Abstract
Introduction:
Large full-thickness helical rim defects following skin cancer resection are a reconstructive challenge. We present a low-morbid, reproducible technique for medium to large helical rim defects and their outcomes.
Methods:
A retrospective review of patients 18 years or older from 2014 to 2024 who underwent helical rim reconstruction using this technique following skin cancer resection was conducted. The technique begins with reducing the helical defect through wedge excision of the adjacent antihelix. Next, an ipsilateral conchal cartilage graft bridges the remaining defect. A local flap provides soft tissue coverage, and flap division occurs 3 to 4 weeks later.
Results:
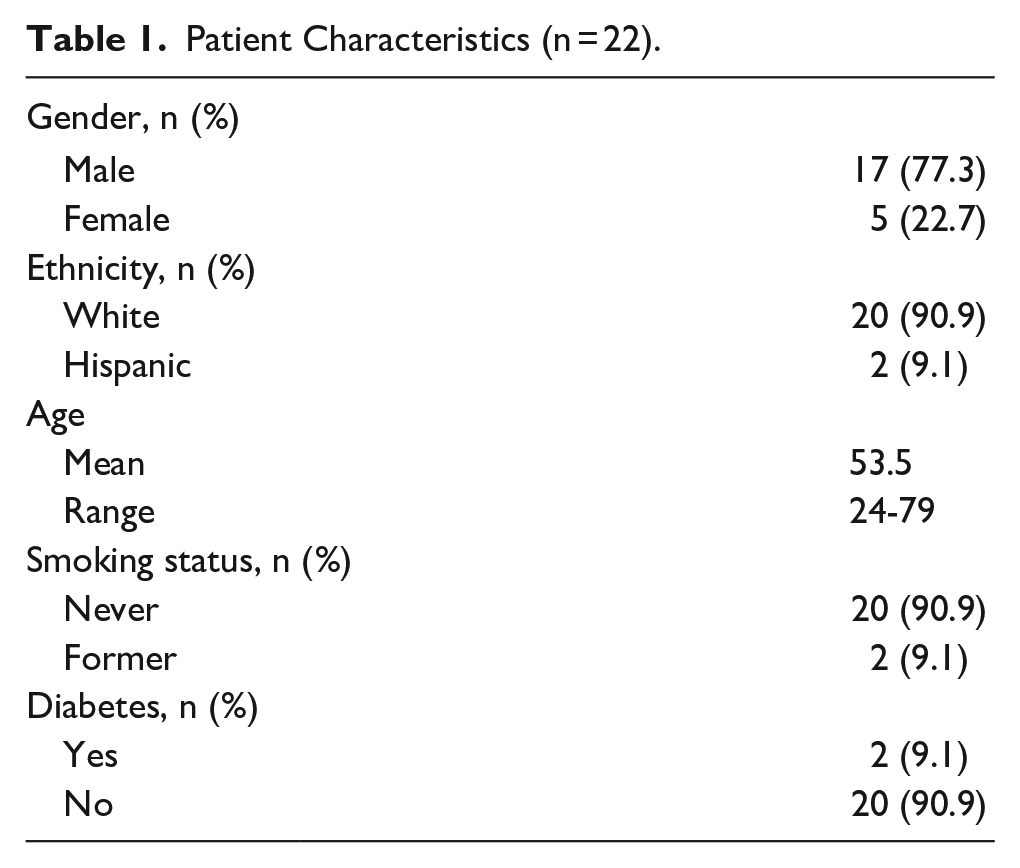
A total of 22 patients, 5 females and 17 males (mean age 53.5) were identified. The average defect size was 3.79 cm by 2.90 cm. Flap coverage was achieved primarily by superiorly based postauricular flaps in 68.2% of cases. Complications included partial flap congestion in 2 patients (9.1%) and infection in 1 patient (4.5%). No flap failures or cartilage graft complications were observed. Three patients (13.6%) underwent in-office flap contour thinning.
Discussion:
The two-stage postauricular flap combined with wedge excision of the ipsilateral adjacent antihelix offers an effective, reproducible, low-morbid technique for auricular reconstruction.
