Abstract
Background:
This systematic review aims to synthesize evidence on primary squamous cell carcinoma (PSCC) of the submandibular gland (SMG), an exceptionally rare malignancy (<1% of salivary gland tumors, ~2% of submandibular malignancies). Key objectives include characterizing its epidemiology, diagnostic challenges, treatment paradigms, and prognostic outcomes to enhance clinical management.
Methods:
Literature search (PubMed, Web of Science, Scopus, Google Scholar) from inception through December 2024 identified English-language studies using MeSH terms (“Carcinoma, Squamous Cell” AND “Submandibular Gland”). After deduplication, 18 full texts were screened; 8 studies (case reports/series, 1992-2023) met the inclusion criteria. Two reviewers independently extracted data on epidemiology, clinical features, management, and outcomes.
Results:
PSCC of the SMG demonstrates a strong male predominance (M:F = 3:1) and a mean age of 57.1 years, with prior cervical radiotherapy representing a key risk factor. Clinically, all patients present with submandibular swelling, though only 25% report pain; symptom duration averages 3.9 months, frequently leading to advanced-stage diagnosis. Definitive diagnosis requires rigorous exclusion of metastatic squamous cell carcinoma and histological mimics, necessitating ultrasound-guided fine-needle aspiration cytology and cross-sectional imaging (CT/MRI). Management universally involves SMG resection, with 50% undergoing neck dissection (supraomohyoid or modified radical). Adjuvant therapy was administered to 75% of patients (radiotherapy: 50%; concurrent chemoradiotherapy [CCRT]: 25%). Outcomes reveal a 62.5% survival rate and 37.5% mortality, with 37.5% developing lung metastases. Limited follow-up data highlights both durable remission (no evidence of disease at 2 years) and aggressive progression, including 1 case of early contralateral recurrence 1 month post-CCRT.
Conclusion:
PSCC of the SMG is an aggressive malignancy requiring multimodal management. Surgical resection with adjuvant radiotherapy/chemotherapy is common, yet high recurrence rates and metastatic risk persist. Diagnostic rigor remains critical to exclude mimics and metastases. Future studies should establish standardized protocols integrating molecular targeting (eg, EGFR/Wnt pathways).
Keywords
Introduction
Primary squamous cell carcinoma (PSCC) of the submandibular gland (SMG) is an exceedingly rare malignancy, representing <1% of all salivary gland tumors and only about 2% of malignant submandibular neoplasms, rendering it exceptionally uncommon.1 -3 It exhibits a marked male predominance and typically presents between the sixth and eighth decades, with a mean age of 61 to 68 years at diagnosis. 1 The primary etiological factor is prior cervical radiotherapy (eg, for head/neck neoplasms or benign conditions), with a median latency of 15.5 years. 4 Genetic alterations activating Wnt/β-catenin are implicated in pathogenesis. 7 Clinically, PSCC usually manifests as a painful, fixed swelling with symptoms lasting <1 year, often leading to an advanced-stage diagnosis due to minimal early signs.8,9 Diagnosis is challenging, requiring rigorous exclusion of metastatic squamous cell carcinoma (SCC; especially from the upper aerodigestive tract or skin), direct extension, and histological mimics like mucoepidermoid carcinoma (periodic acid-Schiff [PAS] negativity) or lymphoepithelial carcinoma (Epstein-Barr virus [EBV] negativity).5,11 Definitive diagnosis relies on characteristic histomorphology and cytonuclear atypia, often necessitating ultrasound-guided fine-needle aspiration cytology (FNAC), cross-sectional imaging (CT/MRI), and sometimes excisional biopsy to avoid pitfalls such as post-radiation metaplasia.5,6,10 Surgical resection is the primary treatment, but exclusive surgery carries high recurrence rates. 11 Consequently, multimodal management, including radical SMG resection combined with ipsilateral neck dissection (due to ~56% nodal involvement risk), and consideration of adjuvant radiotherapy/chemotherapy is common, though the benefit of adjuvant treatment remains debated.12 -14 Metastatic disease portends a poor prognosis with chemoresistance, prompting investigation into EGFR and Wnt/β-catenin targeted therapies. 15 This article shares a collection of all case reports to enhance clinical suspicion and management.
