Abstract
Dermoid cysts, also known as mature cystic teratomas (MCTs), are congenital tumors that originate from all 3 germ layers. While commonly found in the ovary or testis, they are rare occurrences in the floor of the mouth. This case report describes a rare case of neonatal cleft palate with MCT of the floor of the mouth. The report provides a comprehensive analysis of the clinical manifestations, imaging characteristics, pathological findings, and surgical management strategies employed in this rare case to enhance clinicians’ knowledge regarding uncommon tumors in the head and neck of newborns. Through the discussion of this case, the importance of preoperative evaluation, rapid intraoperative diagnosis, and long-term postoperative follow-up is further emphasized.
Mature cystic teratoma (MCT) is a congenital tumor containing tissue components from the 3 embryonic germ layers (ectoderm, mesoderm, and endoderm). While MCT commonly manifests in reproductive organs such as the ovaries and testicles, its occurrence in atypical locations like the neck, oral floor, and sacrococcygeal region is rare. The disease is typically seen in young and elderly people, but is rare in children. 1 The incidence of MCTs in the head and neck of newborns is extremely low, with teratomas in the floor of the mouth being particularly rare. 1 Given the potential for tumors to cause neonatal respiratory distress, swallowing difficulties, and voice changes, prompt diagnosis and intervention are imperative. In this case, the clinical features, diagnostic methods, and surgical treatment of neonatal MCT were discussed in detail, aiming to provide more reference and guidance for clinicians.
Case Introduction
The patient, a full-term female newborn, was transferred to our facility shortly after birth due to respiratory distress, feeding challenges, and the identification of a mass in the floor of the mouth. The neonate was delivered via uncomplicated vaginal birth with a favorable Apgar score of 9. Upon physical examination, a substantial cystic mass was observed at the base of the mouth, accompanied by tongue elevation and mild respiratory obstruction (Figure 1A). In addition, a concealed cleft of the soft palate was noted (Figure 1B). Preliminary consideration of cystic lesions of the floor of the mouth necessitates further imaging examination to confirm the diagnosis. To assess the size, location, and relationship with the surrounding tissue of the tumor, the child underwent magnetic resonance imaging (MRI) of the head and neck. MRI showed a well-defined cystic