Abstract
Objective:
This meta-analysis aimed to assess and compare the results of microwave ablation (MWA) and surgical intervention for the management of solitary T1N0M0 papillary thyroid carcinoma (PTC), including T1A- and T1B-staged tumors.
Methods:
A search was conducted using PubMed, Embase, Cochrane Library, Web of Science, and Scopus. The variables included in the study were oncological outcomes, complications, surgery-related indicators, and tumor volume.
Results:
The findings from the meta-analysis indicated that, in comparison to surgical intervention, MWA treatment for isolated T1N0M0 PTC did not show significant differences in local tumor progression (P = .91), new-onset tumors (P = .88), lymph node metastasis (P = .19), technical success rate (P = 1.00), local recurrence (P = .50), transient hyperthyroidism (P = .60), permanent hyperthyroidism (P = .18), permanent hypoparathyroidism (P = .07), transient hoarseness (P = .29), hematoma (P = .12), infection (P = .27), and airway obstruction (P = .14). In contrast, MWA significantly reduced the overall complication rate, hypothyroidism, transient hypoparathyroidism, permanent hoarseness, dysphagia, and laryngeal edema. MWA also shortened the operation time, bleeding volume, and hospital stay, lowered the treatment cost, reduced the size of the incision, reduced the tumor volume, and improved quality of life (all P < .05). Subgroup analysis based on tumor stages showed similar trends in the outcome indicators within the T1A/T1B subgroup. However, in the new-onset tumors, the risk of new tumors increased in the T1B subgroup (P = .03).
Conclusion:
Ultrasound-guided MWA may be a potential treatment option for highly selected patients with isolated T1N0M0 PTC. It shows advantages in short-term outcomes, but its long-term oncological safety still needs to be confirmed by longer and higher-quality studies. However, in the T1B subgroup, preoperative ultrasound or computed tomography (CT) examinations are required to screen for suspicious lymph nodes. If any suspicious metastasis is found, surgical treatment should be given priority.
Introduction
Papillary thyroid carcinoma (PTC) represents one of the most prevalent forms of malignant tumors within the endocrine system. 1 In recent years, the incidence of PTC has increased annually worldwide. PTC is a significant health burden and an economic pressure for patients and society. 2 Solitary T1N0M0 PTC refers to a single, localized, and nonmetastatic early PTC. 3 Currently, surgical resection remains the primary therapeutic approach; however, this method has several drawbacks, including significant tissue trauma, extended recovery periods, and a range of complications. Notably, these complications may encompass injury to the recurrent laryngeal nerve (RLN), hypothyroidism, and necessitate lifelong thyroid hormone replacement therapy. These can all negatively impact a patient’s quality of life. 4 Hence, more minimally invasive treatment methods are desired. 5
Microwave ablation (MWA) is a minimally invasive surgery. Under ultrasound guidance, microwave electrodes are inserted into the tumor. 6 Through thermal effects, cancerous tissues are coagulated, dehydrated, and necrotized to achieve the therapeutic goal. 6 It has the advantages of small trauma, quick recovery, short hospital stay, and good esthetic effects. 7 As an emerging minimally invasive treatment option, MWA technology has received increasing attention in recent years. 8 Previous studies have shown that MWA significantly reduces surgical risks and shortens recovery time, especially when applied to early PTC. 9 Considering this, MWA may have a broad application in treating isolated T1N0M0 PTC, thus, reducing complications and medical costs. 10
Currently, a growing body of research on MWA as a therapeutic option for larger PTC, particularly for tumors classified at the T1B stage, has emerged. 11 Although MWA is progressively being adopted in clinical settings, the effectiveness and safety of this procedure for patients diagnosed with T1N0M0 PTC remain subjects of debate. Concerns relating to postoperative recurrence, metastasis, and complications have not been addressed adequately. 12 Therefore, this meta-analysis aimed to comprehensively analyze the results of multiple studies to compare the efficacy and safety of MWA and surgery in the treatment of isolated T1N0M0 PTC and to provide guidance for clinical treatment options.
Materials and Methods
This systematic review was conducted in accordance with the guidelines set forth by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses and has been officially registered in the PROSPERO database under the registration number CRD420251110978.
Search Methods
A comprehensive search was conducted across several databases, including PubMed, Embase, Cochrane Library, Web of Science, and Scopus for articles published from the inception of these databases until June 30, 2025. The search was conducted by combining Mesh terms (“Thyroid Cancer, Papillary,” “Ablation Techniques,” and “Thyroidectomy”) with their Entry Terms. Literature and references on MWA and surgery for isolated T1N0M0 PTC were included. This search does not restrict the language. Two reviewers, P.X. and D.L., independently conducted the search and addressed any differences through discussion. In instances where the specifics of the test report were lacking or the data provided were insufficient, we reached out to the authors through email to request additional information, thereby enhancing the comprehensiveness of the literature coverage. Specific search details are provided online (Supplementary File 1).
Inclusion and Exclusion Criteria
Inclusion Criteria: (1) A diagnosis of PTC was made using an ultrasound-guided fine-needle aspiration biopsy. (2) The tumor size measured using ultrasound was ≤2.0 cm. (3) Preoperative imaging (ultrasonography or CT) revealed no evidence of nodal or distant metastases. (4) To directly compare US-guided MWA with surgery (lobectomy or total thyroidectomy) for the treatment of isolated T1N0M0 PTC. (5) No history of neck radiation exposure. (6) The patient had no history of thyroid surgery.
Exclusion Criteria: (1) Case reports, reviews, and conference summaries. (2) Patients with multifocal PTC (≥2 tumor lesions) were excluded in the study. (3) Studies involving overlapping populations. (4) Severe coagulation disorders or bleeding tendencies. (5) Severe heart, lung, or kidney dysfunction, or other serious diseases. (6) History of neck surgery or iodine-131 treatment. (7) Patients who refused treatment or did not cooperate with follow-up. (8) Patients with incomplete data or those lost to follow-up. (9) Patients intolerant of this treatment.
Data Extraction
Subsequent to the elimination of duplicate entries, 2 separate investigators (L.Y. and X.W.) conducted an examination of the titles and abstracts of the relevant articles. Subsequently, the full texts of these articles were subjected to a more thorough assessment based on the established inclusion and exclusion criteria. Employing standardized data extraction templates, the investigators systematically gathered and enumerated pertinent information, which includes (a) Characteristics of the Study: lead author, type of study design, year of publication, demographic details of patients (including number, age, and gender), therapeutic approaches, as well as the clinical status of the patients. (b) Main Results: oncological outcomes (local tumor progression [LTP], new-onset tumors, lymph node metastasis [LNM], distant metastasis [DM], technical success rate, local recurrence [LR]), complications (overall complication rate [OCR], transient hypothyroidism, permanent hypothyroidism, transient hyperthyroidism, permanent hyperthyroidism, transient hypoparathyroidism, permanent hypoparathyroidism, transient hoarseness, permanent hoarseness, bleeding/hematoma, infection, dysphagia/laryngeal edema, airway obstruction). (c) Other Results: surgery-related indicators (surgery time, bleeding volume, hospital stay, treatment cost, incision size, and quality of life score) and tumor volume changes (3, 6, 12, and 24 months after surgery in the ablation group). Any disagreements were resolved through discussions with a senior physician (G.H.).
Quality Assessment
Given that all studies incorporated in this analysis were non-randomized controlled trials, the Newcastle-Ottawa Scale (NOS) 13 was used to appraise the quality of the included literature. Two researchers (W.C. and G.H.) independently used this scale to assess the cohort study quality. When the score of a certain study is ≥7 points, it is regarded as high-quality. In the event of differing viewpoints, the final decision was reached through deliberation or by seeking advice from an external specialist (Z.Z.).
