Abstract
Objectives:
To assess the prognosis of young patients (≤40 years old) with head and neck squamous cell carcinoma (HNSCC), focusing on the preoperative Systemic Inflammation Response Index (SIRI).
Methods:
Between January 2007 and February 2017, 175 young patients with HNSCC (≤40 years old) who underwent radical surgery were retrospectively enrolled in this study. The patients were randomly divided into a training cohort (N = 131) and a validation cohort (N = 44). The SIRI is defined as the absolute neutrophil count (×10⁹/L) multiplied by the absolute monocyte count (×10⁹/L), divided by the absolute lymphocyte count (×10⁹/L) in peripheral blood, all measured within one week prior to radical surgery. Univariate and multivariate Cox regression analyses were conducted to identify variables associated with survival outcomes, which were then used to construct and evaluate a predictive nomogram.
Results:
In both the training and validation cohorts, patients were classified into low- and high-SIRI groups based on a cutoff value of 0.87, which was determined by receiver operating characteristic analysis. This SIRI cutoff effectively stratified patients into two distinct prognostic groups with significant survival differences. Multivariable Cox analysis identified the presence of lymphovascular invasion and the high preoperative SIRI as significant independent prognostic factors associated with poorer cancer-specific survival (CSS) in young patients with HNSCC. Using these variables, a predictive model for 5 year CSS was constructed and visualized as a nomogram. The model demonstrated strong predictive performance, with a C-index of 0.744 [95% CI (0.643-0.845)] in the training cohort and 0.839 [95% CI (0.740-0.938)] in the validation cohort.
Conclusion:
Data from preoperative SIRI assessment, coupled with the presence of pathological adverse features, serve as valuable references for risk stratification in young patients with HNSCC.
Introduction
Head and neck squamous cell carcinoma (HNSCC) is the seventh most common malignancy worldwide, with more than 890,000 cases diagnosed in 2020. 1 It is also one of the most prevalent types of cancer in Taiwan. 2 In addition to the TNM staging system, numerous prognostic factors have been investigated for their impact on treatment outcomes in patients with HNSCC, including adverse pathological features, nutritional status, infections with human papilloma virus.3-9 HNSCC typically occurs in the fifth to eighth decade of life. 10 However, there have been conflicting reports regarding the outcomes of HNSCC in young adults, with some suggesting that tumors are more aggressive in this population and therefore require more aggressive treatment.11,12
Inflammatory biomarkers are considered to reflect the interaction between the tumor microenvironment and the host immune system.13,14 The Systemic Inflammation Response Index (SIRI) is one such inflammatory biomarker that combines the analysis of peripheral blood counts of neutrophils, monocytes, and lymphocytes. The SIRI was first investigated as a novel prognosticator in pancreatic adenocarcinoma in 2016, 15 and a series of studies have shown its clinical significance in the prognosis of various malignancies.16-19 The high SIRI in HNSCC has already been identified as a negative prognostic factor in previous studies. However, these studies typically included patients of all age groups, using a cutoff value between 50 and 65 years.20-22 Currently, limited data are available regarding the impact of the pretreatment SIRI on survival outcomes in young patients with HNSCC. The objective of our study was to evaluate the potential prognostic impact of the preoperative SIRI and to construct a prognostic nomogram aimed at evaluating the prognosis of young patients with HNSCC who have undergone radical surgery.
Materials and Methods
Study Population
Consecutive patients who were histologically confirmed to have first primary HNSCC and underwent curative surgery with/without adjuvant therapy as primary treatment between January 2007 and February 2017 at Kaohsiung Chang Gung Memorial Hospital in Taiwan were retrospectively reviewed. Patients aged ≤40 years were eligible for this study. All the patients included had detailed clinical and pathological information available for review. Patients with (a) a history of any other cancer, (b) presence of distant metastasis at initial diagnosis, (c) prior curative treatment including induction chemotherapy, radiotherapy, or concurrent chemoradiotherapy before radical surgery, (d) clinical evidence of acute infection within 4 weeks prior to the blood tests, and (e) lack of preoperative laboratory data were excluded from this study.
The TNM classification was reclassified according to the 8th edition of the American Joint Committee on Cancer (AJCC) staging system. Adjuvant therapy for these patients after surgery was performed according to American National Comprehensive Cancer Network (NCCN) guidelines.
Study Design and Data Collection
We randomly assigned young patients with HNSCC from the current study to either the training or validation cohort in a 3:1 ratio. The predictive model was developed using patient characteristics and survival outcomes from the training cohort, while the validation cohort was used to evaluate the model’s performance.
Clinical characteristics and pathological variables of interest, such as gender, age, primary cancer location, cancer stage, and pathological features, including perineural invasion (PNI), lymphovascular invasion (LVI), surgical margin status, and extranodal extension (ENE), were collected and analyzed for their influence on survival.
The preoperative SIRI was defined as the absolute neutrophil count multiplied by the absolute monocyte count divided by the absolute lymphocyte count, with all values obtained from peripheral blood tests conducted within one week prior to radical surgery. The outcomes of interest were 5 year cancer-specific survival (CSS) and disease-free survival (DFS) rates. CSS was calculated as the period from the date of radical surgery to the date of death from HNSCC cause or last follow-up. DFS was calculated as the period from the date of radical surgery to the date of tumor relapse or last follow-up.
Statistical Analysis
All statistical analyses were performed using the SPSS 25.0 software (IBM Corp, Armonk, NY, USA) and R Studio. To determine the optimal cutoff for the SIRI, we applied the “survivalROC” package (Version 1.0.3.1) in R. We extracted the true-positive and false-positive predictions from the result object and identified the point on the curve with the highest Youden index (sensitivity + specificity − 1). The Kaplan-Meier method was used to estimate the probability of survival for each categorized factor. The log-rank test was employed to assess the statistical significance of each stratification factor, with two-tailed P < .05 was considered statistically significant. Factors that showed significance in the univariate analysis were included in the Cox regression model to determine whether they were independent risk factors for survival outcomes and estimated their effects on CSS and DFS.
