categories to media outlets serving broad, public audiences rather than more specialized stakeholders. We included local and national media outlets but excluded trade publications and press releases from our search. The specific source categories differed by database and are described below.
Data were collected across three searches using 2015 as a starting point of our date range, because Google Trends indicated Internet searches for the term “CRISPR” began early in that year. Specifically, Nexis-Uni was used to collect media articles published in the United States and in English between January 1, 2015, and May 15, 2019. Source categories included newspapers, blogs, web-based publications, magazines and journals, and news transcripts. LexisNexis Metabase was used to collect articles published worldwide in English between May 11, 2015, and November 3, 2020. Source categories included local and national news. Nexis-Uni was used again to collect media articles published in Europe and in English between January 1, 2015, and December 22, 2020, using the same source categories as the first Nexis-Uni search.
Data were pooled from the three searches. Duplicates were removed and the remaining sample was limited to articles published in the United States or Europe. Articles that only mentioned GEFs once often did so as part of a larger list or as an aside, with little description or discussion, so they were removed from the sample. Articles that discussed GEFs in at least two paragraphs often provided enough context to allow for meaningful coding. Trained coders reviewed the remaining articles to confirm relevance and retain only those articles from outlets serving broad audiences where our intended topic of GEFs were present and discussed in two or more paragraphs. This search criteria resulted in 171 relevant media articles: 92 published in the United States and 79 published in Europe.
Coding
Coders assessed three global factors about each article.
Coders further assessed five additional categories of content-based variables.
Coders also assessed the
Lists of potential
Eight benefits were grouped into two categories. Physical benefits include (a) human health, (b) animal health/welfare, (c) food quality, or (d) the environment. Social benefits include (a) economic, (b) democratization of the technology (easier access to use genetic editing), (c) food security (access to food), or (d) benefits to agriculture.
The fifth and final content-based category describes
Assessment of Intercoder Reliability
Two coders were originally trained to use a codebook using a random sample of ten articles. Based on these initial comparisons, we modified the original codebook to improve reliability. Intercoder reliability was then calculated by comparing the coder’s independent scores on a random selection 20 percent of the total article sample. We calculated Krippendorff’s alpha for these comparisons, resulting in alpha > 0.77 on all factors except for two, which were removed from the codebook. 2 After reaching reliability, a single coder completed the remaining articles.
Results
We explored the frequency distribution of individual variables and used chi-squared tests to assess the likelihood that distributions differed between the US and European samples. Variables with fewer than five counts were infrequent enough to not offer a meaningful comparison and were not included in the comparative analyses between samples. For analyses with more than one degree of freedom, we controlled type 1 error false positives using Bonferroni–Holm corrections (Goeman and Solari 2010).
Global Factors
Domain
Figure 1 shows that consumer foods and agricultural crops were the most frequent domains in which GEFs were portrayed. There were no significant differences between the US and European samples on any of the domains: consumer foods, χ2(1,
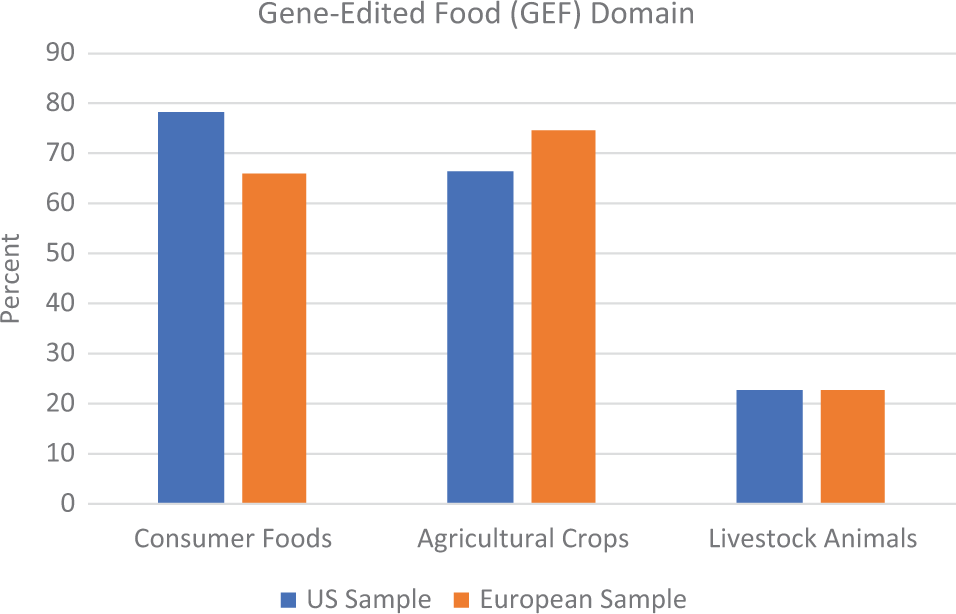
Domains in which gene-edited foods were discussed.
