Abstract
Internal doses are calculated on the basis of knowledge of intakes and/or measurements of activity in bioassay samples, typically using reference biokinetic and dosimetric models recommended by the International Commission on Radiological Protection (ICRP). These models describe the behaviour of the radionuclides after ingestion, inhalation, and absorption to the blood, and the absorption of the energy resulting from their nuclear transformations. They are intended to be used mainly for the purpose of radiological protection: that is, optimisation and demonstration of compliance with dose limits. These models and parameter values are fixed by convention and are not subject to uncertainty. Over the past few years, ICRP has devoted a considerable amount of effort to the revision and improvement of models to make them more physiologically realistic. ICRP models are now sufficiently sophisticated for calculating organ and tissue absorbed doses for scientific purposes, and in many other areas, including toxicology, pharmacology and medicine. In these specific cases, uncertainties in parameters and variability between individuals need to be taken into account.
1. INTRODUCTION
Intakes of radionuclides may occur during routine operations in a range of industrial, medical, educational, and research facilities. They may also occur after an incident involving release of radioactive materials followed by inhalation of gases and particles, or ingestion of contaminated foods.
An adequate assessment of internal exposure resulting from intakes of radionuclides is essential for the optimisation of radiological protection of workers, for the assessment of the health consequences of releases of radioactivity to the environment for members of the public, and for the retrospective demonstration of compliance with regulatory requirements.
Internal and external exposure of workers and members of the public should be assessed in terms of the protection quantity, effective dose. However, after intake of radionuclides, doses received by organs and tissues are protracted; therefore, equivalent and effective doses are accumulated over time. The resulting quantities are referred to as ‘committed doses’. Internal exposure of workers and members of the public is therefore assessed in terms of committed effective dose.
In internal dosimetry, no operational dose quantities have been defined that provide a direct assessment of committed effective dose. Different methods are therefore applied to assess the equivalent or effective dose due to radionuclides in the human body. They are mainly based on various activity measurements, and the application of biokinetic and dosimetric models. Biokinetic models for individual elements and their radioisotopes are used to calculate the total number of transformations (radioactive decays) occurring within specific tissues, organs, or body regions (source regions) over a given period of time (usually 50 y for adults, or up to the age of 70 y for children) by determining the time-integrated activity in each source region. Dosimetric models are used to calculate the deposition of energy in all important organs/tissues (targets) for transformations occurring in each source region, taking account of the energies and yields of all emissions (ICRP, 2008). Many biokinetic and dosimetric models have been produced by the International Commission on Radiological Protection (ICRP) over time and are described hereafter.
In its 2007 Recommendations, ICRP (2007) introduced important changes that affect the values calculated per radiation exposure. Publication 103 (ICRP, 2007) introduced changes to the radiation weighting factors used in the calculation of equivalent dose to organs and tissues, and changes to the tissue weighting factors used in the calculation of effective dose. In addition, an important development was the adoption of reference anatomical computational phantoms (i.e. models of the human body based on medical imaging data), in place of the composite mathematical models that have been used for all previous calculations of organ doses. This process commenced with the adoption of reference adult male and female models (ICRP, 2009), and will be continued with the adoption of paediatric phantoms. Publication 103 also clarified the need for separate calculation of equivalent dose to males and females, and sex-averaging in the calculation of effective dose (ICRP, 2007). In the revision of dose coefficients, the opportunity has also been taken to improve calculations by updating radionuclide decay data (ICRP, 2008), and implementing more sophisticated treatments of radiation transport (ICRP, 2010) using the ICRP reference anatomical phantoms of the human body (ICRP, 2009). These improvements have impacts on dose calculations for external exposures as well as for internal emitters. Work has been conducted by ICRP Committee 2 and its Task Groups to update the current biokinetic and dosimetric models, and provide new dose coefficients. The most recent developments are presented here.
2. GENERAL METHODS FOR THE ASSESSMENT OF INTERNAL DOSES
The methods for calculating the committed effective dose after intake of radionuclides are described below (Fig. 1).