mass measuring ~3.5 cm in diameter within the floor of the mouth. The T2-weighted high signal inside the mass indicated the potential inclusion of adipose tissue (Figure 2A and B). According to the imaging features, a diagnosis of MCT was considered more likely. Given the tumor’s compression on the respiratory tract, impacting the child’s breathing, surgical resection was deemed the optimal treatment approach. The surgery was performed through the oral approach under general anesthesia (Figure 3). The boundary between the tumor and other tissues of the floor of the mouth was distinct, with no evident signs of invasive growth. Complete excision of the entire mass was achieved, leading to prompt alleviation of respiratory tract compression symptoms postoperatively, without obvious complications.
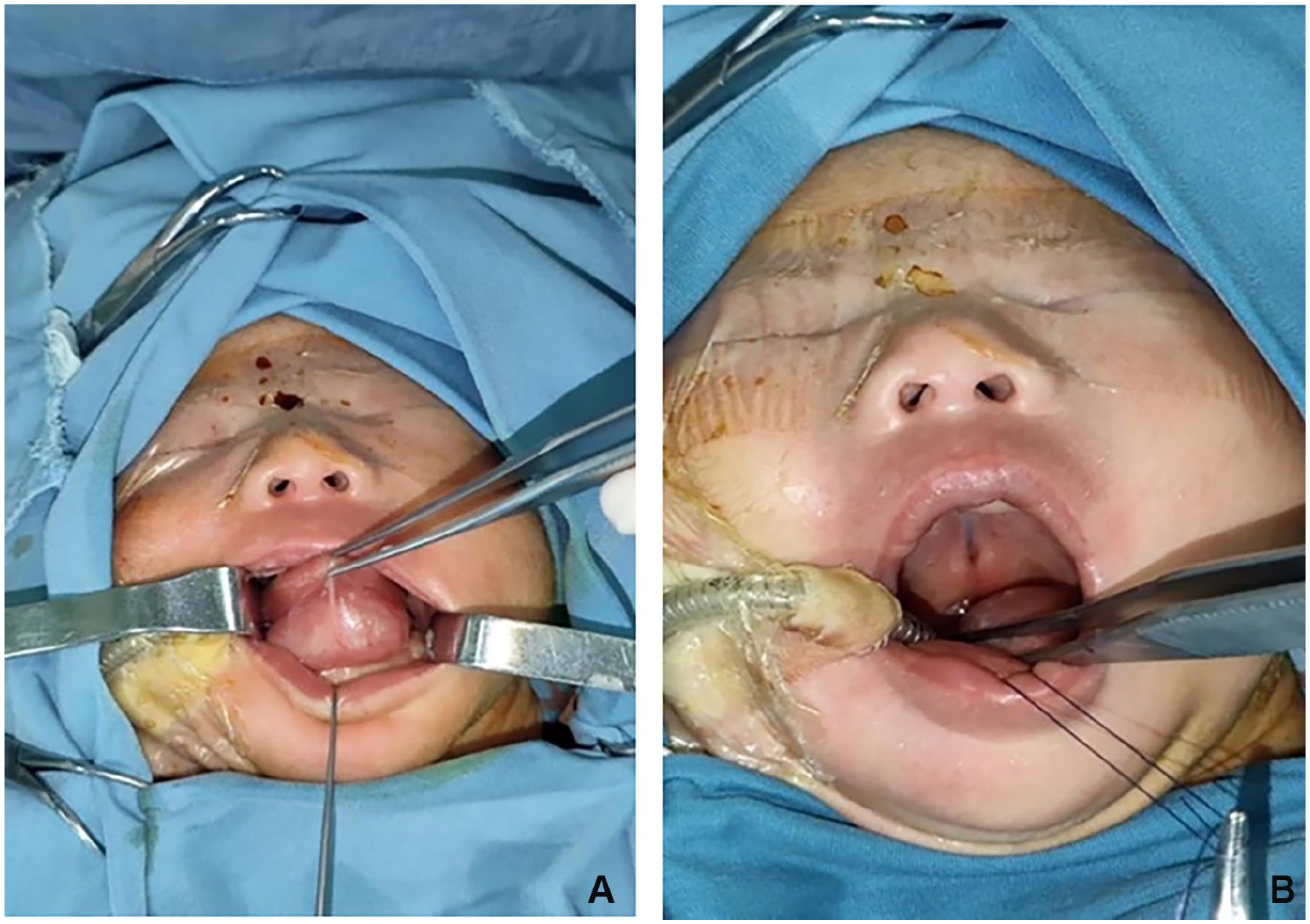
(A) A substantial cystic mass was observed at the base of the mouth, accompanied by tongue elevation. (B) A concealed cleft of the soft palate.
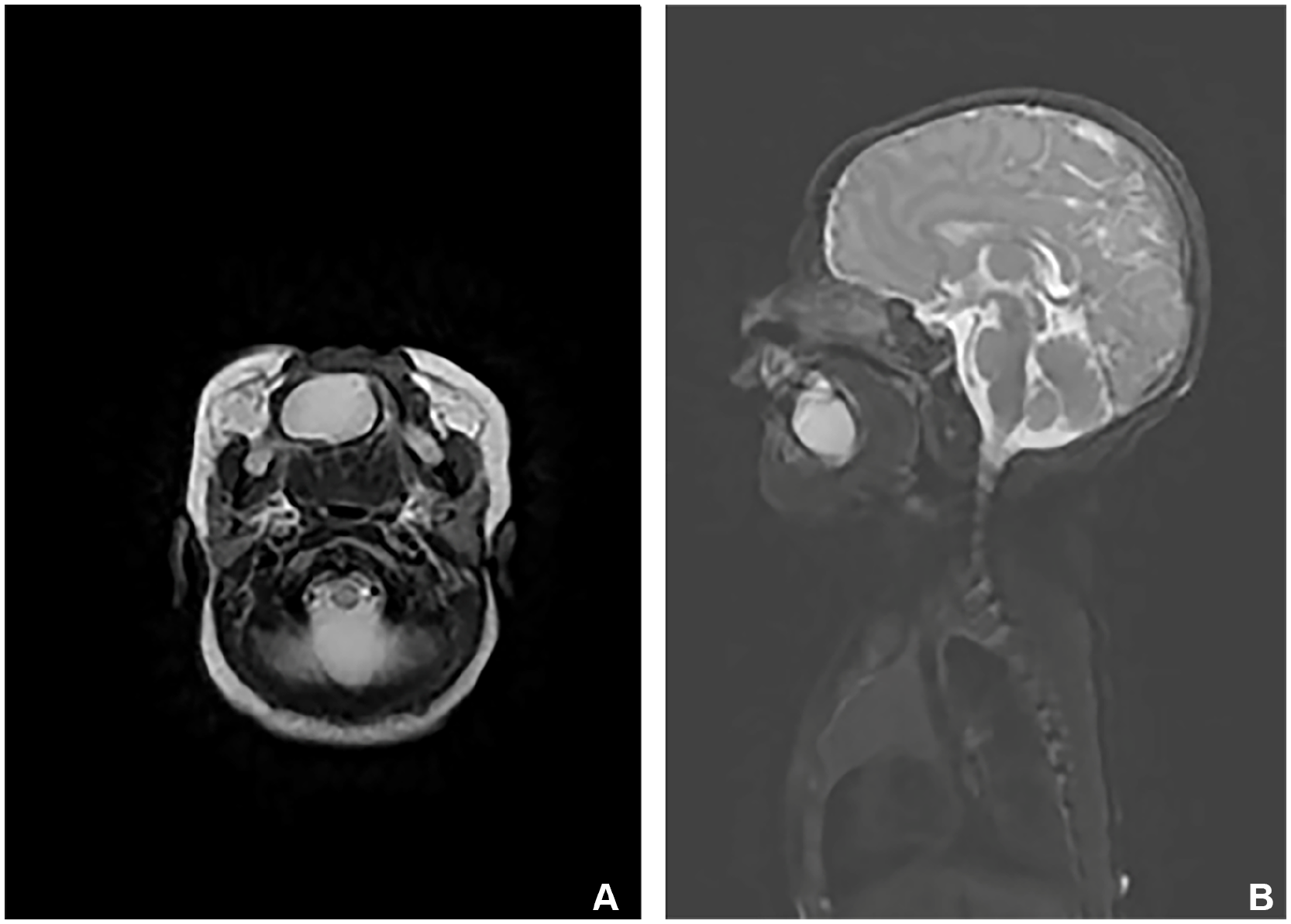
(A, B) MRI showed a well-defined cystic mass measuring ~3.5 cm in diameter within the floor of the mouth. MRI, magnetic resonance imaging.
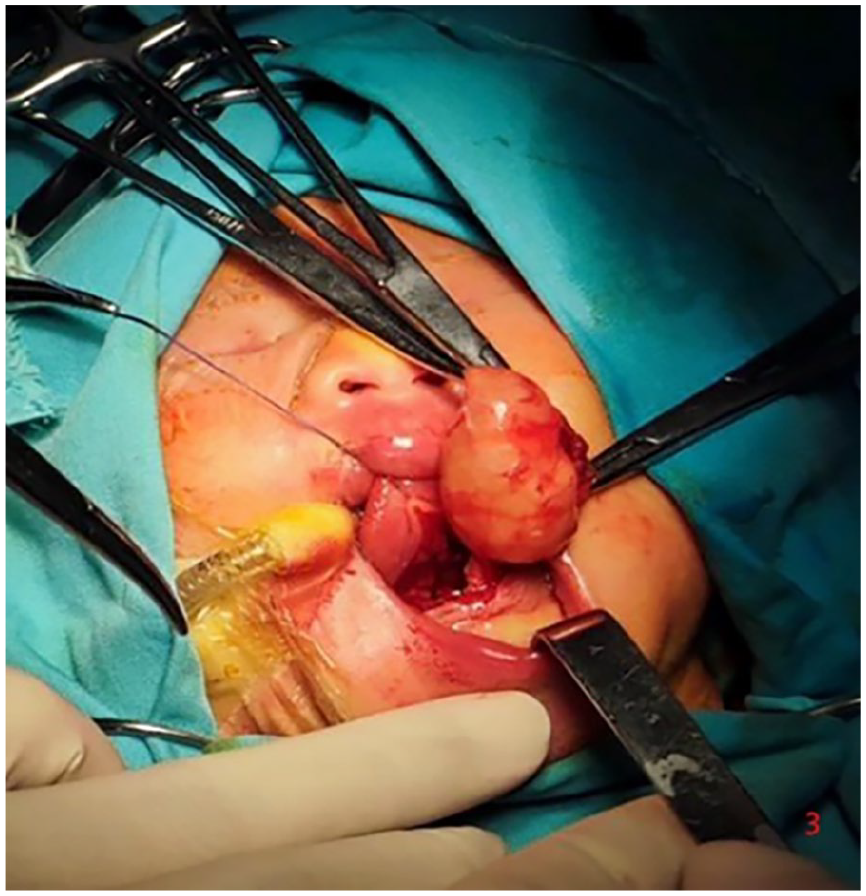
Sublingual cystic mass, yellow–white, complete capsule, about 35 × 25 × 17 mm.