Valence
Figure 2 shows that most articles were neutrally valenced—they presented balanced positive and negative viewpoints about GEFs—and articles leaning positive were also frequent. Articles primarily leaning negative were absent from the European sample and nearly absent from the US sample. A comparison between balanced and positive valanced articles found significantly greater number of positive articles in the US sample, while the European sample had a significantly greater number of balanced articles, χ2(1,
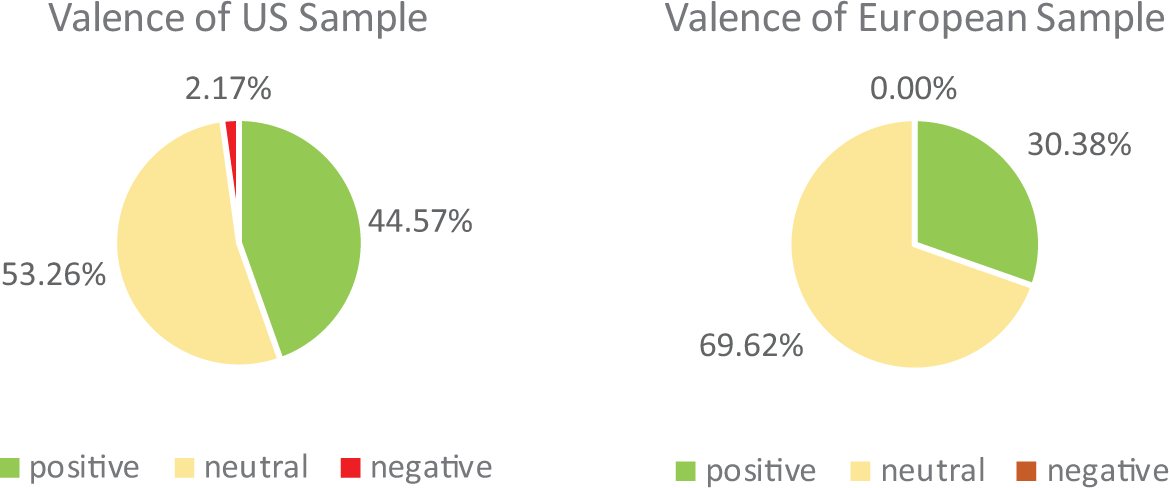
Valence of coverage of gene-edited foods.
