Abstract
This paper does not necessarily reflect the views of the International Commission on Radiological Protection.
1. Introduction
Effective dose was originally introduced in the 1977 Recommendations of the International Commission on Radiological Protection (ICRP, 1977), at which time the over-riding concern was the control of occupational exposures. While the concept has remained essentially unchanged through the 1990 Recommendations (ICRP, 1991) to the 2007 Recommendations (ICRP, 2007), its use has been extended to members of the public of all ages, including in-utero exposures of the embryo and fetus. Particularly problematic is the use of effective dose in the assessment of medical exposures of patient populations of different ages, and where the requirement is for a measure of risk to individuals (Martin, 2007; Brenner, 2008).
Brenner (2008, 2012) proposed that effective dose represents flawed science, and should be replaced with a new quantity, ‘effective risk’, which could be used to provide estimates of risk to individuals from medical procedures, taking account of differences between males and females and at different ages at exposure. Wall et al. (2011) and Balonov and Shrimpton (2012) provided estimates of age- and sex-specific risk per unit effective dose for a range of medical diagnostic procedures to determine variations in comparison with the nominal risk coefficients derived by ICRP.
This paper outlines the purpose of effective dose and the underlying assumptions made in its calculation, and examines the proposal that it should be replaced by effective risk. Results obtained for the risk per unit effective dose for diagnostic x-ray procedures are summarised, with commentary on the use of effective dose as an indicator of risk.
2. Effective dose
The ICRP protection quantities – equivalent and effective dose – enable the summation of doses from internal emitters and from external sources to provide a single number for comparison with dose limits, constraints, and reference levels that relate to stochastic effects of whole-body radiation exposure. Thus, the primary application of effective dose is in the planning and demonstration of compliance in various situations of exposure of workers and members of the public. The calculation of effective dose can be seen as a three-step process, starting with the calculation of absorbed dose to organs and tissues, in gray (Gy; J kg−1). As radiation types differ in their ability to cause cancer per unit of absorbed dose, the second step is to multiply the calculated values of absorbed dose by radiation weighting factors that take account of the greater effectiveness of alpha particles and neutrons compared with beta particles and gamma rays. The results are termed ‘equivalent dose’, and expressed in sieverts (Sv). The final step is to sum the equivalent doses to individual organs and tissues, multiplying each by a tissue weighting factor that represents its contribution to total detriment from uniform whole-body irradiation. The intention is that the overall risk should be comparable, irrespective of the type and distribution of radiation exposure. Effective dose, in sieverts, is the well-known quantity that is often referred to simply as ‘dose’.
It is important to recognise that while effective dose is a risk-related construct, and is ideally suited for use in radiation protection, particularly in planning and optimising protection for workers and members of the public, it does not provide more than a rough estimate of dose and risk to individuals (Dietze and Menzel, 2004; ICRP, 2007; Dietze et al., 2009). Absorbed doses to organs and tissues are calculated in mathematical phantoms and used to provide sex-averaged values of effective dose for a ‘reference person’ (ICRP, 2007; Harrison and Day, 2008). Radiation and tissue weighting factors are chosen as simple sets of values that do not fully reflect our scientific understanding of radiation risks. For example, no account is taken of recognised differences between low-energy mammography x rays and Co-60 gamma rays (Hill, 2004). There is evidence that the relative effectiveness of different radiations is dependent on cancer type and may, for example, be greater for liver cancer than for leukaemia (Harrison and Muirhead, 2004). Tissue weighting factors are age- and sex-averaged values that conceal substantial differences between cancer risk estimates for males and females, and at different ages, dependent on cancer type (ICRP, 2007; Harrison and Day, 2008).
Effective dose and the tissue weighting factors used in its calculation relate to detriment from radiation-induced cancer and hereditary effects. Detriment is calculated as defined by ICRP and explained in detail in Annex A of
The application of effective dose in the control of stochastic effects for protection purposes relies on a number of key assumptions, principally that:
a linear non-threshold (LNT) relationship between dose and risk applies at low doses; acute and chronic exposures are equally effective at low doses; and external dose and internal dose from radionuclides deposited in body tissues can be summed, taking account of radiation quality through simple adjustments using radiation weighting factors.
3. Effective risk
Lifetime attributable risk of specific cancers after irradiation at different ages. Number of cases per 106 exposed to a single dose of 10 mGy. Selected data from BEIR VII (NRC/NAS, 2006).
