Abstract
Objectives:
To investigate and identify the most reliable method for assessing Eustachian tube function (ETF) in pediatric patients with otitis media with effusion (OME).
Methods:
A total of 49 ears from pediatric patients diagnosed with OME and 32 healthy control ears were included. The assessment methods for ETF included acoustic immittance measurement (AIM), tubomanometry (TMM), Eustachian tube score (ETS), sonotubometry (STM), and tubo-tympano-aerodynamic-graphy (TTAG).
Results:
AIM was used as the standard, and the consistency of TMM, ETS, STM, and TTAG was tested with sensitivities of 75.6%, 60.0%, 82.05%, and 53.85%, specificities of 94.4%, 97.2%, 66.67%, and 83.33%, and Youden indices of 0.700, 0.572, 0.487, and 0.372, respectively. ROC analysis revealed an optimal diagnostic threshold for TMM values of 4.5. Significant differences in AIM results were observed between groups stratified by this TMM-based diagnostic threshold (χ2 = 36.690, P = .000). When the TMM values were <4.5, the rate of normal Eustachian tube opening was 5.55%, which increased to 75.56% for values ≥4.5.
Conclusion:
TMM demonstrates superior diagnostic performance compared to ETS, STM, and TTAG. The combination of AIM and TMM is recommended for the evaluation of ETF in pediatric patients with OME.
Introduction
Otitis media with effusion (OME) is a common pediatric disease. 1 The pathogenesis of OME involves infections, gastroesophageal reflux, respiratory allergies, and genetic factors. 2 However, the majority of studies supported that the Eustachian tube dysfunction (ETD) is responsible for the high incidence of pediatric OME and plays a critical role in its prognosis. Currently, the acoustic immittance measurement (AIM) is a widely used in clinical practice for assessing the Eustachian tube function (ETF). Additionally, several new methods have been introduced, including tubomanometry (TMM), Eustachian tube score (ETS), sonotubometry (STM), and tubo-tympano-aerodynamic-graphy (TTAG). However, the most reliable method for evaluating ETF remain unclear, especially in pediatric patients. The aim of this study is to identify the most effective diagnostic tool for ETD in children with OME.
Materials and Methods
Study Population
Forty-one patients aged between 4 and 14 years who visited the ENT outpatient from December 2023 to June 2024 were recruited. Informed consent was obtained from the legal guardian of each participant, and this study was approved by the Ethics Committee of the First Affiliated Hospital of the Air Force Medical University (KY20222070-F-1).
These subjects were divided into the OME group and the normal group according to the following criteria. The inclusion criteria for the OME group included presence of typical clinical symptoms, such as aural fullness, hearing loss, tinnitus, or otorrhea; the tympanic membrane was intact and retracted and had middle ear effusion; and the AIM test showed type B or C tympanograms with negative acoustic reflexes. Subjects without ear symptoms, with type A in the AIM test, and with positive acoustic reflexes were included in the normal group. The exclusion criteria were as follows: a history of nasopharyngeal tumor, cleft palate, Down's syndrome, external auditory canal (EAC) lesions, chronic otitis media, and chronic sinusitis.
ETF Tests
All the participants were seated and sequentially received tests of AIM, TMM, ETS, STM, and TTAG, and a 15-minute break was taken after each test. The JK-05A (RION, Tokyo, Japan) was used for the STM and TTAG tests, whereas the TMM test was performed using a tubomanometer (Spiggle & Theis, Germany).
For the AIM test, a Titan device (International Hearing, Denmark) with a 226 Hz probe recorded tympanograms. The results were classified using the Liden-Jerger system: Type A indicated normal Eustachian tube (ET) function, whereas Types B and C indicated abnormal function.
The TMM test was conducted as follows 3 : a nasal adapter delivered pressures of 30, 40, and 50 mbar while the subjects swallowed water. Changes in the pressure of the EAC were recorded as the ET opening latency index (R value). R ≤ 1 indicated immediate ET opening (2 points), R > 1 indicated delayed opening (1 point), and R < 0 indicated complete obstruction (0 points). A total score across all pressures yielded the TMM value, with scores of 0 or 1 indicating abnormal function.
