Abstract
Keywords
Introduction
Fungal ball sinusitis (FBS) is a chronic inflammatory disease characterized by the noninvasive aggregation of fungal masses within the paranasal sinuses. It predominantly affects middle-aged and elderly women, whose mean age is 52.4 years, and women represent 61% of cases. 1 Epidemiological studies indicate that unilateral involvement is dominant (>97%), with the maxillary sinus being the most frequently affected site (>75%), followed by the sphenoid sinus. 2 Pathogenesis may be associated with anatomical and functional abnormalities in nasal and sinus airflow dynamics (e.g., nasal septum deviation, middle meatal stenosis, and paradoxical middle turbinate), compromised immune function, hypoxic and acidic microenvironments, hyperglycemia, chronic inflammation, or prolonged use of antibiotics or immunosuppressants. 3 Clinical manifestations primarily include unilateral facial pain (79.2%), purulent nasal discharge, and nasal obstruction. CT imaging features include high-density calcifications (median CT value: 351 HU) and sinus wall sclerosis or erosion.1,2 Although functional endoscopic sinus surgery is the primary treatment with a favorable prognosis (recurrence rate <10%), the underlying pathological mechanisms, particularly fungal-host interactions, and the regulation of the local metabolic microenvironment remain poorly understood.1,2
Current metabolomics studies on FBS are extremely limited. Existing research has focused on metabolic characteristics of chronic rhinosinusitis (CRS), such as increased oxidation of unsaturated fatty acids in eosinophilic CRS versus uric acid accumulation in non-eosinophilic CRS. 4 In particular, refractory CRS patients demonstrate elevated glutathione disulfide concentrations that positively correlate with IL-8 mRNA expression. 5 Furthermore, comparative metabolomic analysis of nasal mucosal tissues revealed divergent metabolic patterns among control mucosa, eosinophilic chronic rhinosinusitis with nasal polyps (CRSwNP), and non-eosinophilic CRSwNP, and linoleic acid emerged as the most significant differential metabolite potentially related to eosinophilic infiltration in nasal polyps. 6 Although a recent small-scale study including 12 patients identified significant dysregulation of sphingolipid metabolism and glycerophospholipid metabolism in sinus tissues, FBS, suggesting links to mucosal inflammation and bone destruction, 2 the following unresolved issues persist. Current research lacks systematic comparisons of metabolic profiles between the diseased and healthy sides and does not effectively distinguish localized pathological changes from systemic metabolic responses. The role of abnormal metabolic pathways in driving fungal colonization or host immune responses remains unclear, and currently, there are no validated metabolic biomarkers available for early diagnosis or prognostic evaluation of FBS.
Untargeted metabolomics, which enables high-throughput detection of thousands of small-molecule metabolites, offers a powerful tool to comprehensively uncover disease-related metabolic network perturbations. This approach is particularly advantageous for dissecting host-pathogen interactions and the regulation of the inflammatory microenvironment. 5 This study intends to employ untargeted metabolomics technology to systematically analyze the differences in metabolic profiles among fungal balls in affected sinus cavities, secretions from the middle meatus of both the affected and healthy sides in 30 cases of unilateral FBS, and secretions from the middle meatus of a control group. The aim is to reveal the local metabolic characteristics of fungal ball-type sinusitis and to explore the biological pathways associated with differential metabolites.
Patient and Methods
Participants
This study enrolled 30 FBS patients who underwent initial endoscopic nasal surgery in the Department of Otorhinolaryngology of Beijing Tongren Hospital, Capital Medical University, between February 2023 and April 2024, along with 29 control subjects with a simple nasal septum deviation. All participants met strict exclusion criteria: use of topical or systemic antibiotics/glucocorticoids within the last month; nasal irrigation within the preceding 2 weeks; imaging showing inflammatory changes in the ostiomeatal complex region; comorbid allergic rhinitis; pregnancy or lactation; congenital ciliary dyskinesia; severe respiratory diseases, chronic consumptive diseases, tumors or immunodeficiency; age <18 or >70 years. All clinical specimens were collected from patients with a research protocol approved by the hospital ethics committee. Written informed consent was obtained from all participants, with general clinical data recorded.
