Abstract
Petrous apex cholesteatoma is a relatively rare and chronic aggressive disease that occurs in the petrous part of the temporal bone and can be classified as congenital or acquired. Due to the tendency of petrous cholesteatomas to cause bone destruction and invade important structures such as the facial nerve, cochlea, and semicircular canal, patients present mainly with severe hearing loss, progressive peripheral facial palsy, vertigo, etc. When the lesion invades the intracranial region or is infected, intracranial symptoms such as meningitis, lateral sinus thrombosis, and brain abscess may be present. We report a case of giant petrous cholesteatoma with Pseudomonas aeruginosa infection, with the aim of exploring the corresponding clinical treatment and surgical modalities to provide a reference for the clinical management of this type of disease.
Introduction
Petrous apex cholesteatoma is a group of aggressive diseases that occur in the temporal bone and are caused by the accumulation of keratinized squamous epithelium, which accounts for 4% to 9% of all apical lesions. 1 Petrous apex cholesteatoma can be divided into congenital and acquired types based on etiology. As petrous apex cholesteatoma is prone to cause bone destruction and invasion of important structures such as the facial nerve, cochlea, and hemianopia, patients primarily present with severe hearing loss, progressive peripheral facial palsy, vertigo, etc.2,3. When lesions are combined with a suppurative infection, this creates certain challenges in clinical management for the surgeon. Severe preoperative infection in the operative cavity of the middle ear can lead to diffuse growth of cholesteatoma along the tissue gap, bone suture, and mastoid airspace, which are highly susceptible to residual disease and recurrence. When this occurs, clinicians face the dilemma of controlling infection, preventing lesion recurrence, and choosing the right time for surgery.