In addition, we created a prognostic nomogram that incorporated significant factors identified by multivariate analyses in training cohort using the R software “rms” package (Version 5.1–0; Vanderbilt University, Nashville, TN, USA) with endpoints of 5 year CSS. To ascertain the CSS prediction accuracy of the nomogram, a concordance index (C-index) was derived for the nomogram models. C-index values of 0.5 and 1.0 were considered indicative of random and perfect predictability, respectively. Additionally, a calibration plot was used to determine whether the predicted survival rate was consistent with the observed survival rate. Subsequently, the predictive discriminative ability of the model in cohorts were evaluated using time-dependent receiver operating characteristics (tROC) via the “timeROC” package. This study was approved by the Medical Ethics and Human Clinical Trial Committee of Chang Gung Memorial Hospital (Ethical Application Reference number: 202300725B0). This study was conducted in accordance with the principles of the Declaration of Helsinki.
Results
A total of 175 patients were enrolled in this study, and we randomly divided them into training (n = 131) and validation cohorts (n = 44) in a 3:1 ratio. The baseline clinicopathological characteristics of the two cohorts are presented in Table 1. Most of the patients were male (95.4% in the training cohort and 95.5% in the validation cohort). The most common tumor subsite in the training cohort was the oral cavity (n = 111, 84.7%), followed by the oropharynx (n = 9, 6.9%), hypopharynx (n = 8, 6.1%), and larynx (n = 3, 2.3%). Similarly, in the validation cohort, the most common tumor subsite was the oral cavity (n = 39, 88.6%), followed by the oropharynx (n = 3, 6.8%), hypopharynx (n = 1, 2.3%), and larynx (n = 1, 2.3%). For T classification, the training cohort included 41 patients with T1 tumors, 37 with T2 tumors, 26 with T3 tumors, 26 with T4a tumors, and 1 with a T4b tumor. The validation cohort included 12 patients with T1 tumors, 13 with T2 tumors, 8 with T3 tumors, and 11 with T4a tumors. For N classification, the training cohort was comprised of 85 patients with N0 disease, 13 with N1 disease, 16 with N2 disease, and 17 with N3 disease. The validation cohort included 32 patients with N0 disease, 1 with N1 disease, 5 with N2 disease, and 6 with N3 disease. The presence of PNI was found in 35 patients (26.7%) in the training cohort and in 13 patients (29.5%) in the validation cohort. LVI was observed in 16 patients (12.2%) in the training cohort and 5 patients (11.4%) in the validation cohort. ENE was present in 19 patients (14.5%) in the training cohort and 6 patients (13.6%) in the validation cohort. Additionally, 60 patients (45.8%) in the training cohort and 22 patients (50%) in the validation cohort received adjuvant therapy postoperatively. Regarding surgical margin status, 96 patients (73.3%) in the training cohort had a free surgical margin (≥5 mm), which was significantly higher than the 22 patients (50%) in the validation cohort (P = .011).
Patient Characteristics.
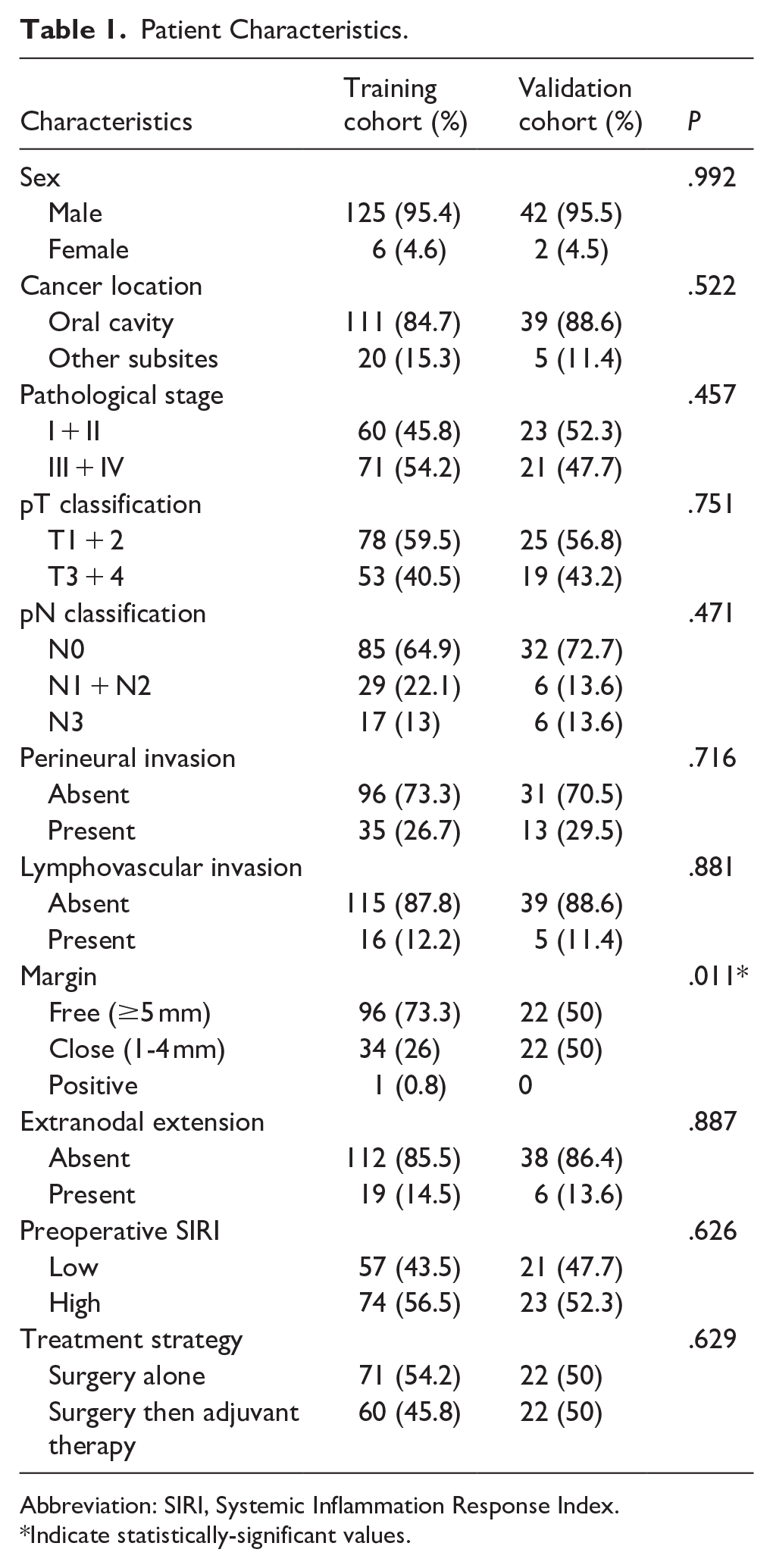
Abbreviation: SIRI, Systemic Inflammation Response Index.