During endoscopic procedures, the samples were collected using ethmoid forceps and nasal transport swabs with protective sleeves, divided into four groups: Group A:fungal ball from the affected sinus cavity; Group B:secretions from the affected middle meatus; Group C:secretions from the contralateral healthy middle meatus; Group D:middle meatal secretions from control subjects. Swabs remained in the middle meatus for 15 seconds before being retracted through protective sleeves to avoid contamination. Swab heads were severed into sterile tubes, immediately placed on ice packs, and transported to freezers <80°C for storage.
UHPLC-MS/MS Analysis
Place the fungal ball sample or the swab sample into a microcentrifuge tube and add 1000 μL of 80% methanol aqueous solution. Vortex thoroughly, incubate on ice for 5 minutes, and centrifuge at 15,000 × g and 4°C for 20 minutes. Transfer the supernatant to a new microcentrifuge tube and lyophilize it to a dry powder. Resuspend the lyophilized powder with a 10% methanol aqueous solution equivalent to the original sample volume and inject the solution into the LC-MS system (liquid chromatography-mass spectrometry) for analysis. HPLC-MS/MS analyses were performed using a Vanquish UHPLC system (ThermoFisher, Germany) coupled with an Orbitrap Q ExactiveTM HF mass spectrometer or Orbitrap Q ExactiveTM HF-X mass spectrometer (Thermo Fisher, Germany)
Data Processing and Metabolite Identification
The raw data files generated by UHPLC-MS/MS were processed using Compound Discoverer 3.3 (CD3.3, ThermoFisher) to perform peak alignment, peak picking, and quantitation for each metabolite. After that, peak intensities were normalized to the total spectral intensity. The normalized data was used to predict the molecular formula based on additive ions, molecular ion peaks, and fragment ions. Then the peaks were matched with the mzCloud,mzVault, and MassList databases to obtain the accurate qualitative and relative quantitative results. Statistical analyses were performed using statistical software R (R version R-3.4.3), Python (Python 2.7.6 version), and CentOS (CentOS release 6.6). When the data were not normally distributed, standardization was performed to obtain relative peak areas. And compounds whose Coefficient of Variation (CVs) of relative peak areas in QC samples were greater than 30% were removed, and finally, the metabolites’ identification and relative quantification results were obtained. Finally, the metabolites are annotated using the KEGG database, the HMDB database, and the LIPIDMaps database. Statistical analysis was performed by SPSS 21 software (SPSS Inc., Chicago, IL), including the independent samples t-test and chi-square test, and correlation analysis.