Introduction
The ear is a complex structure comprised of an intricate cartilage framework and soft tissue covering, important for auditory function and facial harmony. Structural defects related to skin cancer resection are common due to frequent sun exposure. 1 Auricular malignancies represent ~6% to 10% of all head and neck cutaneous cancers. 2 The most common neoplasms of the auricle are squamous cell carcinoma (SCC) and basal cell carcinoma (BCC), followed by melanoma (MM). 1 Among auricular subunits, the helical rim is one of the most vulnerable sites for skin cancer. 2 Full-thickness helical rim defects resulting from oncologic-wide local excision, especially with large margins, pose challenges for reconstruction.
Numerous surgical techniques have been described to address full-thickness helical rim defects. For defects <2.5 cm, options include primary closure, wedge resection, Antia-Buch flaps, interpolated tube flaps, and small advancement flaps. 3 Larger defects (>2.5 cm) often require cartilage grafts from the concha or ribs, porous polyethylene constructs, osseointegrated implants for defects requiring total or subtotal auriculectomy, or larger flaps such as local pedicle or temporoparietal flaps with skin grafts.4-7 However, these approaches increase surgical risk, operative time, and donor site morbidity. 3
For medium to large helical defects, we present a straightforward, systematic, two-stage technique, particularly suited for older patients who are not candidates for extensive reconstructions. The first stage reduces the defect size via adjacent wedge excision of the antihelix, followed by placement of an ipsilateral conchal cartilage graft, and pedicled postauricular flap coverage. The second stage involves flap division and insetting. This method transforms initially large defects into more manageable sizes, minimizing cartilage and flap dimensions required.
Materials and Methods
Study Design, Setting, and Participants
Following institutional review board approval (IRB 00182649), we performed a retrospective chart review of all patients ≥18 years old who underwent helical rim reconstruction between January 2014 and September 2024 at our institution. All helical rim reconstructions were performed by the same surgeon (C.A.). Patients were included in the analysis if they (1) had a history of skin cancer of the helical rim, (2) underwent helical rim reconstruction using our technique during the study period. Patients were excluded if they (1) had concomitant earlobe repair, (2) alternative etiology such as trauma or frostbite, (3) partial-thickness defects. At our institution, a multidisciplinary approach is used, with surgical oncology performing tumor excision and plastic surgery managing soft tissue reconstruction. All patients were referred for plastic surgery after surgical oncology excision.
Variables and Data Sources
Data collection included baseline characteristics such as age, ethnicity, smoking status, history of diabetes, and skin cancer history (ie, BCC, SCC, MM). Surgical characteristics included defect location, defect thickness, original defect size, defect size after antihelix excision, flap type, operative time, and time to second surgical stage. Postoperative outcomes were collected for a follow-up time of at least 1 year from the date of surgery. Postoperative complications included flap congestion, infection, wound dehiscence, operative debridement, partial or total flap loss, hematoma, and keloid formation. Need for flap revisions such as postoperative thinning procedures was collected as well. Surgical variables such as operative time and defect size after antihelix resection were missing for some patients due to transitions in electronic medical record systems during the study period.
Technique Description
The ear is infiltrated using an epinephrine-containing local anesthetic solution. Limited debridement of the compromised soft tissue is performed, and the defect is thoroughly irrigated (Figure 1A–C). The first principle used is to reduce the size of the original helical rim defect by marking out and excising a portion of the adjacent antihelix, oftentimes extending to the conchal bowl for optimal mobilization. The remaining antihelix is then repaired, significantly reducing the size of the defect at the level of the rim. A cartilage graft is harvested from the ipsilateral conchal bowl, either at this stage or sometimes before the antihelix excision, and fashioned to reconstruct the remaining helical rim defect. (Figure 1A–C). Once the cartilage graft has been secured, a pedicled postauricular flap is designed to provide soft tissue coverage. Flap design was based on defect position, prior incisions, and skin laxity. (Figure 1A–C). The flap is raised and transposed to cover the anterior and posterior surfaces of the cartilage graft and sutured in place. (Figure 1A–C). The donor site is closed primarily if possible, often leaving open areas and an exposed pedicle to be closed at a future date. Pedicle division is delayed ~4 weeks to allow for flap incorporation, sometimes longer if there are healing delays with the flap.