Case Report
Summary of Medical History
The patient was a 39-year-old woman with a 30-year history of hearing loss and a 6-year history of facial palsy, accompanied by ear pain and otopyorrhea. In February 2019, she was diagnosed with petrous apex cholesteatoma combined with Pseudomonas aeruginosa infection, underwent subtotal petrosectomy (the cochlea was preserved) to control infection by improving ventilation and drainage, and then achieved complete excision of the lesion in the second stage. However, 4 months after the surgery, she again suffered from left otopyorrhea and pain, and we subsequently performed an expanded subtotal petrosectomy (the cochlea was preserved) and debridement surgery for the same purpose. However, eight months after the surgery, the patient again presented with intermittent and recurrent purulent ear discharge accompanied by ear pain and headache. There were no special circumstances with respect to past history, personal history, or family history.
Examination
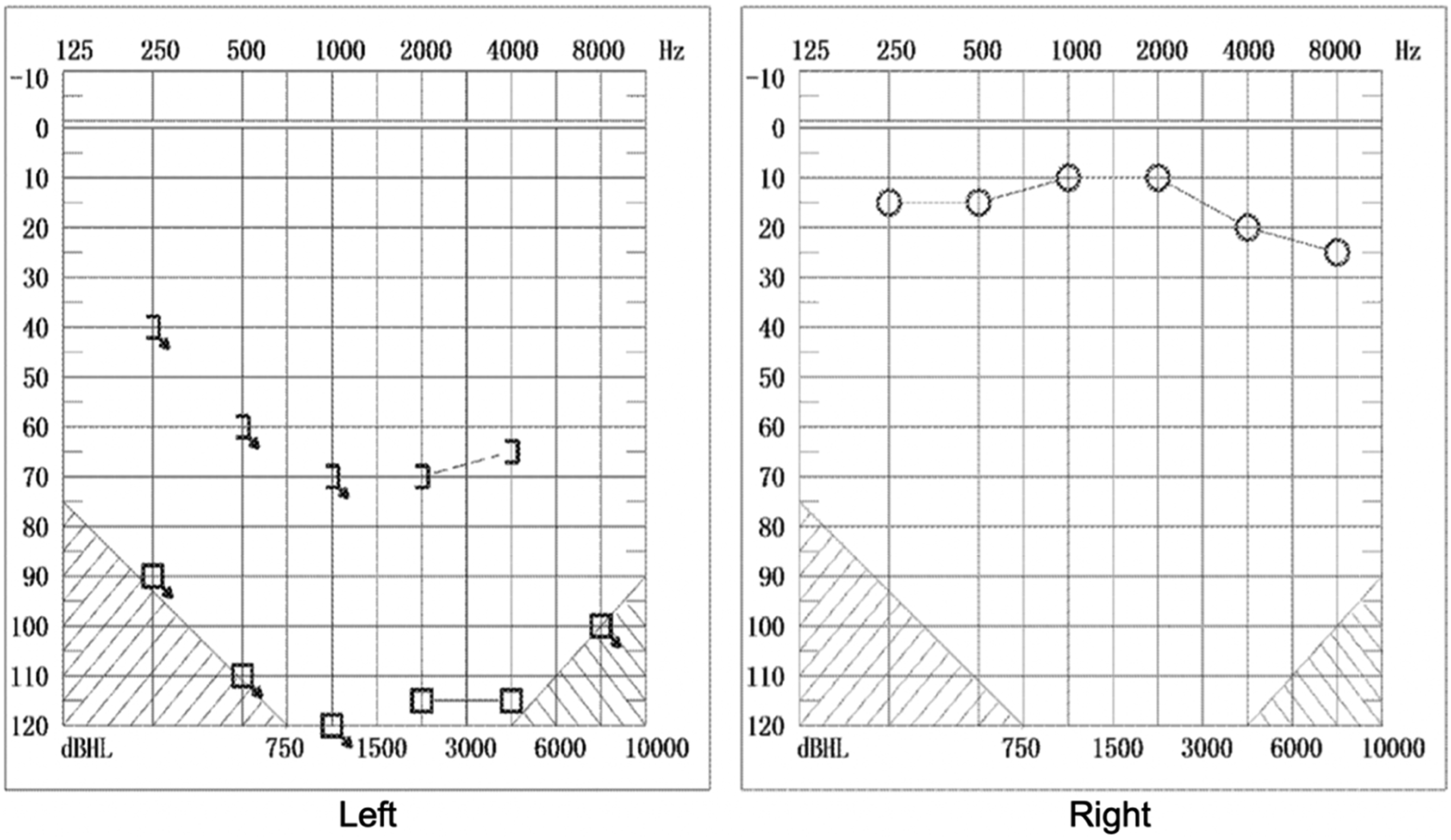
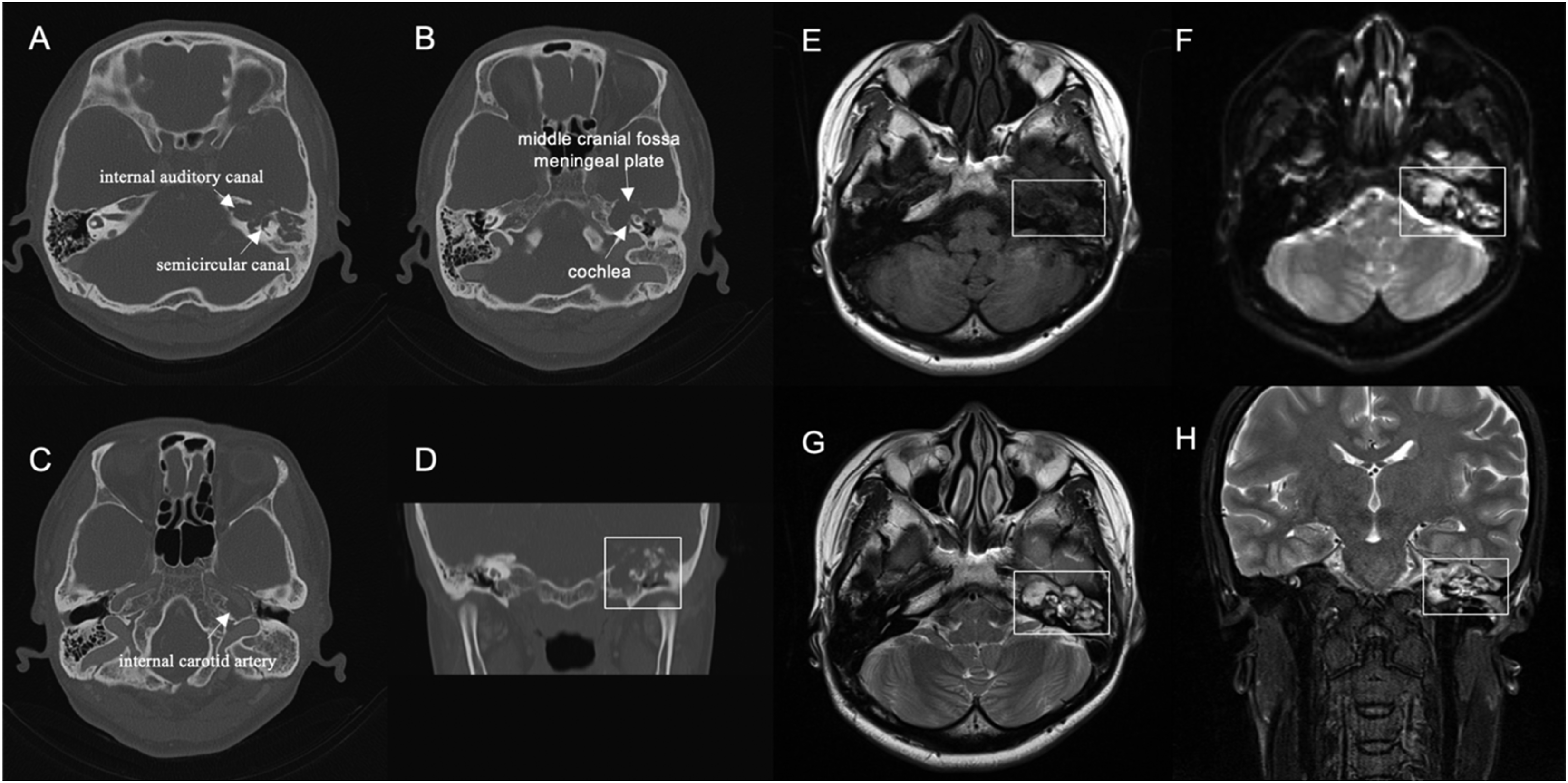
On examination, the patient had grade 5/6 facial function according to the House-Brackmann classification. Endoscopic examination of the ear revealed swelling of the external ear canal, as well as purulent discharge, as shown in Figure 1. The audiogram illustrated profound sensorineural hearing loss in the left ear (Figure 2). The bacteriological culture of the secretion suggested