Abstract
Purpose:
Residual cholesteatoma poses a great challenge in middle ear surgery. Considering that microscopes only offer line-of-sight vision, it was suggested earlier that endoscopes might prove useful to control difficult-to-visualize areas during surgery to ensure total evacuation of squamous epithelium. In this retrospective chart study, we evaluated intraoperative endoscopic findings of difficult-to-access sites in the tympanic cavity and postoperative cholesteatoma recidivism rate in these areas in a patient cohort who underwent primarily microscopic surgery.
Main findings:
Residual cholesteatoma mass was intraoperatively detected in difficult-to-visualize areas using an endoscope during 50% of the procedures (10 cases), mostly in the sinus tympani (6 cases), but also in the supratubal recess (3 cases). During a mean follow-up of 36.1 months (range: 20-59 months, targeted magnetic resonance imaging [MRI] mean follow-up: 31.3 [range: 12-59]), recurrent attic cholesteatoma was found in 2 patients (10%), and residual disease was detected with dedicated MRI sequences in another 2 cases (10%). None of the residual cholesteatomas developed in the endoscopically controlled areas. The mean air-bone gap closure at 1 year was 1.9 ± 10.2 dB (range: −18.3-21.7).
Conclusions:
Intraoperative endoscopic surveillance helps uncovering hidden cholesteatoma mass during routine microscopic surgery and thus offers an effective control of residual disease in these areas during a medium-term follow-up.
Introduction
Certain spaces in the tympanic cavity such as the supratubal recess and the tympanic sinus 1 are difficult to visualize through a traditional microscopic approach during cholesteatoma surgery, and thus, the removal of squamous epithelium cannot be directly supervised in these areas. The sinus tympani is hidden by the facial nerve, while the supratubal recess is challenging to access due to the position of the malleus, the cochleariform process, and the tendon of the tympani tensor muscle. The introduction of endoscopes in otologic surgery allowed surgeons to remove cholesteatomas under direct visual control even from these spaces. 2 Thus, cholesteatomas that do not involve the mastoid cavity can be safely removed by using endoscopes alone, offering a less invasive approach with a similar success rate compared to the traditional microscopic procedure.3,4
Although several studies evaluated the usefulness of endoscopy in cholesteatoma removal showing promising results, data are scarce regarding additional otoendoscopic control in cases where microscopic approach is required due to the involvement of the mastoid bowl.3,5 -9 While most surgeons reported a positive effect for an additional endoscopic inspection in these cases,6,10 -13 some expressed concern over significantly elongated operating times and the use of extra resources. 14
The effectiveness of intraoperative endoscopic control in the microscopically operated cases was mostly evaluated regardless of the original location of the disease or was reported without consistent follow-up. 15 However, the real potential value of otoendoscopy during microscopic surgeries can only be grasped when considering successful disease removal specifically in difficult-to-access sites during follow-up.
Our objective in this project was to retrospectively measure the incidence of residual cholesteatoma in a patient cohort operated using a combined endoscopic-microscopic approach, focusing on endoscopic findings and residual disease rate in those spaces that cannot be directly visualized by microscope alone.
Materials and Methods
Records were screened for patients who underwent surgery with a combined endoscopic-microscopic approach. This procedure was used in cases where cholesteatoma invaded difficult-to-access sites according to the preoperative computed tomography (CT) scan and/or the intraoperative findings. A microscopic approach was preferred when mastoid involvement could be presumed based on the preoperative CT scan.
Patients of any age or sex were included in our study who were operated on from November 1, 2019 to June 30, 2022, using the aforementioned techniques with the goal of cholesteatoma removal. According to our department’s surgical protocol established in 2019, all patients undergo a decade-long follow-up, consisting of annual office visits (physical and otomicroscopic examination), audiological examination and magnetic resonance imaging (MRI) examination (with dedicated cholesteatoma protocol), or second-stage surgery in the year following the initial procedure.
Previous canal-wall-down surgery or radiation treatment in the temporo-cervical region, preexisting craniofacial malformation, and failure to present to follow-up visits were reasons for exclusion.
All patients underwent physical examination, otomicroscopy, audiological evaluation, and high-resolution temporal bone CT scan preoperatively. Procedures were always performed under general anesthesia by 1 of 3 experienced surgeons in our working group who share the same attitude and experience. All patients received cortical mastoidectomy and atticotomy via retroauricular incision except 1 patient, who underwent a canal wall down procedure due to extensive destruction caused by cholesteatoma on the bony posterior wall of the external auditory canal, which was reconstructed at the end of the operation with conchal cartilage, and the mastoid cavity was obliterated with bone pâté. After microscopic dissection, a 45-degree endoscope (Spiggle&Theis GmbH, Overath, Germany) was used to check for cholesteatoma in difficult-to-access areas. In the case of the previously mentioned canal wall down patient, only the supratubal recess needed endoscopic control. If residual cholesteatoma was found, dissection was continued under endoscopic visual control until complete removal. According to the Lee-Cohen classification, these surgeries were classified as class II/a, while those where no squamous epithelium was found during otoendoscopy and thus no endoscopic manipulation was performed were classified as class I. 16 The mastoid cavity and the epitympanic space were obliterated with harvested bone pâté if the cholesteatoma extended into or beyond the mastoid antrum. The tympanic membrane was reconstructed with temporal muscle fascia graft, perichondrium, or thinned conchal cartilage. If needed, the ossicular chain was reconstructed. All patients were followed up at 6 weeks (otomicroscopy and audiology), 6 months (otomicroscopy and audiology), and 1 year postoperatively (otomicroscopy and audiology), then annually (otomicroscopy, audiology, and targeted MRI). Variations in timing occurred in some cases due to patients’ personal scheduling and radiological capacity issues. Unenhanced MRI scans were performed with a Philips Ingenia 1.5T, with dedicated protocol for cholesteatoma follow-up, which included 2 separate series of non-EPI DWI in 2 different planes (axial and coronal), with a slice thickness of 3 mm and b values of b = 0 and b = 1000.