The ETS test combined TMM values with subjective ratings of Valsalva’s and Toynbee’s symptoms. 4 Symptoms were scored as 2 points for “always,” 1 for “sometimes,” and 0 for “never.” A total score ≤5 indicated an abnormal ETF.
For the STM test, 5 a 7-kHz noise was delivered to one nostril, and a microphone in the ipsilateral EAC was used to measure changes in the sound pressure level (SPL) during swallowing. A ≥5 dB increase indicated a normal ETF.
The TTAG test 6 involved placing pressure sensors in the EAC and nostril. The subjects performed a Valsalva maneuver while the opposite nostril was closed. An increase in EAC pressure ≥10 daPa indicated normal function.
Statistical Analysis
SPSS 26.0 software (IBM, Armonk, NY) was used for statistical analysis. T-tests and chi-square tests were used to compare the differences between the two groups. Nonparametric Cochran Q tests were used to compare the ET opening rates of the TMM test results under different pressures. Consistency tests were performed using the kappa consistency test, and the degree of consistency was as follows: extremely strong, 0.81 to 1.00; strong, 0.61 to 0.80; moderate, 0.41 to 0.60; and poor, < 0.20. ROC curves were constructed to establish optimal thresholds for the TMM value. P < .05 indicated statistical significance.
Results
The study included 81 ears from 41 children (22 males and 19 females), with a mean age of 7.22 ± 2.56 years. The OME group consisted of 49 ears from children with a mean age of 7.08 ± 2.48 years, including 28 ears from males and 21 ears from females. The normal group consisted of 32 ears, including 15 from males and 17 from females, with an average age of 7.34 ± 2.67 years. There were no significant differences between the two groups in terms of sex (P = .365) and age (P = .784).
Consistency Comparison
AIM was used as a standard, and the consistency of TMM, ETS, STM, and TTAG was evaluated. The results of the TMM are consistent with those of the AIM at different pressures and show moderately strong agreement with those of the AIM at 30 and 40 mbar (30 mbar: kappa value = .557; 40 mbar: kappa value = .451), and are generally consistent with the AIM at 50 mbar (kappa value = .371). The agreement rates at the pressures of 30, 40, and 50 mbar were 77.78%, 72.84%, and 69.14%, respectively. Furthermore, the results of ETS, STM, and AIM results were also moderately in agreement with 79.01% and 74.07%, respectively (ETS: kappa value = .575; STM: kappa value = .484). The sensitivity of the STM was 82.05%, the specificity was 66.67%, and the Youden index (YI) was 0.487. In contrast, the results of the TTAG agreed well with those of the AIM (Kappa value = .376), with an agreement rate of 69.14%. The sensitivity of TTAG was 53.85%, the specificity was 83.33%, and the YI was 0.372. (Table 1)
Consistency Comparison Between Different ET Tests and AIM.
