Abstract
Objectives:
Allergic rhinitis (AR) is increasingly recognized as a systemic inflammatory condition. While symptom scores are commonly used for disease monitoring, objective biomarkers to predict treatment outcomes remain limited. This study evaluated systemic inflammatory indices (neutrophil-to-lymphocyte ratio [NLR], platelet-to-lymphocyte ratio [PLR], systemic immune-inflammation index [SII], eosinophil count, and total immunoglobulin E [IgE]) as biomarkers in AR over 6 month follow-up.
Methods:
This prospective cohort study enrolled 105 patients with moderate-to-severe persistent AR and 92 matched healthy controls. Baseline and 6 month measurements included complete blood count–derived indices and total serum IgE. Symptom severity was assessed using Total Nasal Symptom Score and visual analog scales. ROC analyses determined optimal cutoff values for predicting treatment response.
Results:
At baseline, AR patients had significantly-higher NLR, PLR, SII, and eosinophil counts than controls (all P < .001). These indices declined significantly in parallel with symptom improvement. Baseline NLR >2.7 showed the strongest predictive value for nonresponse (sensitivity 84%, specificity 76%, OR 4.2). PLR >240, SII >1100, eosinophils >600/µL, and IgE >450 IU/mL were also associated with increased nonresponse risk. Total IgE remained stable and was not useful for short-term monitoring. A mixed-effects longitudinal model confirmed that reductions in NLR, PLR, and eosinophil count were significantly greater in AR patients than in controls, supporting that these biomarker changes were treatment related.
Conclusion:
NLR, PLR, SII, and eosinophil count are promising biomarkers for monitoring disease activity and predicting treatment response in AR. NLR may serve as a simple, cost-effective tool for early risk stratification. These findings support integrating routine hematologic markers into clinical management of AR.
Keywords
Introduction
Allergic rhinitis (AR) is a common IgE-mediated inflammatory disease, affecting approximately 15% to 40% of the global population, with a notably-higher prevalence among children. Approximately 80% of individuals develop AR symptoms before the age of 20 years, peaking in prevalence between ages 20 and 40 before gradually declining thereafter.1 -3 Although AR is traditionally perceived as a localized nasal condition, increasing evidence supports its characterization as a systemic inflammatory disorder affecting multiple immune pathways beyond the nasal mucosa. 4
In the allergic cascade, allergen exposure triggers a T-helper type 2 (Th2) immune response, leading to immunoglobulin E (IgE) production and activation of mast cells and basophils. This response results in the release of inflammatory mediators and subsequent recruitment of eosinophils and other leukocytes, causing both localized nasal symptoms and measurable systemic immune alterations.5,6 Blood eosinophil counts are often elevated in patients with AR compared with those in nonallergic individuals, and total serum IgE levels typically exceed those found in nonatopic populations.5 -7
Despite the well-established roles of IgE and eosinophils in AR pathophysiology, clinically-accessible biomarkers that reliably reflect disease severity or therapeutic responses remain limited. 8 Currently, patient-reported nasal symptom scores are widely utilized, yet objective blood markers correlating consistently with clinical symptoms or predictive of treatment outcomes would significantly improve clinical management. 9 Recently, hematological indices derived from routine blood counts, particularly the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have shown promise as systemic inflammation markers with prognostic relevance across various diseases. 10
Growing evidence suggests that elevated NLR and PLR are associated with moderate-to-severe AR, supporting their potential utility as objective indicators for assessing disease severity and monitoring inflammatory activity in clinical practice.4,11 -14 A recent systematic review and meta-analysis by Khanzadeh et al further confirmed significantly-higher NLR levels in AR patients than in healthy controls, validating the role of NLR as a biomarker for predicting AR severity and comorbidities such as asthma. 4 Additionally, the systemic immune-inflammation index (SII) derived from neutrophil, platelet, and lymphocyte counts (Neutrophils × Platelets/Lymphocytes) has gained recognition for its prognostic value in chronic inflammatory conditions, including cancer and cardiovascular diseases, although its application in AR remains underexplored.15,16 Given the limitations of conventional biomarkers such as total IgE and eosinophil counts, these systemic indices (NLR, PLR, and SII) offer promising, accessible, and cost-effective alternatives for improving diagnostic accuracy, disease monitoring, and therapeutic decision-making in AR.17,18 In AR, although IgE and eosinophils orchestrate the classic allergic response, other immune cells also contribute to the inflammatory milieu: For instance, neutrophils can be recruited during late-phase reactions and severe exacerbations, and activated platelets release mediators that amplify leukocyte recruitment. Thus, composite indices that incorporate neutrophil and platelet counts (like NLR, PLR, SII) may capture this broader AR-specific inflammatory activity, potentially reflecting disease severity more comprehensively than eosinophil counts or IgE levels alone.
Recent evidence also indicates that AR is driven not only by classical IgE-mediated, eosinophil-dominant inflammation but also by upstream epithelial and non-type-2 pathways. Epithelial barrier dysfunction promotes the release of alarmin cytokines such as IL-33, IL-25, and Thymic stromal lymphopoietin (TSLP), which rapidly recruit and activate neutrophils and amplify mucosal inflammation.19,20 Activated platelets further enhance leukocyte trafficking and vascular permeability during allergic airway responses. Consistent with these expanded inflammatory mechanisms, composite complete blood count (CBC)-based indices (including NLR, PLR, and SII) tend to be elevated in AR and correlate with disease severity, capturing neutrophil-driven and platelet-driven activities in addition to traditional type-2 pathways. Therefore, these systemic markers may reflect the multi-pathway inflammatory burden of AR more comprehensively than isolated biomarkers such as eosinophil count or total IgE.
This prospective study aimed to evaluate the clinical utility of systemic inflammatory indices, including NLR, PLR, and SII, as biomarkers for predicting disease severity and monitoring treatment response over a 6 month follow-up period in patients with allergic rhinitis.
Methods
Study Design
This study was designed as a prospective, observational cohort study conducted over a period of 6 months at the Otorhinolaryngology Clinic of a tertiary research and training hospital in Ankara, Turkey, between January 2024 and January 2025. Patients diagnosed with AR were enrolled to evaluate the predictive and monitoring capabilities of systemic inflammatory indices: NLR, PLR, and SII.
NLR: neutrophil count (cells/µL)/lymphocyte count (cells/µL)
PLR: platelet count (cells/µL)/lymphocyte count (cells/µL)
SII: (neutrophil count (cells/µL) × platelet count (cells/µL))/lymphocyte count (cells/µL)
A healthy control group, matched using propensity score matching based on age, gender, and body mass index (BMI), was included for comparative purposes, serving as a baseline reference to confirm the presence of systemic inflammation in AR and to ensure that observed changes in markers were disease-specific.