Materials and Methods
This systematic review was conducted with accordance with PRISMA 2020 guidelines. A comprehensive literature search of PubMed, Web of Science, Scopus, and Google Scholar databases from inception through December 2024 was performed using the MeSH terms (“Carcinoma, Squamous Cell” [Mesh] AND “Submandibular Gland” [Mesh]) supplemented by Boolean operators and keywords, restricted to English-language publications. After initial retrieval of 192 records and duplicate removal, 2 independent reviewers screened titles/abstracts and subsequently assessed 22 full-text articles against predetermined eligibility criteria, which included case reports, case series, and observational studies of PSCC exclusively originating in the SMG, while excluding non-English articles, metastatic SCC to SMG, secondary SMG involvement from oral SCC, and non-original research. Discrepancies were resolved through consensus and third-reviewer consultation, yielding 8 included studies documented in the PRISMA flow diagram (Figure 1). Data extraction was conducted using a standardized Microsoft Word template capturing epidemiology, clinical features, diagnostics, management, and outcomes, with dual independent extraction and cross-verification; study quality was assessed using JBI critical appraisal tools for case reports/series, with full methodology details including complete search strategies and quality assessments. The data extraction focused on epidemiology, etiology, clinical manifestations, diagnostic modalities, management, and outcomes.
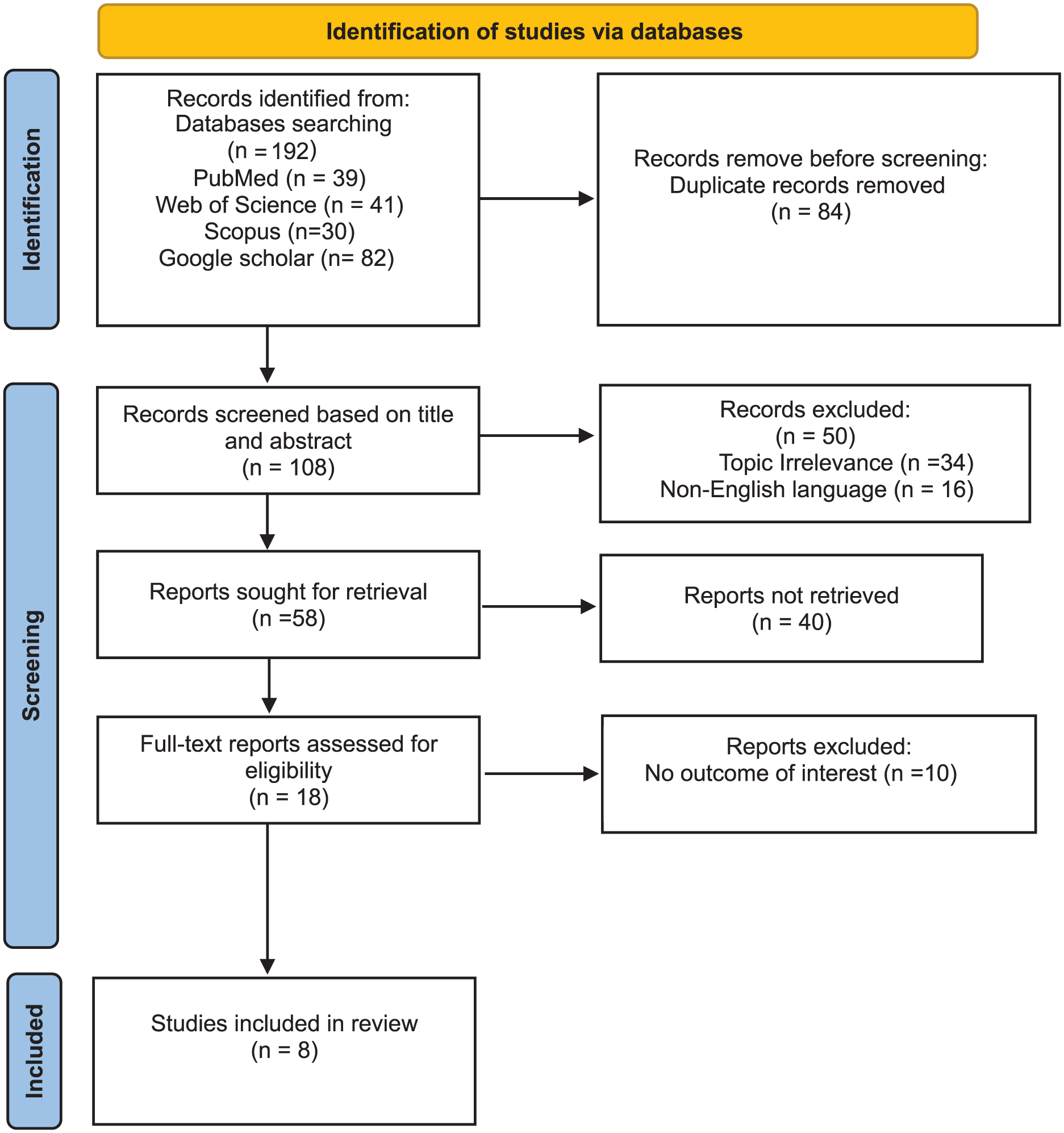
PRISMA flowchart for selection of the articles in the systematic review.