Indicate statistically-significant values.
In the training cohort, the median preoperative SIRI was 0.945 (range: 0.219-4.695). Using ROC analysis, the optimal cutoff value for the preoperative SIRI was determined to be 0.87, with an area under the curve (AUC) of 0.654 (Figure 1). Based on this cutoff value, patients in the training cohort were divided into two groups: high SIRI (n = 74, SIRI ≥ 0.87) and low SIRI (n = 57, SIRI < 0.87). Similarly, patients in the validation cohort were classified using the same cutoff value of 0.87 into the high SIRI (n = 23, SIRI ≥ 0.87) and the low SIRI (n = 21, SIRI < 0.87) groups. In the training cohort, patients with the high preoperative SIRI were more likely to present with nodal positive disease, PNI, ENE, and were more likely to require adjuvant therapy than those with the low preoperative SIRI. In the validation cohort, the high preoperative SIRI was significantly associated with advanced T classification and PNI. These associations between the preoperative SIRI and clinicopathological variables are summarized in Table 2.
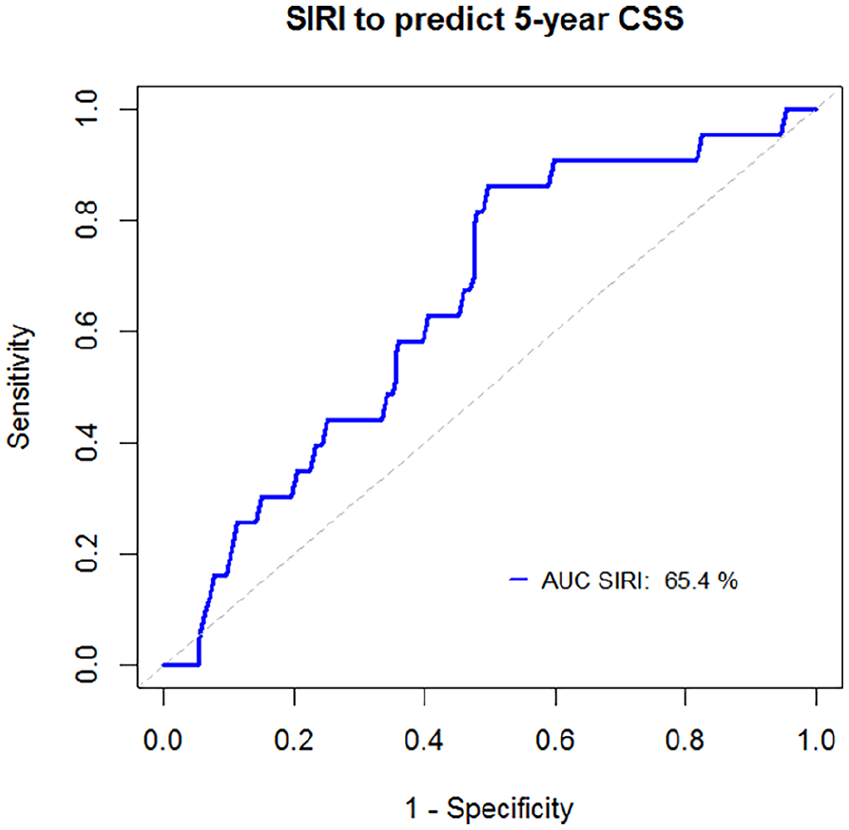
Receiver operating characteristic curve. Receiver operating characteristic curve of the preoperative SIRI for predicting CSS.
Association Analysis Between SIRI and Other Clinical Factors in Young Population of HNSCC.
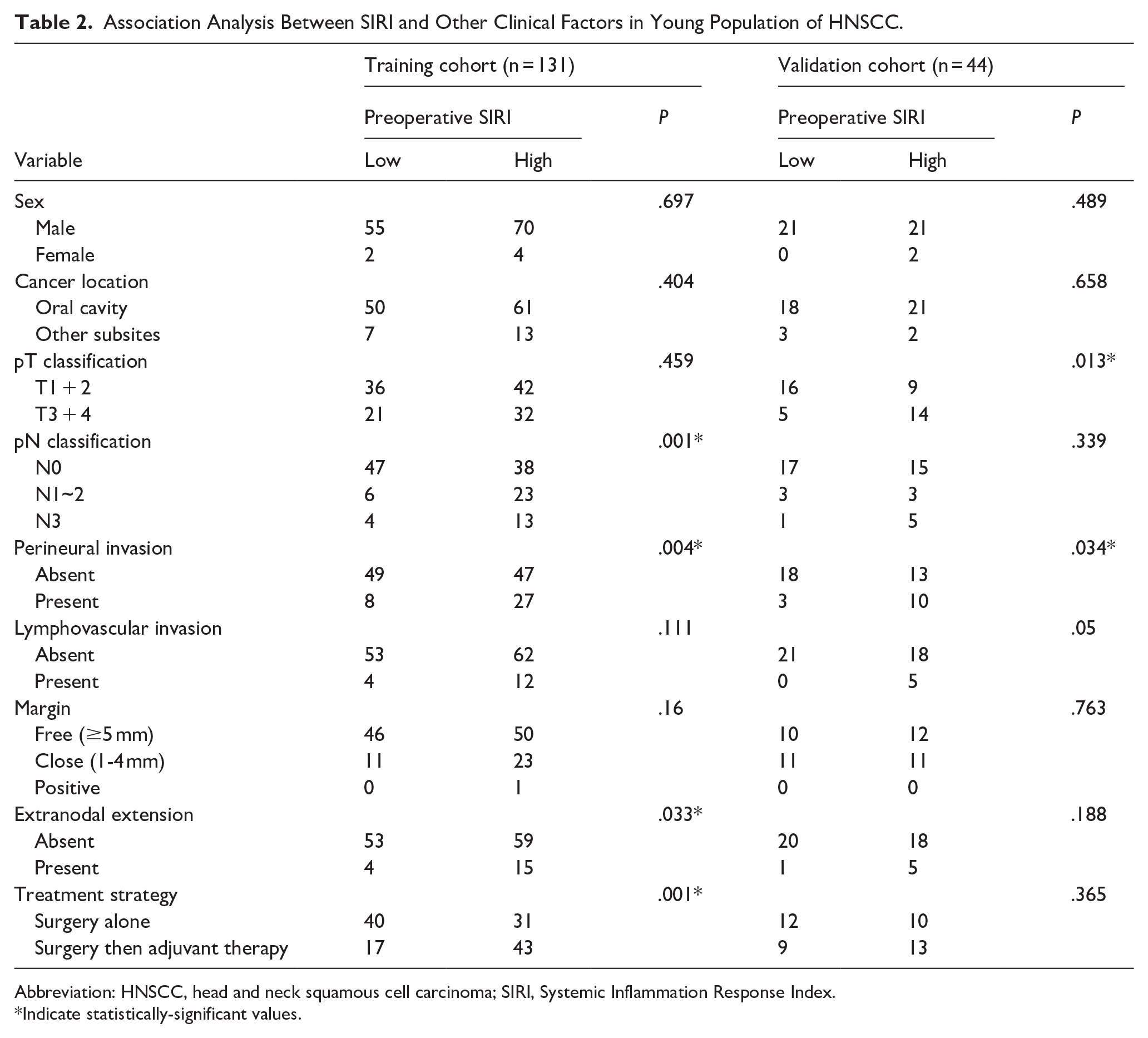
Abbreviation: HNSCC, head and neck squamous cell carcinoma; SIRI, Systemic Inflammation Response Index.