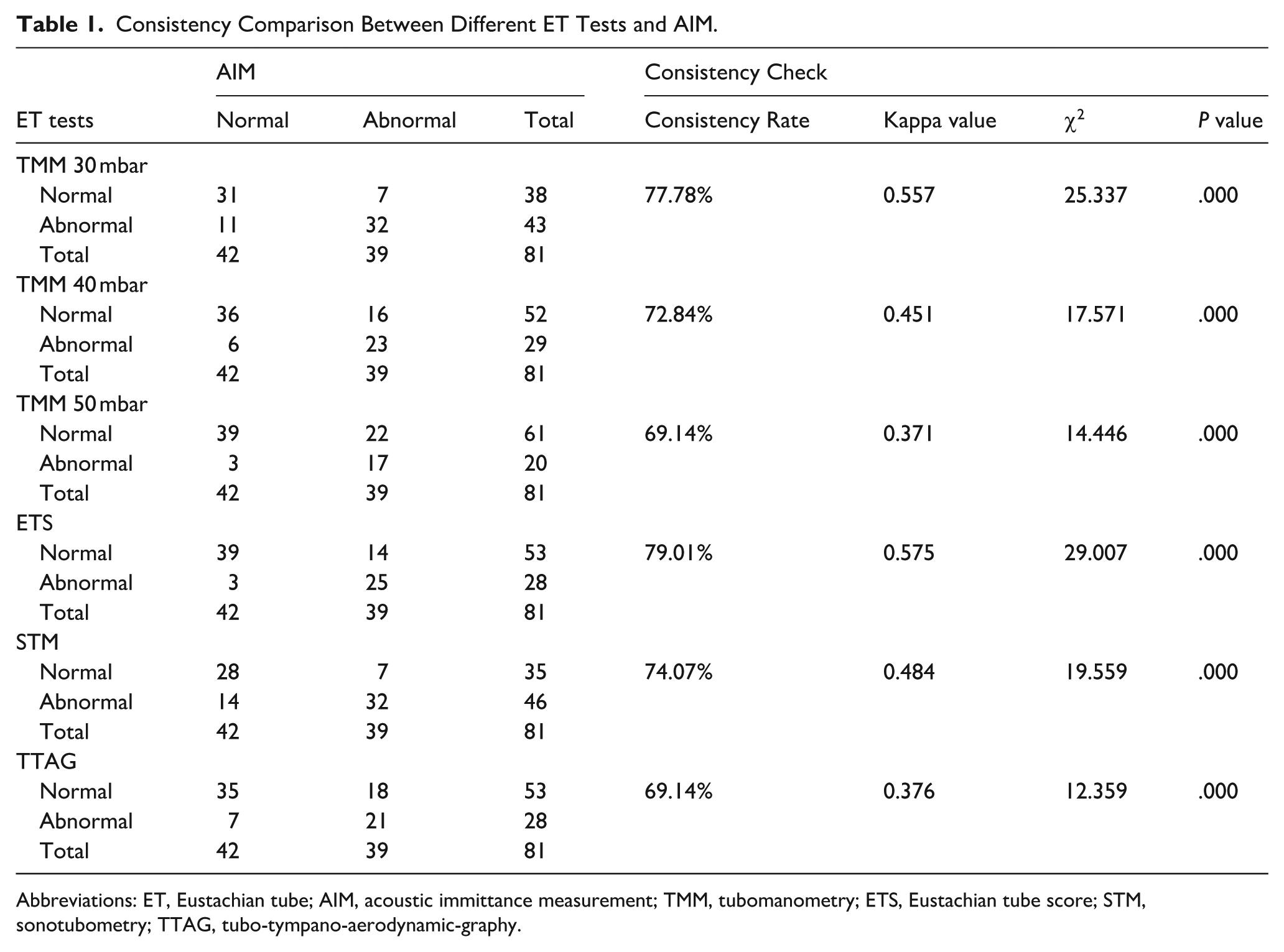
Abbreviations: ET, Eustachian tube; AIM, acoustic immittance measurement; TMM, tubomanometry; ETS, Eustachian tube score; STM, sonotubometry; TTAG, tubo-tympano-aerodynamic-graphy.
Analysis of TMM Results
TMM Results
The incidence of abnormal ETF is associated with TMM pressure. In the OME group, the percentages of abnormal ET opening function were 77.6%, 59.2%, and 40.8% at pressures of 30, 40, and 50 mbar, respectively. In the control group, the percentages were 18.2%, 3.0%, and 3.0%, respectively. Significant differences were observed between the two groups when the results at the three pressures were compared (30 mbar: χ2 = 27.952, P = .000; 40 mbar: χ2 = 26.801, P = .000; 50 mbar: χ2 = 14.779, P = .000). The proportion of normal ET opening significantly increased with increasing pressure in the OME group (χ2 = 13.686, P = .001). Pairwise comparisons within the OME group revealed statistically significant differences only between 30 mbar and 50 mbar (χ2 = 13.686, P = .000). Similarly, the normal group exhibited a significant pressure-dependent increase in normal ET opening rates (χ2 = 5.541, P = .047). Significant pairwise differences in the normal group were observed solely between 30 and 40 mbar (χ2 = 3.995, P = .046), and between 30 and 50 mbar (χ2 = 3.995, P = .046) (Table 2).