Residual disease was defined according to the EAONO/JOS statement. MRI scans were considered positive for residual cholesteatoma if a pronounced diffusion restriction was visualized in the expected area of the surgery with a corresponding moderate signal intensity on T2w and DRIVE sequences.
Audiological outcomes were evaluated according to the recommendations of the Committee of Hearing and Equilibrium. 17 Air conduction and bone conduction were measured pre- and postoperatively (early postoperative: 6 to 12 weeks and late postoperative: 1 year), and the air-bone gap (ABG) was calculated. The pure tone average threshold was determined as the mean value of thresholds at 0.5-1 to 2-3 kHz.
Data was reviewed and coded by the first authors. Demographic features, date of surgery and follow-up visits were collected retrospectively from patients’ charts, as well as results of pre- and postoperative audiological examinations. Data were collected after receiving ethical approval for this study on September 12, 2023 (approval protocol ID: ETT TUKEB BM/24488-1/2023). Data tables were created and completed in a password-protected Microsoft Excel file and were stored on a secure drive, only the data managers, BK and KI, had full access to. Interrater reliability for coding data was consistently monitored throughout the data collection phase through discussion between the authors. When editing the manuscript, we have proceeded according to the recommendations of the STROBE checklist. 18
Descriptive statistical analysis was conducted using Microsoft Excel (Microsoft Corporation, Redmond, WA, USA, 2018; https://office.microsoft.com/excel) to synthesize data on hearing outcomes. Two-sample T-test was used to evaluate whether the average of the preoperative and late postoperative ABG significantly differs. SPSS software (IBM Corp. Released 2023, IBM SPSS Statistics for Windows, Version 26.0; IBM Corp., Armonk, NY, USA) was used to create Kaplan-Meier curves to estimate recidivism-free survival. 19