Statistical Analysis
All analyses were conducted utilizing RevMan 5.3 or R software 4.4.0. The analysis of dichotomous variables was conducted by computing the 95% confidence intervals (CIs) and the risk ratios (RRs). In the case of continuous variables, the mean difference (MD) or standardized MD (SMD) served as the effect size metric, accompanied by the calculation of the corresponding 95% CI. For the entire dataset, the overall effect size was determined with a significance threshold of P < .05. Variability among the studies was assessed using Cochran’s test. A P < .1 indicated the presence of heterogeneity. Additionally, the I2 statistic was employed to quantify the extent of this heterogeneity. When the I2 statistic was <50%, it indicated a low degree of heterogeneity, thus, warranting the application of a fixed-effect model for analysis. Conversely, if I2 is ≥50%, this indicates the presence of heterogeneity or significant heterogeneity among the results.
It is necessary to further analyze the source of heterogeneity and select a random-effects model. Based on tumor dimensions, the research was categorized into 2 subgroups: the T1A subgroup, which includes tumors measuring 1.0 cm or smaller, and the T1B subgroup, encompassing tumors larger than 1.0 cm but not exceeding 2.0 cm.
Results
Literature Screening Process and Results
A comprehensive search of the 5 databases yielded 2039 articles. Ultimately, a total of 9 articles14 -22 were included in the study. A flowchart illustrating the literature retrieval process is shown in Figure 1.
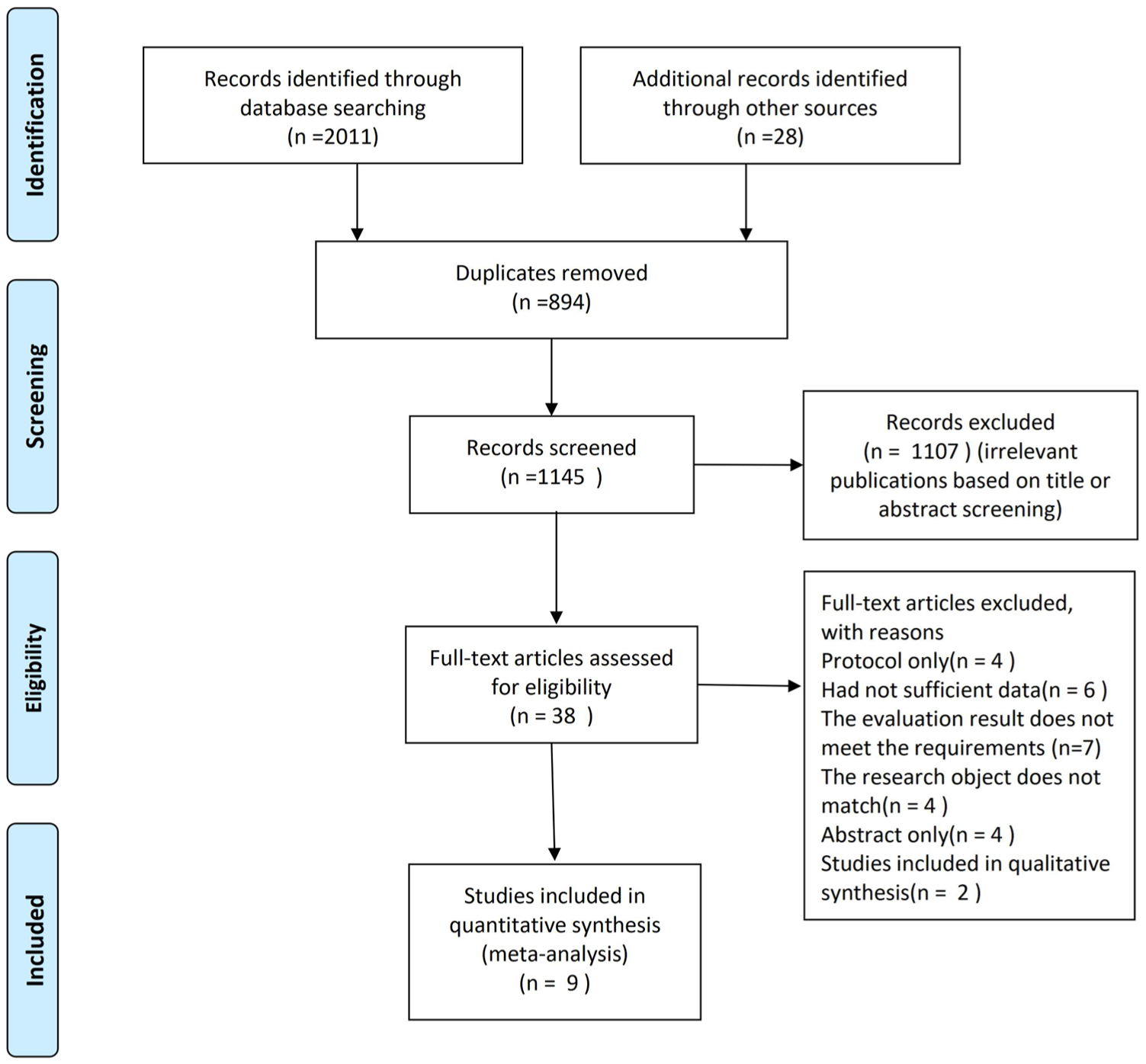
Flow diagram for searches.
Basic Features of Included Literature
A total of 3011 patients were comprehensively enrolled in these investigations, comprising 1494 individuals who underwent MWA and 1517 who underwent surgical procedures. Table 1 presents the quality ratings of the literature.
Characteristics of Included Studies.
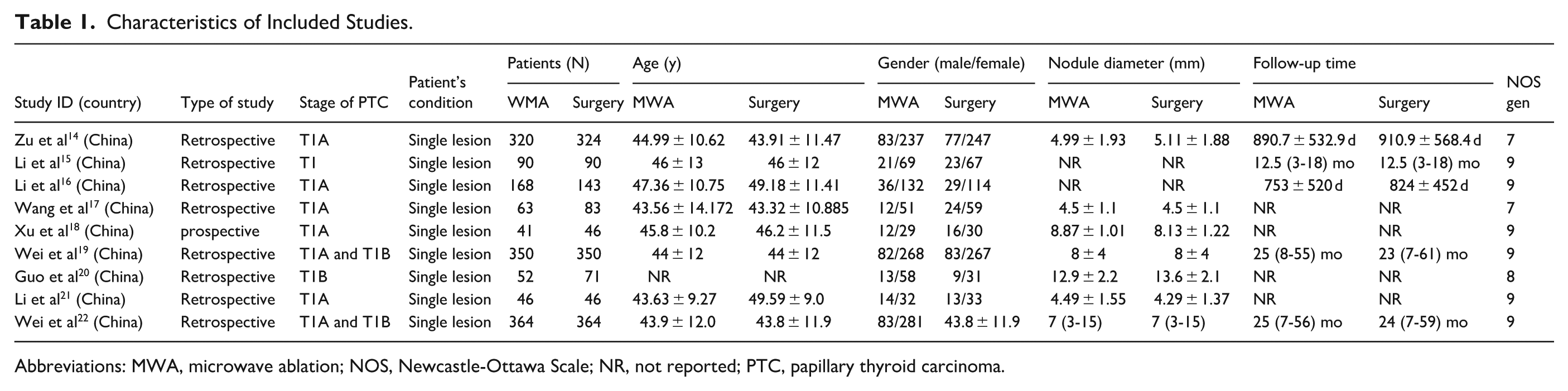
Abbreviations: MWA, microwave ablation; NOS, Newcastle-Ottawa Scale; NR, not reported; PTC, papillary thyroid carcinoma.
Quality Assessment
Nine studies were cohort studies (8 retrospective and 1 prospective). We used the NOS to assess the risk of bias in the included studies. In terms of overall risk of bias, all included studies were considered high-quality literature when their scores were ≥7. The quality scores are shown in Table 1.
Main Results of the Meta-Analysis
Tumor-Related Indicators
Local Tumor Progression
Data related to LTP were extracted from 5 studies14,15,19,20,22 (I2 = 0%, P = .97). The fixed-effects model revealed that there was no statistically significant difference in LTP when comparing the MWA and surgery groups (RR = 0.98; 95% CI = 0.63-1.52; P > .05; Figure 2A).