TMM in the OME Group and Normal Control Group.
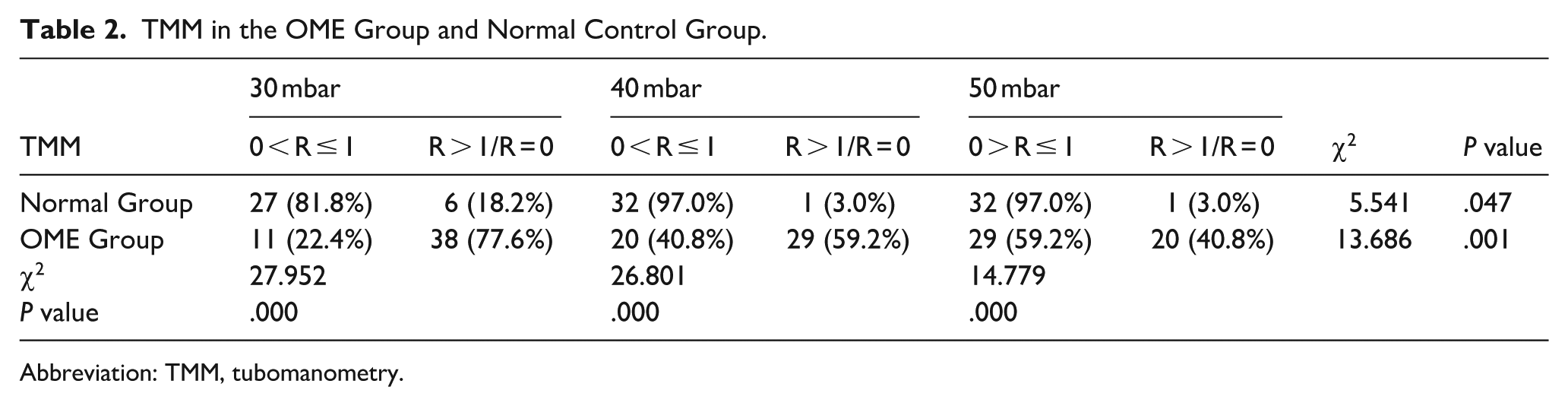
Abbreviation: TMM, tubomanometry.
TMM in Tympanograms of Types B and C
Children with a type B or C tympanogram tend to have impaired ETF. Consequently, the results of the types B and C tympanograms were compared with those of the TMM tests under different pressure conditions. There was no significant difference among the 3 groups with type B tympanograms (χ2 = 1.643, P = .440). With respect to children with type C tympanograms, the rates of abnormal ET opening function were 87.5%, 43.8%, and 18.8% at pressures of 30, 40, and 50 mbar, respectively. When the TMM results were compared between types B and C tympanograms under different pressures, only a significant difference was detected at 50 mbar (χ2 = 6.808, P = .009). Moreover, as the pressure increased, the proportion of children with type B or type C tympanograms who had abnormal ET opening function decreased (Table 3).
TMM in Tympanogram Types B and C.

Abbreviation: TMM, tubomanometry.
Quantification of the Relationship Between TMM Values and ETF in Children
The results of the ROC analysis of the TMM values for all the children revealed that the TMM values were accurate for assessing ETF in children, with an area under the curve of 0.866 (CI = 0.792–0.940), YI = 0.700, an optimal diagnostic threshold of 4.5, a sensitivity of 75.6% and a specificity of 94.4%. (Figure 1). There were significant differences in AIM results between groups categorized by TMM value-based diagnostic thresholds (χ2 = 36.690, P = .000). With a TMM values of < 4.5 (TMM values 1-4), the AIM detection rate of normal ETF was 5.55%; with a TMM values of > 4.5 (TMM values 5-6), the AIM detection rate of normal ETF was 75.56% (Table 4). The ETS is also accurate for assessing ETF in children, with an area under the curve of 0.855 (CI = 0.778–0.932), YI = 0.572, a sensitivity of 60.0% and a specificity of 97.2% (Figure 2).
