Abstract
We report a rare case of epiglottic granulomatosis with polyangiitis (GPA) in a 72-year-old woman presenting with 5 years of progressive dysphagia and dyspnea. Initial laboratory studies revealed leukocytosis (White Blood Cells 14.2 × 10³/µL), eosinophilia (8%), elevated inflammatory markers (C-Reactive Protein 42 mg/L, Erythrocyte Sedimentation Rate 78 mm/hour), and Proteinase 3 Anti-Neutrophil Cytoplasmic Antibody (PR3-ANCA) positivity (65 IU/mL). Laryngoscopy identified supraglottic edema with a right-sided mass, confirmed by Computed Tomography as localized epiglottic thickening without cartilage destruction. Histopathology of the biopsied lesion demonstrated necrotizing granulomas, vasculitis, and multinucleated giant cells, establishing the GPA diagnosis. Treatment with high-dose corticosteroids (1 mg/kg/day prednisone) and rituximab (375 mg/m²/week × 4) induced rapid symptom resolution within 2 weeks. Follow-up laryngoscopy at 1 month showed complete mucosal healing, and the patient remains recurrence-free during 18 month surveillance with maintenance immunosuppression (5 mg/day prednisone). This case highlights 3 critical insights: (1) isolated epiglottic GPA, although rare, should be considered in chronic upper airway obstruction unresponsive to conventional therapy; (2) PR3-ANCA serology and histopathology are indispensable for diagnosis; and (3) rituximab-based regimens achieve excellent outcomes in laryngeal GPA, but prolonged monitoring is essential given relapse risks. Our findings reinforce the need for multidisciplinary management of otolaryngologic GPA manifestations.
Keywords
Key Clinical Message
Epiglottic granulomatosis with polyangiitis demands histopathology/ANCA confirmation, responds excellently to rituximab-based therapy, and warrants prolonged surveillance due to relapse risks. Multidisciplinary coordination optimizes outcomes in this life-threatening localization.
Introduction
Granulomatosis with polyangiitis (GPA) represents a diagnostic challenge in otolaryngology, particularly when manifesting as isolated supraglottic involvement—a rare presentation accounting for <5% of ENT manifestations.1,2 While classic GPA features sinonasal or subglottic disease,1,3 emerging evidence suggests localized laryngeal variants may constitute a distinct phenotype,4,5 often misdiagnosed initially due to nonspecific symptoms. 6 Current knowledge gaps persist regarding optimal management of epiglottic GPA, where diagnostic delays risk airway compromise and treatment strategies must balance efficacy against toxicity.7,8 This case of histopathologically-confirmed epiglottic GPA highlights 3 critical aspects: (1) the indispensability of Proteinase 3 Anti-Neutrophil Cytoplasmic Antibody (PR3-ANCA) testing and biopsy in atypical laryngeal lesions,6,9 (2) the superior outcomes achieved with rituximab-based protocols,7,10 and (3) the necessity for prolonged surveillance given high relapse rates in laryngeal disease.11,12 Through this presentation, we expand upon Belloso et al’s seminal description of supraglottic stenosis 4 and Aono et al’s isolated epiglottic case, 5 while reinforcing recommendation for multidisciplinary management of otolaryngologic GPA. 9
Case Presentation
A 72-year-old woman with no significant medical history presented with a 5 year history of progressive dysphagia, dysphonia, and exertional dyspnea that had recently become debilitating. Indirect laryngoscopy revealed thickening of the epiglottal free edge with characteristic supraglottic sausage-like edema and a smooth, right-sided epiglottic mass, while vocal cord mobility and piriform sinuses remained normal (Figure 1). Initial laboratory studies revealed leukocytosis (White Blood Cells 14.2 × 10³/µL) with eosinophilia (8%), markedly-elevated inflammatory markers (C-Reactive Protein 42 mg/L, Erythrocyte Sedimentation Rate 78 mm/hour). Neck Computed Tomography (CT) demonstrated moderate, right-lateralized epiglottic thickening (Figure 1). Three consecutive days of acid-fast bacillus smears ruled out tuberculosis.