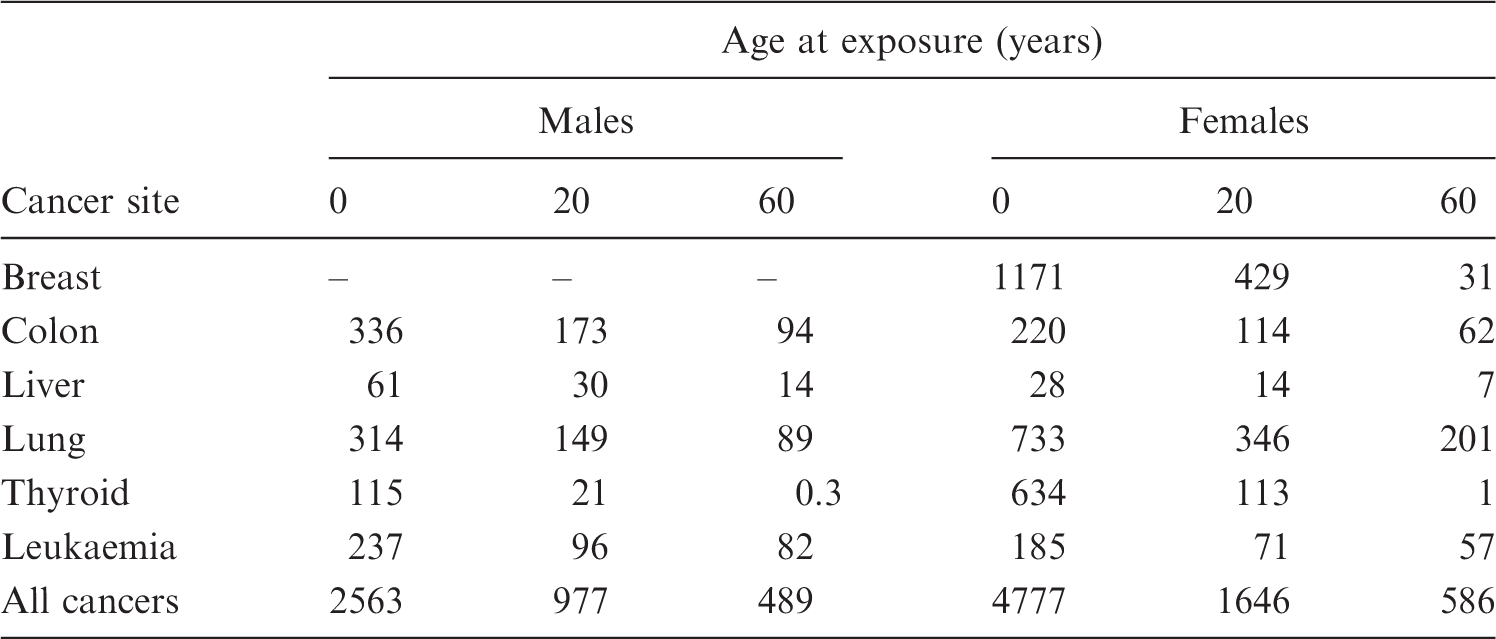
Data of the type shown in Table 1 were also used by ICRP in their calculation of stochastic detriment and tissue weighting factors (see above), although with risks transferred to a composite of seven populations rather than a US population. Effective risk would not be a useful replacement for effective dose for its principal purpose of providing a single metric for control of exposures worldwide. An important argument against the effective risk quantity concerns the dose range to which this quantity would be applied. In radiation protection practice, the effective doses recorded are generally far below the 20-mSv limit for occupationally exposed persons, often as low as 10 µSv or even less. Dose measurements and assessments are well established in this dose range and, particularly for external exposures, quality assurance programmes ensure reliable measurements, even at such low doses. In addition, the operational dose quantities defined for external exposure and used for calibrating dosimeters are taken to provide sufficiently accurate assessments of effective dose for the purposes of radiation monitoring and dose recording. However, while doses can be measured with some precision at such low levels, the corresponding risks are uncertain or even unknown.
The protection system relies on the assumption of an LNT dose–response relationship. The LNT assumption is implicit in the addition of external and internal doses of different magnitudes, with different temporal and spatial patterns of delivery. However, a clear distinction should be drawn between the necessary and justifiable reliance on the LNT dose-response relationship for protection purposes and the assumption of its scientific validity extending to very low doses and all stochastic risks. ICRP (2007) concluded that the validity of the LNT dose–response assumption may prove to be beyond resolution for the foreseeable future. Hence, individual ‘risk monitoring’ at low doses can be judged to be unhelpful from a protection perspective, and scientifically misleading in implying greater knowledge of the risks of low-dose exposures than is currently warranted.
4. Doses and risks from diagnostic procedures
Typical doses from x-ray examinations of adults in the UK (from Balonov and Shrimpton (2012).