Indicate statistically-significant values.
The Prognostic Value of the SIRI for Young Patients with HNSCC
The median follow-up for the entire cohort was 75.2 months (interquartile range: 57.0-111.8 months). The five-year CSS rates were 83.5% in the training cohort and 86.4% in the validation cohort. The five-year DFS rates were 74.2% in the training cohort and 81.8% in the validation cohort. There was no significant difference in CSS nor in DFS between the two cohorts (CSS: P = .824; DFS: P = .259). The Kaplan-Meier analysis revealed that patients in the high-SIRI group had significantly-worse outcomes than those in the low-SIRI group, and this result remained in both the training and validation cohorts. In the training cohort, the five-year CSS and DFS rate were 74.8% and 65.3% for the high-SIRI group versus 94.6% and 85.7% for the low-SIRI group (CSS: P = .001; DFS: P = .011). Similarly, in the validation cohort, the 5 year CSS and DFS rates were 73.9% and 65.2 % for the high-SIRI group compared with 100% of CSS rate and 100% of DFS rate for the low-SIRI group (the log-rank test for CSS: P = .007; DFS: P = .003) (Figure 2).
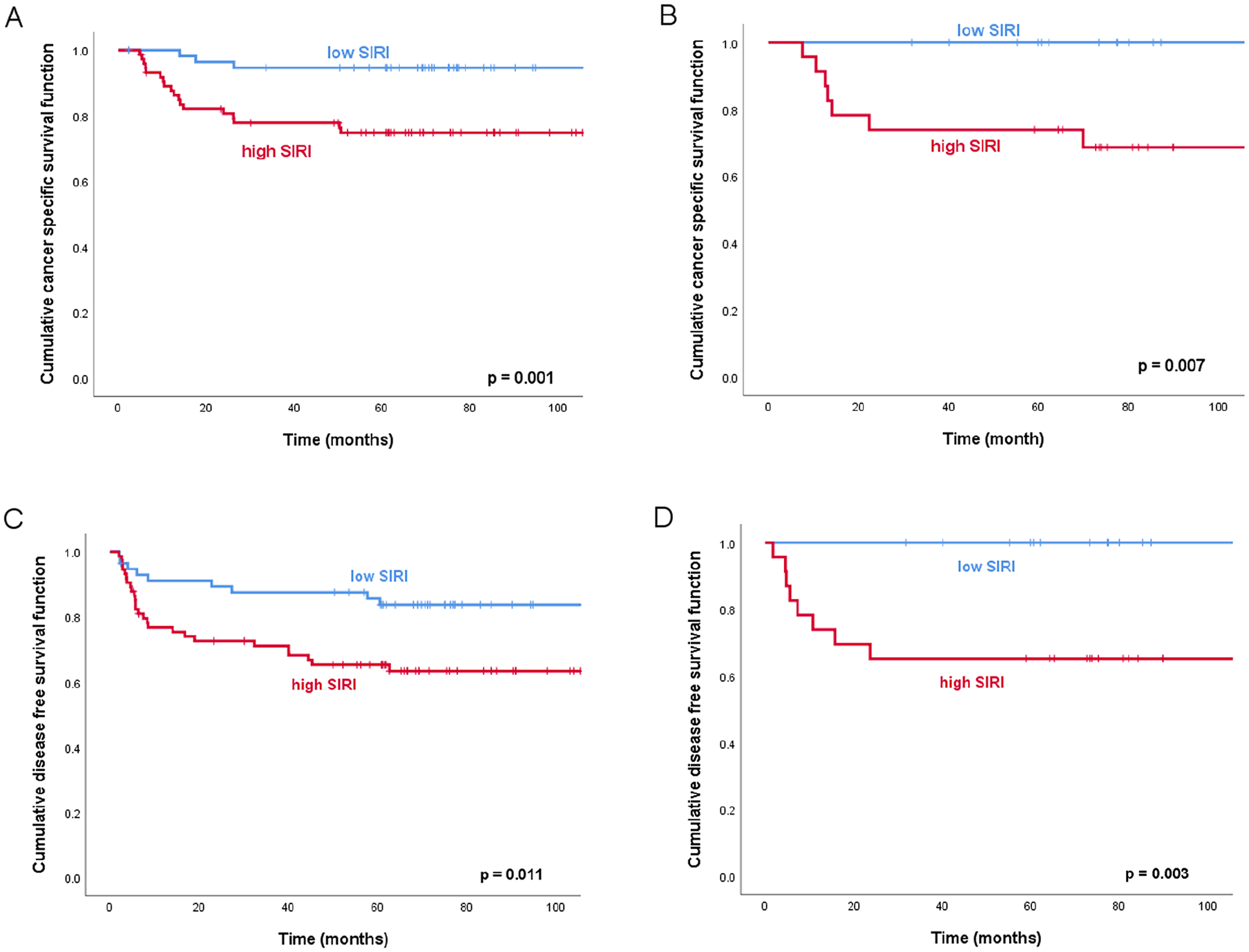
Kaplan-Meier survival curves. Kaplan-Meier survival curves according to the different preoperative Systemic Inflammation Response Index (SIRI) value. (A) cancer-specific survival in training cohort, (B) cancer-specific survival in validation cohort, (C) disease-free survival in training cohort, and (D) disease-free survival in validation cohort.