Calculation of absorbed dose and the International Committee on Radiological Protection’s protection quantities, equivalent and effective dose, for intakes of radionuclides. Source: Publication 130 (ICRP, 2015).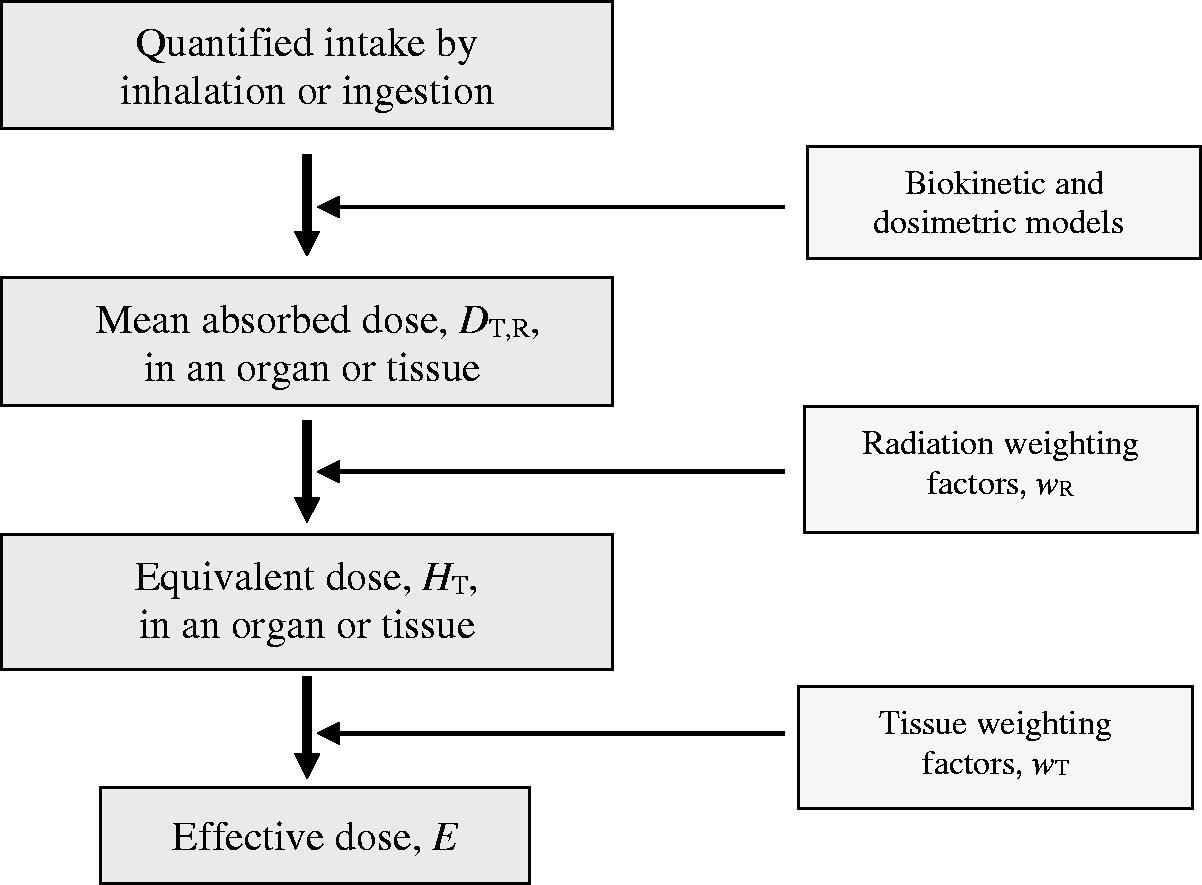
By using the reference biokinetic models, the distribution and retention of radionuclides in body organs and tissues of the reference worker or Reference Person 1 are determined as a function of time after intake by inhalation or ingestion. The total number of nuclear transformations occurring within a 50-y period (or up to the age of 70 y for children) in each source region is calculated. The dosimetric models based on the male and female reference computational phantoms and the Monte Carlo radiation transport codes are used to calculate the sex-specific absorbed dose in each target organ or tissue resulting from a nuclear transformation in each source region. The radiation weighting factors are applied to determine sex-specific committed equivalent doses to each organ or tissue. The sex-specific committed equivalent doses are sex-averaged, and the tissue weighting factors are then applied to determine the sex-averaged committed effective dose.
Calculation of doses after internal contamination is a complex procedure. To simplify the system, ICRP provided dose coefficients (i.e. committed effective dose and committed equivalent doses to organs or tissues per intake) and bioassay functions (i.e. excretion rates or organ retention per intake). Dose coefficients and bioassay functions may be used for both prospective and retrospective assessments of exposure. Prospective assessments provide estimates of intakes and resulting doses for workers engaged in specific activities, or for members of the public exposed in specific circumstances, using information on projected exposures to radionuclides. These assessments generally make use of default assumptions about exposure conditions and default values for parameters describing material-specific properties, such as the particle size distribution of an inhaled aerosol or the absorption characteristics of a material after inhalation or ingestion. Retrospective assessments use the results of individual and workplace monitoring to assess doses in order to maintain individual dose records and demonstrate compliance with regulatory requirements.