Results
Characteristics of the Patient
In this cohort, samples from 30 patients with FBS were collected as the experimental group. For the control group, middle meatus secretions from 29 patients with simple nasal septum deviation were collected. Specific clinical information is shown in Table 1.
Clinical Information for Patients With FBS and the Control Group.
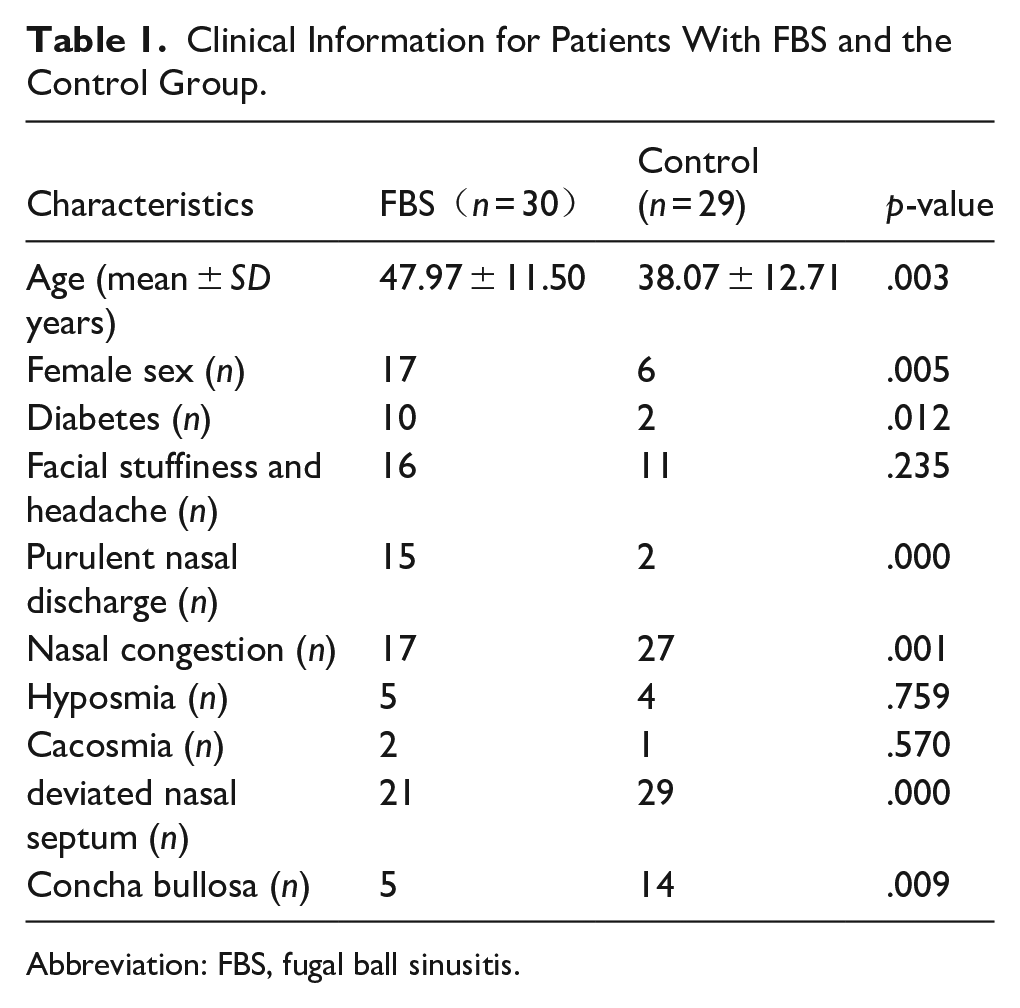
Abbreviation: FBS, fugal ball sinusitis.
Differences in Local Microenvironment Metabolites and Key Metabolites Associated With Fungal Colonization or Host Inflammatory Responses
In this experiment, we performed LC-MS/MS-based metabolomic profiling on a total of 119 samples, identifying 2589 metabolites in total. Among these, 1619 metabolites were detected in positive ion mode and 970 in negative ion mode. As shown in Figure 1a, the positive ion mode analysis revealed different clustering patterns. Group A and a subset of Group B aggregated along the negative T-score[1] axis (−40 to 0), while Groups C/D and the remaining samples of Group B distributed along the positive axis (0 to +40), indicative of the pronounced metabolic disparities between the contents of the diseased sinuses and the healthy/control samples (pR²Y = 0.005, pQ² = 0.005). The partial overlap between groups A and B along the T-score[1] axis suggests minor metabolic divergences between diseased sinus and middle meatus secretions or shared microenvironmental characteristics attributable to anatomical proximity. Negative ion mode analysis, as shown in Figure 1b, demonstrated a marked separation of Group A from all other groups, highlighting the uniqueness of the metabolic signatures specific to fungal ball rhinosinusitis. The overlapping distributions of Groups B and C reflect metabolic similarities between diseased and contralateral middle meatus secretions, while maintaining discernible differential characteristics. The clear delimitation between Groups D and A further validates distinct metabolic profiles between fungal ball rhinosinusitis and isolated nasal septum deviation.