The inclusion of a matched healthy control group was critical for 2 methodological reasons. First, it enabled us to verify that patients with allergic rhinitis exhibited measurably-higher baseline systemic inflammation than with nonallergic individuals, thereby validating the utility of NLR, PLR, SII, and eosinophil count as disease-associated markers rather than normal physiologic variation. Second, by following both cohorts over the same 6 month period, the control group provided a necessary temporal comparison to ensure that reductions in inflammatory indices in the AR group reflected true disease-related and treatment-related improvements rather than spontaneous or seasonal fluctuations. Matching the groups on age, sex, and BMI further minimized confounding, allowing clearer interpretation of biomarker trajectories.
Patient Selection and Criteria
Patients aged between 18 and 60 years presenting with moderate-to-severe persistent AR symptoms, defined according to the Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines, were included. 21 Diagnosis was confirmed clinically based on characteristic symptoms (nasal congestion, rhinorrhea, sneezing, itching) lasting at least 2 consecutive seasons or persistently throughout the year, supported by positive skin prick tests and/or elevated serum-specific IgE levels against relevant aeroallergens. Exclusion criteria comprised mild intermittent AR, uncontrolled asthma, acute or chronic infections, systemic inflammatory diseases, pregnancy or breastfeeding, and the use of immunosuppressive drugs or systemic corticosteroids within the past 2 months.
Sample Size Determination
A total of 210 patients fulfilling the inclusion criteria were initially enrolled. Sample size calculation was based on previous studies evaluating systemic inflammatory indices in AR, estimating at least 100 patients per group to achieve over 80% statistical power with a significance level (α) of .05 for detecting clinically-meaningful differences in NLR. At the end of the 6 month follow-up period, 105 AR patients and 92 matched healthy controls completed the study. Participants lost to follow-up (3 from the intervention group and 10 from the control group) were excluded from final analyses.
Data Collection and Variables
Blood samples for CBC analysis were collected in the morning after an overnight fast to minimize diurnal variation and analyzed using an automated hematology analyzer. From these CBC results, NLR, PLR, and SII were calculated at baseline and again after 6 months. All AR patients received standardized pharmacotherapy consisting of intranasal corticosteroids, antihistamines, and leukotriene receptor antagonists, consistent with routine clinical management. A small subset received allergen-specific immunotherapy as part of their standard treatment. Baseline measurements in this study were obtained at the time of enrollment (pretreatment), and any prior or ongoing AR therapies (including allergen immunotherapy or over-the-counter medications) were recorded; thereafter, all patients continued on the standardized regimen during the follow-up period to maintain consistency. Importantly, this study was purely observational without intervention, and changes in biomarkers were monitored as part of routine clinical care.
Clinical Measurements
Symptom severity was quantified using the Total Nasal Symptom Score (TNSS) and a visual analog scale (VAS) at baseline and after 6 months. TNSS, ranging from 0 (no symptoms) to 12 (severe symptoms), evaluated rhinorrhea, nasal congestion, sneezing, and nasal itching. Treatment response was classified as “responders” if patients showed ≥50% reduction from baseline TNSS and a final score ≤4. Secondary clinical parameters, including disease-specific quality of life measured by the Rhinitis Quality of Life Questionnaire and frequency of rescue medication usage, were recorded but not detailed in this analysis.
Other Variables and Confounders
Demographic data including age, sex, BMI, duration of AR, and presence of comorbid asthma were collected at baseline. Patients were screened for confounding factors such as acute infections or exacerbations of respiratory conditions during the study period. Participants experiencing significant acute infectious or inflammatory events were temporarily excluded from sampling until resolution to avoid confounding influences on systemic inflammatory markers.
Ethical Considerations
The study was conducted in compliance with the Declaration of Helsinki. Ethics approval was obtained from the Ethics Committee of Ankara Bilkent City Hospital (IRB; TABED: 1-25-1250). Written informed consent was obtained from all participants prior to study enrollment.
Statistical Analysis
Statistical analyses were performed using SPSS v.25.0 (IBM Corp, Armonk, NY, USA). Normality of continuous variables was assessed with the Shapiro-Wilk test, and results were summarized as mean ± standard deviation (SD) or median (interquartile range [IQR]) depending on distribution. Categorical variables were reported as frequencies and percentages. Within-group changes from baseline to 6 months were evaluated using paired t-tests or Wilcoxon signed-rank tests, while between-group comparisons (AR vs controls) were performed using independent t-tests or Mann-Whitney U tests. Categorical outcomes were compared with chi-squared tests. Correlations between inflammatory markers and symptom scores were examined using Pearson or Spearman coefficients based on distributional characteristics (normally-distributed variables: Pearson r; non-normal or Δ values: Spearman ρ). ROC curve analyses were conducted to determine optimal baseline cutoff values for predicting treatment response, with sensitivity, specificity, and area under the curve (AUC) reported. Predictive performance of biomarkers was further assessed using multivariate logistic regression incorporating ROC-derived thresholds; odds ratios (ORs), 95% confidence intervals, Nagelkerke R2, and Hosmer–Lemeshow calibration statistics were provided. A repeated-measures mixed-effects model (time × group) was used as a supplementary longitudinal approach to validate biomarker changes over time. Statistical significance was set at P < .05.
Results
Baseline Characteristics
A total of 197 patients completed the study (105 intervention, 92 control). The groups were comparable in demographic and clinical characteristics (Table 1). The mean age was 29.8 ± 9.4 years (range 18-58), and approximately half of participants were female (intervention 49%, control 52%; P = .68). The mean duration of AR symptoms was 7.2 ± 5.1 years, and 21.9% of AR patients had comorbid asthma. All AR patients presented with moderate-to-severe persistent disease.
Baseline Demographic and Clinical Characteristics of Allergic Rhinitis Patients and Matched Healthy Controls.