Frame
Figure 3 shows that GEFs were primarily framed around themes of social/economic/agricultural progress, with public accountability/governance as the second most frequent frame. All the other frames were present in less than 10 percent of the articles. An overall comparison found a significant difference between US and European samples, χ2(2,
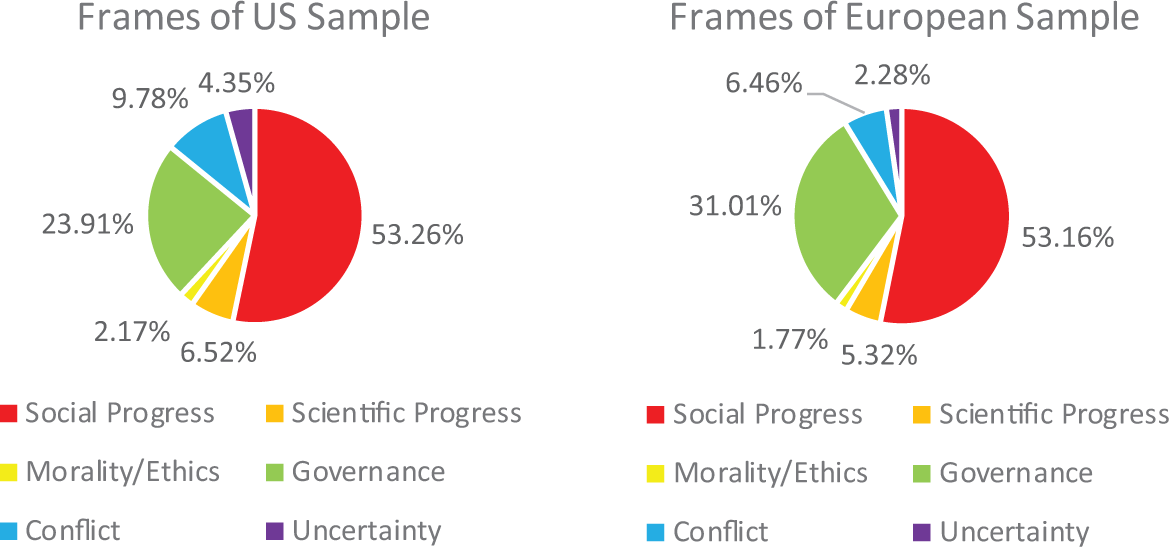
Primary frames within coverage of gene-edited foods.
Content-based Factors
GMO comparison
As seen in Figure 4, GEFs were explicitly portrayed as distinct from GMOs more often than they were equated as similar. There were no significant differences between the US and European samples on either comparison: contrasting GEF/GMO, χ2(1,
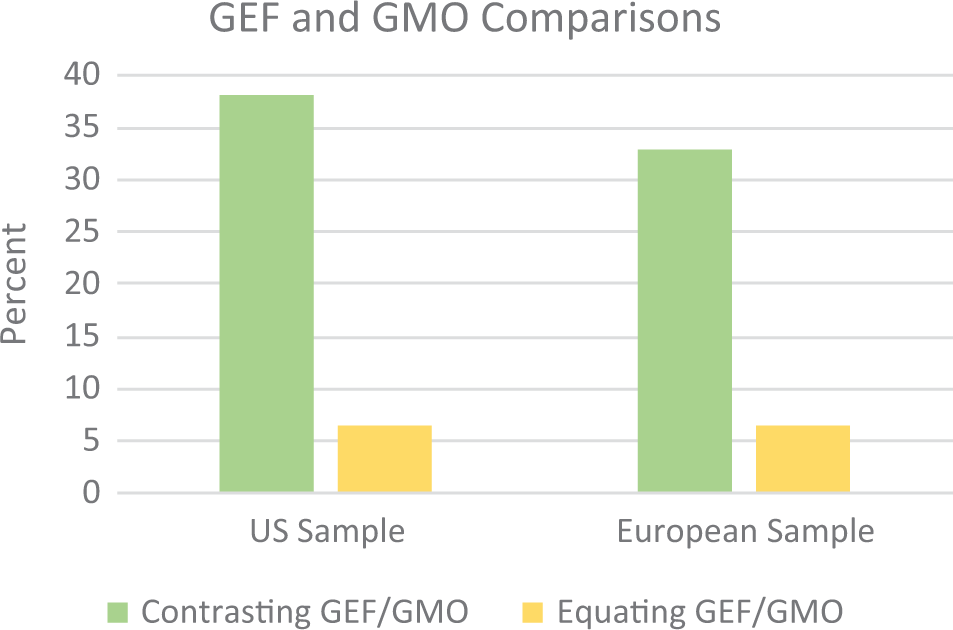
Comparisons between gene-edited foods and genetically modified organisms.