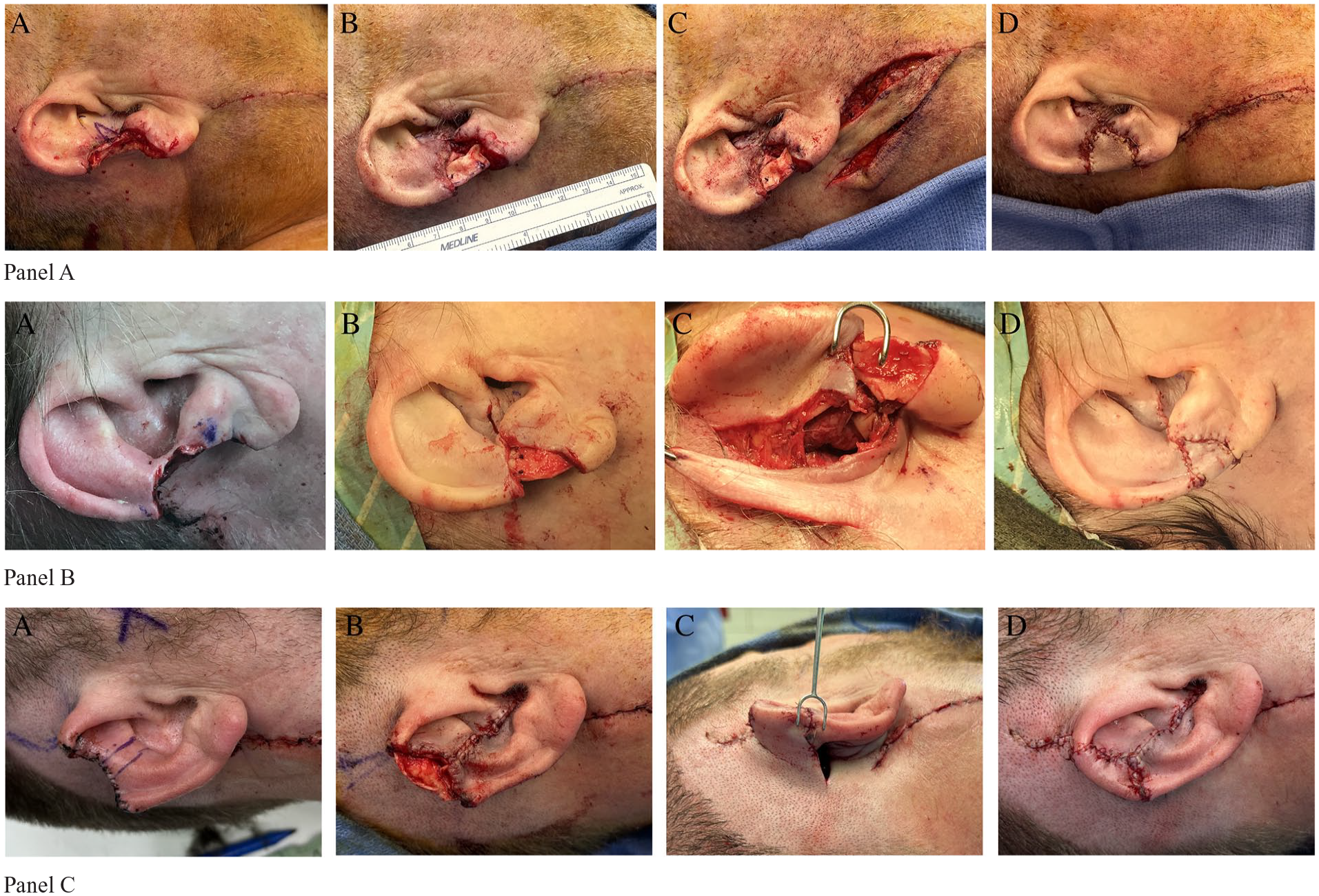
(A) Preoperative image of 3 patients with large defects of the superior and inferior one-third of the helical rim following oncologic resection. Preoperative markings (in purple) of the planned antihelix and/or conchal resection. (B) Ipsilateral conchal cartilage graft bridging remaining defect following the original defect size reduction. (C) Postauricular flap elevation using an inferiorly based (upper), superiorly based (middle), and direct advancement flap (lower). (D) Flap coverage and inset to the helical rim defect.
Statistical Methods
For quantitative variables, we calculated descriptive statistics (means and ranges). We performed all analyses using R version 3.6.1 (R Core Team).