Results
Our systematic review identified 8 cases of primary SMG PSCC, revealing key clinical patterns: The cohort was summarized in Table 1, which demonstrated a strong male predominance with a male-to-female ratio = 3:1. The mean age of 57.1 years (range 11-82 years). Symptom duration before diagnosis averaged 3.9 months (range 1.5-8 months), with all patients (100%) presenting with submandibular swelling, but only 25% (2/8) explicitly reported pain.
Summarized Characteristics of All Reported Cases in the English Literature, Including Demographic, Physical Exam, Management, Outcome, and Follow-Up.
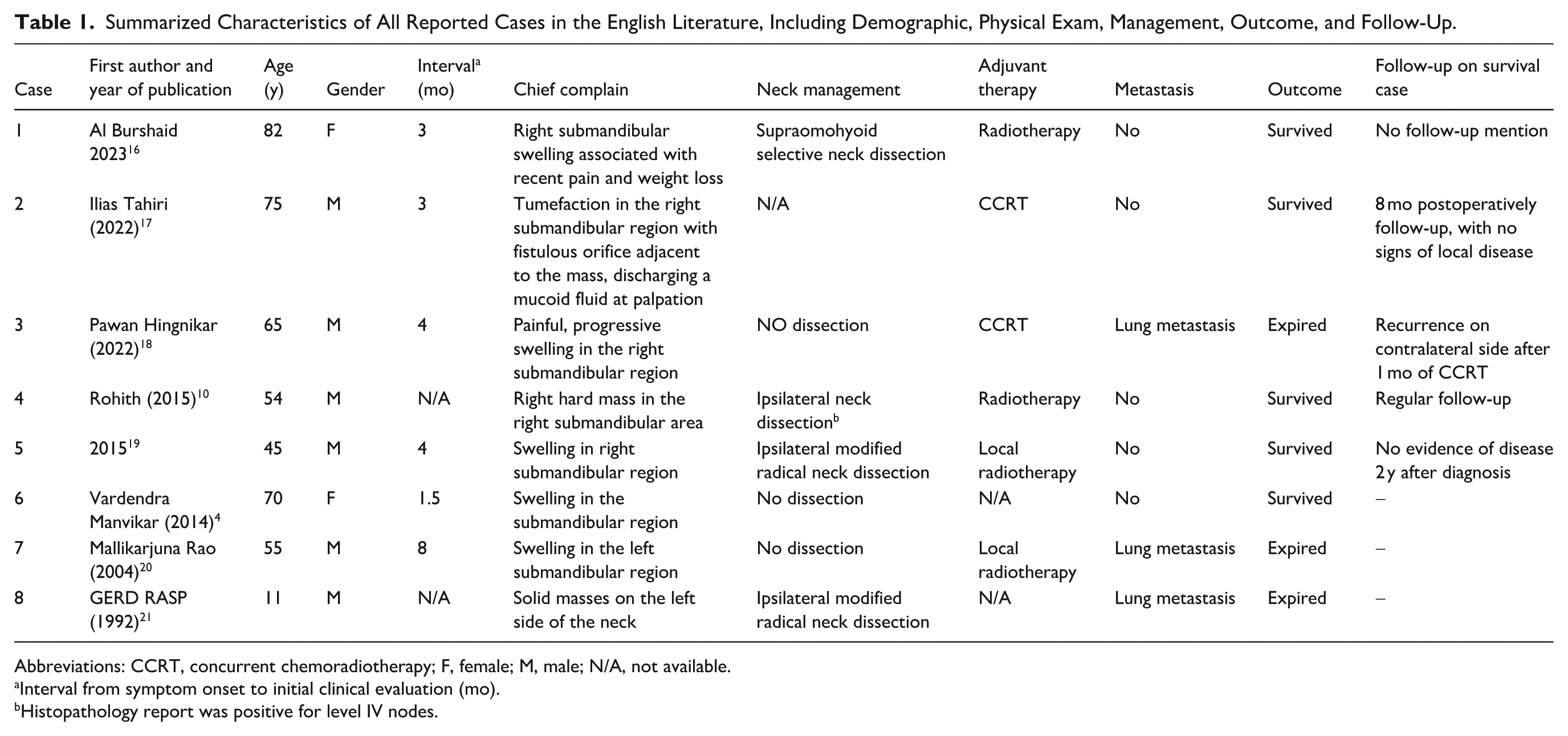
Abbreviations: CCRT, concurrent chemoradiotherapy; F, female; M, male; N/A, not available.