Identification of Independent Predictors
We explored the association between various clinicopathological factors and survival in young patients with HNSCC. In the univariate analysis of the training cohort, nodal positive disease, positive surgical margin, the presence of PNI, LVI, ENE, the need for adjuvant therapy, and high preoperative SIRI values were all associated with poorer 5 year CSS rates (P < .05). Regarding DFS, positive surgical margin, the presence of LVI, the need for adjuvant therapy, and high preoperative SIRI values were all associated with inferior DFS (P < .05) (Table 3).
Univariate Analysis of Survival Outcomes in Training Cohort.
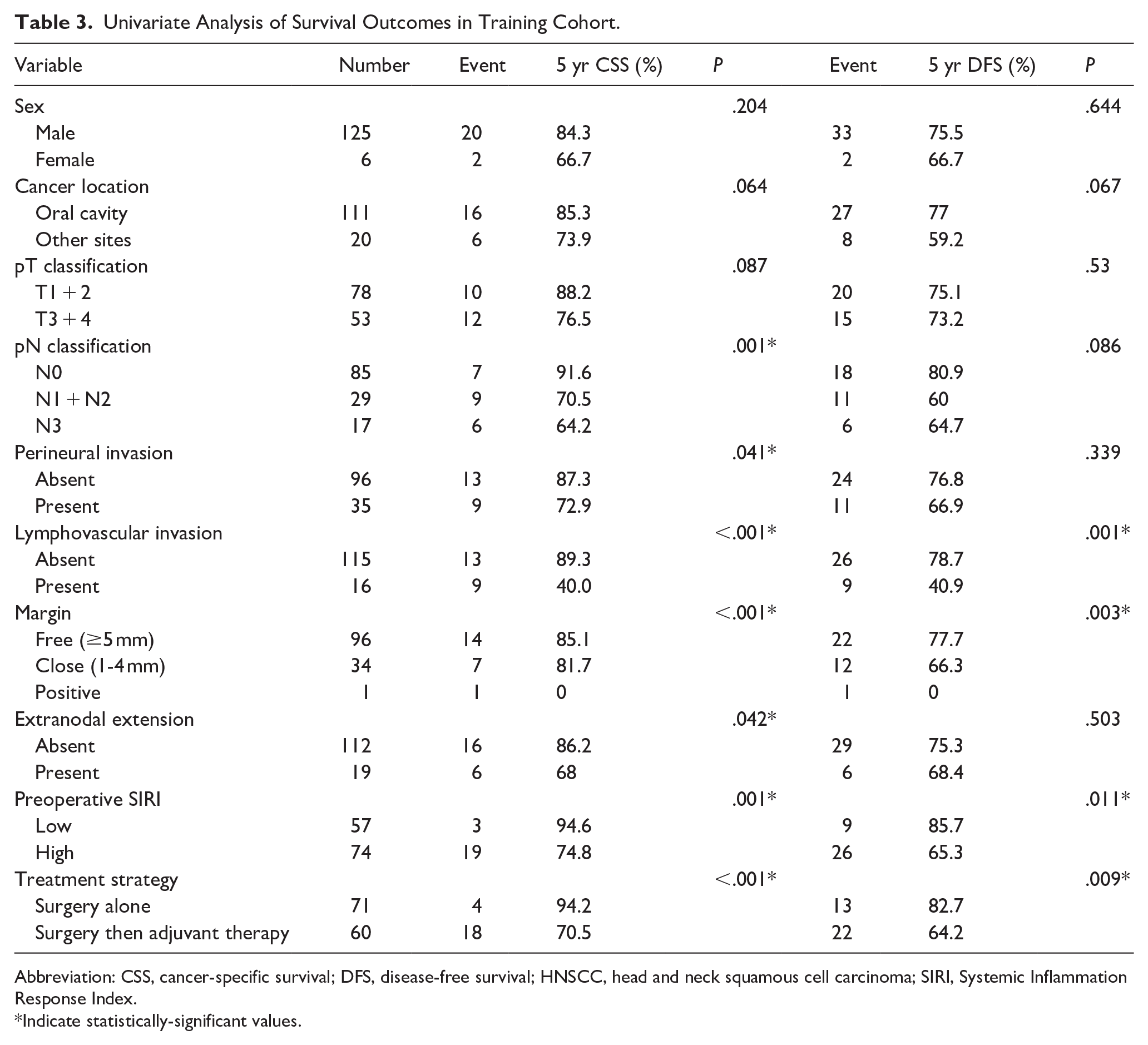
Abbreviation: CSS, cancer-specific survival; DFS, disease-free survival; HNSCC, head and neck squamous cell carcinoma; SIRI, Systemic Inflammation Response Index.
Indicate statistically-significant values.
Subsequently, multivariate Cox regression analysis was performed to evaluate the relationship between survival outcomes and the significant factors identified in the univariate analysis. The results are summarized in Table 4. The analysis demonstrated that a high preoperative SIRI was an independent negative prognostic factor for CSS [Hazard ratio (HR): 4.428, 95% CI (1.183-16.58), P = .027] and DFS [HR: 2.222, 95% CI (1.02-4.84), P = = .044). Additionally, the presence of LVI was identified as another significant independent negative predictor of CSS in this model, with a hazard ratio of 4.334 [95% CI (1.529-12.284), P = .006)
Multivariate Analysis of Factors Impacting Survival Among Training Cohort.