Pseudomonas aeruginosa infection. The patient underwent imaging before the three procedures, including temporal bone enhancement CT and temporal bone magnetic resonance imaging. Temporal bone CT suggested an irregular soft tissue density shadow visible in the left tympanum and petrous apex, and erosion of the horizontal segment of the internal carotid artery, auditory bone chain, semicircular canal, cochlea, and meningeal plate of the middle cranial fossa, which did not show significant improvement. On temporal bone magnetic resonance imaging, there were multiple shadows of short T1 and long T2 signals with high DWI signals in the left middle ear and temporal bone. The lesion was closely related to the cochlea, semicircular canal, internal auditory canal, and internal carotid artery, and the local demarcation was not clear. The preoperative imaging examinations are presented in Figure 3. Endoscopic examination: swollen external ear canal, perforated eardrum, and small amount of purulent discharge. Audiogram: profound sensorineural hearing loss in the left ear. First preoperative image, A-D: temporal bone enhancement CT; E-H: temporal bone magnetic resonance imaging. A: The lesion is located in the patrous apex, surrounding the internal auditory canal, with bony discontinuities in the semicircular canal. B: Bony discontinuities in the cochlea and meningeal plate of the middle cranial fossa. C: The lesion erosion of the horizontal segment of the internal carotid artery. D: Coronal view, the lesion is extensive, no enhancement was seen. E-F: Short signal in T1-weighted image, high signal in DWI image. G: T2-weighted image, the lesion was closely related to the cochlea, the semicircular canal, and the internal auditory canal. H: Coronal view of MRI, the local demarcation was not clear.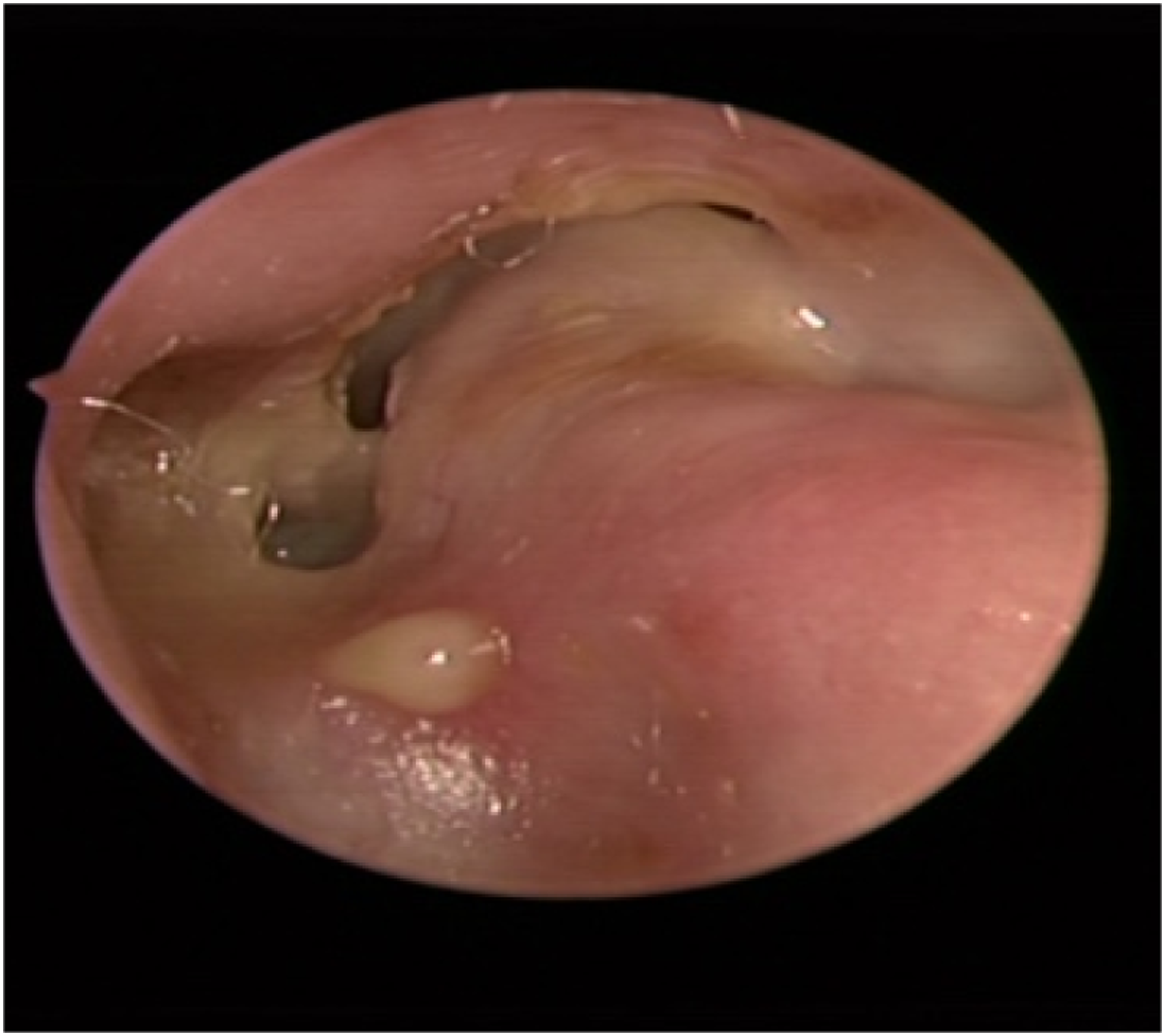
Treatment Process
The patient had undergone two subtotal petrosectomies and still had recurrent postoperative ear discharge with bacterial culture suggesting Pseudomonas aeruginosa infection. Preoperative imaging suggested that lesions surrounded the combination of infection of the cochlea, labyrinth, and internal auditory canal, which led to the obstruction of ventilation and drainage in the petrous region of the temporal bone, making complete control of the infection difficult. Surgery was required to remove the lesion and improve ventilation and drainage before the infection could be further controlled. However, when radical surgery is performed, due to the large extent of the lesion involving the labyrinth, cochlea, and internal auditory canal, leakage of intraoperative cerebrospinal fluid must occur, resulting in a high risk of intracranial infection and even life-threatening complications. Therefore, a meticulous treatment plan must be developed before surgery, and a suitable time for surgery must be chosen.
Multidisciplinary preoperative treatment (MDT) was performed, and after discussion, it was recommended that surgery be performed to remove the lesion and control the infection, but adequate preoperative preparation was needed to reduce the patient's infection load, thus reducing the risk of intracranial infection. Therefore, the patient underwent a long period of repeated debridement and drainage and also received adequate anti-infection treatment, monitoring of hemogram and inflammation index, and strengthening of nutritional input prior to surgery.