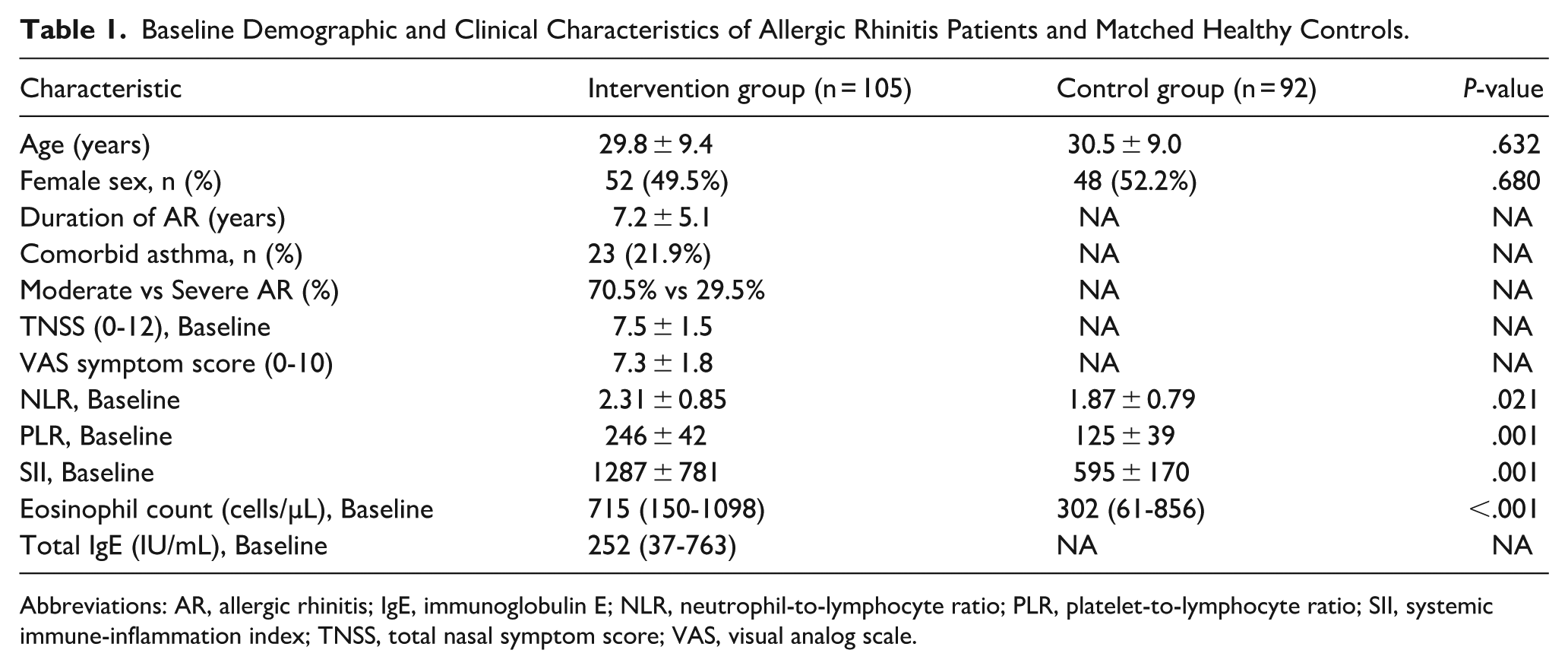
Abbreviations: AR, allergic rhinitis; IgE, immunoglobulin E; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammation index; TNSS, total nasal symptom score; VAS, visual analog scale.
Baseline hematologic indices indicated a clearly-higher systemic inflammatory burden in AR patients. The mean baseline NLR was significantly elevated in the intervention group compared with that in controls (2.31 ± 0.85 vs 1.87 ± 0.79; P = .021), and eosinophil counts were markedly higher (median 715 vs 302 cells/µL; P < .001). Baseline PLR (246 ± 42 vs 125 ± 39; P = .001) and SII (1287 ± 781 vs 595 ± 170; P = .001) values also demonstrated substantially-increased systemic inflammation in AR.
Symptom Changes and Treatment Response
Symptom severity improved significantly over 6 months. In the intervention group, TNSS decreased from 7.5 ± 1.5 to 3.0 ± 1.8 (P < .001), and VAS scores declined from 7.3 ± 1.8 to 2.8 ± 2.1 (P < .001). Based on predefined criteria (≥50% TNSS reduction and TNSS ≤4), 73 of 105 patients (69.5%) were classified as responders.
Diagnostic Performance of Baseline Inflammatory Indices
Baseline inflammatory biomarkers displayed meaningful discriminatory performance for predicting 6 month treatment response. ROC analyses identified optimal cutoff values as follows: NLR >2.7 (sensitivity 84%, specificity 76%, AUC 0.78), PLR >240 (sensitivity 78%, specificity 70%, AUC 0.74), SII >1100 (sensitivity 81%, specificity 73%, AUC 0.75), IgE >450 IU/mL (sensitivity 68%, specificity 75%, AUC 0.69), and eosinophils >600 cells/µL (sensitivity 86%, specificity 77%, AUC 0.80) (Table 2).
ROC-Derived Diagnostic Performance of Systemic Inflammatory Biomarkers for Predicting 6 Month Treatment Response in Allergic Rhinitis.
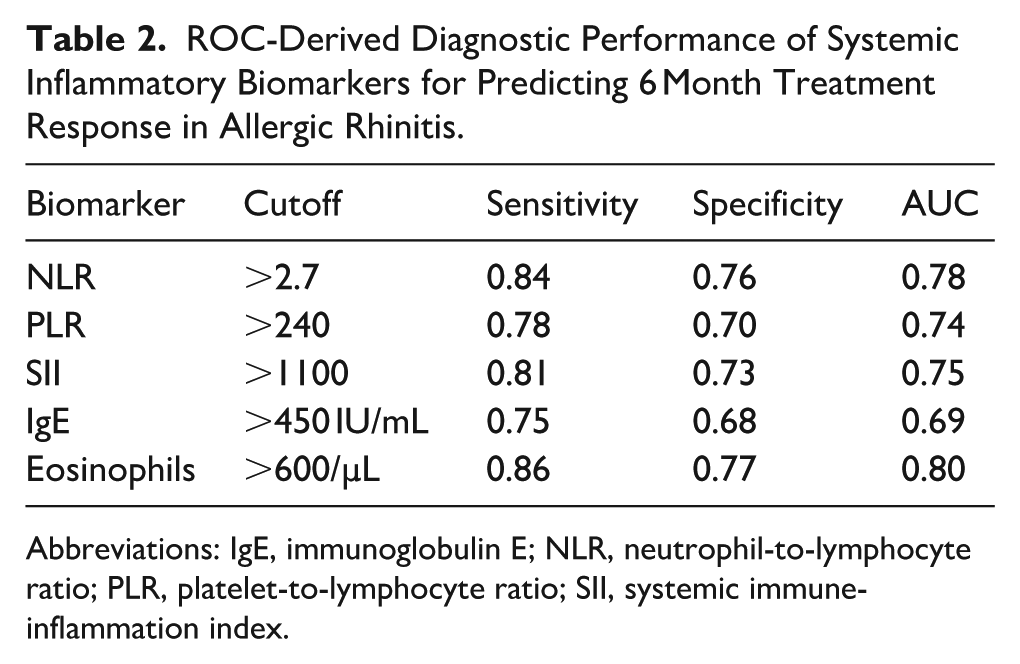
Abbreviations: IgE, immunoglobulin E; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammation index.
Predictors of Nonresponse: Multivariate Analysis
Multivariate logistic regression using ROC-derived thresholds showed that several baseline inflammatory markers were independent predictors of treatment nonresponse. Elevated eosinophil count (>600 cells/µL) demonstrated the strongest association (OR 5.0; 95% CI 2.5-10.0; P < .001), followed by NLR >2.7 (OR 4.2; 95% CI 2.1-8.4; P < .001). PLR >250 (OR 3.8; 95% CI 1.9-7.5; P = .002), SII >1100 (OR 3.2; 95% CI 1.6-6.7; P = .005), and IgE >450 IU/mL (OR 2.9; 95% CI 1.5-5.8; P = .010) were also significant predictors (Figure 1). The model demonstrated moderate explanatory power (Nagelkerke R2 = .26) and good calibration (Hosmer-Lemeshow P > .05). Age, sex, and baseline TNSS were not retained in the final model. NLR emerged as the strongest independent marker of nonresponse, which was further supported by ROC analysis showing an AUC of 0.788 (Figure 2).