Interval from symptom onset to initial clinical evaluation (mo).
Histopathology report was positive for level IV nodes.
Management universally involved surgical resection of the SMG, with neck dissection was performed in 50% (4/8) of patients. The surgical approaches comprised 1 case of supraomohyoid selective neck dissection (levels I-III), 1 case of unspecified ipsilateral neck dissection, and 2 cases of ipsilateral modified radical neck dissection. Neck dissection was not performed in 37.5% (3/8) of cases, while management details were undocumented (N/A) in the remaining 12.5% (1/8). The decision to pursue dissection generally aligned with clinical or histopathological evidence of nodal involvement, though standardized selection criteria were not consistently reported across studies.
Adjuvant therapy was administered to the majority of patients (6 out of 8 cases, 75%). Radiotherapy was the most common modality, used in 4 cases (50%), including 1 specified as local radiotherapy. Concurrent chemoradiotherapy (CCRT) was delivered in 2 cases (25%). The remaining 2 cases (25%) received no adjuvant therapy (N/A).
Metastasis was identified in 3 out of 8 cases (37.5%). All metastatic cases involved the lungs. The remaining 5 cases (62.5%) showed no evidence of metastasis.
In this review, 5 survived (62.5%) while 3 expired (37.5%), indicating a predominant survival outcome overall. This result reflects inherent variability in patient trajectories, although the absence of additional clinical details, such as demographics, specific interventions, or follow-up duration, limits further contextual analysis of the contributing factors. To derive actionable insights, future studies should correlate these survival and mortality outcomes with specific variables like patient age, underlying comorbidities, or treatment protocols.
Detailed follow-up information was available for only a subset of the cases, with substantive data limited to 3 of the 8 patients; the remaining 5 entries lacked specific details, indicating either “no follow-up mention,” unspecified “regular follow-up,” or “none.” Among the patients with documented follow-up outcomes, 1 surviving patient demonstrated no evidence of disease at 2 years post-diagnosis, signifying long-term remission, while another was disease-free at 8 months postoperatively with no local recurrence. Conversely, 1 surviving patient experienced early treatment failure, with recurrence on the contralateral side just 1 month after completing CCRT. The significant absence of detailed follow-up data for the majority of patients (5/8) substantially limits comprehensive analysis of disease progression, recurrence patterns, or long-term outcomes beyond initial survival status, though the available cases highlight both durable remission and rapid recurrence.
Discussion
Definition
PSCC constitutes an exceptionally rare malignancy within the major salivary glands, representing <1% of all salivary gland tumors, with the parotid gland being the predominantly affected site. 1 The reported incidence of PSCC among all major salivary gland malignancies demonstrates considerable variation, ranging from 0.9% to 4.7%. 2
Epidemiology
Within site-specific malignancies, PSCC represents ~9% of malignant parotid tumors while accounting for only 2% of malignant submandibular tumors. This malignancy exhibits a notable rarity in the submandibular salivary gland, constituting an exceptionally uncommon clinical entity. PSCC of the major salivary glands demonstrates a pronounced male predilection and typically presents between the sixth and eighth decades of life, with a mean diagnostic age range of 61 to 68 years.1 -3
Etiologic Assessment
The predominant etiological factor is prior radiation exposure, frequently involving cervical radiotherapy for conditions such as acne, thymic enlargement, thyroid disorders, or head/neck neoplasms. This malignancy exhibits a median latency period of 15.5 years (range: 7-32 years) between irradiation and tumor development. 4 Another risk factor demonstrated was genetic alteration with activation of Wnt/β-catenin, which is linked to the development of aggressive SCC of the salivary glands. 7 When SCC is identified in the SMG, rigorous exclusion of a primary lesion elsewhere is mandatory; in the absence of another primary, the tumor is classified as a primary SMG carcinoma. Early diagnosis remains a critical prognostic determinant. 6
Clinical Presentation
Clinically, these tumors typically present as painful swellings with a symptom duration under 1 year and may exhibit fixation to skin or deep tissues, a presentation often attributable to minimal early symptoms beyond submandibular swelling, leading to frequent diagnosis at advanced stages.8,9
Diagnostic Strategy