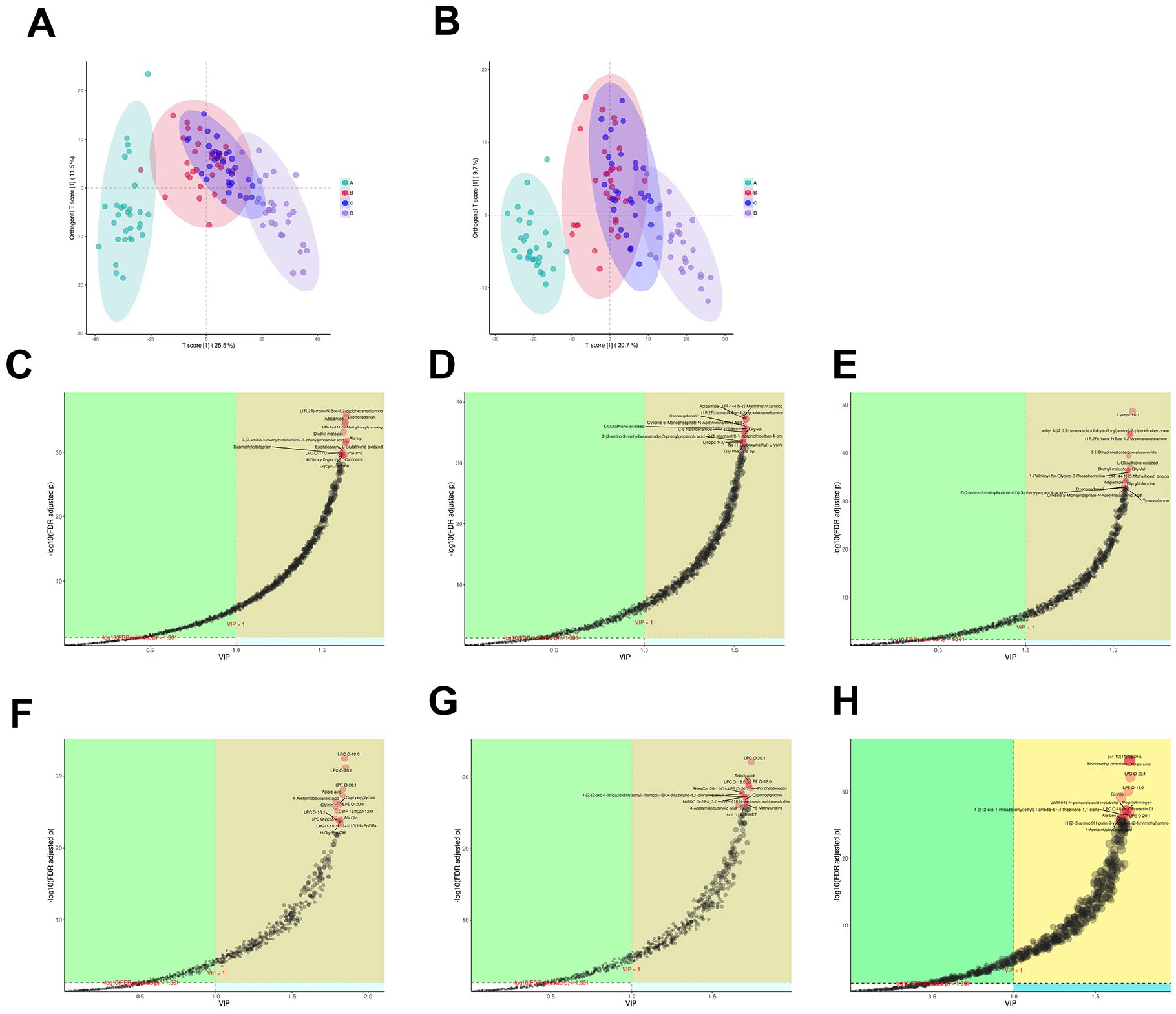
(A) .OPLS-DA score plot of groups A, B, C, and D in positive ion mode (pR2Y = 0.005, pQ2 = .005). (B) OPLS-DA score plot of groups A, B, C, and D in negative ion mode (pR2Y = 0.005, pQ2=0.005). (C) OPLS-DA metabolite importance plot (VIP) between group A and B in positive ion mode, displaying FDR-adjusted p-values. (D) OPLS-DA metabolite importance plot (VIP) between group A and C in positive ion mode, displaying FDR-adjusted p-values. (E) OPLS-DA metabolite importance plot (VIP) between group A and D in positive ion mode, displaying FDR-adjusted p-values. (F) OPLS-DA metabolite importance plot (VIP) between group A and B in negative ion mode, displaying FDR-adjusted p-values. (G) OPLS-DA metabolite importance plot (VIP) between group A and C in negative ion mode, displaying FDR-adjusted p-values. (H). OPLS-DA metabolite importance plot (VIP) between group A and D in negative ion mode, displaying FDR-adjusted p-values.