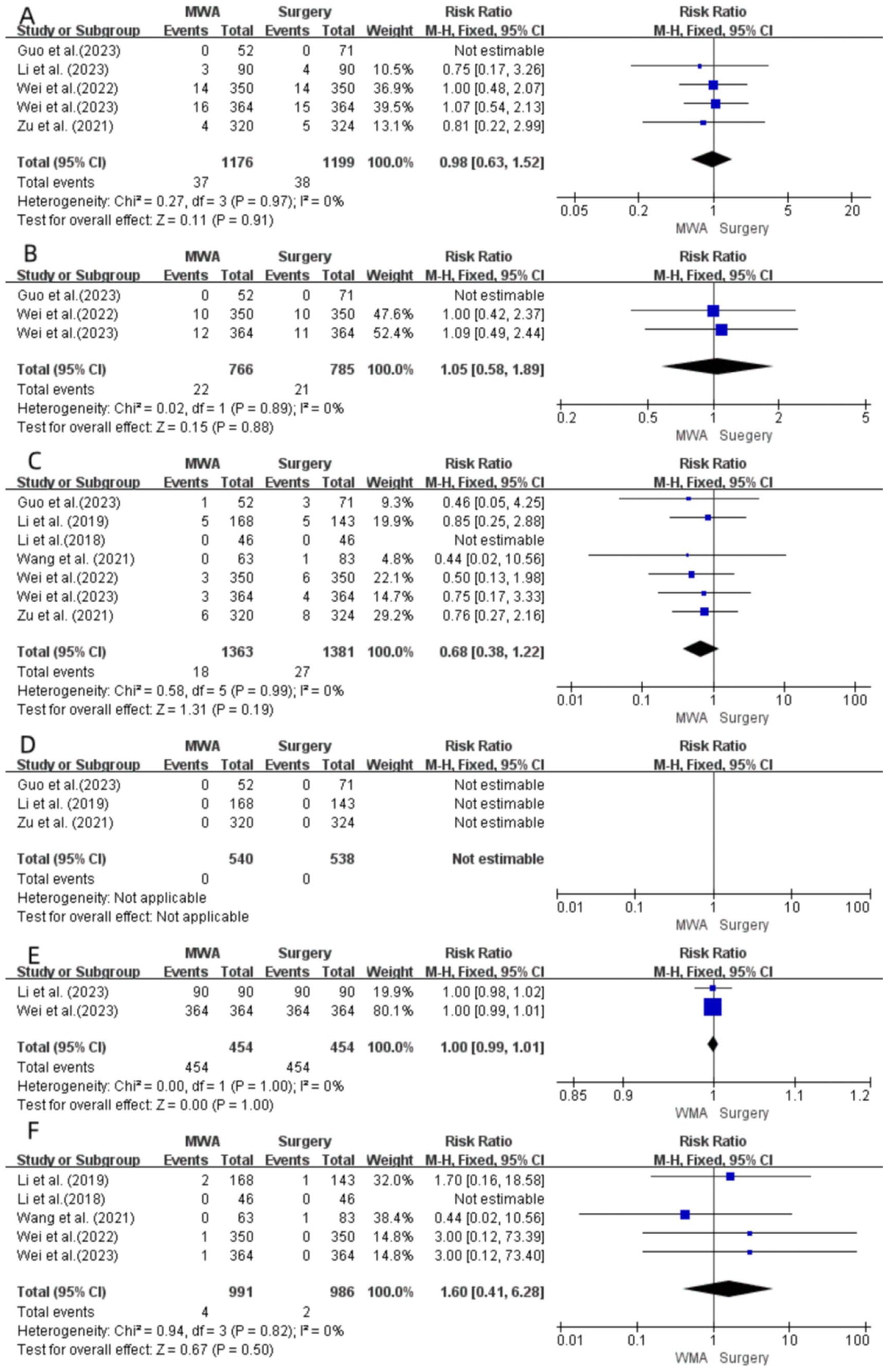
Forest plot of local tumor progression (A), new-onset tumors (B), lymph node metastasis (C), distant metastasis (D), technical success rate (E), and local recurrence (F) between the MWA group and surgery group. MWA, microwave ablation.
New-Onset Tumors
Three studies19,20,22 conducted comparative analyses of data related to newly developed tumors in the 2 groups. The included research indicators exhibited heterogeneity (I2 = 0%, P = .89), prompting the use of a fixed-effects model for the analysis. The findings indicated that there was no statistically significant variation in the occurrence of newly developed tumors between the MWA and surgical groups (RR = 1.05; 95% CI = 0.58-1.89; P > .05; Figure 2B).
Lymph Node Metastasis
The 7 studies14,16,17,19 -22 were documented in LNM (I2 = 0%; P = .99 > .1). A fixed-effects model was used for the analysis. The findings revealed that there was no statistically significant difference in LNM between the MWA and surgery groups (RR = 0.68; 95% CI = 0.38-1.22; P > .05; Figure 2C).
Distant Metastasis
Three studies14,16,20 documented DM (I2 = 0%, P < .1). Distant metastases were not found in the experimental and control groups. Hence, the RR values and 95% CI for the meta-analysis could not be calculated (Figure 2D).
Technical Success Rate
Two papers15,22 compared the technical success rates between the 2 groups. The included research indicators were heterogeneous (I2 = 0%, P = 1.00), and the fixed-effect model was used for analysis. The results showed that there was no significant difference in technical success rate between the MWA and the surgery groups (RR = 1.00; 95% CI = 0.99-1.01; P > .05; Figure 2E).
Local Recurrence
Five studies16,17,19,21,22 conducted comparative analyses of LR data between the 2 groups. The included research indicators exhibited heterogeneity (I2 = 0%, P = .82), and a fixed-effects model were employed for analysis. The findings revealed that there were no statistically significant differences in LR between the MWA and surgical groups (RR = 1.60; 95% CI = 0.41-6.28; P > .05; Figure 2F).
Complications
Overall Complication Rate
Six articles14,16,18,20 -22 compared OCR between the 2 groups. The included studies indicated a high degree of heterogeneity (I2 = 58%, P = .04), and a random-effects model was adopted. The results indicated that OCR in patients who had MWA was significantly reduced (RR = 0.44; 95% CI = 0.25-0.78; P = .005; Figure 3A).
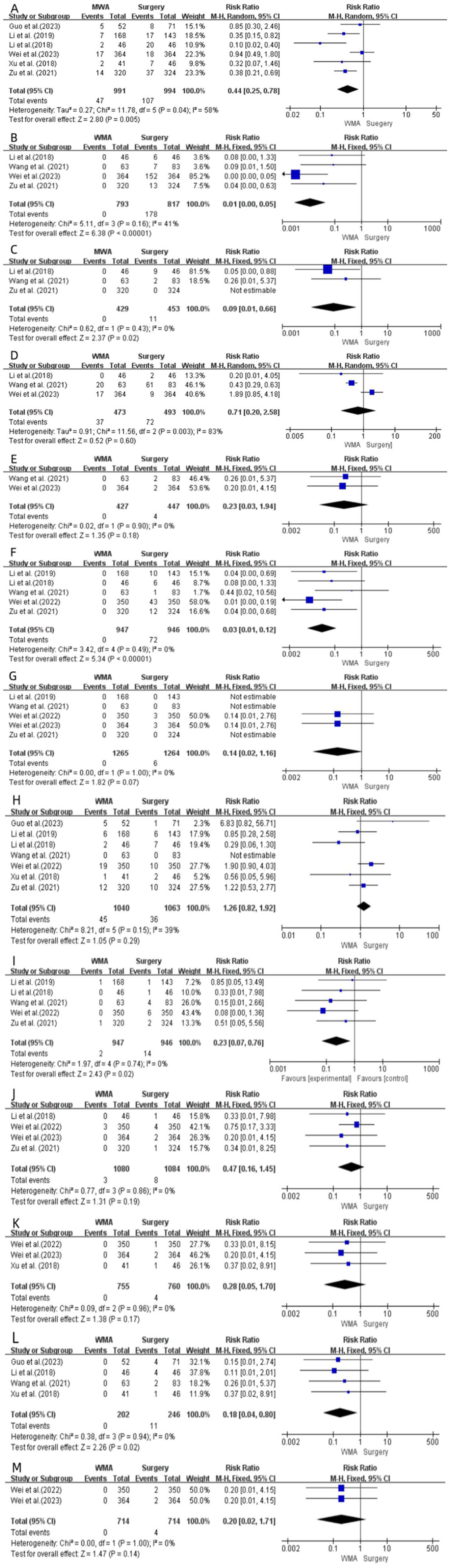
Forest plot of overall complication rate (A), transient hypothyroidism (B), permanent hypothyroidism (C), transient hyperthyroidism, (D) permanent hyperthyroidism (E), transient hypoparathyroidism (F), permanent hypoparathyroidism (G), transient hoarseness (H), permanent hoarseness (I), hemorrhage hematoma (J), infection (K), dysphagia/throat edema (L), and airway obstruction (M) between the MWA group and surgery group. MWA, microwave ablation.