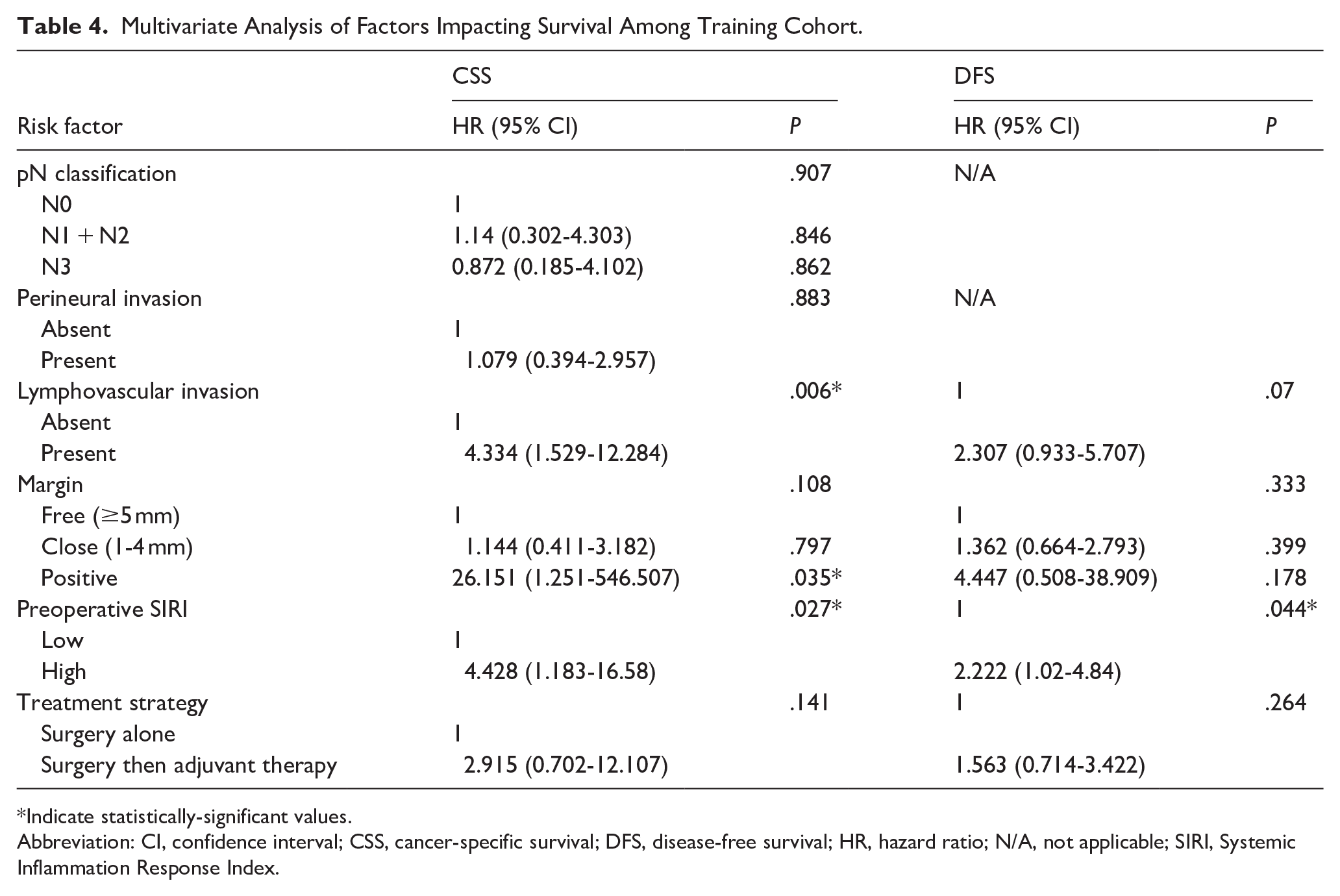
Indicate statistically-significant values.
Abbreviation: CI, confidence interval; CSS, cancer-specific survival; DFS, disease-free survival; HR, hazard ratio; N/A, not applicable; SIRI, Systemic Inflammation Response Index.
Regarding depth of invasion (DOI), among the 150 oral cavity cancer patients with available data, a greater DOI was associated with an increased risk of cancer-specific death and recurrence. In the validation cohort, a DOI > 10 mm significantly worsened 5 year CSS (70.6% vs 100%, P = .035) and DFS (64.7% vs 95.5%, P = .01). In contrast, these differences did not reach statistical significance in the training cohort (CSS: 77.6% vs 89.0%, P = .102; DFS: 75.4% vs 77.9%, P = .497).
Development of the Novel Prognostic Model Based on the SIRI
To enhance the accuracy of survival outcome predictions, a novel prognostic model was developed by integrating the significant prognostic factors identified in the multivariate analyses. The factors, including the preoperative SIRI and the presence of LVI, were combined to construct a nomogram designed to predict 5 year CSS in young patients with HNSCC (Figure 3). In this nomogram, each prognostic factor contributes a specific number of points, and the total score for an individual patient is calculated by summing these points. The 5 year CSS probability is then determined by locating the total score on the nomogram’s survival probability scale. For instance, if a young patient presented with LVI and the preoperative SIRI < 0.87, their total points were as follows: 0 + 71.68 = 71.68, resulting in respective probabilities of 5 year CSS rate 77.4%. In contrast, a young patient with no LVI but a preoperative SIRI ≥ 0.87 would have a total of 100 points, translating into an 84.2% probability of 5 year CSS. Calibration plots (Figure 4A for the training cohort, Figure 4B for the validation cohort) demonstrated good alignment between predicted and actual observed CSS, indicating that the model's predictions were highly accurate. The prognostic model also exhibited strong predictive performance, with a C-index of 0.744 [95% CI (0.643-0.845)] in the training cohort and 0.839 [95% CI (0.740-0.938)] in the validation cohort, surpassing the predictive ability of the traditional TNM staging system, which had C-index of 0.654 [95% CI (0.569-0.740)] and 0.782 [95% CI (0.706-0.933)] in the training and validation cohorts, respectively. Finally, time-dependent ROC curves (Figure 4C and D) further confirmed the superior predictive performance of the SIRI-based model compared with the traditional TNM staging system, highlighting its potential as a valuable tool for risk stratification and clinical decision-making in young patients with HNSCC.