Results
Participants
Of all patients who underwent cholesteatoma surgery at our institution, a total of 23 were potentially eligible to partake in this study. Attrition among included patients was caused by comorbidities that made follow-up visits impossible in 1 case, a subsequent diagnosis of eosinophilic otitis media in another, and noncompliance in the remaining 1 (Figure 1). No complication related to the procedures was detected. Patients included in our cohort had a mean age of 36.2 (range: 7-79), 60% were female, and underwent follow-up for a minimum of 1 year (mean: 36.1 months, range: 20-59 months, mean follow-up with targeted MRI: 31.3 months [range: 12-59 months]; Table 1). Residual cholesteatoma was detected 31 months postoperatively on average. According to the EAONO/JOS cholesteatoma staging system, all included patients had stage II cholesteatoma. Potential bias might originate from the retrospective design of this study, and since operations were conducted during the pandemic, in 1 case (No.3), a 6-week follow-up had to be skipped by the patient due to a case of SARS-CoV-2 infection. Patients who developed residual cholesteatoma were followed after second-look procedures, to this date, no second residuals were detected.
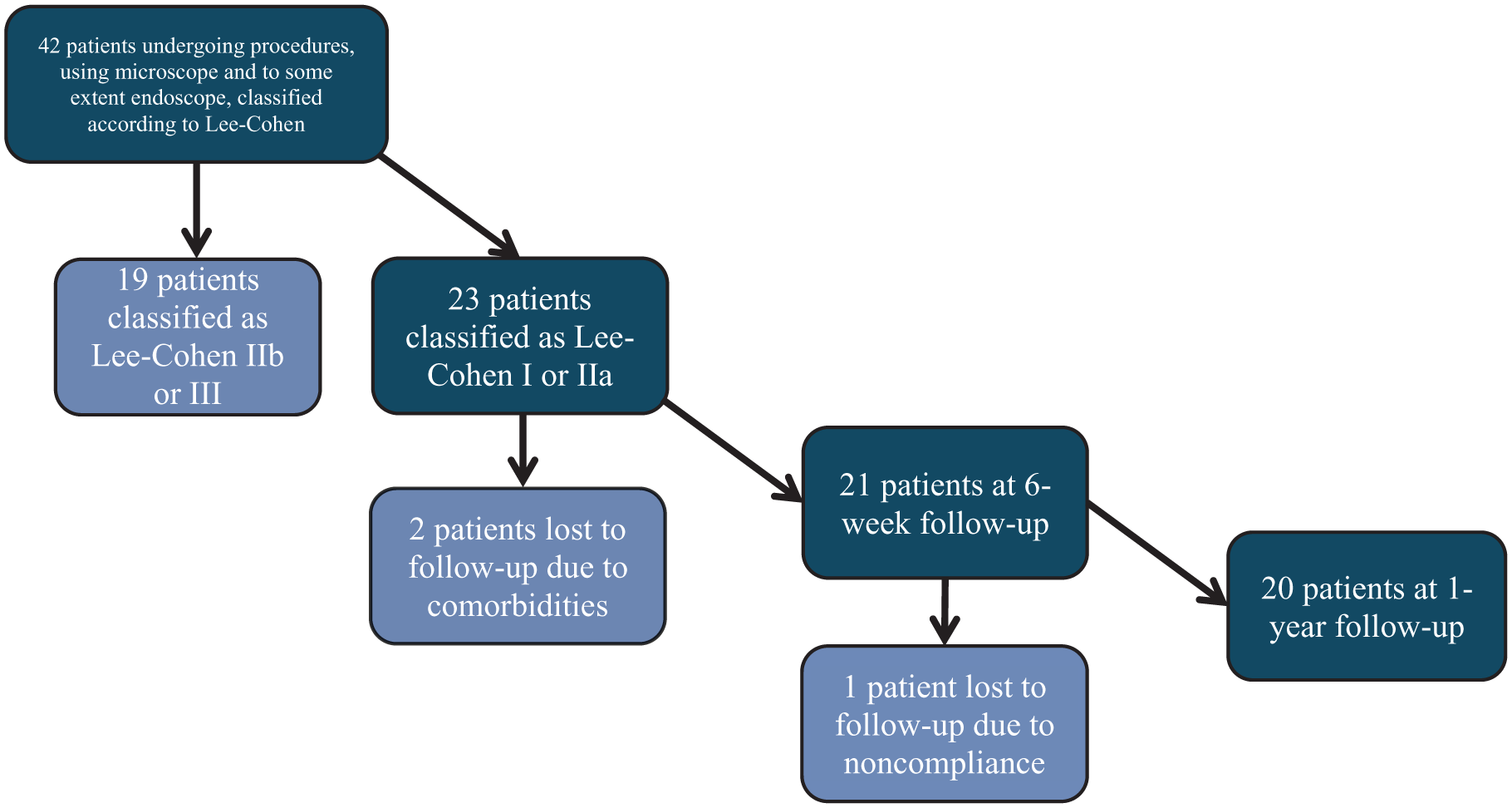
Patient selection and follow-up process.
Patient Characteristics.
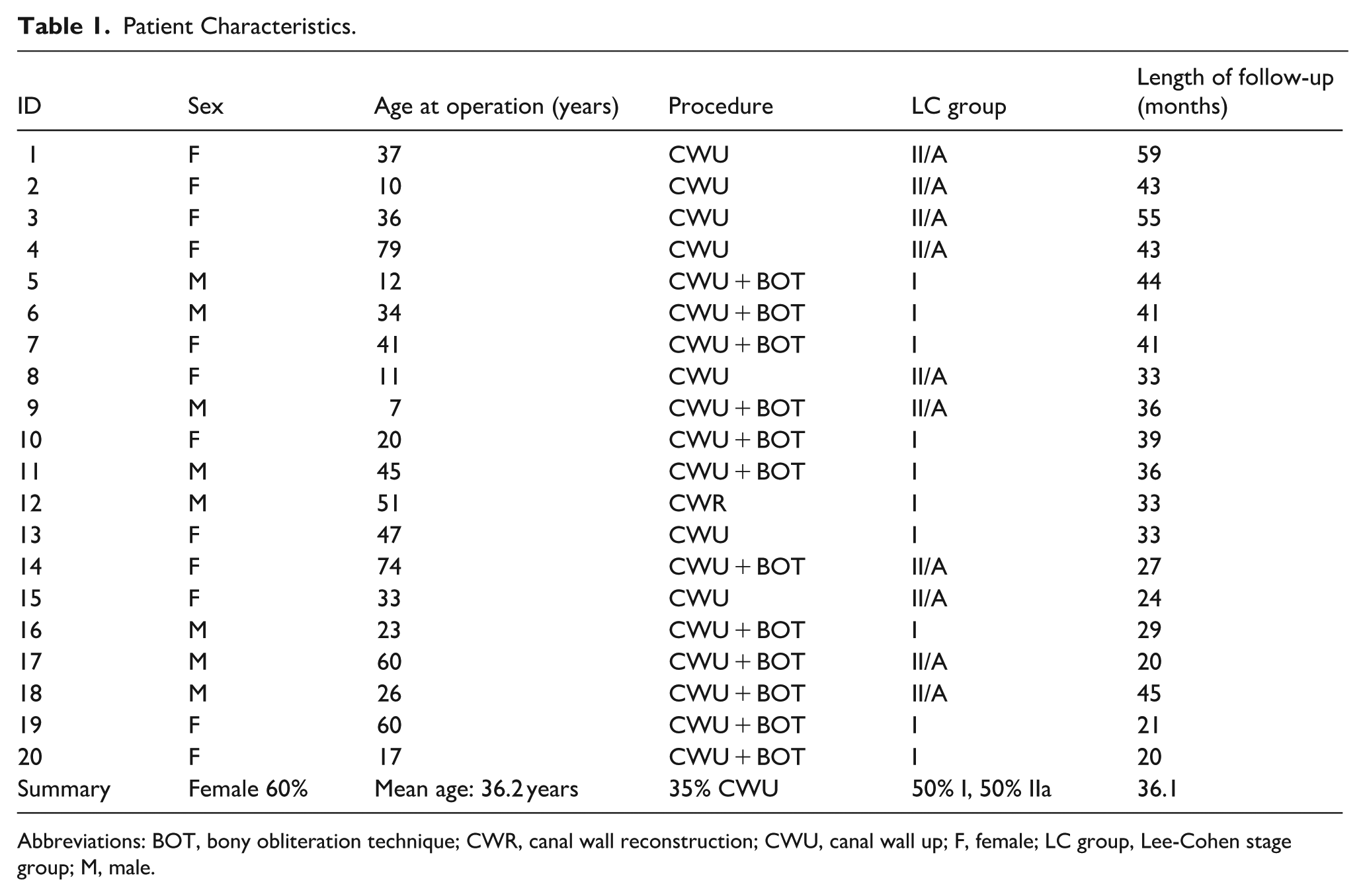
Abbreviations: BOT, bony obliteration technique; CWR, canal wall reconstruction; CWU, canal wall up; F, female; LC group, Lee-Cohen stage group; M, male.