Hypothyroidism
Four studies14,17,21,22 reported transient hypothyroidism (I2 = 41%; P = .16) and 3 studies14,17,20 reported permanent hypothyroidism (I2 = 0%; P = .43). A fixed-effects model was used to analyze transient hypothyroidism. The meta-analysis showed that the risk for patients who had MWA was significantly reduced (RR = 0.01; 95% CI = 0.00-0.05; P < .00001; Figure 3B). For permanent hypothyroidism, using a fixed-effects model, the meta-analysis showed that the risk for patients who underwent MWA was also significantly reduced (RR = 0.09; 95% CI = 0.01-0.66; P = .02; Figure 2C).
Hyperthyroidism
Three studies17,21,22 reported transient hyperthyroidism (I2 = 83%; P = .003) and 2 studies17,22 reported permanent hyperthyroidism (I2 = 0%; P = .90). For transient hyperthyroidism, a random-effects model was applied. The meta-analysis demonstrated no significant difference between MWA and surgery (RR = 0.71; 95% CI = 0.20,2.58; P > .05; Figure 3D). A fixed-effects model was used for permanent hyperthyroidism. The meta-analysis showed no significant difference in risk between MWA and surgery (RR = 0.23; 95% CI = 0.03-1.94; P = .18; Figure 3E).
Hypoparathyroidism
Five studies14,16,17,19,21 reported transient hypoparathyroidism (I2 = 0%; P = .49) and 5 studies14,16,17,19,22 reported permanent hypoparathyroidism (I2 = 0%; P = 1.00). A fixed-effects model was used to analyze transient hypoparathyroidism. Meta-analysis demonstrated a significantly reduced risk in patients undergoing MWA (RR = 0.03; 95% CI = 0.01-0.12; P < .00001; Figure 3F). For permanent hypoparathyroidism, a fixed-effects model was used, and the meta-analysis showed no significant difference in risk between MWA and surgery (RR = 0.14; 95% CI = 0.02-1.16; P > .05; Figure 3G).
Hoarseness
Seven studies14,16 -21 reported transient hoarseness (I2 = 39%; P = .15) and 5 studies14,16,17,19,21 reported permanent hoarseness (I2 = 0%; P = .74). For transient hoarseness, the fixed-effect model showed no significant difference between MWA and surgery (RR = 1.26; 95% CI = 0.82-1.92; P = .29; Figure 3H). Regarding permanent hoarseness, the fixed-effect model demonstrated a significantly lower risk of hoarseness in patients undergoing MWA than in those who received surgery (RR = 0.23; 95% CI = 0.07-0.76; P = .02; Figure 3I).
Hemorrhage/Hematoma
Four studies14,19,21,22 reported hemorrhage/hematoma (I2 = 0%, P = .86). A fixed-effects model analysis showed no significant difference between MWA and surgical approaches (RR = 0.47; 95% CI = 0.16-1.45; P = .19; Figure 3J).
Infection
Data on infections were extracted from 3 studies18,19,22 (I2 = 0%; P = .96). The fixed-effect model indicated no significant difference between MWA and surgery (RR = 0.28; 95% CI = 0.05-1.70; P = .17; Figure 3K).
Dysphagia/Throat Edema
Data related to dysphagia/throat edema were extracted from 4 studies17,18,20,21 (I2 = 0%; P = .94). The fixed-effects model demonstrated that patients who underwent MWA showed a significantly lower risk (RR = 0.18; 95% CI = 0.04-0.80; P = .02; Figure 3L).
Airway Obstruction
Data on airway obstruction were extracted from 2 studies19,22 (I2 = 0%; P = 1.00). The fixed-effect model indicated no significant difference between MWA and surgery (RR = 0.20; 95% CI = 0.02-1.71; P = .14; Figure 3M).
Meta-Analysis of Other Results
Surgical Indicators
Surgery Time
Data related to the surgery time were extracted from 7 studies14,17 -22 (I2 = 95%; P < .00001). Using the random-effects model, the meta-analysis results showed that the surgery time of patients who underwent MWA was significantly shorter than those who had surgery (MD = −58.36; 95% CI = −66.71 to −50.01; P < .00001; Figure 4A).
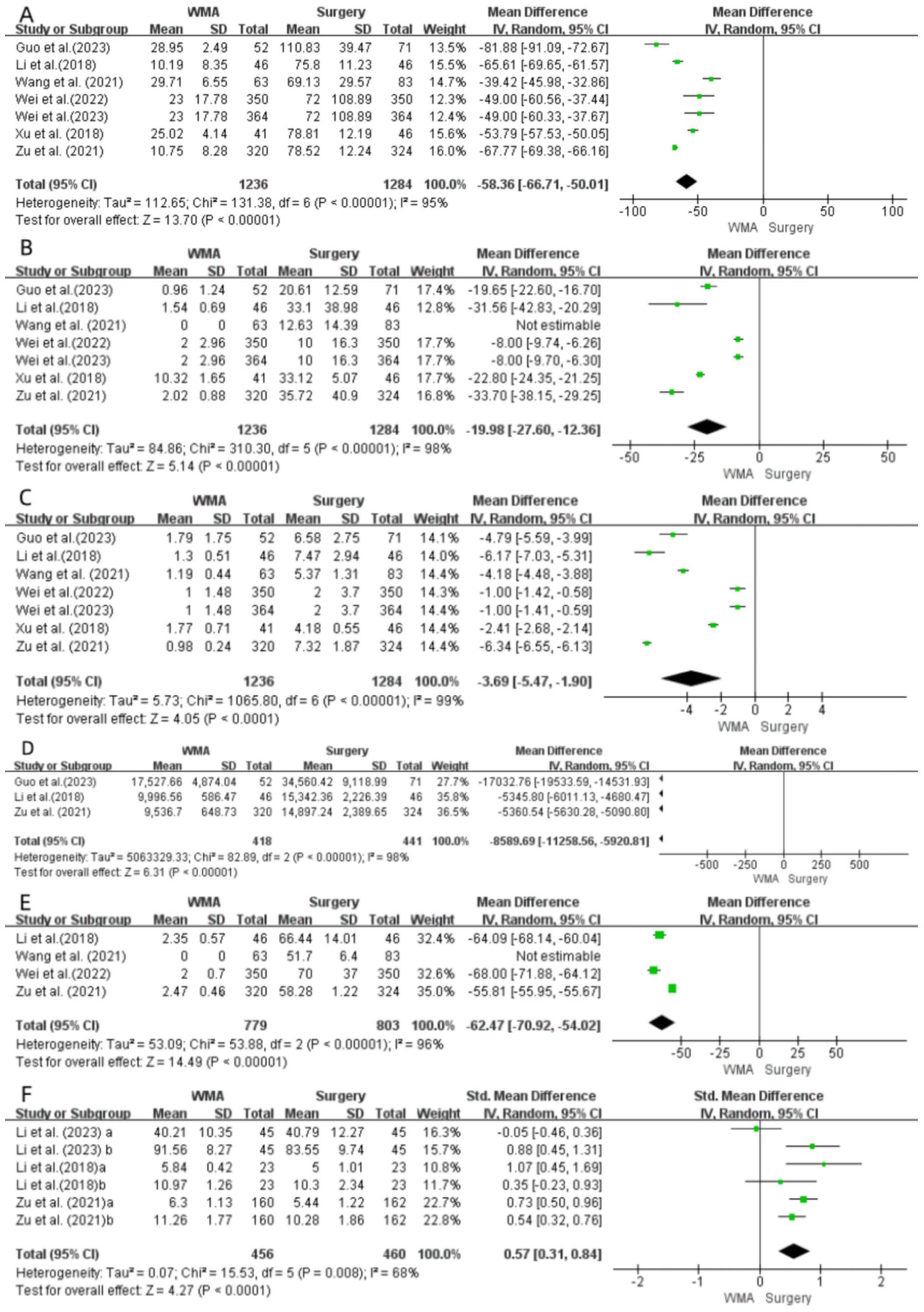
Forest plot of surgery time (A), bleeding volume (B), hospital stay (C), treatment cost (D), incision size (E), and quality of life score (F) between the MWA group and surgery group. MWA, microwave ablation.