Results
Among 28 patients who underwent helical rim reconstruction during the study period, 22 met the inclusion criteria. Of these patients, 22.7% were female and 77.3% were male, with a mean age of 53.5 years (range: 24-79 years; Table 1). The majority of patients (90.9%) identified as White, while 9.1% identified as Hispanic. Two patients (9.1%) had a history of diabetes, and 2 (9.1%) patients were former smokers. All patients had a history of skin cancer, 95.5% with MM and 4.5% with BCC (Table 2). All patients underwent wide local excision for their cancer resulting in a full-thickness defect (100%) with no patients having a history of local radiation. Defects were located on the superior third of the helix in 36.4% of patients, the middle third in 31.8%, and the inferior third in 31.8%. The average original defect size was 3.79 cm by 2.90 cm for an average defect area of 10.9 cm2. Following antihelix wedge excision, the defect size was reduced to an average of 2.39 cm by 2.75 cm, for a total area reduction of 6.5 cm2 (37% reduction).
Patient Characteristics (n = 22).
Oncologic and Defect Characteristics (n = 22).
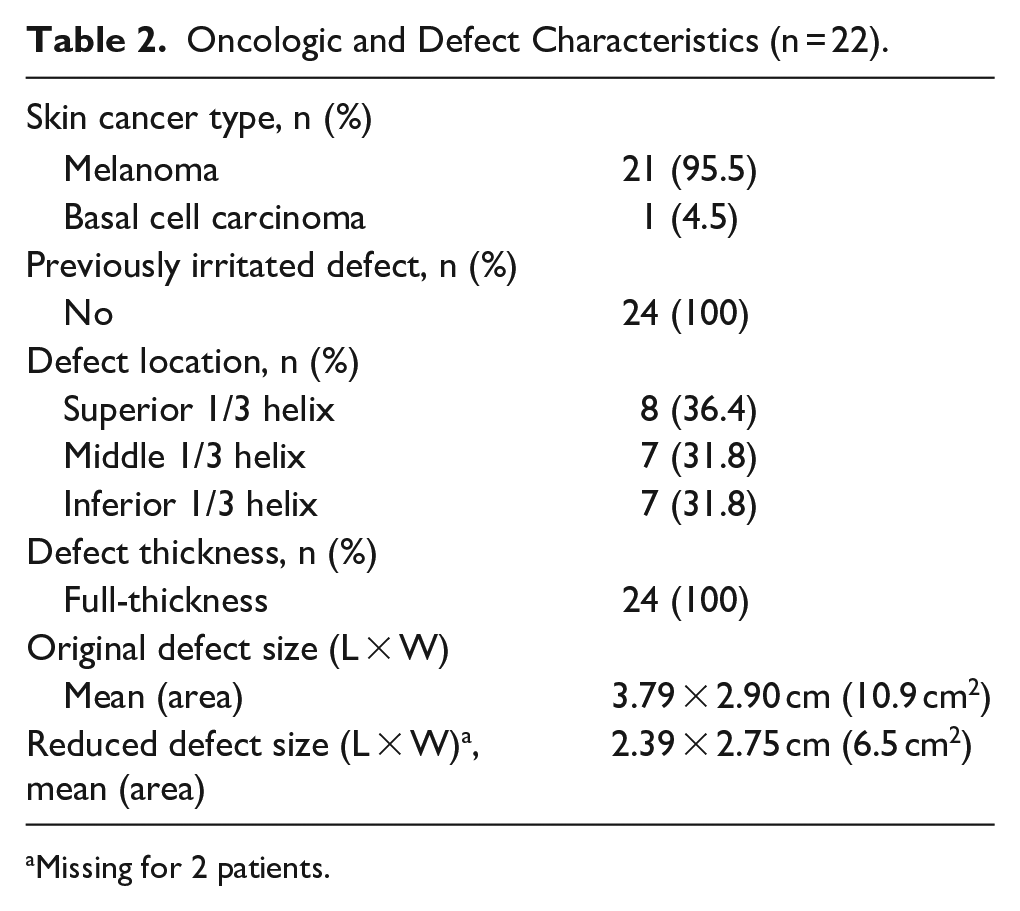
Missing for 2 patients.