Models describing the deposition of energy from transformations of radionuclides in the human body were first published in Publication 2 (ICRP, 1959). More recent reports were published in the 1970s and 1980s, describing the behaviour of radionuclides in different parts of the body. Publication 30 (ICRP, 1979, 1980, 1981, 1988a) and its supplements gave dose coefficients and values of annual limits on intake for workers, for intakes of radionuclides by inhalation and ingestion, referencing the recommendations issued in Publication 26 (ICRP, 1977) and the anatomical and physiological data in Publication 23 (ICRP, 1975). Publication 68 (ICRP, 1994b) provided updated dose coefficients for workers following the 1990 Recommendations in Publication 60 (ICRP, 1991). It applied the Human Respiratory Tract Model (HRTM) for inhaled radionuclides given in Publication 66 (ICRP, 1994a), the updated basic anatomical and physiological data for the skeleton given in Publication 70 (ICRP, 1995b), and revised systemic biokinetic models for selected isotopes of 31 elements given in Publications 56, 67, 69 and 71 (ICRP, 1990, 1993, 1995a,c). Biokinetic models for other elements were taken from Publication 30, and modified by addition of explicit excretion pathways to improve dose estimates for the urinary bladder and colon walls.
A similar approach was taken for calculating doses to members of the public. Models and dose coefficients after ingestion or inhalation of radionuclides by members of the public were published in Publications 56, 67, 69, 71 and 72 (ICRP, 1990, 1993, 1995a,c, 1996). Similarly, models and dose coefficients for the embryo and fetus from intakes of radionuclides by the mother, and for infants from ingestion of radionuclides in mothers' milk were published in Publications 88 and 95 (ICRP, 2001, 2004), respectively.
Doses from intakes of radionuclides can also be assessed retrospectively from bioassay measurements (e.g. daily urinary and faecal excretion) or from direct measurements of the radionuclides in the body. Publications 54 and 78 (ICRP, 1988b, 1997) gave guidance on the design of monitoring programmes and the interpretation of results to estimate doses to workers following radionuclide inhalation or ingestion. The guidance was supported by numerical data to enable the assessment of intakes and doses from bioassay data. These data were provided for a number of radionuclides, selected based on those that are most likely to be encountered in the workplace. Predicted values of the measured quantities for various times after a single intake or for routine monitoring were given in terms of the activity of the intake per activity measured. Standard dose coefficients would then be used to calculate effective dose from the assessed intake.
3. Updates In The Methods And The Models
Publication 103 (ICRP, 2007) introduced changes that affect the calculation of doses. In addition, numerous advances have been made in physiology, dosimetry, and monitoring methods over recent years that support a revision of the models recommended in Publications 30 (doses for workers) and 56 (doses for members of the public) (ICRP, 1979, 1980, 1981, 1988a, 1990). Updates of the models will be presented in the ‘Occupational Intakes of Radionuclides (OIR)’ series and the ‘public exposures’ series, which started being published in ICRP (2015), and are briefly described hereafter. Emphasis is placed here on updates to the biokinetic models of the alimentary tract and respiratory tract, and the biokinetic systemic models. Biokinetic models of the alimentary and respiratory tracts are used to define the movement of radionuclides within these systems, resulting in absorption to blood and/or loss from the body. The behaviour of radionuclides absorbed to blood is described by element-specific systemic models that range in complexity. These models are intended both for the derivation of dose coefficients and the interpretation of bioassay data. At the end of the section, specific attention is given to a new development that allows easier assessment of doses directly from bioassay data.
3.1. Human Respiratory Tract Model
The HRTM, described in Publication 66 (ICRP, 1994a), was updated in Part 1 of the OIR series (ICRP, 2015) to take account of data that have accumulated since issuance of Publication 66, although the basic features of the model remain unchanged. Inhaled particles containing radionuclides deposit in the extrathoracic airways (nose, larynx, etc.), the bronchial and bronchiolar airways of the lung, and the alveolar-interstitial (AI) region, with deposition in each region dependent mainly on particle size (ICRP, 1994a, 2002). Removal from the respiratory tract occurs mainly by dissolution and absorption to blood, and the competing process of transport of particles to the throat followed by their entry into the alimentary tract. The proportions absorbed to blood or cleared by particle transport depend on the speciation and the solubility of the material, and on the physical half-life of the radionuclide. The HRTM is also applied to gases and vapours, and in the OIR series to inhalation of radon and its radioactive progeny.