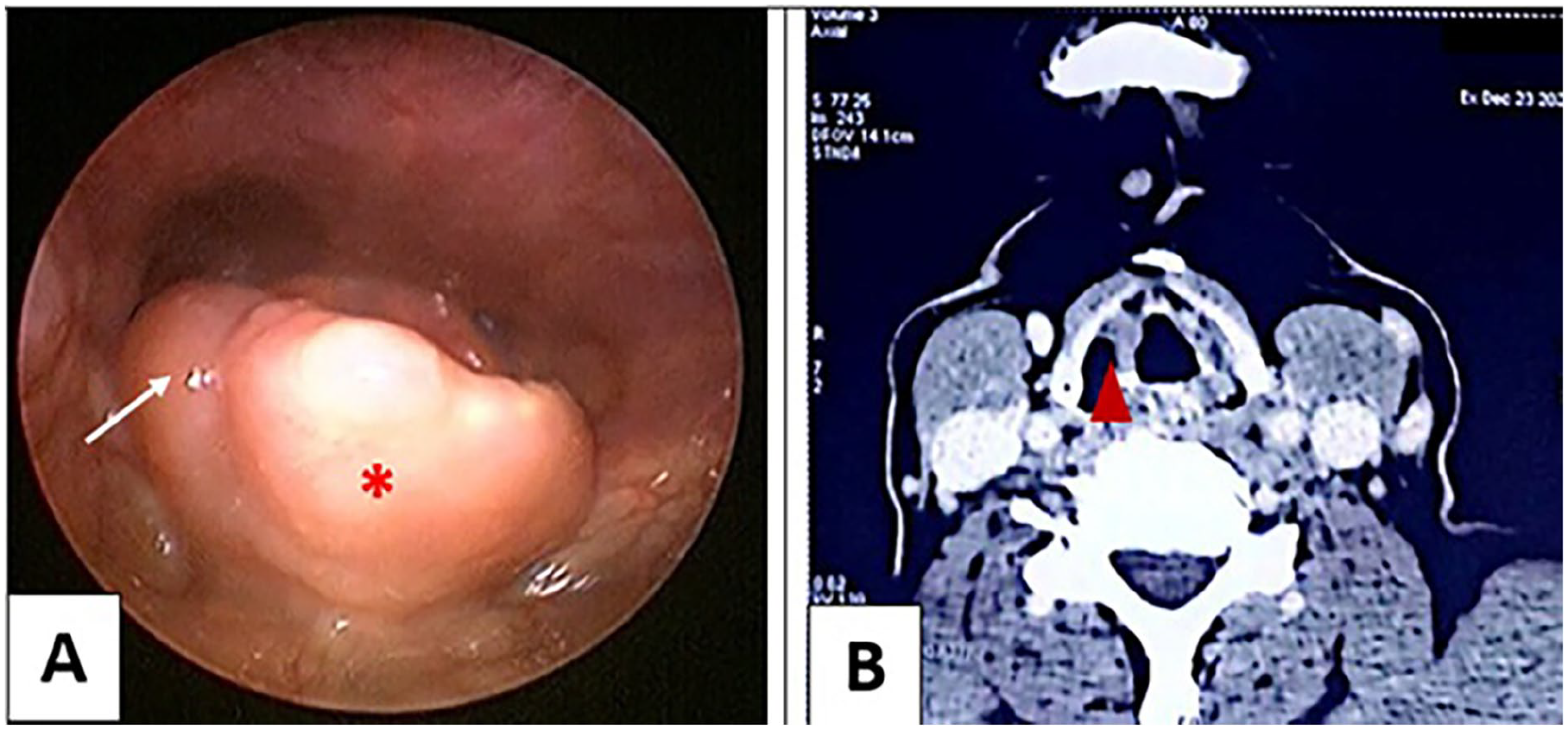
Laryngoscopic and radiologic findings in epiglottic granulomatosis with polyangiitis. (A) Indirect laryngoscopy showing thickened epiglottic free edge with supraglottic “sausage-like” edema (asterisk) and a smooth right-sided mass (white arrow). (B) Axial CT neck with contrast revealing right-lateralized epiglottic thickening (arrowhead) without cartilage destruction.
Direct laryngoscopy with biopsy confirmed GPA through histopathological identification of necrotizing granulomas, vasculitis, and multinucleated giant cells (Figure 2).