According to the schedule, we performed subtotal petrosectomy surgery (without preserving the cochlea) plus transposition and filling of the temporal muscle and the flap of the occipital subcutaneous muscle tissue flap. The cochlea and semicircular canal were drilled away to fully expose the cholesteatoma. We repeatedly flushed the surgical cavity with complex iodine and hydrogen peroxide and then carefully removed the cholesteatoma around the internal auditory canal and the petrous apex. After the cholesteatoma was cleared, membranous closure of the internal auditory canal and a small amount of cerebrospinal fluid leakage were observed; the leakage was halted by electrocoagulation. Finally, transposition of the temporal muscle and the flap of the occipital subcutaneous muscle tissue was performed to fill the cavity. Intraoperative views are shown in Figure 4. A1: After the cochlea and semicircular canal were drilled away, the cholesteatoma was around the internal auditory canal (IAC). A2: The operative cavity after complete removal of the lesion, with a little cerebrospinal fluid leakage. ICA: internal carotid artery; IAC: internal auditory canal; JB: jugular bulb; MP: meningeal plate; FN: facial nerve.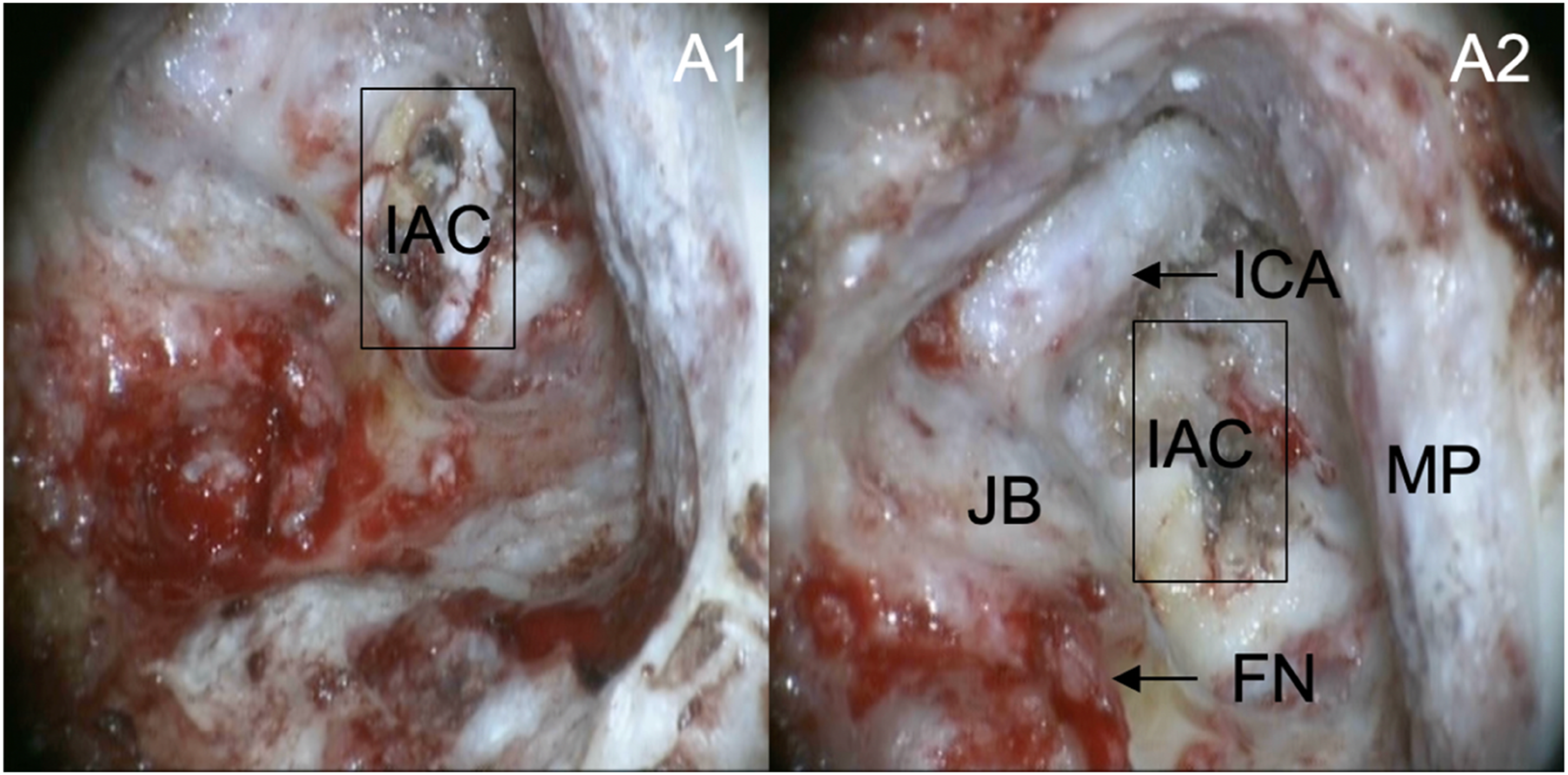
The main purpose of removing the labyrinth and cochlea is to establish sufficient ventilation and drainage in the apical region to control infection. Before the cochlea and labyrinth are removed and the internal auditory lesion is removed, the surgical cavity should be fully flushed with iodophor and hydrogen peroxide, which can also reduce the risk of intracranial infection to some extent. The external auditory canal is also closed due to intraoperative leakage of cerebrospinal fluid to prevent the occurrence of intracranial infections. For the infected operative cavity, a tipped muscle tissue flap should be used to fill the cavity instead of abdominal fat because the tipped muscle tissue flap is enriched with blood supply, has a strong ability to resist infection, and can easily survive. In this patient who exhibited preoperative facial palsy, intraoperative examination revealed severe adhesions of the facial nerve to the lesion and scar of previous surgeries, as well as the absence of the horizontal segment of the facial nerve. Thus, the facial nerve was excised and marked at the severed end to prepare for subsequent facial nerve reconstruction.
Follow-Up