Sources
Figure 5 shows academic sources were most frequently cited, with food and agriculture industry as the second most frequently cited source in both the United States and Europe. All other source types together amount to 10 percent of sources cited. Source categories featured differently in US and Europe publications: food/agriculture/environment nonprofit sources were more frequent in the US sample, χ2(1,
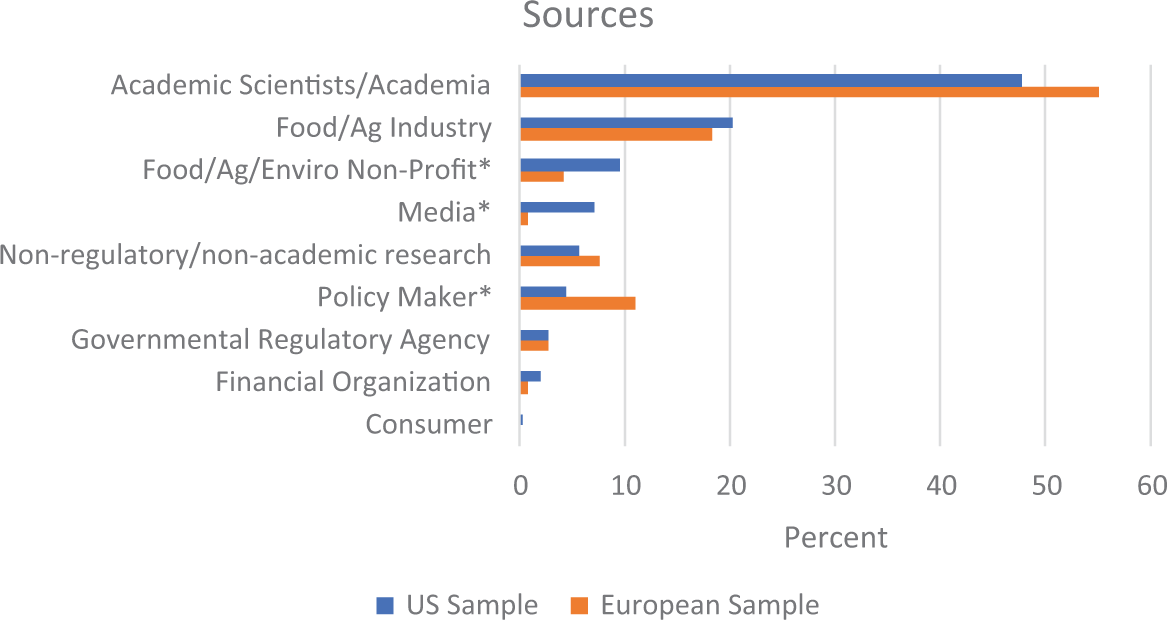
Types of sources cited in coverage of gene-edited foods. *
Benefits
Figure 6 lists various benefits associated with GEF grouped by benefit category. Within the physical benefit category, improved food quality was the most frequent benefit mentioned across both the US and European samples. Improved food quality, χ2(1,
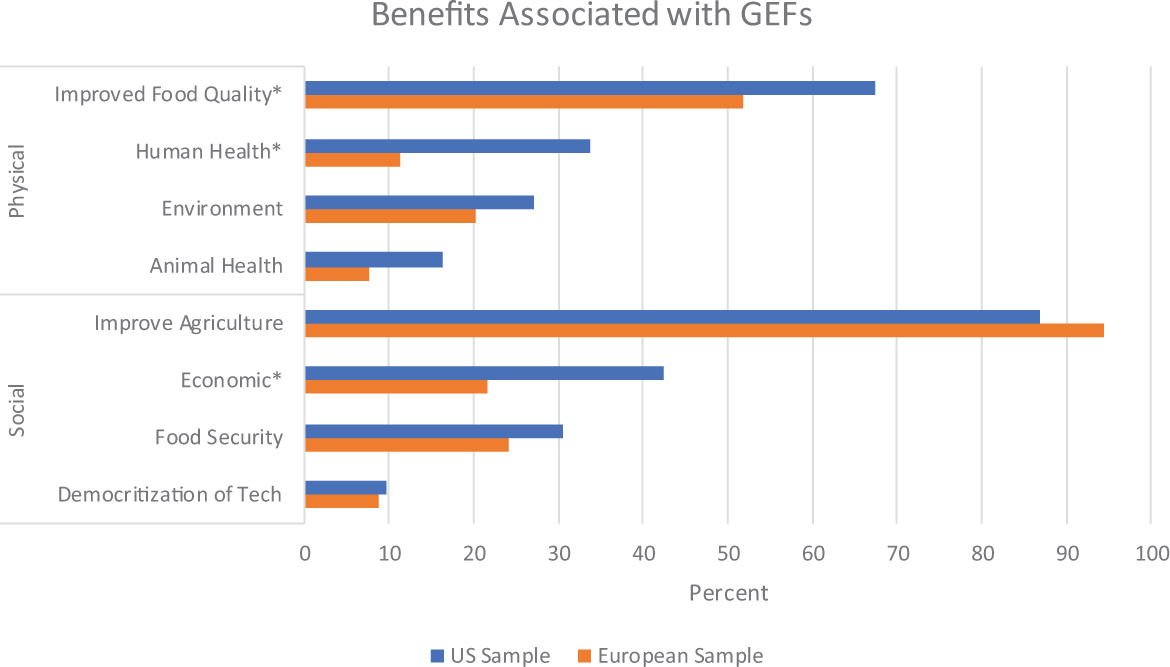
Presence of benefits associated with gene-edited foods. *