Outcome Data
Primary Outcome: Endoscopic Intraoperative Second Look
Residual cholesteatoma was intraoperatively found by using angled endoscope in hidden areas during 10 procedures (50%), of which squamous epithelium was found in multiple locations in 1 patient. The most frequently affected site was the sinus tympani (6 cases), but cholesteatoma remnants were also discovered in the supratubal recess (3 cases), and in the attic (1 case; Table 2).
Intraoperative Residual Disease and Recidivism on Follow-Up.
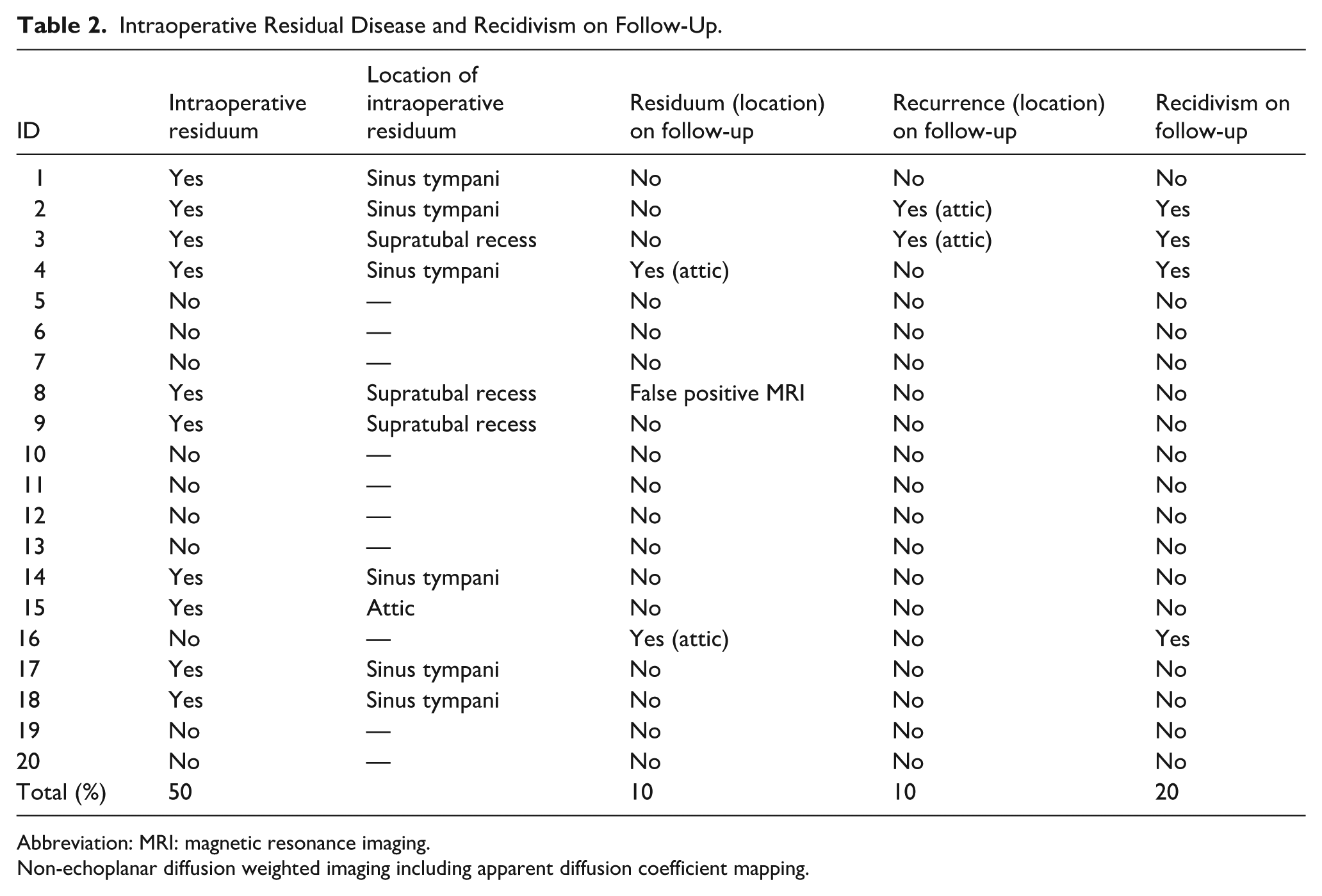
Abbreviation: MRI: magnetic resonance imaging.