Bleeding Volume
Seven studies14,17 -22 reported bleeding volume (I2 = 98%; P < .00001). The random-effects model showed that the amount of bleeding volume in patients who underwent MWA was significantly less than those who had surgery (MD = −19.98; 95% CI = −27.60 to −12.36; P < .00001; Figure 4B).
Hospital Stay
Seven studies14,17 -22 reported on the length of hospital stay (I2 = 99.2%; P < .00001). The random-effects model indicated that the length of hospital stay for patients who underwent MWA was significantly shorter than those who had surgery (MD = −3.69; 95% CI = −5.47 to −1.90; P < .00001; Figure 4C).
Treatment Cost
Data related to treatment costs were extracted from 3 studies14,20,21 (I2 = 98%; P < .00001) and indicated that the treatment cost of MWA was significantly lower than that for patients undergoing surgery (MD = −8589.69; 95% CI = −11 258.56 to −5920.81; P < .00001; Figure 4D).
Incision Size
Data related to incision size were extracted from 4 studies14,17,19,21 (I2 = 96%; P < .00001). The random-effects model indicated that the incision size in patients undergoing MWA was significantly smaller than that in surgical treatment (MD = −62.47; 95% CI = −70.92 to −54.02; P < .00001; Figure 4E).
Quality of Life Score
Data related to quality of life scores were extracted from 3 studies14,15,21 (I2 = 68%; P = .008). The random-effects model showed that patients undergoing MWA had significantly higher quality of life scores than those who underwent surgical treatment (WMD = 0.57; 95% CI = 0.31-0.84; P < .00001; Figure 4F).
Tumor Volume Changes in the MWA Group
Three Months Postoperatively
A total of 7 articles14,16,17,19 -22 investigated changes in tumor volume in the MWA group 3 months after surgery. The heterogeneity of the included research indicators was high (I2 = 99%, P < .00001), and a random-effects model was adopted. After comparing the operation before and after in the MWA group, the analysis concluded that the tumor volume was greater than that before surgery 3 months after operation, and the difference was statistically significant (SMD = 1.89; 95% CI = 1.08-2.70; P < .00001; Figure 5A).
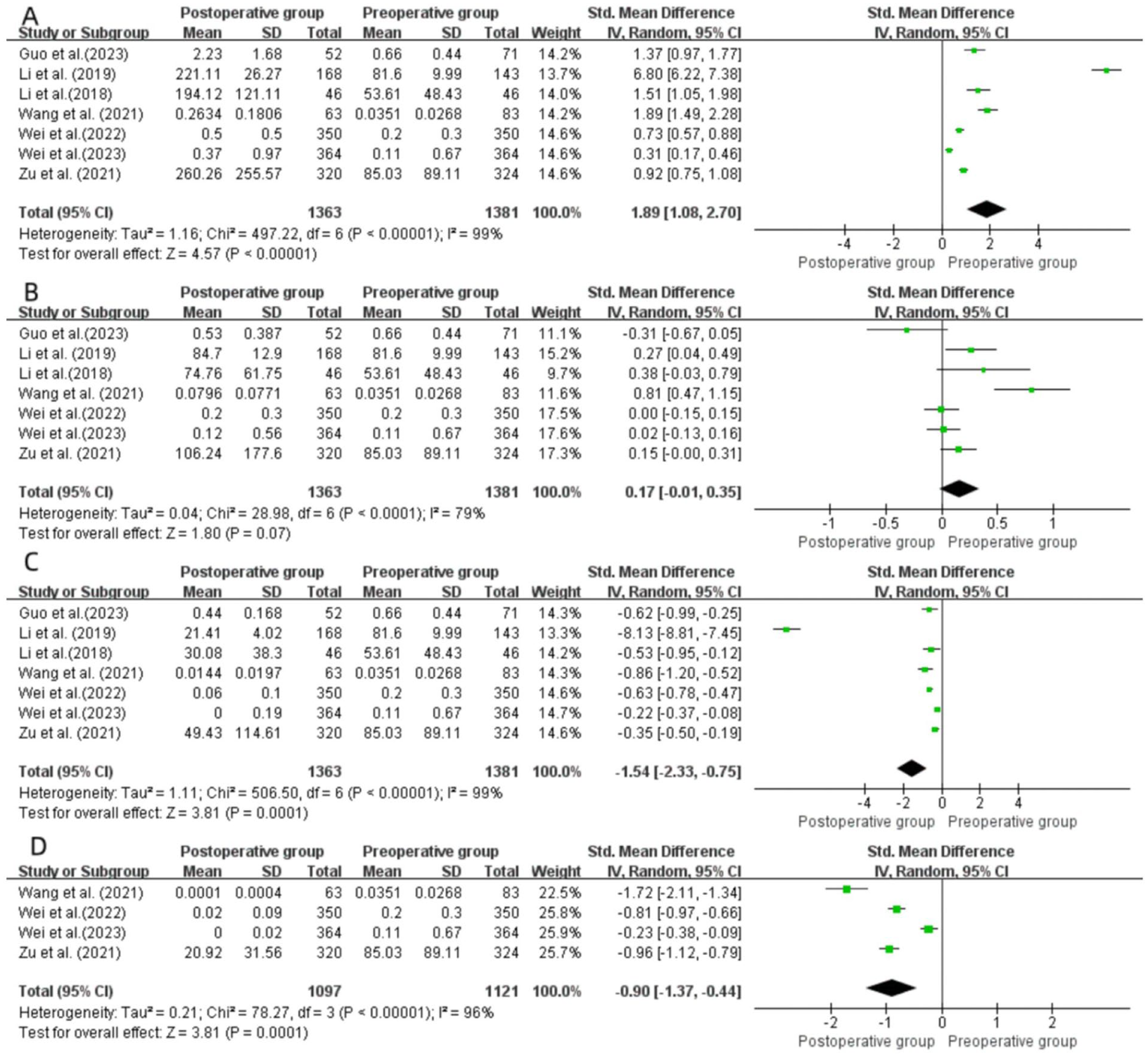
Forest plot of 3 months (A), 6 months (B), 12 months (C), and 24 months (D) after MWA. MWA, microwave ablation.
Six Months Postoperatively
A total of 7 studies14,16,17,19 -22 investigated tumor volume changes in the WMA group 6 months postsurgery. The included studies exhibited significant heterogeneity (I2 = 79%, P < .00001), and a random-effects model was employed. Through intragroup pre- and postoperative comparisons within the WMA group, the analysis concluded that there was no significant difference in tumor volume between 6 months postsurgery and preoperative measurements (SMD = 0.17; 95% CI = −0.01 to 0.35; P = .07; Figure 5B).
Twelve Months Postoperatively
A total of 7 studies14,16,17,19 -22 investigated tumor volume changes in the MWA group at 12 months postsurgery. The included studies exhibited significant heterogeneity in research metrics (I2 = 99%, P < .00001), and a random-effects model was employed. Meta-analysis suggested that the volume of the tumor was smaller than that before surgery after 12 months, and the difference was statistically significant (SMD = −1.54; 95% CI = −2.23 to −0.75; P = .0001; Figure 5C).