We conducted a secondary analysis to explore differences in the overall presence of benefit categories between the US and European sample. Presence of each benefit within a category was summed to create a value for each article representing how many mentions of each benefit category were present. The US sample mentioned a greater number of benefits overall: both for physical benefits, United States:
Risks
Figure 7 lists various risks associated with GEF, grouped by risk category. Within the physical risk category, environmental risks, and risks to human health were the most frequent risks mentioned, yet none of these risks significantly differed between the US and European samples. Within the social risk category, lack of consumer acceptance was the most frequent risk mentioned and was significantly more frequent in the US sample, χ2(1,
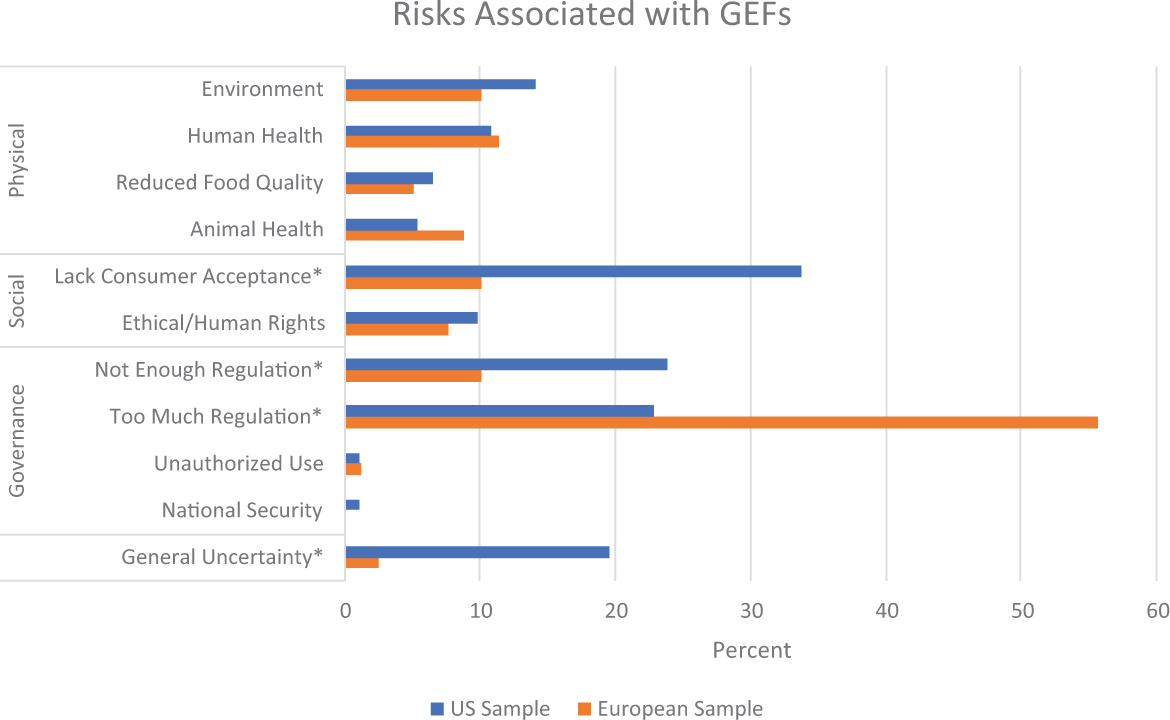