Biomarker prioritization was performed using variable importance in projection (VIP) scores (threshold > 1.0) combined with a false discovery rate (FDR)-adjusted p < .05. The 15 top metabolites with the lowest adjusted p-values were selected as key candidates. Pairwise comparisons of positive ion modes pairwise comparisons (Figure 1C–1E) identified seven differential consensus metabolites in Group A versus B/C/D. Five metabolites exhibited significant downregulation in Group A compared to B/C/D: (1R,2R)-trans-N-Boc-1,2-cyclohexanediamine, Oxohongdenafil, Adipamide, UR-144 N-(5-methylhexyl) analog, and oxidized glutathione. Two metabolites demonstrated A-specific upregulation: 2-(2-Amino-3-methylbutanamido)-3-phenylpropanoic acid and Glycyl-L-leucine. Negative ion mode analyses, as shown in Figures 1F to 1H, revealed six consensus biomarkers. Three lysophospholipids showed pronounced upregulation in Group A: lysophosphatidylcholine (LPC) O-18:0, LPC O-20:1, and LPE O-20:1. The remaining three metabolites were downregulated in Group A relative to the others: Adipic acid, 4-acetamidobutanoic acid, and citrinin.
All differential metabolites demonstrated robust model validity after permutation testing (n = 200 iterations, Q² intercept < 0.05). Fold change (FC) analysis with FDR-adjusted p-values confirmed the magnitude and directionality of these metabolic changes. This multilevel profiling approach provides novel insights into microenvironment-specific metabolic perturbations in the pathogenesis of fungal ball rhinosinusitis.
Enrichment Analysis and Topological Analysis of Metabolic Pathways
In this study, the results of the Over-Representation Analysis (ORA) enrichment analysis of fungal balls of affected sinuses in Group A (FBS) and the middle meatus secretions in control group D demonstrated that, in positive ion mode, as shown in Figure 2a and b, ORA revealed significant enrichment (high FC, low p-value) in nucleotide metabolism, arginine/proline metabolism, and histidine metabolism, suggesting their pivotal roles in the pathogenesis of FBS. Topological analysis placed nucleotide metabolism in the upper right quadrant (high −log10(p) and Impact value), indicating both statistical significance and substantial biological