Twenty-Four Months Postoperatively
A total of 4 articles14,17,19,22 investigated the changes in tumor volume in the MWA group 24 months after surgery. The heterogeneity of the included research indicators was high (I2 = 96%, P < .00001) and a random-effects model was adopted. By comparing the operation before and after within the MWA group, it was concluded that the tumor volume 24 months after surgery was smaller than that before surgery, and the difference was statistically significant (SMD = −0.90; 95% CI = −1.37 to −0.44; P = .0001; Figure 5D).
Subgroup Analysis
Based on tumor size, we classified T1-stage tumors into T1A and T1B subgroups. Compared with patients in the T1 group, those in the T1A subgroup demonstrated a statistically significant advantage in transient hyperthyroidism (P < .00001), while other outcome measures within the subgroups showed similar trends (Supplementary File 2). Compared with the T1 group, patients in the T1B group showed higher incidence rates of new-onset tumors as key indicators (P = .03). However, the advantages of the MWA group in terms of OCR, transient hoarseness, and hospitalization duration were no longer significant (P = .67, P = .05, P = .12, respectively). The other indicators showed similar trends (Supplementary File 3).
Sensitivity Analysis and Publication Bias
A funnel plot was drawn to detect publication bias in the MWA comparative study of isolated T1N0M0 PTC surgery. The overall funnel plot was symmetrical, and the results indicated no significant bias in the analysis (Figure 6). The Egger test further showed (Table 2) that in most of the main results, P > .05, and no significant bias was found in the analysis. Considering LTP, LNM, and dysphagia/throat edema (P = .0175 < .05, P = .0270 < .05, P = .0496 < .05, respectively), after processing the publication bias data using the trimming method (P = .8695 > .05, P = .3138 > .05, P = .0083 < .05, respectively), it was suggested that dysphagia/throat edema had publication bias. For indicators with relatively high heterogeneity, the source of heterogeneity was analyzed, and the methods included in the study were removed one by one (leave-one-method). The result indicated that the data included in the study had little influence on the combined indicators. The findings of this meta-analysis revealed both stability and reliability.
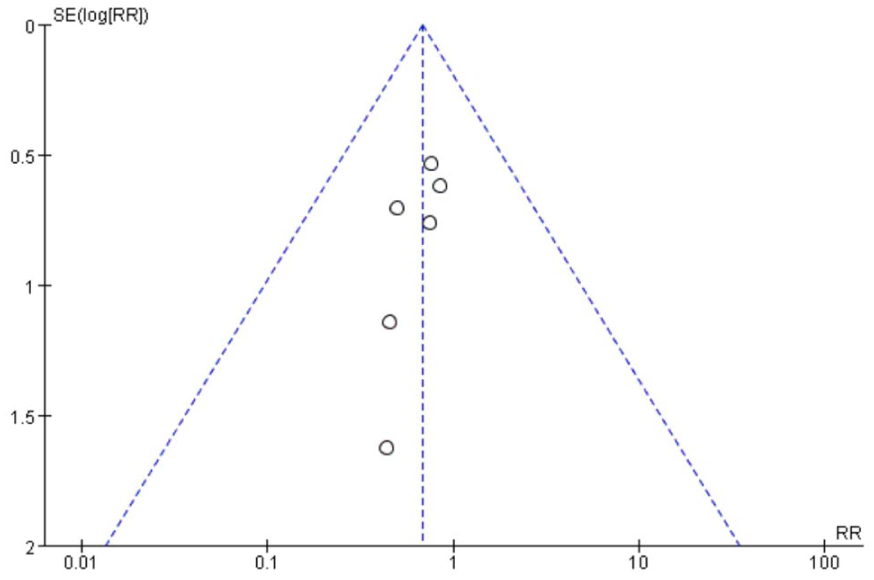
Funnel plot of lymph node metastasis.
Results of Egger Test.
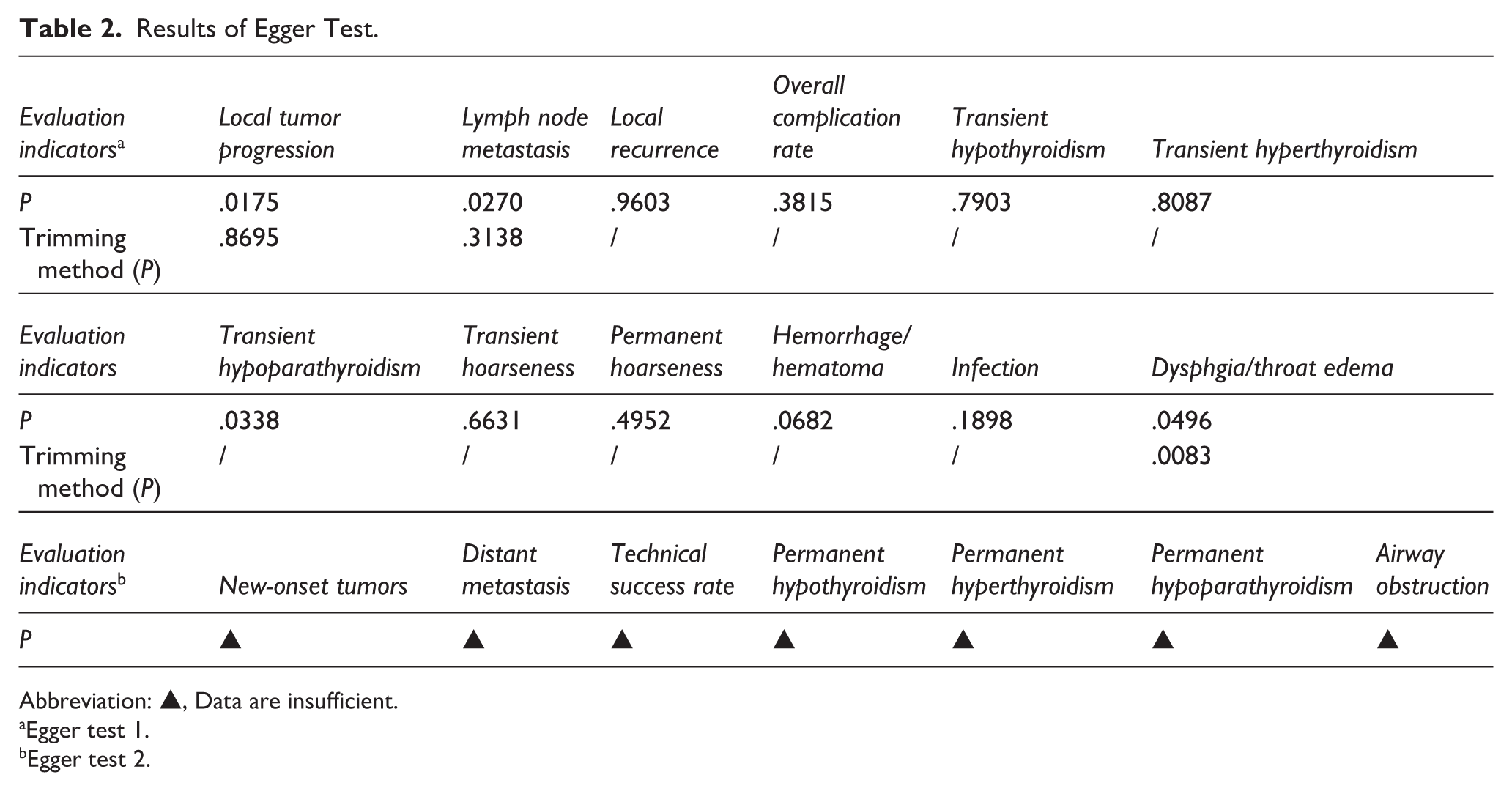
Abbreviation: ▲, Data are insufficient.
Egger test 1.
Egger test 2.
Discussion
In recent years, there has been a growing body of research dedicated to MWA as a treatment option for low-risk papillary thyroid microcarcinoma (PTMC). MWA is acknowledged as a reliable and safe therapeutic approach for patients with PTMC.23 -25 The majority of these investigations have concentrated primarily on PTMC or have not included a comparative analysis with surgical interventions, which markedly diverges from our intended study population and intervention methodology. We comprehensively compared the efficacy and safety of MWA versus surgery for solitary T1N0M0 PTC for the first time, including the T1A and T1B subgroups.