Presence of risks associated with gene-edited foods. *
As before, we conducted a secondary analysis to explore differences in the overall presence of risk categories between the US and European sample. The average number of physical risks mentioned in the US sample (
Regulation
As seen in Figure 8, regulation of GEFs featured frequently articles about GEFs, but it was significantly more frequent in the European sample, χ2(1,
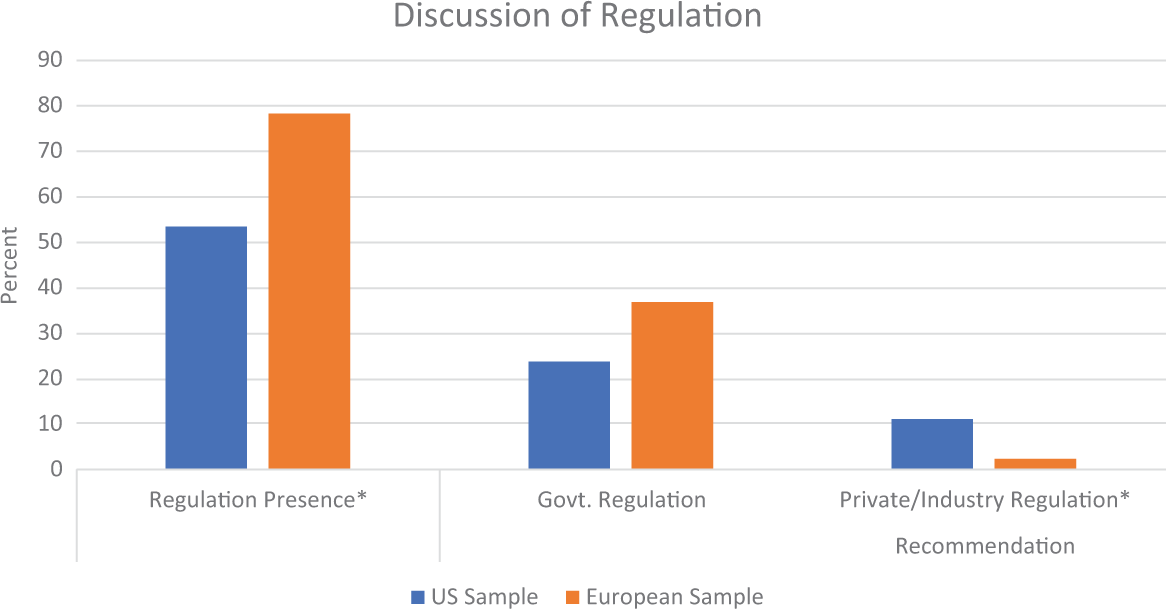
Discussion of regulation associated with gene-edited foods. *
Discussion
This study set out to examine for the first time how GEFs are portrayed in public facing media in the contrasting regulatory environments of the United States and Europe. Meaningful differences can be observed and are discussed further below—but it is also noteworthy that the form and content of media messages were remarkably similar across the United States and Europe.