For absorption to blood, the main changes introduced in the new model are as follows.
Redefinition of Type F, M and S absorption default parameter values: larger rapid dissolution fractions (fr) for Types M and S of 0.2 and 0.01, replacing 0.1 and 0.001, respectively, with lower rapid dissolution rates (sr) of 3 d−1 for Types M and S, and 30 d−1 for Type F, replacing 100 d−1. Material-specific parameter values for fr, sr, and the slow dissolution rate (ss) in selected cases where sufficient information is available (e.g. forms of uranium and plutonium). Element-specific values of sr and the bound state parameters, fb and sb, where sufficient information is available.
For clearance by particle transport, the main changes are as follows.
More realistic clearance from the nasal passage, including transfer from the anterior region to the posterior region, based on recent human experimental studies. Revised characteristics of slow particle clearance from the bronchial tree based on recent human experimental studies. It is now assumed that it only occurs in the bronchioles, rather than as a particle-size-dependent phenomenon throughout the bronchial tree. Longer retention in the AI region of the lung, with a revised model structure based on recent data including very-long-term follow-up of groups of workers exposed to insoluble Co-60 particles and plutonium dioxide.
3.2. Human Alimentary Tract Model
The Publication 30 (ICRP, 1979) model of the gastrointestinal tract has been replaced by the Human Alimentary Tract Model (HATM) described in Publication 100 (ICRP, 2006). The main features of the HATM are summarised as follows.
Inclusion of all regions of the alimentary tract: oral cavity, oesophagus, stomach, small intestine, right colon, left colon, and rectosigmoid (the sigmoid colon and rectum). A default assumption that absorption of an element and its radioisotopes to blood occurs exclusively in the small intestine (i.e. the total fractional absorption, fA, equals the fractional absorption from the small intestine, fSI). A model structure that allows for absorption in other regions, where information is available. A model structure that allows for retention in the mucosal tissues of the walls of the regions of the alimentary tract, and on teeth, where information is available. Explicit specification of the location of target regions for cancer induction within each region of the alimentary tract.
3.3. Systemic models
A systemic model describes the time-dependent distribution and retention of a radionuclide in the body after it reaches the systemic circulation, and its excretion from the body. In contrast to the current and past ICRP biokinetic models describing the behaviour of radionuclides in the respiratory and alimentary tracts, the ICRP systemic models have generally been element-specific with regard to model structure as well as parameter values. A single generic model structure that depicts all potentially important systemic repositories and paths of transfer of all elements of interest in radiological protection would be too complex to be of much practical use. However, generic model structures have been used in previous ICRP reports to address the systemic biokinetics of groups of elements, typically chemical families, known (or expected) to have qualitatively similar behaviour in the body. For example, Publication 20 (ICRP, 1973) introduced a generic model formulation for the alkaline earth elements calcium, strontium, barium and radium, but provided element-specific values for most model parameters. In Parts 1–3 of Publication 30 (ICRP, 1979, 1980, 1981), a model developed for plutonium, including parameter values as well as model structure, was applied to most actinide elements. The use of generic systemic model structures was increased in ICRP reports on doses to members of the public from intake of radionuclides (ICRP, 1993, 1995a,c), and is further expanded in new reports because it facilitates the development, description and application of systemic biokinetic models. An important development is that, as the availability of data allows, models have been made more physiologically realistic with regard to the dynamics of organ retention and excretion, so that they are applicable to the interpretation of bioassay data as well as the calculation of dose coefficients.
3.4. Interpretation of bioassay data
As described in Chapter 2, doses may be assessed retrospectively by direct measurement of activity in organs, in the body, or by activity measurement in excreta of biological samples. The system of dose assessment from bioassay data that is generally applied involves evaluation of the intake of a radionuclide either from direct measurements (e.g. external monitoring of the whole body or of specific organs and tissues) or indirect measurements (e.g. of urine, faeces, or environmental samples). Predicted values of these measured quantities for intake of a radionuclide are recommended by ICRP, and these values can be used to estimate the intake (ICRP, 1997). The committed effective dose resulting from any intake is then calculated using the appropriate dose coefficient recommended by ICRP, or determined using ICRP’s recommended methodology.