Regarding the primary outcome tumor-related indicators (LTP, new-onset tumors, lymph node metastases, distant metastases, technical success rate, and LR), there was no significant difference in the risk between patients treated with MWA and those who underwent surgery. The heating process induced by MWA results not only in localized coagulative necrosis but also contributes to the development of tissue scarring and fibrosis. 26 MWA induces alterations in both the texture and vascularization of the tumor as well as the adjacent tissue, resulting in the formation of a physical barrier. Furthermore, the thermal effects lead to modifications in cellular architecture and nuclear characteristics, potentially diminishing the capacity of any remaining tumor cells to grow and spread. 27 Crucially, MWA also leads to vascular blockage within the neoplasm, thereby effectively restricting blood flow, which, if left unchecked, could facilitate tumor proliferation and metastasis. 28 In the T1A and T1B subgroups, most indicators showed the same trend. However, the incidence of new tumors was higher in the T1B subgroup than in the surgical group. This might be due to tumors in the T1B subgroup being larger in volume or more closely adhered to the capsule. As such, MWA might not fully cover the lesion, thereby increasing the risk of new tumors. 29 This study revealed an increased risk of new tumor development after MWA in T1B subgroup (1-2 cm diameter) PTC patients. However, this finding is based on limited research and may be influenced by multiple factors: (1) T1B tumors may have longer follow-up periods. (2) Their biological behavior may be more aggressive. (3) Monitoring bias may exist. (4) The likelihood of insufficient ablation scope is higher. Therefore, this finding requires cautious interpretation. Patients who received MWA experienced markedly reduced complication rates (OCR, transient hypothyroidism, permanent hypoparathyroidism, permanent hoarseness, and dysphagia/laryngeal edema) than those who underwent surgery. MWA precisely locates the lesion through image guidance (ultrasound or CT), and only a 1 to 2 mm needle tract is needed to treat, avoiding the extensive tissue incision and the risk of nerve and vascular damage that are inherent to traditional surgery, thereby reducing the incidence of traumatic complications.30,31 Furthermore, MWA eliminates the need for thyroid gland excision, preserving over 90% of the functional tissue. By selectively ablating tumor tissue while preserving most normal thyroid tissue, MWA may reduce the risk of permanent hypothyroidism and avoid the need for lifelong thyroid hormone replacement therapy.32,33 However, the degree of thyroid function preservation is affected by many factors, including ablation range, basal thyroid function, etc., and more accurate evaluation criteria are needed.
Our investigation indicated that there was no statistically meaningful distinction in the incidence of temporary hoarseness when comparing the MWA group with the surgical cohort. It is possible that thermal damage from MWA can potentiate transient hoarseness. Therefore, liquid isolation technology to reduce thermal damage and promote the recovery of hoarseness during follow-up is recommended.34,35 Following MWA, the likelihood of developing hypoparathyroidism was markedly reduced compared with that associated with surgical intervention. Notably, none of the patients who underwent the MWA had hypoparathyroidism. Alteration of the microenvironment surrounding the remaining parathyroid cells was identified as the primary factor contributing to the occurrence of postoperative hypoparathyroidism. 36 No significant variation was detected in the incidence of postoperative hyperthyroidism among patients who received MWA compared to those who underwent surgical interventions.
MWA usually causes thermal damage leading to the rupture of thyroid follicles, resulting in a transient release of stored hormones, and thus, triggering a temporary hyperthyroidism, which typically resolves on its own within 1 to 3 months after the operation. 37 In contrast, if the ablation range is insufficient, it may lead to the continuous secretion of hormones by residual lesions. 38 The situation of surgery-related hyperthyroidism is more complex, possibly because of inadequate preoperative antithyroid drug preparation, hormone release caused by compression of the thyroid during the operation, or insufficient resection range, leaving the remaining hyperplastic or adenomatous tissue still active. 39 Additionally, if the dosage of levothyroxine supplementation is excessive after surgery, it may cause drug-induced hyperthyroidism, which requires thyroid function monitoring and medication adjustments. 40 Overall, the incidence of hematoma, infection, airway obstruction, and hyperthyroidism in both groups of patients was very low, and there were no significant statistical differences. The subgroup analysis showed that T1A and T1 showed similar trends. In group T1B, the OCR and the advantage of transient hoarseness were no longer observed after MWA. This may be due to the diameter of T1B tumors being 1 to 2 cm, which may be closer to the RLN, trachea, or blood vessels, increasing the risk of thermal injury. 41 Local inflammation after MWA may stimulate the laryngeal tissue and cause temporary hoarseness. 42 Certain investigations that advocate for the benefits of MWA often involve limited sample sizes or lack adequate follow-up durations, potentially leading to an underestimation of the likelihood of long-term complications. Therefore, extensive observational studies are necessary to provide more comprehensive insights.
In terms of surgery-related indicators and postoperative volume changes of the tumor, MWA has significant advantages in terms of operation time, blood loss, hospital stay, treatment cost, incision size, and quality of life score because it is minimally invasive.
MWA requires only local anesthesia, and the single ablation time is 5 to 15 minutes, whereas open surgery takes 60 to 90 minutes. With real-time ultrasound guidance and accurate localization of lesions, extensive tissue stripping is avoided. 43 The bleeding amount during the operation is very low (<5 ml), and the microwave energy can achieve synchronous hemostasis by blocking blood vessels through high temperature, while the bleeding amount in traditional surgery is often up to 50 to 100 ml due to direct tissue cutting. 44 Of note, the MWA group numerically showed shorter procedure time, less estimated blood loss, and shorter length of hospital stay, but these results were heterogeneous (I2 > 90%) and should be interpreted with caution. These differences may be influenced by differences in surgical techniques, health system characteristics, and lack of global applicability.
Moreover, MWA usually involves day surgery or 1 to 2 days of hospital admission. Patients can leave the hospital after observation without abnormalities, whereas traditional surgery requires 5 to 7 days of hospitalization, which significantly reduces the risk of nosocomial infections. MWA is also more cost-effective because of its simplicity, shorter theater time, and shorter length of hospital stay. There is no need for lifelong medication; thus, the long-term cost is also lower.14,20,21 The size of the MWA incision is only 1 to 2 mm, and there is no surgical incision scar, whereas surgery requires a 5 to 10 cm neck incision. Furthermore, more than 90% of patients are satisfied with the esthetic outcome after MWA.14,15,21
In terms of quality of life score, postoperative pain tended to be mild after MWA, and thyroid function tended to remain intact without medication intervention. In contrast, 30% to 50% of patients in the surgical group needed to take medication for life.14,15,21 MWA, with its minimally invasive, precise, and efficient technical characteristics, comprehensively optimizes safety, functional preservation, and esthetics in the treatment of thyroid diseases. It is particularly suitable for patients with high esthetic requirements, weak underlying conditions, or those who refuse traditional surgery. The subgroup analysis suggested a similar trend in the T1A and T1B groups. The hospitalization time in the T1B group did not show a statistically significantly different from that in the surgical group. This lack of variation could be attributed to a limited sample size or inadequate follow-up duration. In the MWA group, changes in tumor volume showed a specific pattern. Three months after the operation, the tumor volume temporarily increased compared with that before the operation. However, 6 months after the operation, the tumor volume decreased to a level close to that before the operation, and during the follow-ups at 12 and 24 months after the operation, the tumor volume further decreased significantly. This indicates that MWA can significantly reduce the tumor size in patients.
Of note, according to American thyroid association (ATA) and National comprehensive cancer network (NCCN) guidelines, surgical resection is still the standard treatment option for low-risk PTMC (eg, solitary, ≤1 cm, and no metastasis). 45 Although some guidelines (such as ATA) mention that nonsurgical treatment (including thermal ablation) can be considered for low-risk patients, it is clear that patients need to be strictly screened and based on multidisciplinary evaluation. 46 Studies have shown that MWA has good short-term efficacy in carefully selected low-risk PTMC patients, with a technical success rate of 98% to 100%, and a significantly lower complication rate than surgery (such as hoarseness, hypocalcemia). 20
Although the results were encouraging, this study has some limitations. Primarily, studies in this meta-analysis were mostly retrospective in design, and there was the possibility of selection bias and confounding bias (such as indication confusion). While we performed NOS scoring and sensitivity analysis of the included literature, these approaches can only somewhat mitigate the constraints associated with retrospective data. There are differences in the exploration of the ablation extent of cancer tissue, but the lack of detailed description of ablation power and energy per unit volume in most studies may account for this difference. However, all the included studies were from China, which limits the generalizability of the findings to external populations, particularly those from different health care systems and ethnic backgrounds.