The excised mass, upon postoperative examination, appeared as a complete cystic structure with a yellowish–white hue, measuring 35 × 25 × 17 mm. It exhibited a smooth surface, a cystic interior containing slightly turbid grayish–white mucus, and a cyst wall thickness ranging from 0.1 to 0.2 cm. Histological observation showed that the tumor was composed of 3 germ layers. The cyst wall was lined with skin squamous epithelium and gastrointestinal mucus columnar epithelium, alongside discernible skin appendages, adipose tissue, and striated muscle fibers. Notably, no immature components or malignant changes were observed (Figure 4). Subsequent follow-up examinations were conducted every 6 months, with a 3-year follow-up revealing no signs of recurrence.
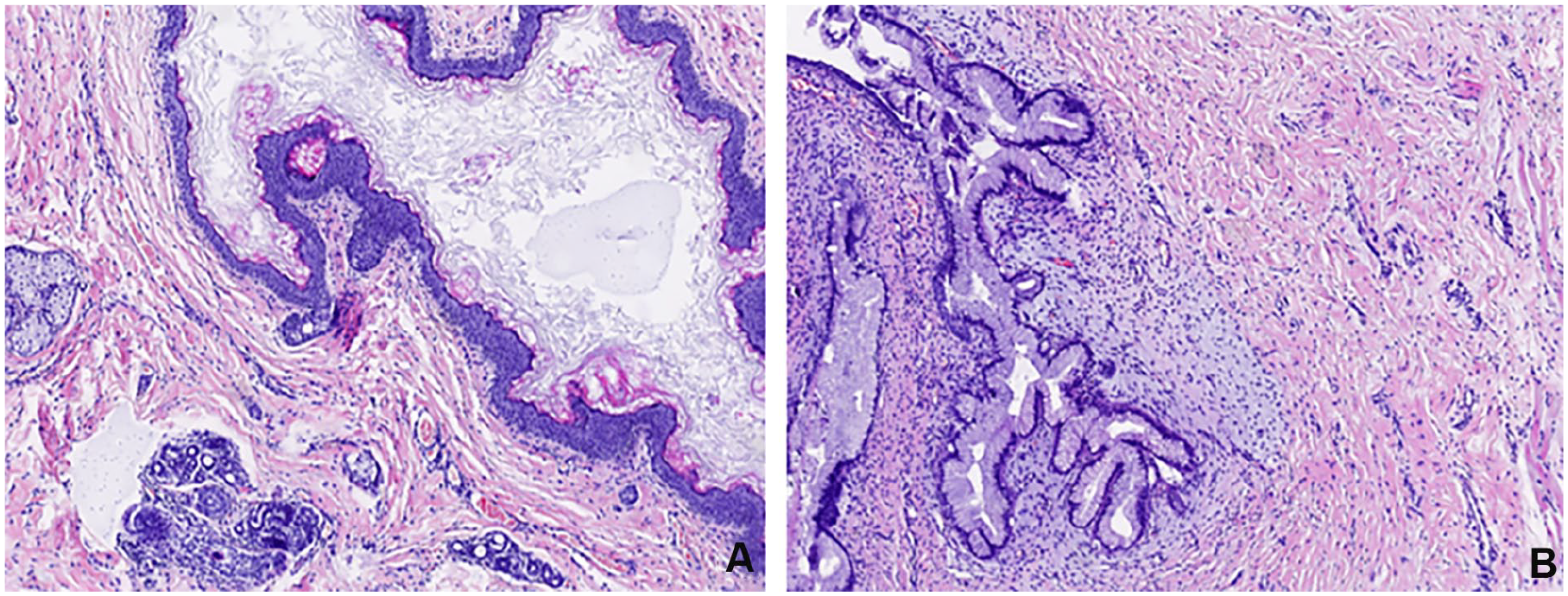
(A) Skin appendages, fat, and striated muscle tissue can be seen around the squamous epithelium of the cyst wall lining skin. (B) Viscous columnar epithelium and striated muscle tissue of the gastrointestinal tract.
Discussion
The incidence of MCT in the head and neck of newborns is relatively rare, especially in the floor of the mouth. 2 Teratomas in this area can significantly impact respiratory function and feeding due to their special location, thus increasing the complexity of diagnosis and treatment. In cases like the one presented here, the presence of a large MCT in the floor of the mouth can lead to a “heavy tongue” presentation, occupying oral cavity space and resulting in respiratory symptoms postnatally. Oropharyngeal cystic teratomas typically develop between the seventh and ninth weeks of gestation. Around 6% of these teratomas are associated with other congenital anomalies, such as bifid tongue, cleft lip, and cleft palate. Among these, cleft lip and cleft palate are the most common. This occurs because the tumor’s growth interferes with the normal fusion of the bilateral palatine processes. A literature review by Liang et al identified 16 cases of teratomas associated with cleft lip and palate. 3 Therefore, during clinical examinations, it is crucial to carefully inspect the palatal anatomy to avoid missing this condition. Timely imaging studies are crucial to delineate the extent of the lesion for prompt intervention. In this case, the operation through the oral approach not only successfully removed the tumor but also avoided damage to other structures. The postoperative recovery was favorable, with a prompt improvement in respiratory function noted.