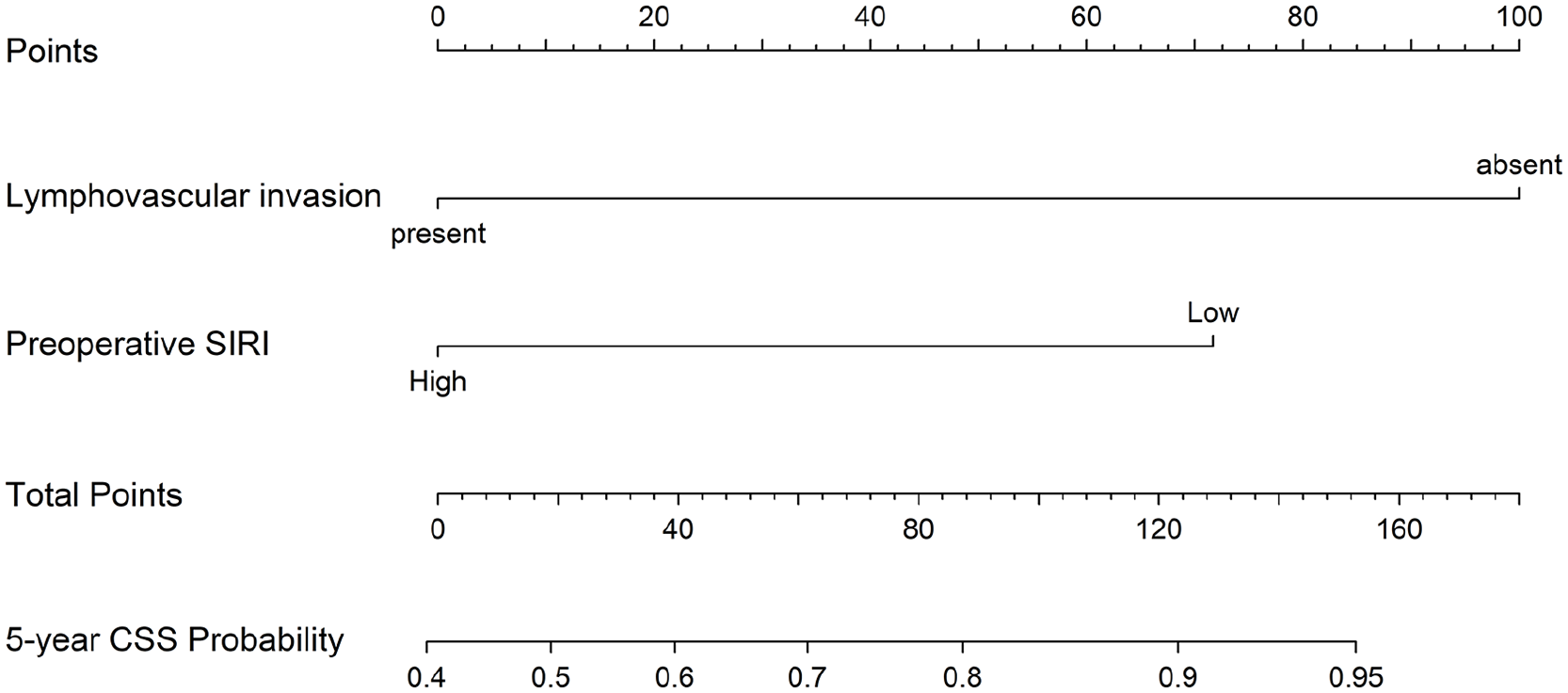
Nomogram and survival predictions. Nomogram for predicted CSS rate. A vertical line is drawn from each factor to the point score. By adding the points from all the factors, a total points score is reached, which is translated into five-year CSS rates by drawing a vertical line to its axis.
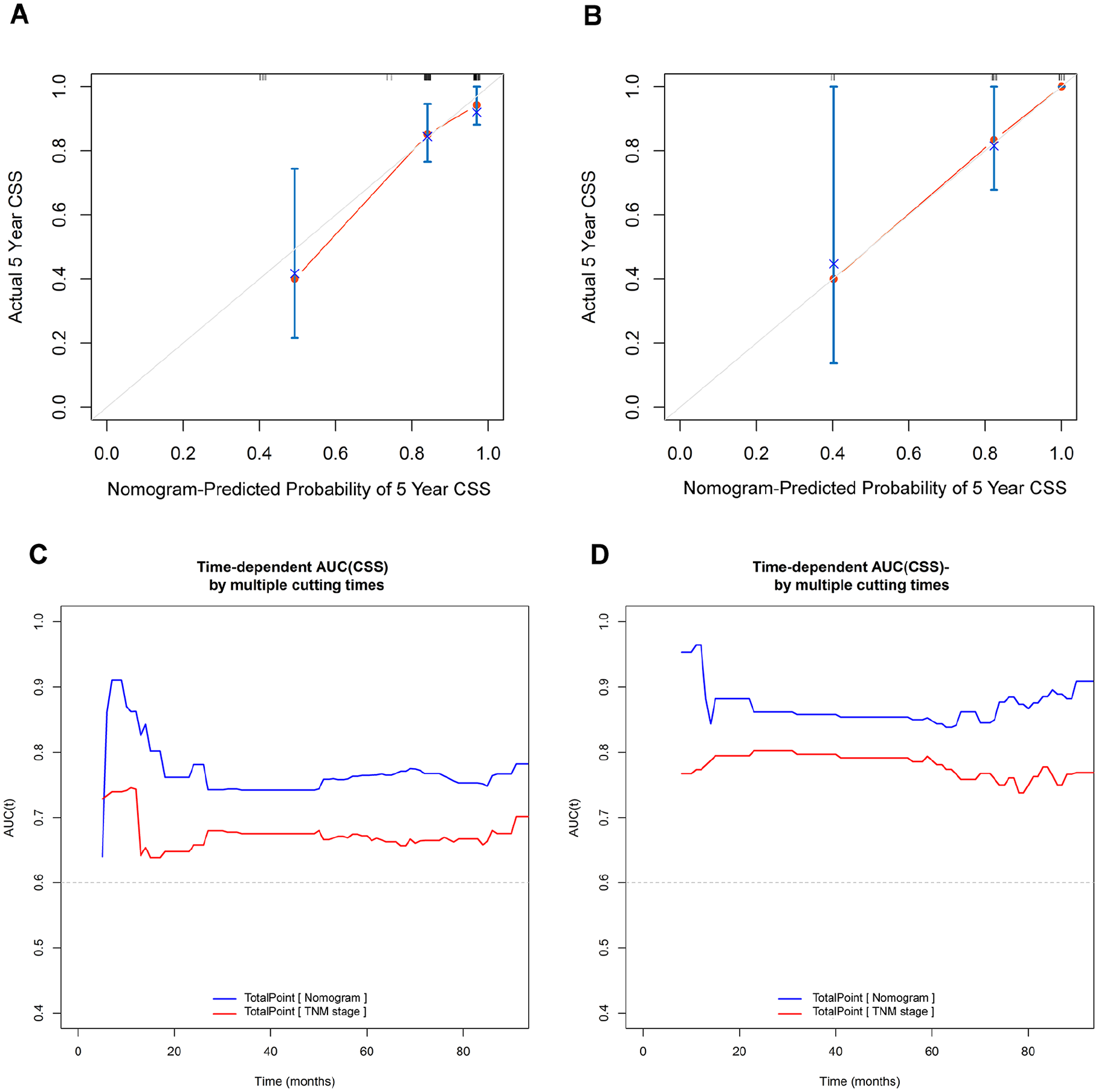
The calibration and comparison of the predictive capabilities of this SIRI-based prognostic model. (A) Calibration plot in the training cohort. (B) Calibration plot in the validation cohort. (C) Time-dependent ROC curves comparison between the current prognostic model and traditional TNM staging system in the training cohort. (D) Time-dependent ROC curves comparison between the current prognostic model and traditional TNM staging system in the validation cohort.