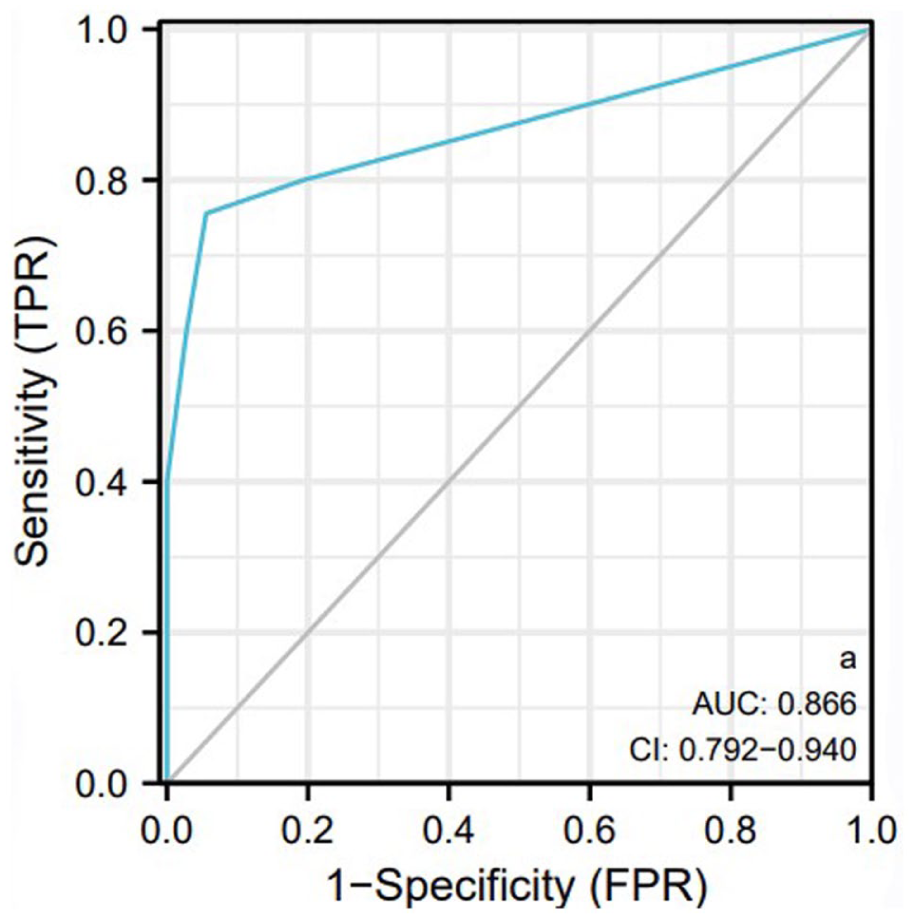
ROC analysis of TMM values.
Quantification of the Relationship Between TMM Values and ETF in Children.