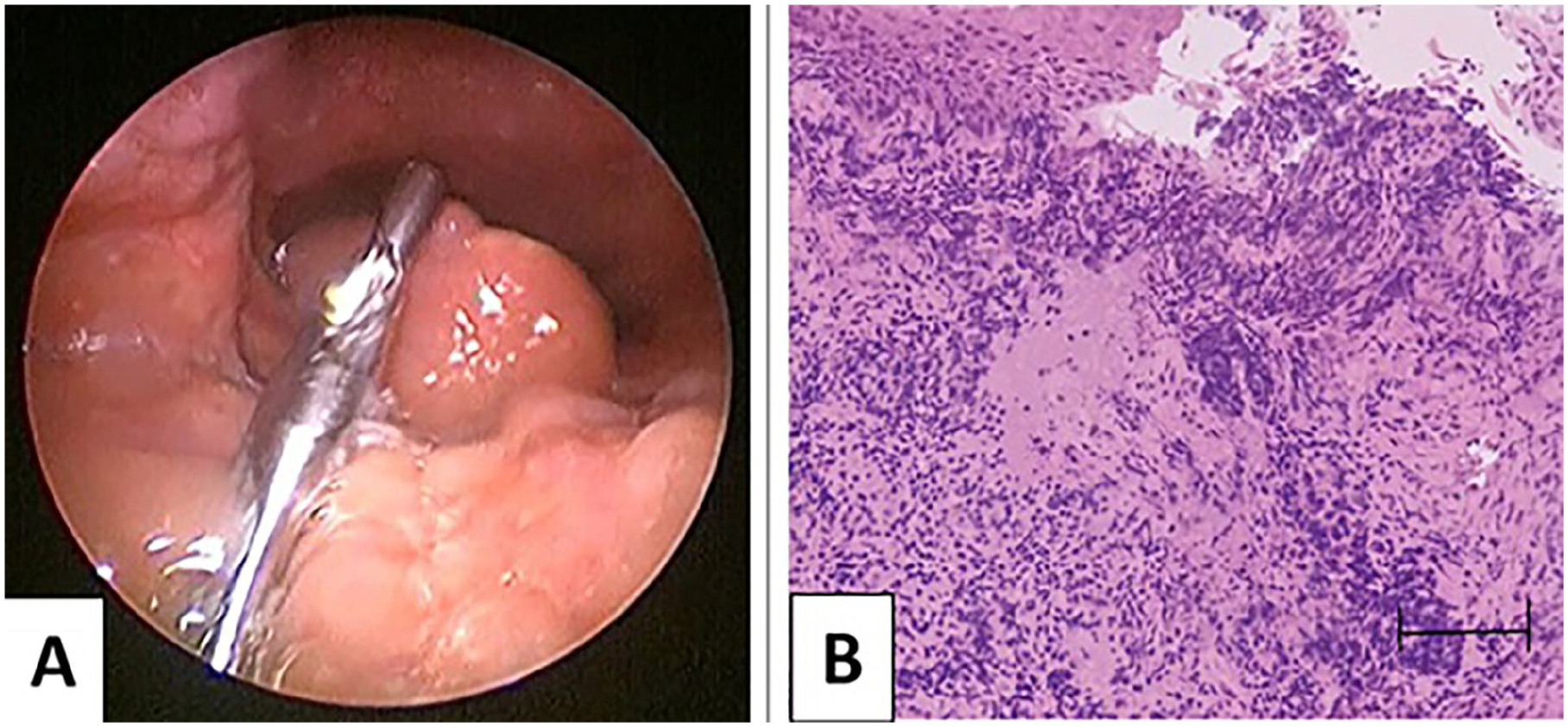
(A) Direct laryngoscopy with biopsy site at the right epiglottic mass. (B) Hematoxylin & eosin (H&E) staining (40×) demonstrating necrotizing granulomas, vasculitis, and multinucleated giant cells (black arrow).
Supportive diagnostic findings included positive cytoplasmic-ANCA (c-ANCA) with anti-proteinase 3 antibodies (PR3-ANCA) at 65 IU/mL (normal <3). Treatment was initiated with high-dose corticosteroids (1 mg/kg/day prednisone equivalent) combined with rituximab (375 mg/m² weekly for 4 weeks), followed by a carefully-tapered regimen: reduction by 10 mg every 2 weeks until 20 mg/day, then by 5 mg monthly until reaching a 5 to 7.5 mg/day maintenance dose for 18 to 24 months to prevent relapse in this critical anatomic site. The patient showed dramatic symptomatic improvement within 2 weeks, and 1 month follow-up laryngoscopy demonstrated complete resolution of the epiglottic lesions. The patient remains under regular follow-up to date, with excellent disease control and no evidence of epiglottic recurrence.