Discussion
In this study, we identified the preoperative SIRI as an independent prognostic factor for young patients with HNSCC at our center. Based on this finding, we developed a novel prognostic prediction model, which demonstrated strong predictive performance in both the training and validation cohorts. Compared to invasive, experience-dependent histological examinations and costly, less accessible genetic testing methods, subparameters derived from routine laboratory tests such as complete blood count offer significant clinical advantages. According to our data, the SIRI-based nomogram showed superior prognostic accuracy compared with the traditional TNM staging system. This suggests that the SIRI-based model could serve as a convenient, noninvasive, affordable, and reliable tool for improving prognostic predictions and aiding clinicians in making informed treatment decisions for young patients with HNSCC undergoing radical surgery.
The number of studies investigating the clinical effects of the SIRI on HNSCC prognosis has increased recently. Valero et al. discovered that a high SIRI was associated with inferior CSS and recurrence-free survival rates in a cohort of 824 patients with HNSCC. 20 Saroul et al. retrospectively reviewed the prospectively-collected data of patients with HNSCC and observed that low SIRI values were associated with better 5 year OS compared with those of the control group. 23 Chuang et al. studied a cohort of 141 patients with hypopharyngeal/laryngeal cancer who received chemoradiation/radiation and showed that the pretreatment SIRI served as a good predictor for evaluating treatment response and clinical outcomes. 24 Consistent with these findings, our study demonstrated that decreasing preoperative SIRI levels in young patients with HNSCC were associated with improved CSS rates.
The mechanism by which an increased systemic inflammatory response promotes tumor cell invasion is not yet clearly understood. One possible explanation could be attributed to the antitumoral roles played by lymphocytes, which inhibit tumor cell proliferation and migration while reinforcing the human immune response to cancer. 25 It has been observed that a lower presence of infiltrating lymphocytes is associated with a poorer prognosis. 26 In contrast, higher levels of monocyte-derived macrophages and neutrophils have been linked to increased tumor aggressiveness and worse survival outcomes.27,28 This is thought to occur through the action of tumor microenvironment mediators such as TNF-a, vascular endothelial growth factor, and epidermal growth factor.28,29 A high SIRI implies a relative increase in the count of neutrophils and/or monocytes, as well as a decrease in lymphocytes. It is possible that the prognostic ability of the SIRI stems from its function as a crude marker for the dynamic interplay between pro-tumor and antitumor responses within the immune system.
In addition, the presence of LVI was another unfavorable independent predictor of CSS. LVI is a pathological phenomenon in which tumor cells invade an endothelium-lined space of vascular or lymphatic vessels without underlying muscular walls and has been well recognized as a common risk factor for locoregional recurrences and poor survival chances in patients with head and neck cancer. 30 The results echo our previous study that revealed the presence of LVI as an inferior oncologic outcome in patients with locally-advanced oral cancer undergoing radical surgery. 31
The nomogram combines demographic and pathological data for improved patient stratification and the estimation of individualized treatment outcomes. 32 To the best of our knowledge, few studies have incorporated the SIRI variable into the predictive nomogram for young population with HNSCC. Our proposed SIRI-based nomogram demonstrated statistically-significant results and showed a closer correspondence to actual survival rates than the TNM staging system. Based on these observations, the nomogram based on the SIRI and various clinicopathological features may be a convenient and effective tool to help clinicians execute individualized estimations of cancer-specific mortality risk of young patients with HNSCC after surgery and improve personalized therapy; however, further research is required.
This study had some limitations. First, this was a retrospective study, and all patients underwent surgical procedures at a single institution performed by different head and neck surgeons. Therefore, this study was prone to selection bias. Second, our cohort focused on patients’ upfront surgery with/without adjuvant therapy; thus, our results might fail to extend to the patient's upfront definitive chemoradiotherapy. Thirdly, the results were not validated using an independent dataset. External validation of the results using an independent patient cohort may strengthen this evidence.
Conclusions
Our results suggest that the preoperative SIRI serves as an independent survival predictor for young patients with HNSCC. The findings suggest that other than TNM staging, the preoperative SIRI, along with the presence of LVI, could be utilized as key factors for risk stratification in young patients with HNSCC.
Footnotes
Ethics Considerations
The study was performed in accordance with the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Chang Gung Medical Foundation in Taiwan (Ethical Application Reference number: 202300725B0).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by the Kaohsiung Chang Gung Memorial Hospital in Taiwan with the grant number: CMRPG8N0151.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