There was no significant difference about media frames. Both samples equally framed GEFs in terms of social progress (56 percent of US articles, 53 percent of European articles) and governance (23 percent of US articles, 31 percent of European articles), with few news stories framing the topic in other ways. This may point to the fact that discussions about the utility and oversight of gene-editing technology are still in the early days.
Similarly, media portrayals of GEFs in both the United States and Europe have been generally positive or balanced, with benefits discussed more often than any associated risks. This may be because the primary sources allowed to interpret GEFs in media stories are the same stakeholders who have either been involved in their development (academic scientists) or those who stand to benefit financially from the technology (agricultural industries). GEFs have thus far avoided many of the negative views previously associated with GMOs.
Emphasizing this point, another similarity was the more frequent portrayal of GEFs as distinct from GMOs across both samples. While this definitional difference is still on the table for regulating GEFs in both the United States and Europe, some 30-40 percent of media messages across both samples explicitly claimed that GEFs and GMOs are distinct from one another and should be treated as such. In contrast, only a few messages equate GEFs and GMOs—this may signal an instance where the GMO debates of past years are extending into contemporary discussions and public perceptions regarding GEFs. For instance, one article stated in its headline: “Critics of the technology fear gene editing is just ‘GMO 2.0’—a new and not-yet-regulated way to play with plant genes.” It will be interesting to observe future GEF messaging, to understand its role in shaping a public agenda that equates GEFs with GMOs, and whether bringing the two closer together will also lead to more negatively valanced messaging.
This precautionary perspective is already present elsewhere when we look deeper. More than 50 percent of the articles used the frames of social and scientific progress. Two other frames that emphasize a more questioning outlook on GEFs were used by more than 30 percent of the media stories: conflict and public accountability/governance. These frames account for many of the articles that provided a neutral or negative valence on GEFs. The second most frequent grouping of risks echo these broader concerns about gene-editing technology and its role in society, focusing on questions of morality/ethics, uncertainty, and the desire for regulation and accountability. It is likely that these broader questions will grow more frequent if a greater diversity of voices related to GEFs are included, such as sources from the media. And even though very few articles were negative toward GEFs, previous work suggests that balanced coverage of science may lead the public to believe scientists are conflicted, which can contribute to greater belief in nonscientific claims (Dixon and Clarke 2012). This may indicate that the significant proportion of articles we found to be neutrally valenced may actually be interpreted by audiences as representing increased skepticism.
These similar patterns suggest where the coverage of GEFs is following a similar media attention cycle across both US and European samples. Prior research has documented how media coverage of other scientific technologies progresses through stages defined by the dominance of specific frames, sources, and risk/benefit descriptions (Dudo, Dunwoody, and Scheufele 2012; Nisbet and Huge 2006). GEFs are a novel technology, so it is not surprising that our findings place current coverage within a stage dominated by early market development and regulatory precedents. Continuing to track the portrayal of GEFs over the next few years will help map when, if at all, this early stage in the attention cycle begins to transition more toward the themes of conflict and social protest that are already present but not yet dominant.