In the OIR series, ICRP will also give dose per content functions (calculated from the dose coefficient divided by the relevant bioassay function). They enable committed doses to be calculated directly from bioassay measurements as a function of time after intake. The main advantage of this approach is that the user does not perform the intermediate step of calculating the intake in order to evaluate the dose. In some cases, the assessed dose is less sensitive to the choice of parameter values than it is to the assessed intake. However, care is still needed in determining the most appropriate measurement data and defining the time of the intake.
Effective dose assessed from bioassay measurements is relatively insensitive to the choice of parameter values when the measured quantity is directly related to an organ dose that makes a dominant contribution to the effective dose (e.g. in the case of lung retention measurements after inhalation of an insoluble Co-60 compound, where lung dose dominates the effective dose). However, sensitivity to parameter values may be much higher when the measured quantity is less closely related to the effective dose, such as when lung dose makes a dominant contribution to effective dose and urine monitoring is employed. For such a case, the results of urine monitoring can provide a reliable measure of doses to systemic organs, but assessed lung dose is sensitive to the choice of absorption parameter values. An example includes the assessment of effective dose from urine monitoring data after inhalation of an insoluble uranium compound.
4. UNCERTAINTY AND VARIABILITY IN THE ASSESSMENT OF EFFECTIVE DOSE
The effective dose calculated for protection purposes is determined from the equivalent doses to organs and tissues of the human body, which are in turn calculated from the mean absorbed doses to those organs and tissues. Effective dose provides a value that takes the given exposure conditions into account, but does not consider the characteristics of a specific individual. In particular, the tissue weighting factors that are used to determine effective dose are selected, rounded values that represent averages over many individuals of different ages and sexes. The equivalent doses to each organ or tissue of the reference male and the reference female are averaged, and these averaged doses are both multiplied by the corresponding tissue weighting factor to determine the sex-averaged effective dose for the Reference Person (ICRP, 2007). It follows that effective dose does not provide an individual-specific dose, but rather provides a dose for a Reference Person under given exposure conditions (ICRP, 2007). The reference models and necessary reference parameter values are established and selected from a range of experimental investigations and human studies through judgements. For regulatory purposes, these models and parameter values are fixed by convention and are not subject to uncertainty.
However, thanks to a considerable amount of effort in the revision and improvement of models to make them more physiologically realistic, the ICRP models are now sufficiently sophisticated so that they can also be used to calculate organ and tissue absorbed doses for scientific purposes, and in many other areas, including toxicology, pharmacology and medicine. In these specific cases, uncertainties in parameters and variability between individuals may need to be taken into account. Reviews of the sources and levels of uncertainties have been published (Leggett et al., 1998; Harrison et al., 2001; Leggett, 2001; NCRP, 2009). They can be summarised as below.
‘Uncertainty’ here refers to the lack of knowledge of a central value for a population, and ‘variability’ refers to the quantitative differences between different members of a population. Although uncertainty and variability are distinct concepts, the variability in biokinetic characteristics within a population is often an important factor contributing to the uncertainty in a central estimate of a biokinetic quantity. This is because such variability contributes to the problem of identifying the central tendency of these characteristics in the population due to the small number of observations generally available, and because subjects of biokinetic studies are not usually selected at random. Variability in the biokinetics of radionuclides, pharmaceuticals or chemicals in human populations appears to result from many different physiological factors or modulating host factors of an environmental nature, including age, sex, pregnancy, lactation, exercise, disease, stress, smoking and diet. Large interindividual biokinetic variations sometimes persist in the absence of appreciable environmental differences, and suggest that these variations may be genetically controlled. In real-world situations, genetic and environmental factors may interact dynamically, producing sizable variations in the behaviour of substances taken into the human body.
The uncertainty in an internal dose assessment depends on: uncertainties associated with measurements used to determine the activity of a radionuclide in vivo or in a biological sample; uncertainties in the exposure scenario used to interpret the bioassay results, including factors such as the route of intake, the time pattern of intake, the specific radionuclide(s) taken into the body, and the chemical and physical form of the deposited radionuclide(s); and uncertainties in the biokinetic and dosimetric models used to interpret the bioassay results.
Uncertainties in time and route of intake are important in retrospective dose assessment using bioassay or body measurement. If an intake is not recognised for some time after an incident, and if total body retention and urinary and faecal excretion rates would diminish quickly, the assumed time pattern of intake could be the dominant uncertainty in the dose estimate. On the other hand, if a worker is exposed in the vicinity of an immediately recognised accidental release, or if total body retention and excretion rates are fairly constant, the time pattern of intake may be a negligible source of uncertainty in the dose estimate.