Non-echoplanar diffusion weighted imaging including apparent diffusion coefficient mapping.
Primary Outcome: Recidivism
Dedicated MRI examinations showed residual cholesteatoma in 2 patients (10%; patient No. 4 at 35 months; patient No. 16 at 10 months). MRI results were indeterminate in 3 cases (15%; small diffusion restriction in the previously operated area but not specific for cholesteatoma usually next to materials like cartilage that decrease the reliability of MRI sequences and/or showed no growth on subsequent MRI scan). 20 Imaging follow-up provided 1 false-positive result. In this case, a second-look surgery was performed because a signal enhancement specific for residual cholesteatoma was registered with MRI in the mastoid antrum, but during surgical exploration, no intratympanic squamous epithelium was found. During otomicroscopic follow-up of a minimum of 12 months, 2 patients (10%) developed recurrent cholesteatoma, both in the attic (patient No. 2 at 41 months; patient No. 3 at 38 months). Through Kaplan-Meier survival analysis, the estimated mean recidivism-free survival was 49.9 months (range: 42.6-57.3 months [95% confidence interval]), cumulative (Figure 2).
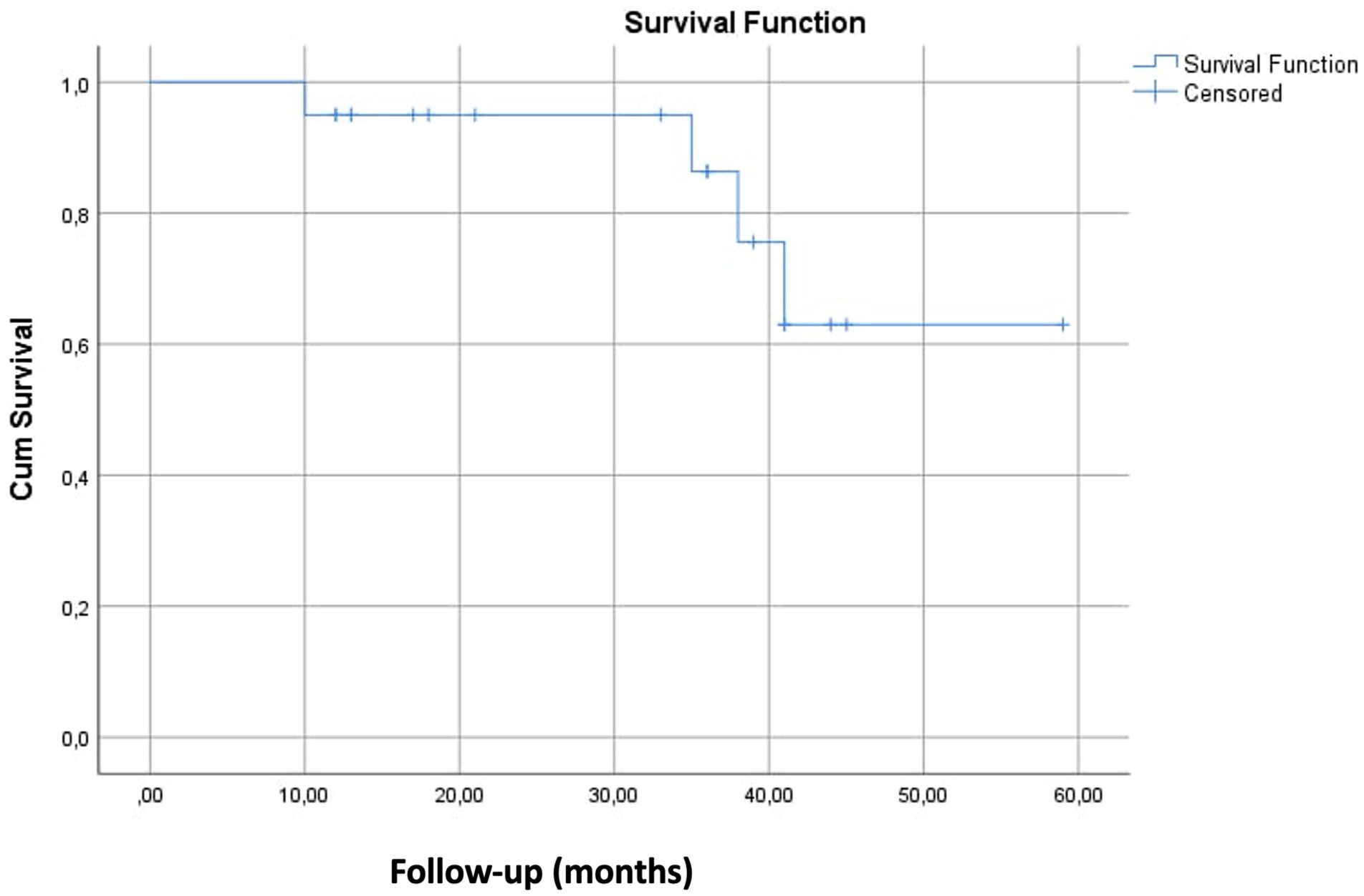
Kaplan-Meier survival analysis for recidivism-free survival.