The radiological investigations are necessary due to the fact that the identification of the site of origin is often not obvious at the time of presentation. Diagnostic workup necessitates ultrasound-guided FNAC alongside cross-sectional imaging (CT and MRI) for accurate lesion characterization, staging, and therapeutic planning; preoperative fine-needle aspiration (FNA) biopsy is strongly recommended to establish reliable histology. 6 Histopathological diagnosis of PSCC in the SMG poses significant challenges, particularly due to the diagnostic pitfall of ductal squamous metaplasia in patients with prior head and neck cancer treatment; FNA biopsies from irradiated fields frequently yield squamous cells in fluid samples, risking false interpretation and underscoring the necessity of excisional biopsy with comprehensive pathological examination for definitive confirmation. 10 Key discriminative features include cytonuclear atypia organized in islands and cords (absent in metaplasia), alongside round uniform metaplastic ducts, acinar atrophy, and infiltrative inflammation—findings highly suggestive of PSCC. 10 Definitive diagnosis is further supported by microscopic features including polygonal cells with prominent nucleoli, abundant tonofilaments, and well-developed desmosomal junctions; critically, the presence of any concurrent SCC in the upper aerodigestive tract or skin definitively excludes a primary submandibular origin. PSCC of the SMG remains a diagnosis of exclusion, mandating rigorous elimination of key differentials: (1) metastasis from distant primaries, (2) mucoepidermoid carcinoma, and (3) direct extension of cutaneous SCC. Histopathological distinction between PSCC and metastatic SCC is particularly challenging, as both present as keratinizing lesions without primary tumor-specific features. 5 Consequently, critical differentials require targeted exclusion methods: mucoepidermoid carcinoma is ruled out by the absence of mucin-producing cells/keratinization with PAS negativity, while lymphoepithelial carcinoma is excluded via negative EBV immunohistochemistry. 11 PET scan is not routinely used, as it is reserved for cases with diagnostic difficulty. 6 In this article, we aim to share our experience with such an entity, to raise clinical suspicion while facing similar presentations.
While FNAC is emphasized, its limitations in distinguishing radiation-induced metaplasia from true malignancy in previously irradiated patients warrant explicit discussion. Core needle biopsy may offer superior architectural assessment and reduce false positives by allowing evaluation of tissue invasion and stromal reaction, features often ambiguous on cytology alone. Recommending core biopsy as a complementary tool in high-risk patients (prior radiation history) could mitigate diagnostic delays, particularly when excisional biopsy is deferred due to comorbidities.
Management
Treatment Paradigm for SMG PSCC
While specific guidelines for SMG PSCC are lacking, surgical resection constitutes the primary therapeutic modality. Exclusive surgery, however, correlates with substantial recurrence rates (~50% parotid, ~60% submandibular), necessitating multimodal management to mitigate postoperative failure. This entails radical surgery combined with adjuvant radiotherapy and chemotherapy. For parotid PSCC without perineural invasion, facial nerve-sparing total parotidectomy achieves durable local control.11 -13
Neck Dissection
Given frequent nodal involvement (regions I-III), ipsilateral modified radical neck dissection addresses occult metastases undetectable by MRI; bilateral dissection is reserved for clinically palpable bilateral adenopathy. 13 Neck dissection in addition to primary tumor resection is necessary due to the high risk of nodal involvement, which has been reported to be as high as 56% for SCC of the major salivary glands. 12 Treatment includes resection of the involved submandibular salivary gland with concurrent neck dissection, if cervical metastases are detected or suspected. Follow‑up information in the literature showed regional recurrence occurring in 66% of cases within 1 year. There appears to be some evidence that post‑operative radiotherapy may help to control local recurrence. 12
Given the 56% nodal involvement risk, the absence of standardized criteria for elective neck dissection (END) in clinically N0 patients remains a critical dilemma. Our finding that 50% underwent neck dissection—primarily driven by clinical/histologic suspicion—highlights inconsistent selection. Proposing a risk-stratified approach (eg, END for T2+ tumors, prior radiation, or poor differentiation) could standardize practice. This contrasts with parotid PSCC, where END is more universally accepted, underscoring site-specific management needs.