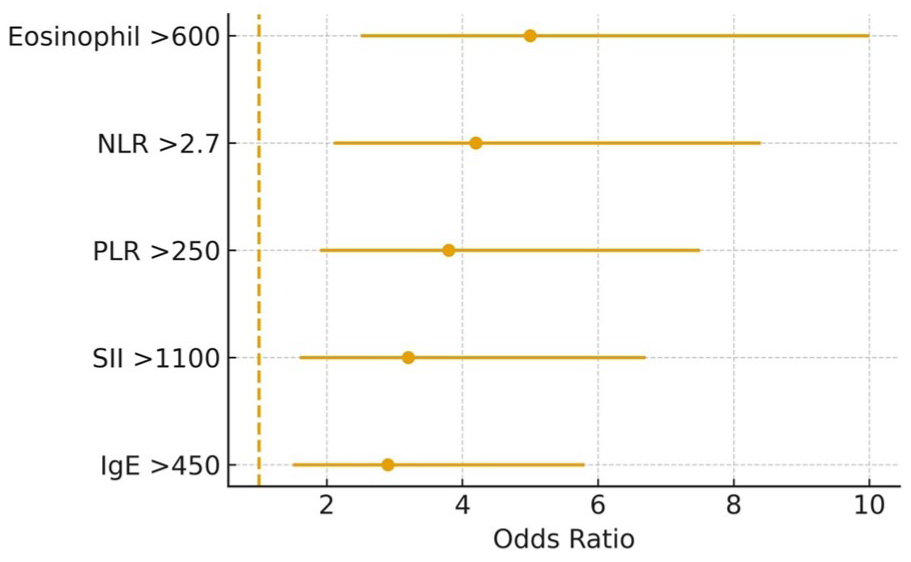
Forest plot of multivariate logistic regression predictors.
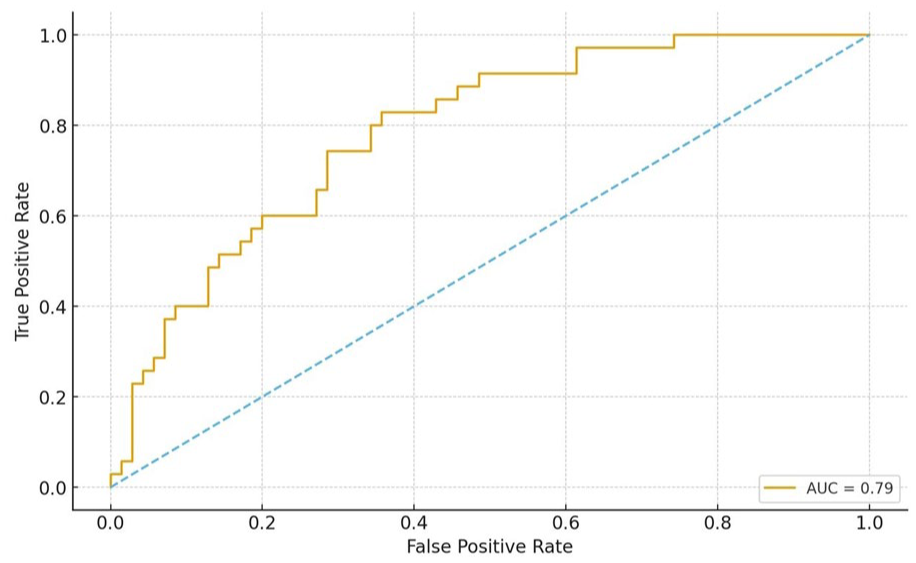
ROC curve for baseline NLR. NLR, neutrophil-to-lymphocyte ratio.
Responder Versus Nonresponder Differences
Responders exhibited substantially-lower baseline inflammatory indices than nonresponders. Specifically, responders had lower NLR (1.86 vs 2.79; P < .001), PLR (218.5 vs 277.7; P < .001), SII (1037 ± 300 vs 1563 ± 420; P < .001), eosinophil counts (543 ± 180 vs 954 ± 310 cells/µL; P < .001), and total IgE (329 ± 140 vs 642 ± 210 IU/mL; P = .002) (Figures 3 and 4; Table 3). Responders also showed milder baseline symptoms, including lower TNSS (6.6 ± 2.5 vs 8.1 ± 3.0; P = .015) and VAS (6.2 ± 3.6 vs 7.0 ± 2.1; P < .001).
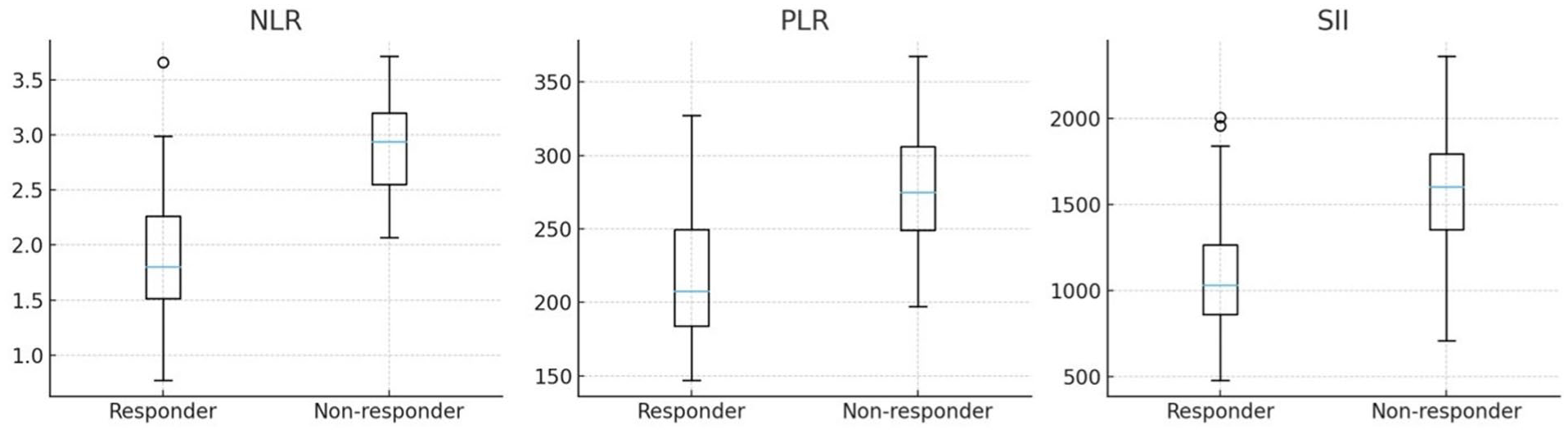
Boxplots of NLR, PLR, and SII in responders versus nonresponders. NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammation index.