influence. Pathways such as histidine metabolism and phenylalanine/tryptophan biosynthesis showed moderate significance, which warrants further investigation. Under negative ion mode, as shown in Figures 2d and e, nucleotide metabolism, pyrimidine metabolism, and arginine biosynthesis emerged as the most enriched pathways with high impact values. The metabolism of taurine/hypotaurine and 2-oxocarboxylic acid displayed notable regulatory potential. In summary, nucleotide metabolism demonstrated significant enrichment in both positive and negative ion modes during the intergroup comparison between Group A and Group D.
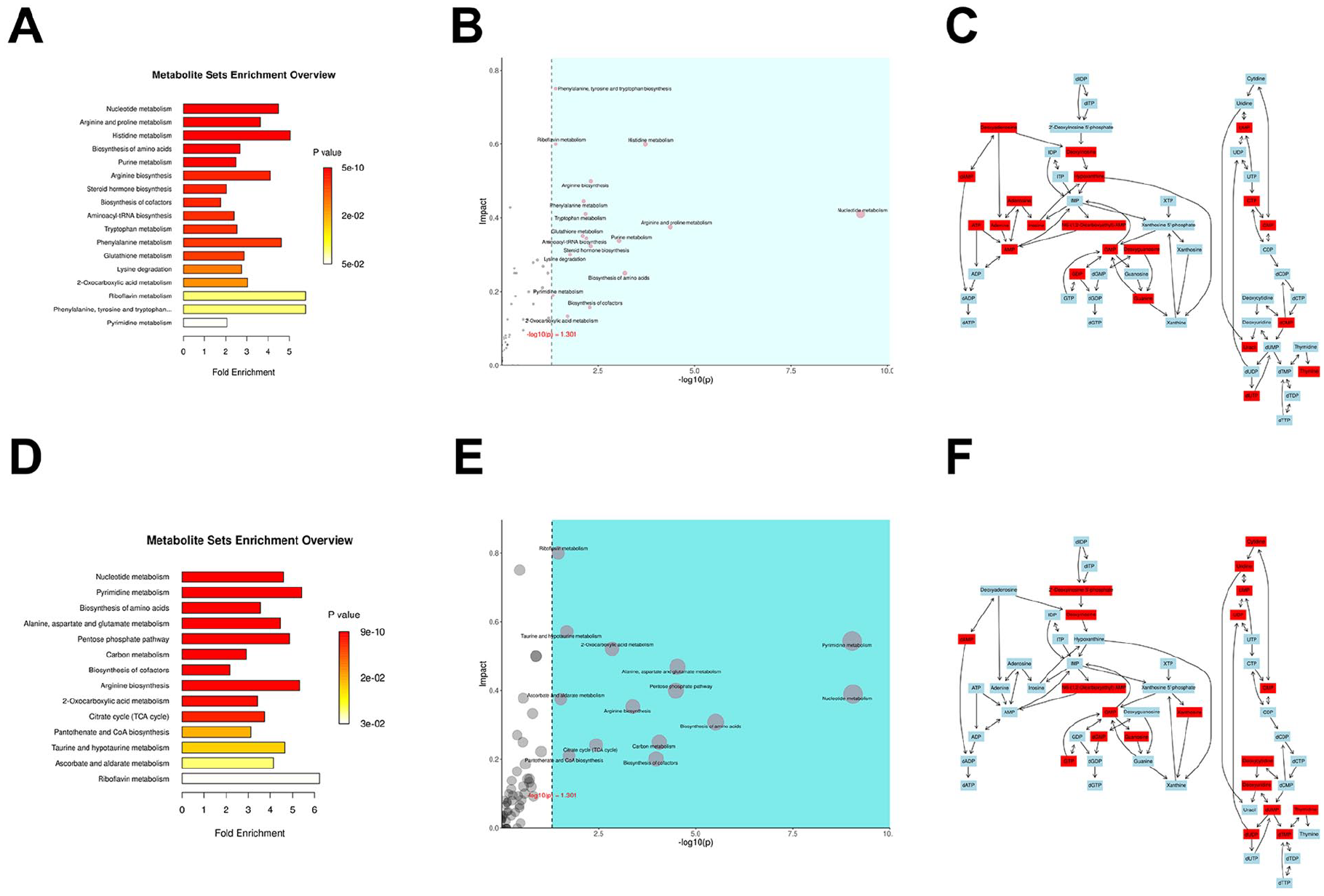
(A) ORA enrichment analysis between Group A and Group D in positive ion mode. (B) Integrated ORA enrichment and topological analysis of Group A versus Group D in positive ion mode. (C) Compound reaction network of nucleotide metabolic pathway in positive ion mode. (D) Comparative ORA enrichment analysis between Group A and Group D in positive ion mode. (E) Combined ORA enrichment and topological profiling of Group A versus Group D in positive ion mode. (F) Nucleotide metabolism pathway reaction network under positive ion mode.
As shown in Figure 2c and d, which represent the metabolic reaction networks of compounds of the nucleotide pathway under positive and negative ion modes, respectively, the diagram illustrates the metabolic processes of purine metabolism, pyrimidine metabolism, and deoxynucleotide metabolism, as well as the interconversions of compounds within these three metabolic pathways. By calculating the FC of metabolites combined with the p-values, it was revealed that under positive ion mode, compounds such as adenine, adenosine, and thymine were upregulated in Group A, while guanine, cytosine, and deoxyadenosine showed relative downregulation in Group A. In negative ion mode, compounds such as cytidine and thymidine were upregulated in Group A, whereas guanosine, xanthosine, and uridine exhibited relative downregulation in Group A.
Discussion
Among the numerous metabolites and metabolic pathways detected in our study, we have selected the following five differential metabolites and one characteristic metabolic pathway for detailed discussion.