Compared with common cystic masses such as dermoid cysts and epidermoid cysts, the complexity of MCT lies in its diverse tissue components and irregular growth patterns, leading to more heterogeneous clinical and imaging presentations. MRI in this case indicated a cystic lesion containing fat-like elements, which is a typical feature of teratomas. Gordon et al outlined the distinctions in 2013 as follows 4 : (1) epidermoid cyst – characterized by complexity with a squamous epithelial lining lacking dermal appendages; (2) dermoid cyst – featuring a stratified squamous epithelial lining with skin appendages present in the underlying connective tissue, including sebaceous glands, hair follicles, and sweat glands; (3) MCT – in addition to the skin appendages, it also contains mesodermal components, such as bone, muscle, respiratory tract, and gastrointestinal tissue, with the possibility of malignant transformation. 5 Therefore, consistent postoperative monitoring is of paramount importance. In neonatal cases, postoperative follow-up should primarily concentrate on monitoring tumor recurrence, observing the growth and development of children, and screening for other potential teratomas or undiscovered lesions.
Imaging examinations, including ultrasound, computed tomography (CT), and MRI, are of great value in the evaluation of cystic tumors in the head and neck. Ultrasound may help to distinguish solid, vascular, and cystic lesions. CT and MRI imaging offer precise localization, with MRI T2-weighted hyperintensity and T1-weighted hypointensity typically indicating the presence of cystic structures. 6 In this case, MRI not only provides information on the size, boundary, and internal structure of the tumor but also suggests the fat content of the tumor, which is a common feature of mature teratoma. Although imaging provides important clues to the nature of cystic tumors, the final diagnosis still depends on pathological examination.
Pathological examination plays a pivotal role in the accurate diagnosis of MCT, particularly in distinguishing it from immature teratoma. The histological analysis in this case revealed the presence of skin squamous epithelium, adipose tissue, muscle, gastrointestinal mucous columnar epithelium, skin appendages, and other tissues, consistent with the classical features of mature teratomas. Importantly, no immature components or malignant changes were found. This diagnosis is essential for the development of follow-up treatment strategies.
Since MCT involves complex diagnosis and treatment processes, multidisciplinary collaboration holds significant importance. The coordinated efforts of the imaging, pathology, and surgical teams were instrumental in swiftly and effectively diagnosing and treating the presented case. In particular, prompt intraoperative pathological assessment is critical for characterizing the tumor’s nature and informing the extent of surgical intervention. This case also highlights the importance of interdisciplinary cooperation in the diagnosis and treatment of rare head and neck tumors in neonates.
Conclusion
Neonatal MCT of the floor of the mouth is a rare congenital tumor that poses challenges in clinical recognition due to its resemblance to other cystic lesions, thus increasing the risk of misdiagnosis. Through comprehensive imaging evaluation (such as MRI) combined with pathological analysis, early and accurate diagnosis can be achieved. The success of the operation depends not only on the technique of tumor resection but also on the comprehensive evaluation before operation and close follow-up after operation. In addition, the presence of concurrent cleft palate in affected individuals necessitates a more comprehensive diagnostic approach by healthcare providers to prevent diagnostic oversights. Clinicians should adopt individualized strategies when dealing with rare tumors, while maintaining sensitivity to the literature, referring to similar cases reported in the past, and constantly accumulating experience in diagnosis and treatment.
Footnotes
Ethics Considerations
The studies involving human participants were reviewed and approved by the ethics committee of Qingdao Women and Children’s Hospital.
Consent to Participate
The patient’s legal guardian provided written informed consent to participate in this study.
Author Contributions
Junwei Wang: conception and design of study; acquisition of data, data analysis, and interpretation; drafting of manuscript and critical revision; approval of final version of manuscript. Yuelin Qin: conception and design of study; acquisition of data; data analysis and interpretation; drafting of manuscript and critical revision; approval of final version of manuscript. Fang Wang: acquisition of data. Xuecai Yang: drafting of manuscript and critical revision; approval of final version of manuscript. Cuimei Zhang: drafting of manuscript and critical revision; approval of final version of manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Qingdao Key Medical and Health Discipline Project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