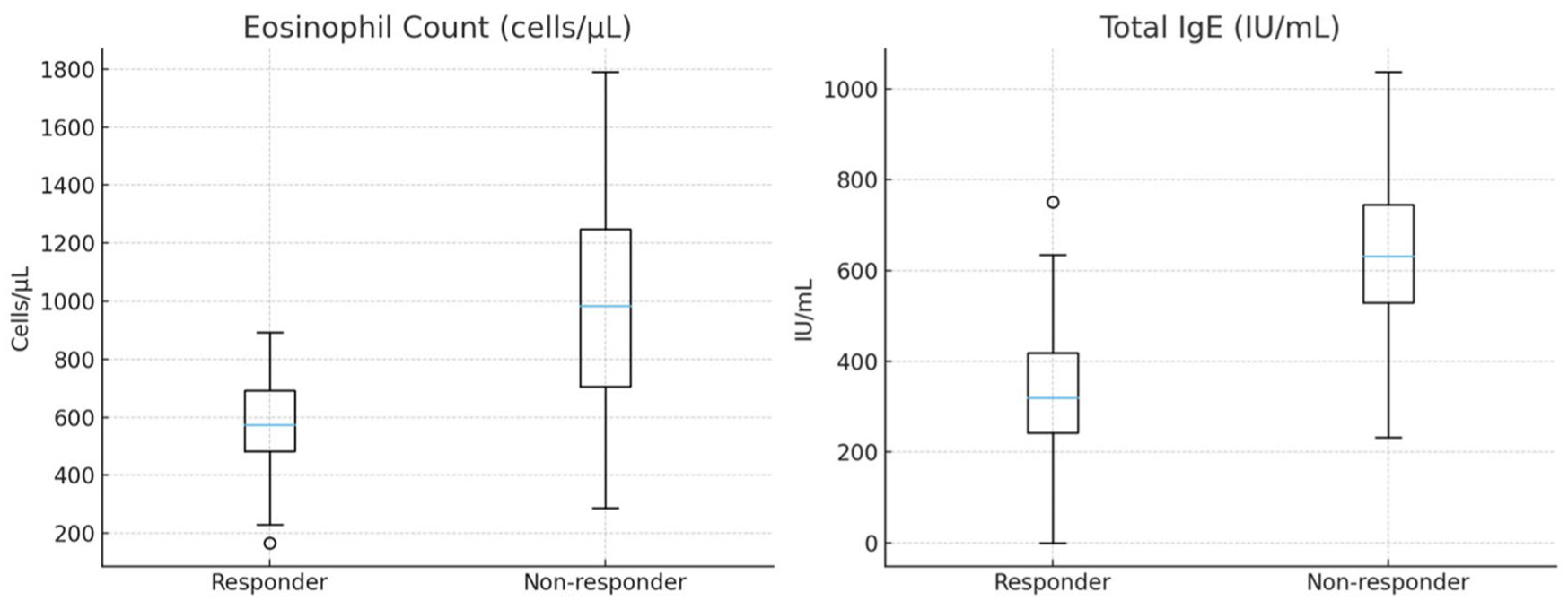
Boxplots of eosinophils and IgE in responders versus nonresponders.
Baseline Inflammatory Indices and IgE Levels Comparing Treatment Responders and Nonresponders in Allergic Rhinitis Patients.
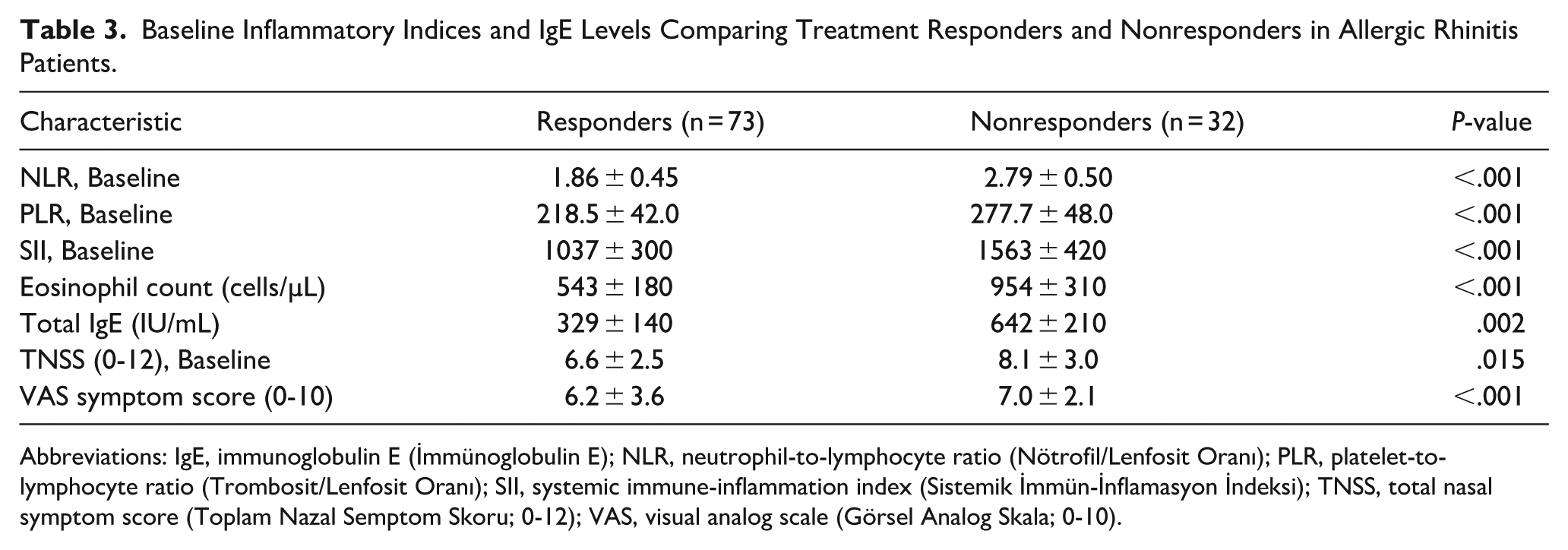
Abbreviations: IgE, immunoglobulin E (İmmünoglobulin E); NLR, neutrophil-to-lymphocyte ratio (Nötrofil/Lenfosit Oranı); PLR, platelet-to-lymphocyte ratio (Trombosit/Lenfosit Oranı); SII, systemic immune-inflammation index (Sistemik İmmün-İnflamasyon İndeksi); TNSS, total nasal symptom score (Toplam Nazal Semptom Skoru; 0-12); VAS, visual analog scale (Görsel Analog Skala; 0-10).