In OPLS-DA comparisons between Group A (affected sinus fungal ball) and Groups B/C (ipsilateral/contralateral middle meatal secretions from FBS patients), differential metabolic patterns revealed a significant elevation of 4-acetamidobutanoic acid in Groups B/C compared to Group A, with the following mechanistic interpretations: The observed upregulation is consistent with polymicrobial coinfections in FBS, where microbial communities frequently include Gram-negative pathogens such as Pseudomonas aeruginosa. 7 4-Acetamidobutanoic acid represents an acetylated derivative of putrescine, classified under polyamines, aliphatic nitrogenous compounds with more than two amino groups. In living organisms, one of the most common polyamines is putrescine. 8 Studies show that in sepsis caused by Gram-negative bacterial infections (such as Pseudomonas aeruginosa), P. aeruginosa metabolizes putrescine to produce N-acetylputrescine, which is further converted to 4-acetamidobutanoate. These metabolites are significantly elevated in patient plasma and mouse models and are correlated with disease severity. 9 4-Acetamidobutanoate and 4-acetamidobutanoic acid represent different ionic forms of the same chemical entity. Under physiological conditions where the pH approximates neutrality, this metabolite exists predominantly in its deprotonated salt form (4-acetamidobutanoate) due to the chemical equilibrium established between the carboxylic acid and its conjugate base. The strictly aerobic nature of Pseudomonas aeruginosa prevents it from acetylating putrescine in the hypoxic environment of sinus cavities affected by FBS, where low oxygen levels result from fungal mass obstruction and inflammatory responses. 10
Glutathione (GSH), a tripeptide composed of glutamate, cysteine, and glycine, functions primarily through its antioxidant properties and maintenance of thiol redox homeostasis. As the predominant intracellular antioxidant, GSH regulates redox-sensitive transcription factors (e.g., NF-κB, HIF-1α) and signaling pathways, acting as a molecular switch in inflammatory responses. Within the glutathione redox cycle, GSH serves as a substrate for glutathione peroxidase (GPx), catalyzing the reduction of reactive oxygen species (ROS) such as hydrogen peroxide (H₂O₂) to water, while oxidizing to glutathione disulfide (GSSG). Subsequently, GSSG is regenerated to GSH by glutathione reductase utilizing Nicotinamide Adenine Dinucleotide Phosphate (NADPH), thus maintaining cellular redox equilibrium. 11 In OPLS-DA pairwise comparisons between Group A and Groups B/C/D, upregulated expression of GSSG was observed in Groups B and C. This phenomenon may be attributed to active oxidative stress and immune defense mechanisms in the middle meatus, a primary region for nasal inflammatory responses. This anatomical site harbors abundant immune cells (e.g., neutrophils and macrophages) 12 that generate substantial ROS upon recognition of fungal antigens, driving GSH oxidation to GSSG and subsequent accumulation. In particular, fungal antigens may also induce localized oxidative stress in non-colonized contralateral areas through nasal circulation or mucosal lymphatic immune activation. 13 Elevated levels of GSSG in Group D likely reflect physiological oxidative stress in the middle meatus, a non-sterile upper respiratory compartment with resident microbiota and cumulative inflammatory burden during respiration. On the contrary, the relative downregulation of GSSG in Group A could be due to hypoxia-induced immunosuppression in the maxillary sinus containing the fungal ball. Specifically, anatomical constraints (e.g., narrow ostium of the maxillary sinus) create localized hypoxia and acidic microenvironments that suppress mitochondrial respiratory chain activity. Concurrently, the physical barrier formed by fungal biofilms may restrict immune cell infiltration, collectively diminishing ROS production and resulting in lower levels of GSSG.
Glycerophospholipids, as the main constituents of cellular membranes, serve dual roles as reservoirs for bioactive compounds and precursors for signaling molecules. These lipids play a pivotal role in fundamental physiological processes that include cell proliferation, motility, signal transduction, and programmed cell death. 14 Through enzymatic modification by phospholipase A2 (PLA2), glycerophospholipids undergo metabolic conversion to generate lysophospholipids such as LPC and lysophosphatidylethanolamine (LPE).15,16 Evidence suggests a correlation between elevated LPC levels and decline in lung function in patients with chronic obstructive pulmonary disease, possibly mediated by oxidative stress pathways. 17 In contrast, specific LPC subspecies and LPE have demonstrated anti-inflammatory properties in other experimental models.18,19 In our OPLS-DA comparing Group A with Groups B, C, and D, three lysophospholipids, LPC O-18: 0, LPC O-20: 1, and LPE O-20: 1, showed significant upregulation in Group A. This phenomenon may be mechanistically related to fungal cell degradation processes, since fungal membranes predominantly consist of sterols, glycerophospholipids, and sphingolipids. 20 The observed elevation probably reflects: enzymatic breakdown of fungal cell walls and sinus mucosal epithelial cells within fungal balls; cytotoxic effects of certain LPC derivatives that exacerbate mucosal apoptosis; 21 compensatory production of anti-inflammatory LPE and unsaturated LPC species under chronic inflammatory conditions. In particular, anatomical abnormalities (e.g., middle meatal stenosis and deviated nasal septum) and inflammation-induced mucosal edema may synergistically alter mucociliary clearance, 3 creating a retention niche for these lipid metabolites and altering maxillary sinus fungal aggregates.