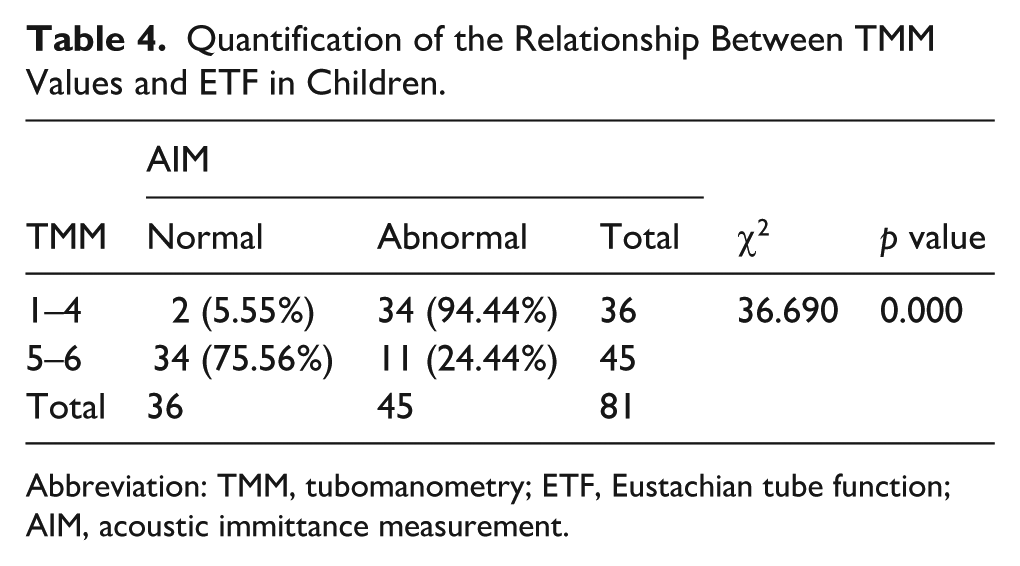
Abbreviation: TMM, tubomanometry; ETF, Eustachian tube function; AIM, acoustic immittance measurement.
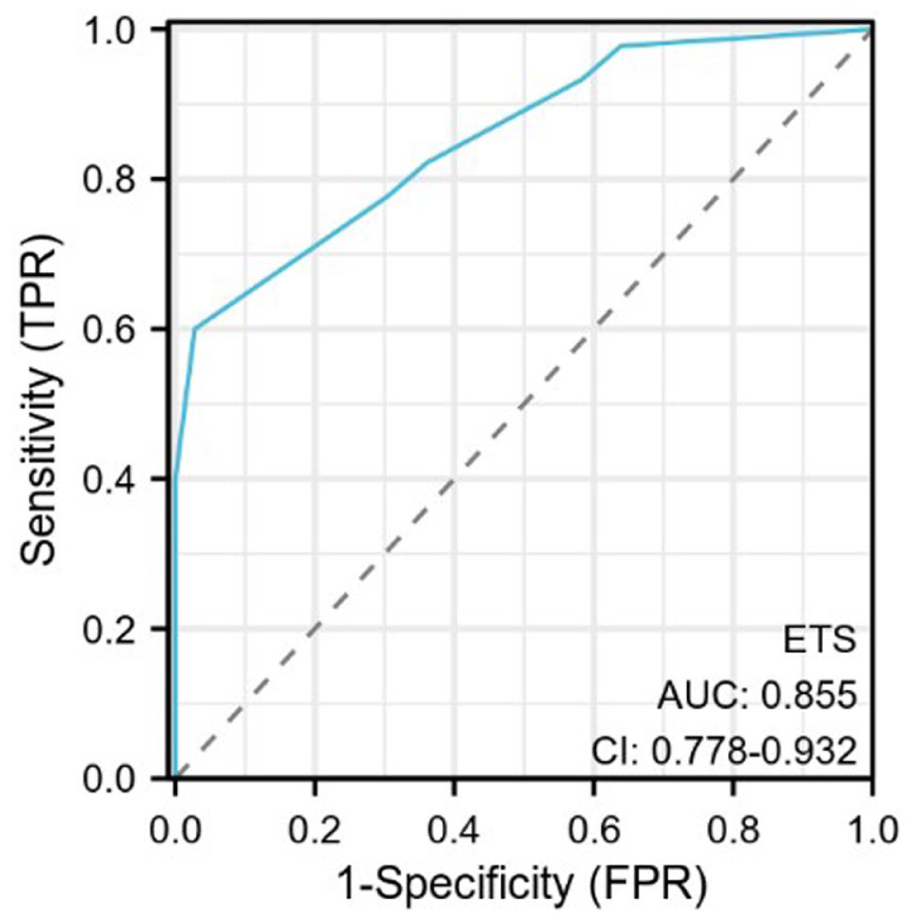
ROC analysis of ETS. ETS, Eustachian tube score.