Defect coverage after cartilage repair was achieved using superiorly based postauricular flaps in 68.2% of cases, inferiorly based postauricular flaps in 13.6%, and posteriorly based direct advancement flaps in 18.2% (Table 3). The mean operative time for the first stage was 106.4 minutes (range: 40-173 minutes). Postoperatively, 2 (9.1%) patients experienced flap congestion, 1 (4.5%) patient had a postoperative infection, and 1 (4.5%) patient had partial flap loss. The 2 flaps that were congested resolved with the application of topical dimethyl sulfoxide (DMSO). No cases of total flap failure, hematomas, wound dehiscence, or complications necessitating operative debridement were observed. The time to second stage flap division and insetting was 6 weeks on average (range: 3-15 weeks). The mean operative time of the second stage was 47 minutes (range: 26-89 minutes). A total of 3 patients (13.6%) had in-office flap thinning procedure performed several months after the flap division, and 1 (4.5%) patient had a keloid requiring further management, including corticosteroid injections, which were inadequate, ultimately requiring surgical excision.
Reconstructive Characteristics and Outcomes (n = 22).

Missing for 1 patient.
Discussion
Among the auricular subunits, the helix is commonly affected by skin cancer, often requiring surgical excision that can result in a sizeable defect. 2 Full-thickness helical rim defects following oncologic resection, especially after cancers requiring large margins such as MM, present a reconstructive challenge. The helix is the most visible component of the ear with an intricate cartilaginous framework that establishes a harmonious facial profile. Additionally, it serves as the support for eyeglasses and hearing aids, making it an important structure for daily functioning. Loss of these features may impact self-esteem and social well-being.8-10 The use of a postauricular flap combined with wedge excision of the adjacent ipsilateral antihelix and conchal bowl graft offers a simple, effective, and reproducible technique for medium to large helical rim defects following oncologic resection. This approach minimizes donor site morbidity, preserves auricular integrity, and achieves favorable outcomes.
While no consensus exists on the optimal management of helical rim defects, surgical reconstruction goals remain consistent. These principles involve re-establishing the cartilaginous framework, maintaining auricular dimensions, and using tissue that matches the color, texture, and thickness of the surrounding ear.11-13 Numerous surgical techniques have been described to repair full-thickness helical rim deformities. Wedge excision is used for 1.5 to 2 cm defects but can result in a cupping deformity of the helical rim and vertical ear shortening.13-15 The Antia-Buch flap, though a well-established reconstructive technique, is typically limited to 2.5 cm defects on average.16-19 Larger defects (>2.5-3 cm) are usually not amenable to local tissue rearrangement and require regional tissue coverage with or without cartilage grafting. Cartilage is often necessary in full-thickness cases to maintain ear contour and prevent contracture. 19 Interpolated tube flaps can be used for deformities >2.5 cm, but they lack structural cartilaginous support, create a bulky helical rim, and require 3 surgical stages. 20 The temporoparietal fascial flap can provide vascularized coverage of large ear defects when combined with an autogenous rib cartilage and skin graft.6,19 However, this technique can result in significant donor site morbidity and may not be suitable for older patients. For very large defects involving over half of the ear, total auriculectomy with either porous polyethylene reconstruction, costal cartilage reconstruction, or osteointegrated implant is sometimes desirable.4-6 It should be noted that non-surgical options, including local radiation therapy, intralesional therapy, and systemic treatments (eg, PD-1 inhibitors), have emerged as valuable alternatives for managing helical rim malignancies in select patients, particularly the elderly or those with unresectable or advanced disease.21-23
Regional cutaneous flaps based on the postauricular skin, as used in our described technique, are good alternatives for reconstructing full-thickness helical rim defects. 24 These flaps offer substantial soft-tissue coverage with excellent color matching and skin thickness that closely matches the surrounding defect. These flaps can be harvested superiorly, inferiorly, or as an advancement flap based on a rich vascular network that can create random patterned flaps with reliable blood supply.25,26 The postauricular skin can be raised with cartilage from the ipsilateral ear (ie, chondrocutaneous flap) or cover cartilage grafts harvested from the ribs, nasal septum, or conchal bowl.12,27-29 Compared with these methods, conchal bowl grafts match the flexible elastic cartilage framework of the native ear and can be harvested in large quantities without auricular distortion. 3 Additionally, these grafts can be taken from the ipsilateral ear, minimizing donor site morbidity. Although not utilized in our series, the earlobe-based advancement flap represents an alternative reconstructive option for medium-sized full-thickness helical rim defects, offering effective coverage as single-stage technique. 30
While there are several studies describing the use of a postauricular flap for auricular reconstruction, few have used this flap with a free ipsilateral conchal bowl graft. Youssif et al used a superiorly based postauricular skin flap with an ipsilateral conchal bowl graft in 20 patients with full-thickness upper-third helical rim defects following trauma, human bites, and thermal injury. 31 Of these patients, 2 (10%) developed flap congestion, 1 (5%) had an infection, and 2 (10%) experienced hypertrophic scarring. 31 Similarly, in a case report by Kang et al, an inferiorly based postauricular perforator flap was combined with a diced ipsilateral conchal bowl graft in a 25-year-old trauma patient. 32 In a case series of 10 patients, Hu et al used a postauricular advancement flap with an ipsilateral conchal bowl graft for trauma, bite, and frostbite defects. 33 Although these techniques achieved acceptable cosmetic and functional outcomes, they were used for small to medium helical rim defects measuring 0.88, 1.46, and 5 cm2 on average.31-33 In contrast, our average defect size was 10.9 cm2, making our technique suitable for addressing larger helical rim defects (area range: 4-24.8 cm2). Performing a wedge excision of the ipsilateral conchal bowl not only serves as a reliable cartilage source for the helical rim but it effectively reduces the length of the primary defect, making it a more manageable size for flap coverage. In our series, this resulted in a 37% reduction in defect length on average. Though the reconstructed ear is smaller, we have found this to be well tolerated and acceptable to patients.