Longitudinal Changes in Inflammatory Indices
Over the 6 month follow-up, the intervention group demonstrated significant reductions in all major inflammatory markers. NLR decreased from 2.31 ± 0.85 to 1.89 ± 0.62 (P < .001), PLR from 246 ± 42 to 140 ± 30 (P < .001), SII from 1287 ± 781 to 872 ± 140 (P < .001), and eosinophils from 715 to 150 cells/µL (P < .001). In contrast, the control group showed no meaningful longitudinal changes (all P > .05). At the 6 month assessment, the intervention group demonstrated significantly-lower SII (872 ± 140 vs 346 ± 150; P = .026) and eosinophil counts (150 vs 97 cells/µL; P < .001) than controls, reflecting a return toward normative inflammatory activity. Between-group comparisons for NLR (1.89 ± 0.62 vs 1.87 ± 0.75; P = .107) and PLR (140 ± 30 vs 121 ± 36; P = .085) approached significance. Total IgE remained largely unchanged over 6 months (252 ± 140 vs 240 ± 130 IU/mL; P = .285), indicating poor utility as a short-term response marker (Table 4).
Baseline and 6 Month Outcomes in the Intervention Versus Control Groups.
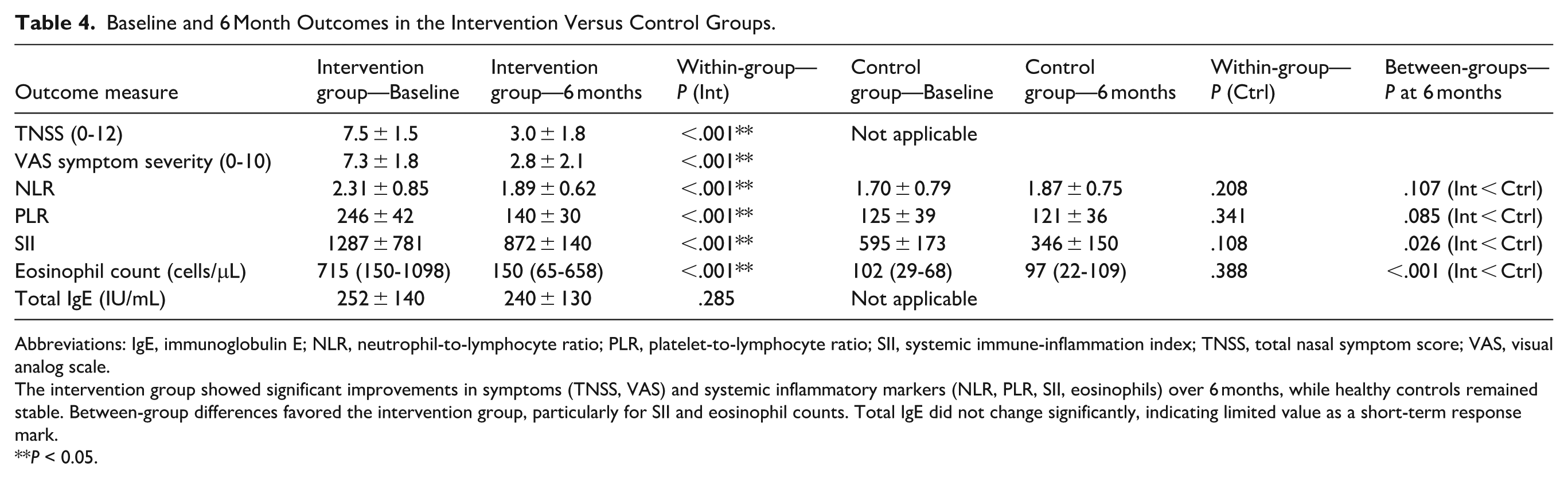
Abbreviations: IgE, immunoglobulin E; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammation index; TNSS, total nasal symptom score; VAS, visual analog scale.
The intervention group showed significant improvements in symptoms (TNSS, VAS) and systemic inflammatory markers (NLR, PLR, SII, eosinophils) over 6 months, while healthy controls remained stable. Between-group differences favored the intervention group, particularly for SII and eosinophil counts. Total IgE did not change significantly, indicating limited value as a short-term response mark.
P < 0.05.
Correlation Analyses
At baseline, higher systemic inflammatory markers were associated with greater symptom severity. Baseline TNSS correlated with NLR (r = .41; P < .001), PLR (r = .30; P = .001), SII (r = .21; P = .004), and eosinophil count (Spearman ρ = 0.26; P = .002), while total IgE showed no significant association (ρ = 0.12; P = .105).
Longitudinally, improvements in inflammatory markers correlated with improvements in symptoms. Reductions in ΔNLR (Spearman ρ = 0.36; P < .001) and ΔPLR (ρ = 0.28; P = .006) were associated with reductions in TNSS. ΔSII showed a weaker but significant correlation (ρ = 0.20; P = .03). ΔEosinophils demonstrated the strongest association with ΔTNSS (ρ = 0.52; P < .001), consistent with eosinophils’ central role in allergic inflammation.
Mixed-Effects Longitudinal Model
To validate these Δ-based findings and minimize the risk of mathematical coupling, a repeated-measures mixed-effects model (time × group) was conducted. This model demonstrated that reductions in NLR, PLR, and eosinophil counts were significantly greater in the intervention group than in controls (all P < .01), confirming that biomarker improvements reflected true treatment effects rather than temporal variation.
Discussion
In this prospective 6 month study of patients with allergic rhinitis, we investigated the utility of several systemic inflammatory markers—NLR, PLR, SII, eosinophil count, and total IgE—as tools for disease monitoring and outcome prediction. To our knowledge, this is one of the first studies to comprehensively evaluate these indices longitudinally in AR with a controlled intervention. Our findings can be summarized as follows: (a) At baseline, AR patients exhibited elevated NLR, PLR, SII, and eosinophil counts (consistent with a state of systemic inflammation accompanying their allergic condition), and these markers correlated with symptom severity. (b) Effective treatment (demonstrated by reduced TNSS and VAS scores) led to significant reductions in NLR, PLR, SII, and eosinophil counts over 6 months, in parallel with clinical improvement, suggesting these indices have value in monitoring disease activity. (c) Among the evaluated markers, baseline NLR emerged as an independent predictor of which patients would achieve good symptom control with treatment (ie, higher NLR was associated with poorer outcomes), whereas baseline eosinophil counts and total IgE levels did not reliably predict short-term outcomes. We discuss the implications of each of these points below, in context with the existing literature.