Discussion
This case of isolated epiglottic GPA expands the limited understanding of supraglottic involvement in this disease, which accounts for fewer than 5% of otolaryngologic manifestations according to recent analyses.1,2 While subglottic stenosis remains the most well-documented airway complication,1,3 our patient’s presentation with purely-epiglottic disease aligns with rare cases described by Belloso et al 4 and Aono et al, 5 reinforcing that GPA should be considered in atypical laryngeal lesions unresponsive to conventional therapy. The diagnostic challenges we encountered mirror those reported by Srouji et al, 6 where 40% of patients with GPA experienced delayed diagnosis due to nonspecific initial symptoms, particularly when limited to single anatomic sites. Histopathological confirmation proved critical in our case, as emphasized by Borner et al, 13 who demonstrated that even ANCA-negative localized disease (seen in 20%-30% of cases) can be reliably diagnosed through biopsy demonstrating necrotizing granulomas and vasculitis. The presence of PR3-ANCA antibodies in our patient supported the diagnosis, consistent with findings by Trimarchi et al 9 showing 85% to 90% seropositivity in otolaryngologic GPA. Our treatment approach using rituximab followed Stone et al’s 7 landmark protocol demonstrating superior efficacy to cyclophosphamide for remission induction, with Miloslavsky et al 11 further establishing its role in preventing relapse—particularly relevant given the 50% to 70% relapse rate in ENT-localized disease noted by Martinez Del Pero et al. 10 The rapid mucosal healing we observed correlates with Comarmond and Cacoub’s 8 findings that 78% of otolaryngologic manifestations show a significant improvement within 3 months of rituximab initiation. However, the need for prolonged maintenance therapy in our case reflects Wojciechowska and Kręcicki’s observation that laryngeal GPA requires extended immunosuppression (typically 18-24 months) compared with sinonasal disease. 12 The absence of systemic involvement in our patient aligns with Comarmond and Cacoub’s 8 description of localized GPA variants that may progress systemically without treatment. Our multidisciplinary approach echoes Reinhold-Keller et al’s 14 demonstration that coordinated ENT/rheumatology care improves long-term outcomes, reducing mortality from 28% to 8% in their 15 year follow-up. The excellent response in our case supports Polychronopoulos et al’s 3 conclusion that early-biologic therapy prevents irreversible airway damage in GPA. This case underscores recommendation for prompt biopsy of atypical laryngeal lesions, 9 particularly given Morales-Angulo et al’s finding that 30% of GPA patients present initially with only ENT symptoms. 15 While our patient maintained remission, data suggest ongoing vigilance is needed, as 60% of relapses occur within 2 years. 16 The anatomic specificity of epiglottic involvement in our case expands upon Alam et al’s 1 classification of upper airway GPA manifestations, suggesting this rare localization may represent a distinct phenotypic variant requiring tailored management strategies.
Conclusion
This case reinforces that isolated epiglottic GPA, although rare, demands prompt recognition given its potential for airway obstruction.1,5 The diagnostic triad of histopathology, Anti-Neutrophil Cytoplasmic Antibody (ANCA) serology, and characteristic imaging proved essential, while rituximab demonstrated remarkable efficacy as both induction and maintenance therapy.10,11 Long-term remission achieved in our patient supports most of the literature’s conclusions,7,11 although the 60% relapse risk noted by Greco et al 16 necessitates ongoing vigilance. Our experience confirms assertion that localized disease requires tissue diagnosis 13 and aligns with the call for early-biologic therapy in airway involvement. 3 Future studies should address the need for phenotype-specific treatment algorithms, 12 particularly for rare manifestations like epiglottic GPA that challenge conventional diagnostic frameworks.
Footnotes
Acknowledgements
All authors approved the final version and have the agreement to be responsible for all aspects of the work in ensuring that questions related to the accuracy.
Ethical Considerations
Our institution does not require ethics approval for reporting individual cases.
Consent to Paticipate
Written consent was obtained from the patient.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