The patient was followed for 1.5 years after surgery, and there was no recurrence of the lesion or infection. The postoperative temporal bone magnetic resonance imaging is illustrated in Figure 5. Temporal bone MRI: Muscle tissue flap filling is visible in the operative cavity, and there was no recurrence of the lesion or infection. A: Axial view. B: Coronal view. C: DWI.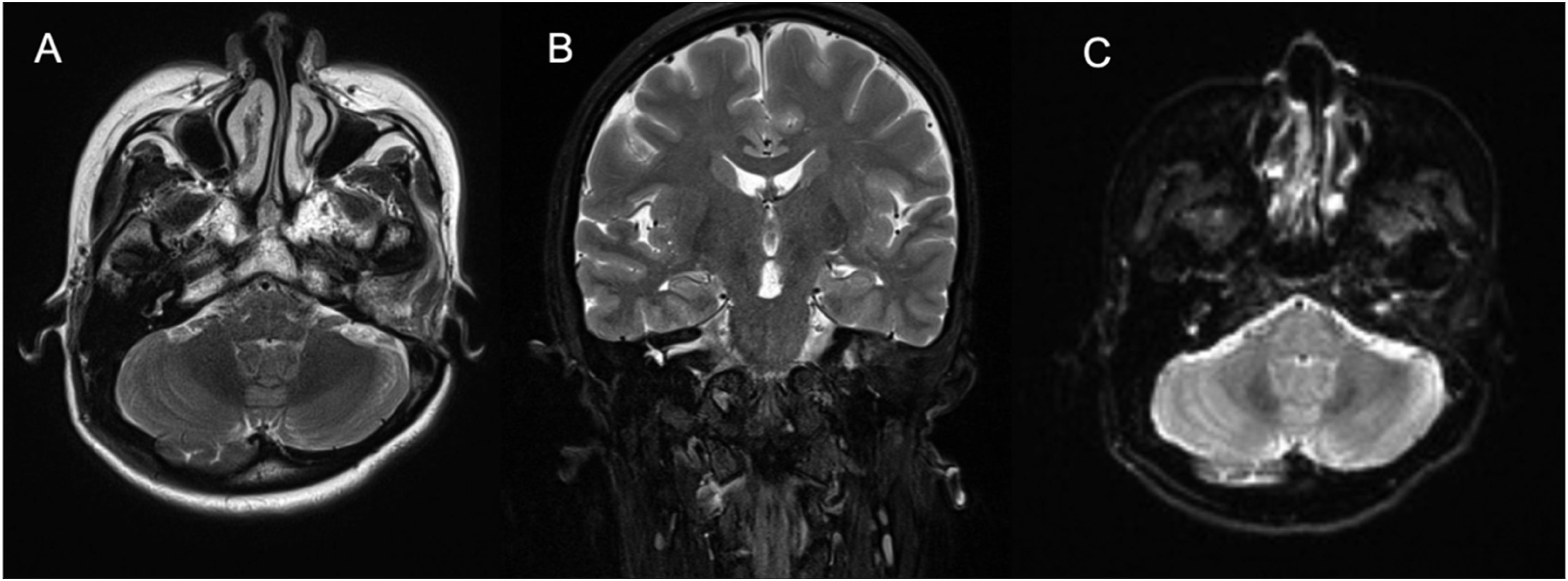
We present a case of extensive petrous apex cholesteatoma involving the cochlea, facial nerve, carotid artery, and internal auditory canal in combination with infection. Because petrous apex cholesteatoma is prone to bone destruction and invasion of important structures, such as the facial nerve, cochlea, and semicircular canal, patients present primarily with severe hearing loss, progressive peripheral facial palsy, vertigo, etc. The patient presented preoperatively with severe facial paralysis and profound hearing loss due to erosion of the lesion. Because the clinical manifestation of apical cholesteatoma is not specific, the clinical diagnosis is usually based on its typical imaging features. A high DWI signal is an important feature in distinguishing cholesteatoma from other apical lesions. 4 Treatment options for petrous apex cholesteatoma remain controversial because cases are relatively rare in clinical practice. Several surgical approaches for petrous apex cholesteatoma are possible depending on the patient's preoperative hearing and degree of facial palsy, as well as the location and extent of lesion invasion. The literature suggests that for petrous apex cholesteatoma located in the labyrinth, a limited middle cranial fossa approach can be used to facilitate the preservation of hearing and facial nerve function, although this approach is prone to cerebrospinal fluid leakage and poor vision due to exposure. When cholesteatoma affects vestibular structures and the cochlea, subtotal petrosectomy can fully expose the lesion and facilitate its removal.5 -7 In contrast, when the lesion invades the structures surrounding the internal auditory canal, there is a general consensus that radical resection of the petrous apex cholesteatoma can be achieved by sacrificing the ear capsule.8,9 Currently, endoscopic and combined microscopic-endoscopic surgery is gaining increasing attention and achieving good results. 10 Cases of severe preoperative infection in the operative cavity of the middle ear can lead to diffuse growth of cholesteatoma along the tissue gap, bone suture, and mastoid airspace, which are highly susceptible to residual disease and recurrence. According to Prof. Fisch’s experience, in patients with more severe infection or suspected residual lesions, the external auditory canal can be temporarily closed to facilitate drainage and control infection. 