In our cohort, baseline NLR was significantly elevated in patients with AR compared with that in healthy controls, which is consistent with emerging evidence highlighting systemic inflammation in AR. A recent meta-analysis by Khanzadeh et al reported a modest but statistically-significant elevation of NLR in AR patients relative to healthy individuals (pooled standardized mean difference ≈0.2), supporting its potential role as a marker of systemic inflammatory activity in AR. 4 Similarly, Rohila et al demonstrated a mean NLR of approximately 1.9 in AR patients versus 1.5 in controls, reinforcing that even modest elevations in NLR are a characteristic of active AR. 12 In contrast, Selçuk et al found no significant difference in NLR between AR and nonallergic rhinitis groups, indicating that variability in patient populations or concurrent inflammatory states may influence baseline NLR values. Pathophysiologically, an elevated NLR in AR likely reflects increased neutrophil-driven inflammatory responses, including enhanced cytokine production and oxidative stress, accompanied by a relative reduction in circulating lymphocytes, possibly secondary to stress-induced redistribution or immune regulatory modulation.22,23 Thus, higher NLR values observed in AR patients plausibly mirror systemic immune activation extending beyond the nasal mucosa, highlighting the broader inflammatory burden associated with this traditionally-localized allergic condition.13,24 -26 Additionally, emerging evidence indicates that epithelial-derived cytokines (“alarmins” such as IL-33, TSLP, and IL-25), released from the upper airway mucosa upon allergen or viral stimulation, can drive a T-helper 17 (Th17) immune response in AR.27,28 This Th17 pathway contributes to neutrophilic inflammation in the airways, providing a mechanistic basis for the neutrophil recruitment and activation reflected by an elevated NLR in some AR patients. 29
Baseline PLR was also significantly higher among AR patients than among healthy controls, reflecting a state of systemic inflammation involving platelet activation alongside relative lymphocyte suppression. These findings align with previous studies highlighting elevated PLR as a characteristic of allergic diseases. Rohila et al recently reported significantly-higher mean PLR values in AR patients, emphasizing the potential role of PLR as a systemic inflammatory biomarker in allergic airway conditions. 12 Similarly, a pediatric study by Cansever et al found a positive correlation between elevated PLR and increasing severity of AR symptoms, suggesting its applicability across different age groups. 14 It is thought that elevated PLR in allergic rhinitis may result from increased platelet activation mediated by allergic inflammation and platelet interactions with leukocytes, endothelial cells, and mast cells, ultimately enhancing inflammatory mediator release, tissue edema, and eosinophil recruitment.10,12 -14,30,31 Relative lymphopenia, as observed in elevated NLR, may reflect immune cell redistribution or impaired regulatory T-cell activity in the context of chronic allergic inflammation, which in turn contributes to increased PLR values.22,23
Importantly, beyond its baseline elevation, our findings demonstrate that PLR also carries prognostic relevance in AR. Responders exhibited substantially-lower PLR values at baseline than nonresponders, and reductions in PLR over 6 months correlated with symptom improvement (ΔTNSS). This dual behavior (both as a cross-sectional marker of inflammatory load and as a dynamic marker reflecting therapeutic response) suggests that PLR captures platelet-mediated amplification of allergic inflammation in a clinically-meaningful manner. Although its predictive performance was more modest than that of NLR (AUC 0.74), the independent association of PLR with symptom improvement supports its potential role as an adjunctive biomarker in AR monitoring.
Consistently, baseline SII was significantly higher in AR patients than in controls, suggesting a pronounced systemic inflammatory state in AR. Although literature on SII in AR populations remains limited, our findings demonstrate parallels to observations in related chronic inflammatory airway diseases.6,9,30,32 Tian et al recently reported elevated SII values in asthma patients that correlated positively with disease activity and severity, highlighting this index’s capacity to reflect systemic inflammation beyond isolated hematologic ratios. 32 The elevated SII observed in our AR patients likely represents an integrated inflammatory response involving neutrophil-driven inflammation, platelet-mediated amplification of immune signals, and concurrent lymphocyte suppression indicative of chronic immune dysregulation. Notably, each component of the SII has a distinct role in allergic inflammation: neutrophils release inflammatory cytokines and reactive oxygen species, platelets enhance leukocyte recruitment and mediator release, and lymphocytes orchestrate adaptive immunity. Thus, the composite nature of SII potentially offers enhanced sensitivity in capturing the systemic inflammatory profile of AR patients.
Our study adds clinically-meaningful evidence to this emerging concept by demonstrating that SII not only differentiates AR patients from healthy controls but also distinguishes responders from nonresponders at baseline. Furthermore, SII declined significantly following treatment and correlated with symptom improvement, indicating that it reflects dynamic shifts in systemic inflammatory burden. Notably, the mixed-effects model confirmed that reductions in SII were predominantly treatment-related rather than attributable to temporal variation. These findings highlight SII as a promising, multifaceted biomarker for assessing disease activity and therapeutic response in AR, warranting further evaluation in larger, multicenter cohorts.
Peripheral blood eosinophil counts were significantly elevated in AR patients compared with those in controls, corroborating eosinophilia as a hallmark feature of atopic inflammation.7,8,33 This observation aligns with numerous studies underscoring the critical role of eosinophils as primary effector cells in allergic diseases.7,33,34 For instance, Xu et al reported higher circulating eosinophil levels among chronic rhinitis patients with more severe clinical manifestations, emphasizing the correlation between eosinophil count and disease severity. This study demonstrated significantly-increased peripheral eosinophil counts in AR patients, particularly those with comorbid allergic conjunctivitis, thus supporting eosinophilia as a systemic reflection of overall allergic burden. 7 Peripheral eosinophilia in AR results from IL-5–driven bone marrow production and mobilization of eosinophils, reflecting a systemic shift toward type 2 (Th2-mediated) inflammation.34,35 Consequently, elevated eosinophil counts not only serve as an indicator of atopic inflammation but also signify broader immunological dysregulation beyond the nasal mucosa, providing clinicians with an accessible biomarker for assessing disease activity and potentially guiding therapeutic interventions. Furthermore, the eosinophil-to-lymphocyte ratio (ELR) has also been proposed as a useful inflammatory indicator in AR. Recent reports indicate that patients with more severe AR symptoms tend to have higher ELR values, reflecting an elevated eosinophil burden relative to lymphocytes. 8 For example, one study found that adults with severe AR had significantly-greater ELR values than those with milder disease. 36 Although we did not analyze ELR in our cohort, it may warrant consideration in future studies to further capture the eosinophil-dominant aspect of allergic inflammation (Figure 3).
Finally, total IgE levels were markedly elevated in our AR patients relative to controls, reaffirming the crucial role of IgE-mediated sensitization in allergic pathophysiology.6,17,18,37 This observation is in line with extensive literature identifying elevated total IgE as a hallmark of atopy, closely correlated with allergen-specific sensitization profiles.37 -39 Notably, patients with polysensitization have been shown to exhibit substantially-higher total IgE levels than those sensitized to a single allergen, underscoring IgE’s ability to reflect the overall allergic burden and breadth of sensitization. Despite this clear association with atopic status, the correlation between total IgE level and the clinical severity of AR symptoms remains inconsistent across studies.6,10,18,37 -39 Some individuals with very high IgE may have moderate symptoms and vice versa, indicating that while IgE confirms atopic predisposition, it is not a direct gauge of current disease severity.