Primary Outcome: Location Specific Residual Disease
None of the patients developed recurrent or residual cholesteatoma in the same area where intraoperative otoendoscopy revealed residual squamous epithelium during the first procedure. In both patients with residual cholesteatoma, lesions were discovered in the epitympanic space during MRI follow-up. In one of them, intraoperative endoscopy revealed no hidden cholesteatoma during the first surgery, while in the other case, residual epithelium was found during the operation in the sinus tympani via endoscopy. In 2 patients, recurrent cholesteatoma was found in the attic (Table 2).
Secondary Outcome: Hearing Results
In 5 cases, early postoperative, and in 3 cases, late postoperative audiology results were not available. The ossicular chain was intact during the surgery in only 1 case, and 19 patients needed ossicular reconstruction. Mean preoperative ABG was 30.7 ± 9.3 dB (range: 11.7-48.3 dB), mean early postoperative (6-12 weeks) ABG was 32.0 ± 12.8 dB (range: 11.7-51.7 dB), and mean late postoperative ABG was 28.6 ± 10.7 dB (range: 14-52.5 dB; Supplemental Figure 1). Mean ABG closure at 6 to 12 weeks was −1.7 ± 0.2 dB (range: −25.8-17.5 dB) while mean late (at least 12 months postoperatively) ABG closure was 1.9 ± 10.2 dB (range: −18.3-21.7 dB; Table 3, Figures 3 and 4). Mean ABG change at 1 year was not significant (P = .27; Figure 5). Raw data of all audiology results are available in the Supplemental Tables 1–3).
Summary of Pre- and Postoperative Audiology Results.
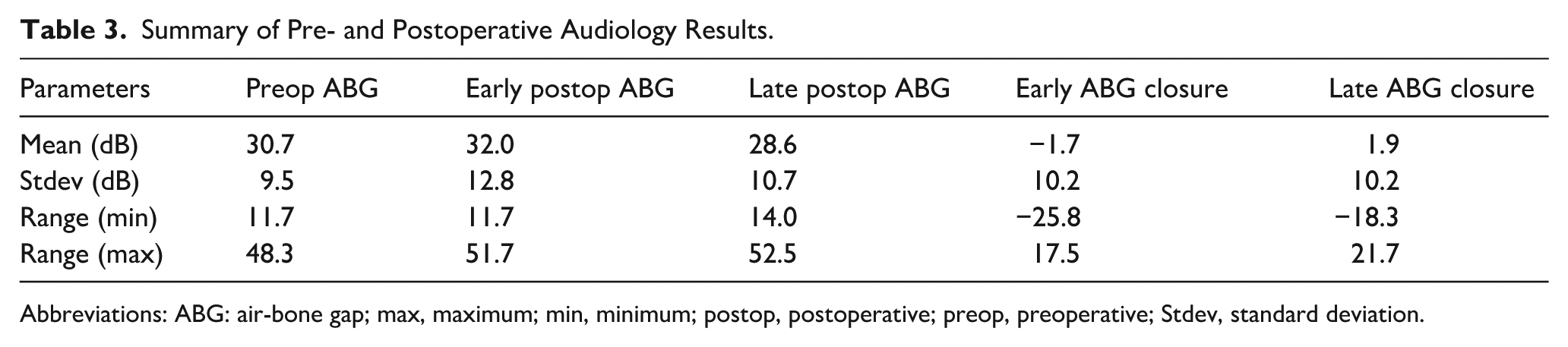
Abbreviations: ABG: air-bone gap; max, maximum; min, minimum; postop, postoperative; preop, preoperative; Stdev, standard deviation.
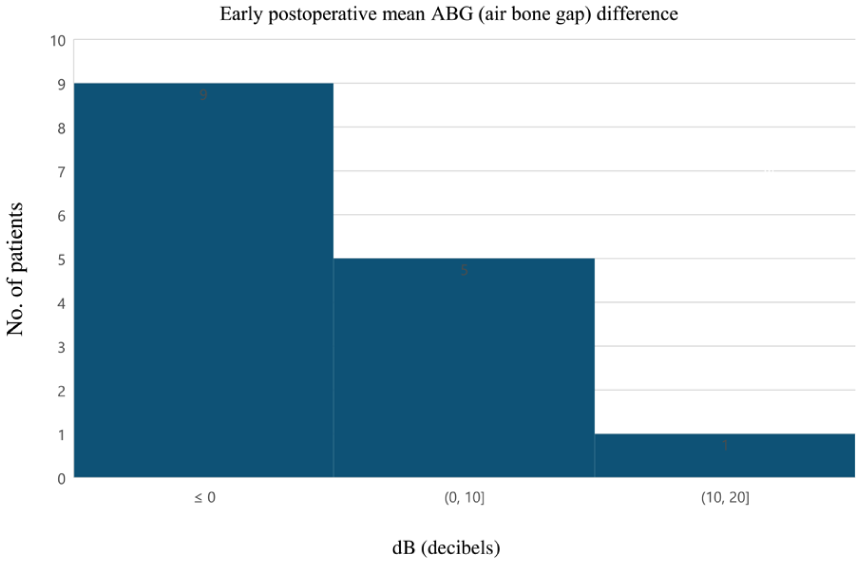
Hearing outcomes: Early postoperative mean ABG difference. ABG, air-bone gap.