An additional and important source of uncertainties, for both prospective and retrospective dose assessment, is linked to the biokinetic models. The confidence that can be placed in predictions of a biokinetic model for an element or compound depends not only on uncertainties associated with parameter values of the model, but also on uncertainties associated with the model structure. Such uncertainties may arise because the structure provides an oversimplified representation of the known processes, because unknown processes have been omitted from the model, or because part or all of the model formulation is based on mathematical convenience rather than consideration of processes. Some combination of these limitations in model structure is virtually associated with all biokinetic models for radionuclides.
Other sources of uncertainties in biokinetic models are associated with the types of information used to construct the models for the elements. Available data may be of different types, including: (i) human data involving quantitative measurements of the element; (ii) observations of the behaviour of chemically similar elements; (iii) observations of the behaviour of the element in non-human species; and (iv) observations of the behaviour of one or more chemically similar elements in non-human species. Direct information on humans is the preferred type of information on which to base a biokinetic model. To some degree, this type of direct information is available for most essential elements, as well as for some important non-essential elements, such as caesium, lead, radium, uranium, americium and plutonium.
In some cases where information is missing, data on animals and chemical analogues may be used as surrogates, leading to uncertainties in the model parameters. Interspecies extrapolation of biokinetic data is based on the concept of a general biological regularity across the different species with regard to cellular structure, organ structure, and biochemistry. However, despite the broad structural, functional, and biochemical similarities among mammalian species, interspecies extrapolation of biokinetic data has proven to be an uncertain process. Similarities across species are often more of a qualitative than quantitative nature, in that two species which handle an internally deposited radionuclide in the same qualitative manner may exhibit dissimilar kinetics with regards to that substance. Moreover, there are important differences among the mammalian species, including differences in specialised organs, hepatic bile formation and composition, level of biliary secretion, urine volume and acidity, amount of fat in the body, magnitude of absorption or secretion in various regions of the alimentary tract, types of bacteria in the digestive tract, and microstructure and patterns of bone remodelling.
Similarly, biokinetic models for elements are often constructed partly or wholly from data for chemically similar elements, on the basis of empirical evidence that chemical analogues often exhibit close physiological similarities. For example, the alkaline earth elements calcium, strontium, barium, and radium exhibit many physiological as well as chemical similarities (ICRP, 1993), and the alkali metals rubidium and caesium closely follow the movement of their chemical analogue, potassium. There are, however, counterexamples to the premise that chemical analogues are also physiological analogues. For example, the alkali metals potassium and sodium share close physical and chemical similarities but exhibit diametrically opposite behaviours in the body, with potassium being primarily an intracellular element, and sodium being primarily an extracellular element. Moreover, some of the chemically similar elements that behave in a qualitatively similar fashion in the body may exhibit quite different kinetics. For example, caesium follows the behaviour of potassium in the body in a qualitative sense, but is distributed somewhat differently from potassium at early times after intake, and exhibits a substantially longer whole-body retention time.
The level of confidence that can be placed in a model value based on human data for a chemically similar element depends on the quality and completeness of the data for the analogue, as well as the expected strength of the analogy for the given situation. Whatever the quality of the data for the chemical analogue, the confidence interval should reflect the fact that some confidence in the predictive strength of the data is lost when the data are extrapolated across elements.
The strength of the chemical analogy for a given element depends largely on the extent to which the chemically similar elements have also been found to be physiologically similar. That is, the analogy would be considered strong for a pair of elements if a relatively large set of experimental data indicates that these elements have essentially the same qualitative behaviour in the body, and that their quantitative behaviour is either similar or differs in a predictable fashion. In view of counterexamples to the premise that chemically similar elements are necessarily physiologically similar, the chemical analogy does not provide high confidence if the elements in question have not been compared in animals or humans.
5. CONCLUSIONS
Determination of doses after internal exposure is a complex procedure that requires the use of models describing the behaviour of the radionuclides and the deposition of their energy in the tissues. Recent advances have provided models with increased physiological realism that in turn allows more realistic dosimetry. For radiological protection purposes (i.e. optimisation and demonstration of compliance with dose limits), these models are regarded as reference tools that are not subject to uncertainty. When these models are used in other areas such as toxicology, pharmacology, medicine, and dose reconstruction for epidemiological studies, uncertainty and variability need to be taken into account.