Discussion
ETD is clinically prevalent. The prevalence of ETD is 4.6% in adults in the United States and is even higher in children.7,8 Owing to the unique anatomy of the ET in children, 9 they are more prone to edema in response to certain bacterial and/or viral infections, mechanical obstruction and immune responses. 10 ETD is a major cause of recurrent OME. Although more than 100 years ago Politzer proposed that abnormal ETF might be a significant factor in middle ear disease, 11 there is still no clinical “gold standard” for evaluating ETF. However, several tests have emerged, such as TMM, ETS, STM, and TTAG. Multiple studies have compared the correlations among these tests or explored their values as predictive factors for treatment outcomes.12 -14 However, their validity and reliability remain unclear. Moreover, most related studies are focused primarily on adults, and studies on pediatric patients are limited.14 -16 Therefore, identifying the most effective diagnostic tool for ETD in children is crucial.
As a simple method for measuring middle ear pressure, the AIM is currently widely used in clinical practice. Kim et al 7 in their study on the prevalence and population estimates of ETD among American adolescents, proposed that AIM alone can provide a conservative estimate of ETD prevalence in the general population. Shekelle et al 17 from overseas reported that its sensitivity and specificity were 94% and 95%, respectively. Additionally, a multitude of studies focusing on ETD-related diseases have adopted AIM as one of the primary measures.13,18,19 In this study, we used the AIM as a reference standard to evaluate the consistency of the TMM (at 30, 40, and 50 mbar), ETS, STM, and TTAG. All methods demonstrated some diagnostic capability for ETD in children. However, as the pressure increased, the agreement between the TMM and AIM decreased, likely because passive ET opening under excessive pressure led to false-negatives. We computed the sensitivity, specificity, and Youden’s index for each test. STM demonstrated the highest sensitivity, indicating considerable value in screening for patients with potential ETD. Notably, high sensitivity is typically accompanied by an elevated false-positive rate; thus, this method holds significant clinical relevance for excluding ETD comorbidity in patients with OME. The ETS exhibited highest specificity, suggesting prominent advantages in confirming ETD diagnosis. However, the high specificity may be associated with an increased risk of missed diagnoses. In contrast, TMM yielded the highest Youden’s index, indicating superior diagnostic value for pediatric ETF, which is consistent with Herrera M’s findings. 16 A comparative analysis of the TMM results under different pressure levels between the normal group and the OME group revealed that the proportion of ET opening remained largely consistent across pressure levels in normal subjects. In contrast, the OME group showed a statistically significant difference, which may be attributed to the passive opening of dysfunctional ET under higher pressure. In the normal group, 6 cases did not open at 30 mbar; 5 of these opened normally at higher pressures, possibly due to suboptimal swallowing timing. One case failed to open at all pressures, potentially because of recent upper respiratory infection causing transient mucosal swelling or obstruction. This finding highlights the importance of screening for recent infections, rhinitis, or immune conditions before testing to avoid misdiagnosis. Furthermore, such subjects may also be affected by abnormal pressure regulation of the ET. 16 Aiper et al 3 argue that although the TMM test is sensitive, it may lack specificity in detecting ET opening, especially when it is only conducted on healthy subjects and does not cover all TMM pressure ranges.
AIM is generally recognized for its high sensitivity and specificity in detecting middle ear effusion. Individuals with middle ear effusion and tympanic membrane retraction typically exhibit type B or type C tympanograms. When these findings were compared with those of the TMM, we found that a statistically significant difference was observed only at a pressure of 50 mbar. In subjects with type B tympanograms, the TMM results did not significantly vary with increasing pressure. This may be due to the passive opening of the ET under elevated pressure in type C cases, whereas more severe ETD in type B cases could resist opening even at high pressures. Another study by Alper et al 20 indicates that in TMM, a pathological R value at 30 to 40 mbar may indicate the passive opening of a normal ET. Only when a pathological result occurs at 50 mbar or the ET fails to open across all pressure levels can it suggest ETD.