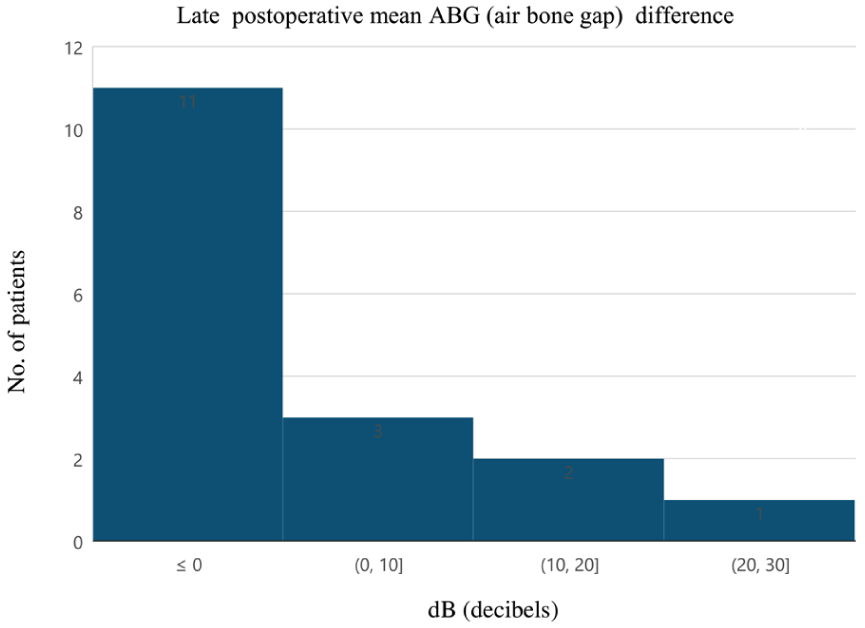
Hearing outcomes: Late postoperative mean ABG difference. ABG, air-bone gap.
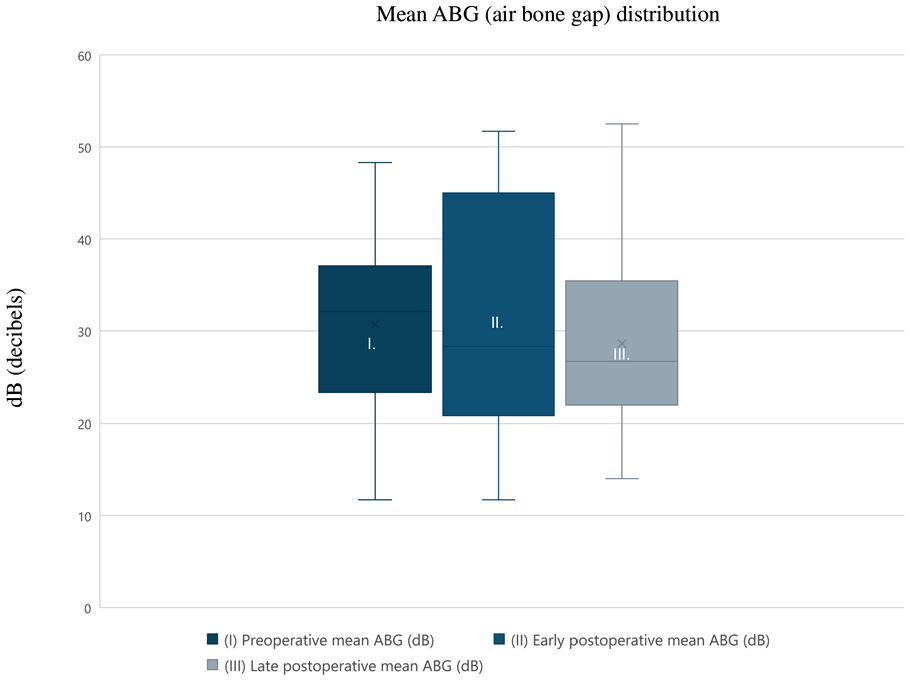
Mean ABG distribution. ABG, air-bone gap.
Discussion
Although preserving the posterior bony wall of the ear canal allows us to create a functional middle ear within its original dimensions, cholesteatoma removal can be challenging due to reduced intraoperative visibility of the disease, which may contribute cholesteatoma recidivism being as frequent as 30% to 40%. 21 In the past few decades, among other techniques, 22 otoendoscopy has become an important tool in cholesteatoma surgery, offering direct visual control in canal wall up surgery when removing squamous epithelium from the spaces that are difficult to access with a microscopic approach, leading to a decrease in residual disease23,24 and shorter operation time. 25 All dedicated studies concurred that hidden, remnant cholesteatoma tissue may be discovered using endoscopes after a seemingly complete microscopic evacuation; however, the rate of uncovered residual disease varies largely in the literature. Authors who recorded low incidences of residual cholesteatoma using endoscopy debate the procedure’s cost-effectiveness due to the increased operating time the combined technique brought about. 14 In comparison, other studies reported an intraoperative residual rate of 50% or above, 26 and a further systematic review concluded that the average rate of endoscopically uncovered cholesteatoma after microscopic surgery is 15.82%. 27 In our case series, we also detected residual cholesteatoma in half of the surgeries, which is in the higher range of reported rates. Our explanation for this phenomenon is that as endoscopic surveillance has become a common practice at our institution, we do not routinely perform posterior tympanotomy.