Comparative ORA and topological examination between Groups A and D revealed significant nucleotide metabolic dysregulation, attributable to three interconnected pathobiochemical mechanisms: (1) fungal-host metabolic competition:fungal balls are primarily formed by entanglement of hyphae from filamentous fungi such as Aspergillus. Their rapid proliferation requires substantial nucleotide precursors for DNA/RNA synthesis. This may lead to the activation or compensatory enhancement of local nucleotide metabolic pathways in the host to meet the fungal metabolic demands. Furthermore, fungi may secrete hydrolases (e.g., nucleases) to degrade nucleic acids released by host cells, competitively depleting nucleotide resources and initiating host metabolic reprogramming. 22 (2) Chronic inflammation and cellular catabolism: the chronic presence of fungal balls exerts persistent mechanical and biochemical stimulation on the sinus mucosa, establishing a self-perpetuating inflammatory cascade. Histopathological analysis reveals substantial leukocyte infiltration dominated by neutrophils and lymphocytes in affected regions. These activated immunocytes secrete pro-inflammatory cytokines, including TNF-α and IL-6, 23 which transcriptionally upregulate enzymes that govern nucleotide metabolism, particularly adenosine deaminase. This enzymatic activation drives accelerated ATP catabolism, generating adenosine metabolites that skew the purinergic signaling balance. 24 Gleichzeitig, the inflammatory environment triggers massive release of nucleic acids from host cellular damage and metabolic reprogramming through the induction of the salvage pathway. 25 (3) Microenvironment-driven metabolic rewiring: The hypoxic and acidic microenvironment created by the formation of fungal balls may inhibit mitochondrial oxidative phosphorylation, forcing host cells to rely on nucleotide metabolism (e.g., the purine nucleotide cycle) to sustain the energy supply. 26
This study has the following limitations: Small sample size, which may introduce bias into the results. Potential blood contamination that interferes with the detection of lysophospholipids in fungal balls. Future research should include large-scale analyses and multiomics integrated approaches.
Conclusion
In summary, in this study, we analyzed metabolic differences between fungal balls in the affected sinus cavity, secretions from the middle meatus of the affected side, secretions from the middle meatus of the healthy side, and secretions from the middle meatus of the control group in patients with FBS using UHPLC-MS/MS. By exploring these variations from a metabolomic perspective, our aim was to elucidate the pathogenesis of this disease. Our findings revealed that the metabolite composition within the fungal ball in the affected sinus cavity of patients with FBS shows significant differences compared to the secretions of the middle meatus on the affected side, the contralateral side, and the control group. In the future, we plan to further investigate the pathogenesis of FBS through multi-omics integrative analysis and explore potential therapeutic approaches.
Footnotes
Ethical Considerations
This study protocol adhered to the ethical guidelines of the Declaration of
Helsinki of 1975 and was approved by the Ethics Committee of Beijing Tongren Hospital, Capital Medical University.
Consent to Participate
Informed consent was obtained from each patient for this study.
Consent for Publication
All authors have consented to publication.
Author Contribution
Yi Wang: Contributed to specimen collection, data curation, formal analysis, investigation, project administration, and drafted the manuscript. Hongzheng Wei: Contributed to data collection, formal analysis, project administration, critically revised manuscript, gave final approval, and agreed to be responsible for all aspects of the work, ensuring integrity and accuracy. Ting Yang: Contributed to data curation and project administration. Chengyao Liu: Contributed to data curation and project administration. Pengyu Shi: Contributed to data curation and project administration. Jie Zhou: Contributed to data curation and project administration. Yunchuan Li: Contributed to design, acquisition, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work, ensuring integrity and accuracy. All authors gave their final approval and agreed to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Beijing Natural Science Foundation of China, 'Research on the Pathogenesis of Fungus Ball Sinusitis (FBS) Based on Microbiomics and Metabolomics' (7222026).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