Because of the significant differences between thyroid lobectomy and total thyroidectomy in terms of complications, we performed detailed statistics of surgical interventions in the 9 included studies (see Table 3). However, due to the relatively insufficient data available on surgical methods, we were unable to perform subgroup analyses by different surgical methods, which limited the comprehensiveness and accuracy of our findings to some extent.
The Surgical Methods of Surgical Group of Included Studies.
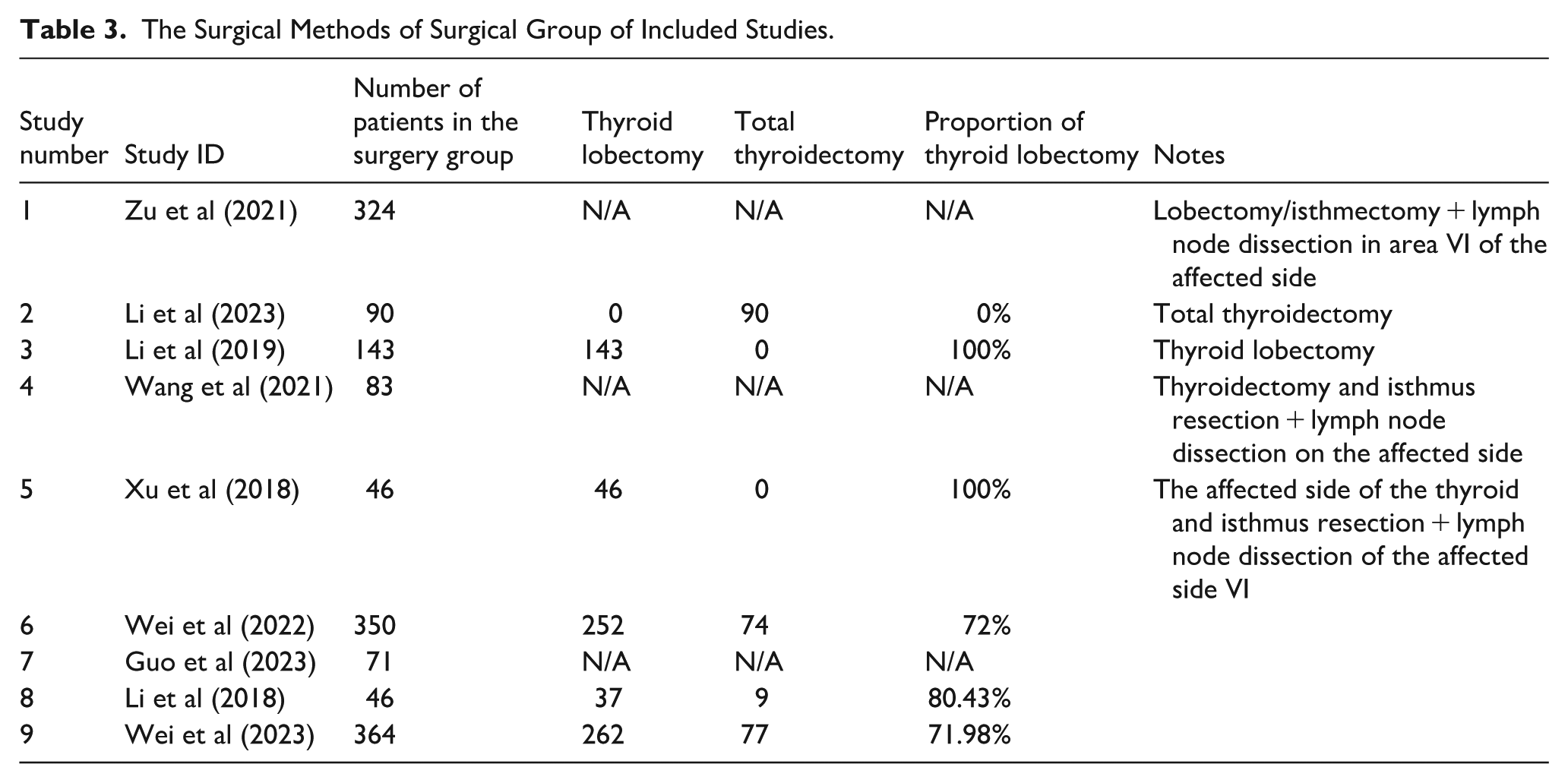
Therefore, future studies should adopt a prospective observational design to enhance validity. Additionally, the follow-up periods of some studies were relatively short, potentially limiting their ability to fully assess possible long-term or delayed outcomes. Oncology equivalence could not be established because of the current follow-up duration of the included studies. Finally, the small sample size could influence the precision of the analytical outcomes. To supplement the sample size, future studies should include a greater number of participants.
Conclusion
The results of this meta-analysis indicate that (1) ultrasound-guided MWA may be a potential treatment option for highly selected patients with isolated T1N0M0 PTC. It shows advantages in short-term outcomes (such as operation time, hospital stay, and some complications), but its long-term oncological safety still needs to be confirmed by longer and higher quality studies. (2) However, in the T1B subgroup, preoperative ultrasound or CT examinations are required to screen for suspicious lymph nodes. If any suspicious metastasis is found, surgical treatment should be given priority. (3) The conclusions of this study are mainly applicable to the situation similar to that of the Chinese patient population.
Supplemental Material
sj-docx-2-ear-10.1177_01455613251415512 – Supplemental material for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-ear-10.1177_01455613251415512 for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis by Peng Xiang, Dan Liu, Lin Yang, Xianwen Wang, Ge Hu, Wei Chang and Zhenhua Zhu in Ear, Nose & Throat Journal
Supplemental Material
sj-docx-3-ear-10.1177_01455613251415512 – Supplemental material for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-3-ear-10.1177_01455613251415512 for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis by Peng Xiang, Dan Liu, Lin Yang, Xianwen Wang, Ge Hu, Wei Chang and Zhenhua Zhu in Ear, Nose & Throat Journal
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251415512 – Supplemental material for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-1-ear-10.1177_01455613251415512 for The Efficacy and Safety of Ultrasound-Guided Microwave Ablation Versus Surgery for Solitary T1N0M0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis by Peng Xiang, Dan Liu, Lin Yang, Xianwen Wang, Ge Hu, Wei Chang and Zhenhua Zhu in Ear, Nose & Throat Journal
Footnotes
Author Note
The information contained in this manuscript has not been published nor submitted for review to any other forum.
Ethical Considerations
Approval complied with ethical standards.
Author Contributions
Peng Xiang was responsible for screening the research, data entry, statistical analyses, and writing the paper. Dan Liu participated in the research design, screening, and writing of the article. Lin Yang participated in data extraction, statistical analysis, and article writing. Xianwen Wang participated in data extraction, statistical analysis, and article writing. Wei Chang participated in the bias risk assessment, data entry, statistical analysis, and article writing. Ge Hu participated in bias risk assessment, study design, and article writing. Zhenhua Zhu participated in the bias risk assessment, study design, and manuscript writing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the first batch of the Chinese Medicine Discipline Leader Project of Hunan Province during the 14th Five-Year Plan period (No Xiang CM Medicine [2022] 4), the National Advantage Specialty Construction Unit of Chinese Medicine (No [2024] No 90, 498 Department of Otolaryngology), Hunan Clinical Research Center of Otolaryngology of Chinese Medicine (No 2021SK4024), Changsha Natural Science Foundation (No kq2403105), Outstanding Youth Project of Hunan Provincial Education Department (No 23B0386), and the Joint Fund Project of Hunan University of Chinese Medicine (No 2025XYLH040).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