Although there are no prospective randomized studies providing a high level of evidence in the field, there are reports that unambiguously make a stand for intraoperative otoendoscopic control during microscopic CWU cholesteatoma surgery. In the highly cited study of El-Meselaty et al 26 residual disease was detected during second look surgery only in those cases, where previously intraoperative endoscopy was not used. In a large case series, application of endoscopy during CWU surgery resulted in a similar recidivism rate when compared to CWD tympanoplasties, which is a more effective approach to disease control than CWU surgery, 12 although it has significant functional and cosmetic disadvantages.28,29 In a retrospective cohort of 110 cholesteatoma patients, otoendoscopy helped uncover hidden squamous epithelium in 20 cases with only 3 patients suffering recidivism after at least 2 years of follow up. 6 In contrast to this, a retrospective chart review of 350 patients showed no influence of intraoperative otoendoscopy on recidivism, although with a low level of evidence. 30 A recent study by Hu et al 14 concluded that endoscopic control takes extra time, but does not lower residual disease rate, however, they always performed posterior tympanotomy, which also takes time and increases the risk of facial nerve injury. In our case series, although were had no opportunity to directly evaluate the effectiveness of otoendoscopy by comparing groups with and without endoscopic control, we found intraoperative residual cholesteatomas in 10 patients, and none of them developed residual disease during the follow-up at the same location, which also supports the utility of intraoperative endoscopic surveillance. Operating time and the learning curve of endoscopic techniques also would have been clinically relevant outcomes to pursue but due to the retrospective nature of our project we were not able to collect data on these in our setting.
According to the literature, dedicated MRI protocols (non-EPI DWI MRI with ADC mapping) show high sensitivity and specificity for detecting residual or recurrent cholesteatoma,31,32 increasingly rendering the previously obvious need for second-look surgery obsolete. Cholesteatomas under 3 mm however may not appear on these images or yield false negative results because the slice thickness of these imaging protocols are set between 2 and 4 mm. 33 Our cohort included 2 patients with radiologically and surgically confirmed residual cholesteatomas. However, some factors were shown to affect evaluation of imaging findings. Cartilage reconstruction, ossiculoplasty using cortical bone columella, movement artifacts, cholesterol granulomas or tympanosclerosis may result in false-positive, or indeterminate reports.20,34 A typical example for the latter is one of our patients with an intact novomembrane, and an aerated, healthy tympanic cavity, whose MRI 4 years postoperatively showed a 4 mm indeterminate, but cholesteatoma suspect lesion behind a cartilage-reconstructed scutum, however, during second-look surgery, no residual squamous tissue was found. Our cohort also included 3 patients with indeterminate diffusion inhibiting entities without growth on repeated imaging results. In cases with indeterminate results, characteristically lesions below the size of 5 mm, further imaging follow-up was proposed to increase specificity and decrease the number of unnecessary second-stage surgeries. 35
This project focuses on the role of intraoperative endoscopy in eradicating cholesteatoma, but we also evaluated our audiological results. Although there was a slight improvement in mean ABG after 1 year, the change was not significant. Our hypothesis for the modest results is that most of the cholesteatomas were extensive, which is a negative predictor for audiological outcomes, 36 and also nearly all of our patients needed ossiculoplasty as well.37,38
Our study is based on well-documented cases with contemporary follow-up using targeted MRI or second-look surgery within 1 year for all enrolled patients with individual patient data included. We could directly point to the advances of intraoperative otoendoscopic control both immediate and long term. The retrospective, 1-arm design of this study with a relatively low number of cases abates evidential value.
Conclusion
Based on our findings, the areas named in the EAONO/JOS consensus statement as difficult-to-access were indeed likely to contain residual squamous epithelium that was challenging to visualize with a microscope. Although our study was conducted in a single center with a limited sample size, which may affect generalizability, endoscopes proved to be excessively useful in our practice for removing these residues under direct visual control and ensuring low rates of residual disease in these areas.
Although multicenter prospective studies with large sample sizes are needed to determine the true efficiency of a combined approach in cholesteatoma surgery, our results based on a small patient cohort can be considered promising.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251383676 – Supplemental material for Endoscopic Control of Hard-to-Access Sites During Microscopic Cholesteatoma Surgery
Supplemental material, sj-docx-1-ear-10.1177_01455613251383676 for Endoscopic Control of Hard-to-Access Sites During Microscopic Cholesteatoma Surgery by Borbála Körmendy, Kata Illés, Bence Bajor, Judit Kálmán, Hunor Sükösd and Tamás Horváth in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
The authors would like to thank Bálint Liktor, MD Sr., Bálint Liktor, MD Jr., Zsuzsanna Várgedő, MD, and Zsuzsanna Keresztély, MD for their efforts in this project.
Ethical Considerations
Data was collected after receiving ethical approval for this study on September 12, 2023 (approval protocol ID: ETT TUKEB BM/24488-1/2023), and written participant consent was also collected.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All anonymized clinical data are shared either in the manuscript or the Supplemental Material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