11 For surgical cavities at risk of infection, a tipped muscle tissue flap instead of abdominal fat can be used for filling. Based on our clinical experience and the relevant literature, temporal and suboccipital muscle tissue flaps are suitable for operative cavities with a risk of infection due to their rich blood supply and resistance to infection. When petrous apex cholesteatoma is combined with infection, surgical establishment of ventilation and drainage, as well as removal of the cholesteatoma lesion, is an effective way to address recurrent infection episodes, but surgeries leading to cerebrospinal fluid leakage increase the risk of intracranial infection. Once intracranial infection occurs, the consequences are catastrophic; thus, adequate preoperative preparation to reduce the infection load and selection of the appropriate time for surgery are crucial, and preoperative multidisciplinary consultation plays an important role. In addition, careful intraoperative manipulation, protection of the brain meninges, adequate irrigation, use of tipped muscle tissue flaps for tamponade, and adequate preoperative and postoperative courses of anti-infective therapy are of great importance for such stained operative cavities.
Conclusions
This case presents a rare giant cholesteatoma with extensive destruction of the otic capsule, the internal auditory canal, and the carotid artery combined with Pseudomonas aeruginosa infection. Patients with a petrous apex cholesteatoma combined with severe infection are highly susceptible to residual disease and recurrence. Surgical establishment of ventilation and drainage, as well as removal of the cholesteatoma lesion, is an effective way to control infection. Therefore, radical resection of cholesteatoma and control of infection can be achieved by sacrificing the ear capsule, especially in patients with poor hearing. However, if surgery is bound to cause cerebrospinal fluid leakage, adequate preoperative preparation and a suitable time for surgery are necessary.
Footnotes
Acknowledgments
Author Zhiqiang Gao is the corresponding author. Thanks for the opinions about the revision from Dr Yang Zhao.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fundamental Research Funds for the Center Universities (3332021010) and Beijing Union Medical College Hospital Sinking Research Fund (ZC201901017)
Consent to Participate Patient Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Consent for Publication
All of the authors provided consent for the publication.