We evaluated the prognostic value of baseline systemic inflammatory markers in predicting treatment response in AR. Among all indices, NLR showed the strongest predictive utility. A baseline NLR >2.7 was associated with a fourfold increased risk of persistent symptoms, with high sensitivity (84%) and specificity (76%), suggesting its clinical utility for early risk stratification and treatment planning. PLR >240 also showed some predictive value, but its performance was modest (sensitivity 78%, specificity 70%), and its independent use as a prognostic marker remains uncertain. Similarly, SII >1100 was associated with poor outcomes (sensitivity 81%, specificity 73%), indicating that elevated systemic immune burden may reflect more treatment-resistant phenotypes, though its role as a predictive marker requires further validation. Eosinophil counts >600 cells/µL were linked to a higher risk of treatment failure, with strong sensitivity (86%) but moderate specificity (77%). While this supports the value of baseline eosinophilia in identifying high-risk patients who may benefit from more aggressive therapy, variability in outcomes among eosinophil-high individuals limits its use as a stand-alone predictor. In contrast, total IgE >450 IU/mL was associated with increased odds of poor symptom control (~3-fold), but its ability to discriminate between responders and nonresponders was limited. Although high total IgE confirms atopic predisposition, it lacks sufficient specificity for predicting short-term treatment success. Detailed allergen-specific IgE profiles and additional markers may be required to guide personalized therapy decisions. In our multivariate model, baseline eosinophil count remained a significant predictor alongside NLR, suggesting that NLR’s prognostic power is complementary to (and not a replacement for) the established eosinophil-mediated pathways of AR inflammation.
Taken together, these findings indicate that NLR and eosinophil count provide complementary—but biologically-distinct—prognostic information. Although NLR demonstrated strong discriminatory accuracy in ROC analyses, baseline eosinophil count remained an equally-important independent predictor in the multivariate model. This distinction reflects the dual inflammatory pathways involved in AR, where NLR captures neutrophil-driven systemic activation and relative lymphocytopenia, whereas eosinophils reflect Th2-mediated allergic inflammation. Patients exhibiting simultaneous elevations in both markers represented the highest risk phenotype for poor treatment response, while those with low NLR and low eosinophil counts experienced the most favorable outcomes. Thus, NLR does not supersede eosinophils; rather, both markers together enhance clinical risk stratification more effectively than either alone.
Beyond these biomarker-specific analyses, the inclusion of a repeated-measures mixed-effects model provides important methodological strength to our study. While change-change correlations are informative, they can be susceptible to mathematical coupling and regression-to-the-mean effects. The mixed-effects model, which evaluates change within individuals while simultaneously accounting for differences between groups over time, confirmed that reductions in NLR, PLR, and eosinophil counts were significantly greater in AR patients than in controls. This reinforces that the observed biomarker improvements were treatment-related rather than attributable to temporal variability. Accordingly, the consistency between the mixed-effects model and our Δ-based analyses strengthens the validity of our longitudinal findings and supports the clinical relevance of these biomarkers as dynamic indicators of therapeutic response.
This study has several notable strengths. First, the prospective, controlled design (with a matched healthy control group) provides robust evidence by allowing longitudinal comparisons and minimizing potential biases. We used standardized diagnostic criteria based on ARIA guidelines and objective symptom scoring (TNSS and VAS), which ensures consistency, reproducibility, and precision in evaluating disease severity and treatment responses. Additionally, the 6 month follow-up period is sufficiently long to capture meaningful changes in both clinical symptoms and inflammatory parameters, enabling us to observe the temporal effects of treatment on systemic inflammation in AR.
However, several limitations should be acknowledged. First, as a single-center study, the generalizability of our findings to broader populations or different clinical settings may be limited; multicenter studies involving more diverse patient groups would enhance the external validity of these results. Second, although 6 months of follow-up provided valuable insight, this duration might still be insufficient to capture longer term inflammatory fluctuations or delayed treatment effects, so extended follow-up studies are warranted. Third, the systemic inflammatory markers examined (NLR, PLR, SII) are nonspecific indicators of inflammation and could be influenced by extrinsic factors such as intercurrent infections, stress, or comorbid conditions; we attempted to control for obvious confounders, but their influence cannot be entirely excluded. Fourth, our predictive cutoff values and multivariate model were derived from a single-center cohort without internal or external validation. This limitation raises the possibility that the reported sensitivities, specificities, and odds ratios may be optimistically biased. Direct comparison of our thresholds with those from other AR cohorts is also challenging due to differences in study populations and methodology. Larger multicenter studies incorporating validation cohorts or statistical techniques such as bootstrapping or k-fold cross-validation will be essential to confirm the generalizability and robustness of these predictive findings.
Lastly, the standardized treatment regimen in our study (while clinically appropriate) might not reflect the full range of therapeutic approaches in real-world practice, which could affect the applicability of our findings. Future research addressing these limitations will help in establishing more definitive clinical guidelines for incorporating these biomarkers into the management of allergic rhinitis.
Conclusion
In this prospective 6 month cohort study, we demonstrated that systemic inflammatory indices derived from routine blood counts (NLR, PLR, and SII) provide clinically-meaningful information for assessing disease activity and predicting treatment response in allergic rhinitis. All 3 markers were significantly elevated at baseline in AR patients, correlated with symptom severity, and decreased in parallel with clinical improvement following standard therapy. Among them, NLR showed the strongest predictive performance, while SII and PLR offered additional insights into the broader inflammatory burden. Eosinophil counts remained a robust complementary predictor, reflecting the Th2-dominant pathway of allergic inflammation. Together, these findings highlight the value of integrating easily-accessible hematologic indices into the routine evaluation of AR to enhance risk stratification, monitor treatment response, and inform individualized management strategies. Larger multicenter studies are warranted to validate these biomarkers and further refine their clinical applicability.
Footnotes
Acknowledgements
The authors thank the clinical and laboratory staff of Ankara Bilkent City Hospital for their support during data collection and patient follow-up.
Ethical Considerations
The study was conducted in full compliance with the Declaration of Helsinki. Ethics approval was obtained from the Ethics Committee of Ankara Bilkent City Hospital (IRB; TABED: 1-25-1250). All procedures adhered to institutional and national ethical standards, and participant confidentiality was fully protected throughout the study.
Consent to Participate
Written informed consent was obtained from all participants prior to study enrollment.
Consent for Publication
Participants provided informed consent for the publication of anonymized data derived from this research.
Author Contributions
All authors contributed to the study conception, design, data collection, analysis, interpretation, and manuscript preparation. All authors approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Artificial Intelligence Usage Disclosure
Artificial intelligence tools (ChatGPT developed by OpenAI) were used only to assist with language refinement, grammar correction, structural editing, and improving the clarity of the manuscript. AI tools were not used for data analysis, statistical computation, hypothesis generation, or drafting of original scientific content. All scientific interpretations, conclusions, and data analyses were performed solely by the authors. The authors reviewed and verified all AI-assisted edits to ensure accuracy and integrity.