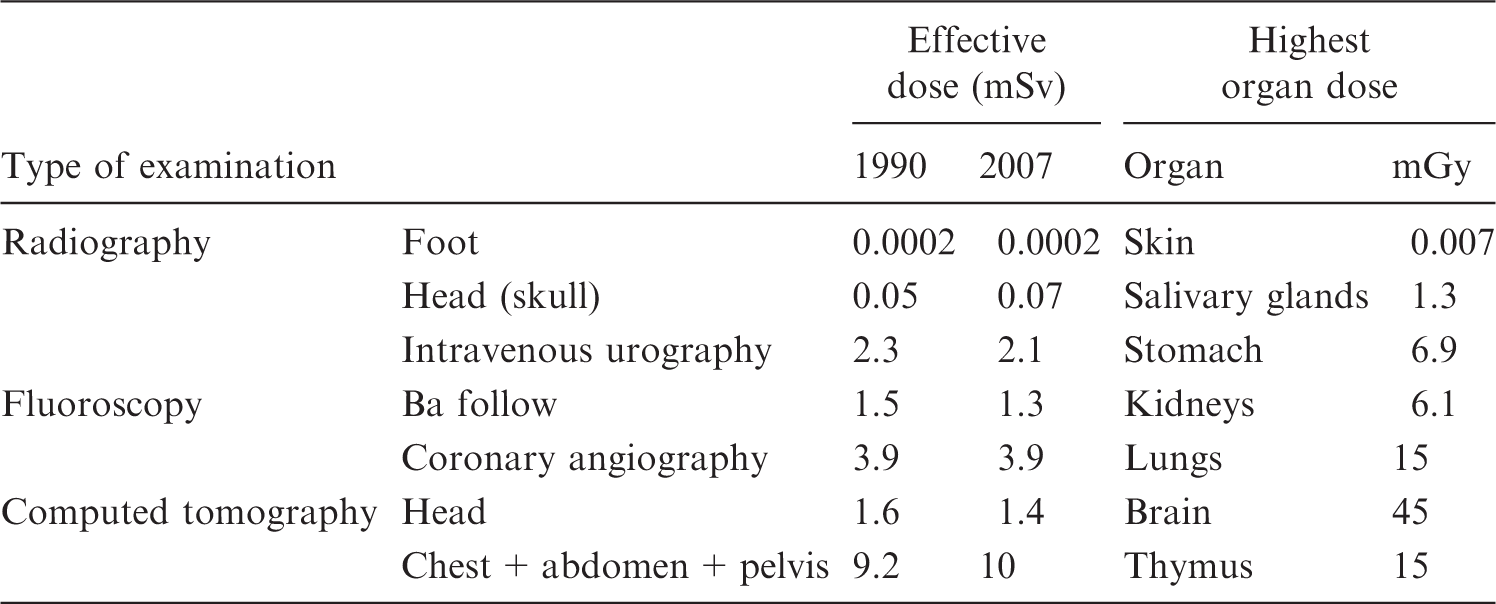
5. Discussion
Effective dose is a radiation protection quantity that is used to set limits, constraints, and reference levels that apply to reference workers or reference members of the public. It provides an elegant solution to the requirement for a single quantity that enables the summation of all radiation exposures, from external exposures and from radionuclides entering the body by inhalation or ingestion. Effective dose is used as a risk-related quantity for the optimisation of protection below constraints and reference levels. For protection purposes, the reasonable assumption is made of an LNT dose–response relationship down to very low doses of a few microsieverts or even less. However, while doses can be calculated with reasonable reliability at such low levels, the associated risk is uncertain.
Effective dose has proved to be a useful tool in controlling exposures received by patients undergoing medical diagnostic and interventional procedures. However, its use to provide estimates of risk to individual patients goes beyond its intended use (ICRP, 2007; Menzel and Harrison, 2012). Brenner (2008, 2012) suggested that effective dose should be replaced by effective risk as a more scientifically based quantity. Unfortunately, this approach ignores the uncertainties associated with the LNT dose–response relationship as applied to all cancer types, and gives a misleading impression of our ability to quantify risks at low doses.
A clear distinction should be drawn between the application of science in the definition of the risk-adjusted protection quantity, ‘effective dose’, which has proved to be of considerable practical utility, and the use of best scientific information to calculate risks to individuals and population groups. Risk estimation can be specific to individuals of known body mass and dimensions, applying age- and sex-specific risk factors derived for the most appropriate population group. An example referred to by Harrison (2015) in this issue is the National Aeronautics and Space Administration’s estimates of risk to individual astronauts. Such scientific best estimates of risk should also consider associated uncertainties.
Although effective dose was not intended as a measure of risk to individuals, it might reasonably be used as an approximate indicator of risk. Considering a range of medical x-ray examinations and comparing best estimates of risk with values obtained using effective dose and nominal risk coefficients, Balonov and Shrimpton (2012) showed that effective dose might underestimate risk by up to a factor of four for children, and overestimate risk by approximately one order of magnitude for older adults. Making such simple adjustments to the risk associated with effective dose could help make informed judgements on the justification of examinations without implying greater knowledge of radiation risks at low doses than is warranted.
ICRP currently has a task group responsible for preparing a report on the use of effective dose (Harrison, 2015). This task group, led by Committee 2, has membership from Committees 1, 3, and 4, as well as external experts. Useful guidance on restrictions on the use of the quantity is provided in Annex B to the 2007 Recommendations (ICRP, 2007). This guidance needs to be expanded and proposals made for the control of exposures and risk management in situations where ‘effective dose’ should not be used. An important focus of the report will be medical exposures.