Complication rates using our technique were low and are in line with previous auricular reconstruction studies.24,26 There were no flap losses, hematomas, or wound dehiscence. Three patients experienced flap congestion that resolved with DMSO, 1 patient had a postoperative infection treated with antibiotics, and 1 patient had a keloid requiring surgical removal. The partial flap loss observed in 1 patient was presumed to be secondary to subzero weather exposure, based on patient-reported timeline and photos showing flap necrosis after a day of working outside in the cold, despite initial flap viability. Timing to the second stage was 1.5 months on average but ranged from 3 to 15 weeks. Our standard protocol to perform flap inset is 4 weeks following the first surgical stage; however, variations in the timing to the second stage were influenced by patient preference, scheduling conflicts, or delayed flap healing. Interestingly, all 4 patients who had a posteriorly based advancement flap, opted to not undergo a second-stage surgery and were satisfied with the results following the primary procedure.
The technique described is a simple, two-stage, outpatient procedure that is reproducible over a large range of defect sizes. It can be used for large full-thickness defects of the superior, middle, and inferior one-third of the helix and for those that extend into the scapha or antihelix. Due to the rich vascular supply of the posterior ear, these flaps can be designed in multiple orientations. Although the vertical height of the ear is reduced, this difference is well-tolerated and generally acceptable to the patient. Due to the conchal bowl resection for cartilage grafting, some patients noticed that the inner part of the concha was smaller when wearing earbuds. However, the postauricular groove is preserved for patients to wear hearing aids or glasses. Esthetically, the donor site scar is hidden within or behind the ear and there is minimal donor site morbidity. Only 3 patients requested flap thinning postoperatively, suggesting that the majority of patients (86.4%) were satisfied with the overall outcome of their ear reconstruction. Of the patients who wanted flap thinning, this was performed in-office during a postoperative clinic visit.
There are limitations to this study. While our series was sufficient to illustrate the feasibility and outcomes using this surgical technique for skin cancer resections, our small sample size limits broad conclusions for all helical rim reconstructions. Furthermore, we did not directly compare the outcomes of this technique with other alternative techniques. Lastly, we did not include patient-reported outcomes as part of our outcome analysis. Future research with larger sample sizes and patient-reported satisfaction measures is necessary to further evaluate this technique and its outcomes.
Conclusion
For large full-thickness helical rim defects, the initial reduction of the defect with wedge excision of antihelix followed by ipsilateral conchal cartilage graft and a local postauricular flap offers an effective and reproducible technique for helical rim reconstruction With the reduced donor site morbidity, this technique is a good option to consider for patients who are not candidates for larger, more extensive reconstructions or who do not desire a larger reconstruction that aims to preserve the entire height of the ear. Our results demonstrate favorable outcomes across diverse defect sizes, with a low complication rate.
Footnotes
Ethical Considerations
This study received ethical approval from the University of Utah IRB (approval #00182649) on October 15, 2024.
Consent to Participate
All patient information was de-identified, and patient consent was not required as this is an IRB-approved retrospective study. Patient data will not be shared with third parties.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are not publicly available due to ethical and legal restrictions.