STM is the only method that takes into account the physiological status of subjects. It determines whether the Eustachian tube is open by judging whether sound can be transmitted through the ET during a dry swallow. 15 TTAG applies the principle of aerodynamics to measure the pressure changes generated when air flows into and out of the middle ear cavity through the ET. 21 ETS combines subjective scores such as those of swallowing and the Valsalva maneuver with those of the TMM. 4 Both STM and TTAG rely on maneuvers such as swallowing, nose-pinching, and air-blowing, which exhibit considerable interindividual and intraindividual variability. This dependency on subjective cooperation limits reproducibility and requires substantial comprehension and operational ability from subjects, making these methods suboptimal for pediatric ETD assessment. Similarly, subjective scoring based on self-reported perceptions of ear sounds is unsuitable for children.
In contrast, TMM shows significant advantages in the detection of ETF in children. 22 A prospective validation 23 has clearly pointed out that TMM is a reliable tool for measuring ETF in children, and the normal limit of its R value should be set at 1.12. A retrospective study 14 indicates that adult patients with acute OME who have a TMM value of 2 to 6 and a type C tympanogram may benefit from pharmacotherapy. The TMM value is an independent predictor of treatment outcomes. We performed a ROC analysis to compare the TMM and ETS results, which indicated that although ETS offers complementary diagnostic value when combined with the TMM, the TMM values alone possesses superior diagnostic performance for children. Meanwhile, the optimal TMM diagnostic threshold was identified as 4.5, with scores of 0 to 4 indicating abnormal function and scores of 5 to 6 indicating normal function. On the one hand, TMM values eliminate the uncertainty of subjective scoring, making the test results more objective. On the other hand, it is easy to perform and can conduct a semiquantitative assessment of the ETF of children. Therefore, the TMM is a reliable tool for evaluating the ETF of children.
Conclusions
OME in children is closely linked to ETD. AIM TMM, ETS, STM, and TTAG can all be used to assess ETF in pediatric patients. Among these, the TMM is a simple and accurate approach for the objective and semiquantitative evaluation of ETF. It demonstrates superior diagnostic performance compared to with ETS, STM, and TTAG in children. A TMM value of ≤4 is strongly associated with ETD and may serve as a meaningful diagnostic threshold. For a comprehensive and accurate assessment of ETF in children, the combined use of the AIM and TMM is recommended.
Limitations
Given that the research participants were children, the sample size included in this study was relatively small. Additionally, due to coordination considerations, the participation of children under 4 years old was restricted. Furthermore, the absence of long-term follow-up data precluded confirmation of the association between ETF and the efficacy and prognosis of children with OME. Our research team will continue to conduct prospective follow-up for this study. Finally, a critical limitation is the lack of a universally accepted “gold standard” for the diagnosis of ETD.
Footnotes
Acknowledgements
The authors thank all the participants and guardians for their consent. Thank them for their great contributions.
Ethical Considerations
This study was approved by the Ethics Committee of the First Affiliated Hospital of the Air Force Medical University (KY20222070-F-1).
Consent for Publication
Written informed consent has been obtained from the parents of these patients.
Author Contributions
Rui Li: data collection, statistics and manuscript writing, manuscript submission. Jiawei Chen: English edition and supervision. Runqin Yang: manuscript proofreading, English edition and supervision. Xinhui Wang: manuscript proofreading. Lin Li: management of data collection, project conception. Xuewei Liu: management of data collection, manuscript proofreading. Lizhi Sun: management of data collection, manuscript proofreading. Dingjun Zha: project conception, manuscript proofreading. Yu Han: project conception, manuscript proofreading, English edition and supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the National Natural Science Foundation of China (82471167), and National Clinical Research Center for Otorinolaryngologic Diseases (2024KF010).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