Adjuvant Therapy Considerations and Post-Treatment Surveillance
Adjuvant therapy for SMG-SCC (SMG-SCC) typically combines radiotherapy with cisplatin-based chemotherapy; however, modality selection (including monotherapy) requires careful evaluation of comorbidities, physiological age, and performance status. 14 Critically, the evidence supporting a survival benefit from adjuvant therapy remains limited. This approach is subject to ongoing debate: while some case reports advocate for systematic adjuvant treatment,1,3 prospective analyses demonstrate no statistically significant improvement in local control when comparing surgery alone versus surgery with adjuvant radiotherapy or chemoradiotherapy. 12 Importantly, the small cohort in this review (n = 8) is insufficient to contribute meaningfully to this debate or to draw any conclusions regarding therapeutic efficacy. Consequently, therapeutic planning demands careful individualization based on patient comorbidities, performance status, and multidisciplinary team consultation.
Metastatic Disease Challenges
Metastatic PSCC portends poor survival due to chemoresistance and rapid progression, with no standardized protocols. 14 Systemic agents include cisplatin, 5-fluorouracil, and anthracyclines. Emerging molecular insights implicate EGFR and Wnt/β-catenin pathways in carcinogenesis, suggesting immunotherapy potential for metastatic disease. Further research should elucidate immune interactions with salivary carcinomas and evaluate targeting EGFR/Wnt proteins. Assessing RAS-mediated anti-EGFR resistance may enable molecularly guided therapies. 15 Early hematogenous dissemination complicates management, underscoring the criticality of local control. Elevated distant metastasis risk with nodal involvement mandates regional lymphadenectomy during submandibular resection, particularly in radiotherapy-exposed patients.
The predominance of lung metastases (37.5%) and chemoresistance necessitates dedicated palliative frameworks. While molecular targets (EGFR/Wnt) are explored, symptom control remains paramount. Highlighting early integration of supportive care—such as palliative radiotherapy for symptomatic lesions or bronchoscopic interventions for airway-obstructing metastases—could improve quality of life. This complements systemic therapy discussions by addressing unmet needs in terminal disease trajectories.
Limitations
These findings must be interpreted with significant limitations inherent to studying this exceptionally rare malignancy. The small sample size (n = 8) precludes robust statistical analysis or definitive conclusions regarding optimal management. Reliance on heterogeneous case reports/series introduces selection bias and limits data granularity (eg, inconsistent reporting of tumor stage, margin status, or detailed treatment protocols). Publication bias likely favors cases with unusual presentations or positive outcomes. The retrospective nature and lack of controlled studies hinder assessment of treatment efficacy, particularly the debated role of adjuvant therapy. Furthermore, the extended timeframe of included studies (1992-2023) introduces potential historical bias in diagnostic and therapeutic approaches. These constraints warrant cautious interpretation and highlight the critical need for multi-institutional collaboration to establish registries for rare salivary malignancies.
The inclusion of an 11-year-old (Case 8) introduces unique biologic variables: pediatric salivary SCC is exceptionally rare and may reflect genetic syndromes (eg, radiation-naïve pathogenesis). Similarly, outcomes in elderly patients (eg, 82F) may be confounded by competing mortality risks. This age disparity limits extrapolation of management principles across age groups, emphasizing that adult-derived data cannot guide pediatric care and vice versa. Future registries should stratify analyses by age and prior radiation exposure.
Conclusion
PSCC of the SMG is an exceptionally rare and aggressive malignancy, characterized by strong male predominance, prior cervical radiotherapy exposure (median latency: 15.5 years), and frequent advanced-stage diagnosis due to non-specific symptomatology. Diagnosis mandates rigorous exclusion of metastatic disease and histological mimics through immunohistochemical profiling (PAS/EBV negativity) and multimodal imaging. While universal surgical resection forms the treatment cornerstone, high recurrence rates persist despite frequent adjuvant therapy (75% of cases). Outcomes remain guarded, with 37.5% developing lung metastases and 62.5% overall survival, though rapid recurrence occurred in 1 case within 1 month post-chemoradiation. The limited evidence base (n = 8) underscores the imperative for multi-institutional collaboration to establish diagnostic standardization, validate multimodal protocols integrating molecular targeting (EGFR/Wnt pathways), and improve survival for this historically neglected malignancy.
Footnotes
Ethical Considerations
This study is a systematic review based on already published data, where no human participants are involved; therefore, ethical approval not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are embedded in the manuscript as it’s secondary research.