The unique deviations between the US and European samples point to where the attention cycles of GEFs are not in sync and emphasizes how the contrasting regulatory environments and governance initiatives in these regions influence the media’s attention cycle. The most stark and provocative divergence was how drastically different the two regions prioritized the risks of over- and underregulation. Contrary to their dominant regulatory orientation, the US sample featured more frequent discussions of the risk of not enough regulation regarding GEFs than the European sample. Meanwhile the risk of too much regulation was discussed among the European sample at more than double the frequency of the US sample. Should these trends continue with increasing public-facing media stories about GEFs, we anticipate that articles discussing the risk of not enough regulation may set an agenda in the United States to influence public opinion and policy to restrict the currently open regulatory environment and call for increased oversight to minimize any physical or social risks (e.g., environmental impacts, fair distribution of benefits). The same may be argued in Europe where we might anticipate increased discussion that seeks to advance the agenda that GEFs are overly regulated, and thus the potential physical, social, and financial benefits of the agrotechnology may not eventuate. Should media coverage trends continue similarly, we may anticipate an increase in formal debates along these lines as media influences public perceptions which in turn may set new policy agendas regarding the degree to how these distinct regulatory structures around GEF continue to be developed for use.
These differences in current regulation regimes extend further to impact other deviations in coverage. In the United States where GEFs have already entered the market, the risk of consumer rejection is portrayed as the predominant risk associated with GEFs, present in over 30 percent of all the US sample. The generalized risk of uncertainty is also portrayed as a frequent risk in almost 20 percent of the US sample, likely connected to the more frequently portrayed risk of not enough regulation. These risks are significantly less frequent in the European sample. Even though the two most frequently cited source categories align across the United States and Europe (academic scientists followed by food and agricultural industries), we start to see interesting deviations beyond that. The third most cited source category in the European sample was policymakers, which is likely keeping regulatory issues and debates more salient and playing a role in the significantly greater proportion of European articles that discuss regulation of GEFs. In contrast, NGOs represented the third most cited source category in the United States, likely introducing more issues connected to social conflict into the media environment. It is possible these deviations will reinforce each other and amplify differences across the US and European media environments as GEFs continue to be covered by the media into the future.
This study reveals how public-facing mass media across different risk cultures have portrayed GEFs, identifying the broad directional pressure upon public opinion formation coming from the current media environment. The quantitative nature of our methods captures the frequencies of relevant categorizations of coverage but do not provide an interpretive analysis of the specific connotations involved or how they may relate to larger cultural understandings. For instance, our measure of valence represented a ratio between the number of positive and negative paragraphs on each article, rather than an overall magnitude of sentiment. Similarly, we coded only for the primary frame and cannot speak to the presence or absence of secondary frames in an article. A direct future study could focus on the novel findings identified in our study—specifically on the question of regulation—and conduct a qualitative study to further explore how the specific language and arguments used may be interpreted in a larger cultural context.
Similarly, individuals retain power to amplify or attenuate any media influence on their own belief structures based on their own communication choices. We aggregated articles from all source categories to explore broad patterns, and we hope that future studies can explore how the broad coverage patterns identified in public-facing media shifts as individuals selectively share and comment upon GEFs within reinforcing communities on social media. A network analysis exploring what aspects of our broad patterns of coverage are picked up and amplified by certain clusters of online communities would be useful.
It is also important to repeat that the patterns from our European sample are not meant to be representative of the entire European media environment. Comparing US coverage with English-language media articles published in European outlets allowed us to explore coverage within the contrasting regulatory environments of interest for this study but does not permit generalizing claims applicable the wider media environment across all of Europe. Future studies could compare coverage of GEFs across different European countries and EU member states because despite shared regulatory environments, their diverse histories and experiences with biotechnology will likely reveal theoretically useful heterogeneity.
Finally, a content analysis cannot be used as a measure of public opinion, nor should the patterns of coverage be assumed to influence audiences directly or consistently. Our systematic examination of the media portrayals of GEFs provides a foundation for improving understanding of information available in the US and European media environments that audiences use to form their attitudes about the issue.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by USDA National Institute of Food and Agriculture, Social Implications of Emerging Technologies (grant # 2018-67023-27679).
