Abstract
Background
Cutaneous malignant melanoma (CMM) is one of the most aggressive skin tumors. Sentinel lymph node biopsy (SLNB) is an important test before thorough treatment of melanoma. The aim of this study was to investigate cancer-specific survival (CSS) in patients with head and neck CMM after negative SLNB and to analyze predictors of decreased survival.
Methods
Based on the Surveillance, Epidemiology and End Results (SEER) database, a study was conducted using data from patients with head and neck CMM after negative SLNB. The demographic, clinical, and pathological characteristics of the case population were analyzed. Cox univariate, Kaplan–Meier analysis, and multivariate Cox regression models were used to explore predictors of decreased survival; propensity score matching (PSM) analysis was used to reduce confounding bias, and outcomes were compared between the wide margin excision and narrow margin excision groups.
Results
A total of 1597 confirmed head and neck CMM patients with SLNB-negative were found. A Breslow>4.0 mm was the highest independent risk predictor for patients (HR 3.82, 95% CI 2.04-7.16, P < .001), and significant risk independent predictors also included a high mitotic rate >4 (HR 1.54, 95% CI 1.06-2.25, P = .023). Age< 60 years old was a significant survival predictor (HR 0.56, 95% CI .37-.85, P = .007), and not scalp and neck CMM were also important factors for longer survival (auricle skin: HR .51, 95% CI .29-.90, P = .02; unspecified parts of face: HR .59, 95% CI .40-.87, P = .007). After harmonizing baseline data by PSM, it was found that the extent of surgical resection did not affect patient survival.
Conclusion
This study analyzed the risk factors affecting CSS in patients with CMM of the head and neck region with SLNB-negative and observed a statistically significant difference in the prognosis of patients with CMM in different aesthetic subunits of the head and neck region. Close clinical follow-up for this population is necessary, and periodic medical examinations should be carried out.
Introduction
Cutaneous malignant melanoma (CMM) is a malignant tumor derived from neural crest melanocytes that is highly malignant and prone to lymphatic metastasis.1,2 For CMM patients with clinically negative lymph node metastasis, sentinel lymph node biopsy (SLNB) is the most important and accurate prognostic factor in the pathological evidence of lymph node metastasis.3-5 Sentinel lymph node biopsy has high application value in the auxiliary diagnosis of malignant melanoma, which can early determine the lymphatic metastasis of tumor patients, accurately assess the stage of malignant melanoma, predict prognosis and formulate follow-up treatment plans. 6 However, the lymphatic drainage of the head and neck is the most complex and highly variable, usually involving multiple lymph node drainage regions, and the location of the sentinel lymph node group is relatively close to the primary tumor, so the implementation of SLNB is more difficult, and there is also the possibility of false-negative SLNB.7-9 A false-negative SLNB was defined as a negative SLNB that recurred in the sampled lymph node region after several years of follow-up. True-negative SLNB was defined as negative SLNB without regional recurrence in previously sampled lymph node basins. According to statistics, up to 21.0% of negative SLNB patients may develop metastasis in the draining lymph node area during the follow-up period. 10 Studies have recommended lymph node dissection in patients with SLNB-negative at regional recurrence. 11
It is currently recognized that the factors affecting the survival of patients with CMM involve age, sex, tumor Breslow thickness, ulceration, and mitotic rate. Other studies have shown that the status of sentinel lymph nodes is also the strongest predictor of survival in patients with intermediate-thickness melanoma.12,13 However, the multicenter selective lymphadenectomy trial-I (MSLT-I) demonstrated that SLNB-based management extended disease-free survival in all patients or in patients with medium-thick melanoma compared with clinical lymph node observation, and the management also prolonged melanoma-specific survival in patients with lymph node metastases. 14
There are currently few studies on prognostic factors in patients with CMM after negative SLNB. Therefore, the purpose of this retrospective study was to analyze the predictors of tumor-specific survival in patients with clinically node-negative head and neck CMM after SLNB and to evaluate the surgical benefit of patients with different surgical resection extents after propensity score matching (PSM) analysis.
Materials and methods
Collection of baseline information
We performed a population-based cross-sectional analysis according to the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER). We identified head and neck CMM by selecting the year of diagnosis from 2010 to 2015, the International Classification of Diseases in Oncology, Third Edition (ICD-0-3) histology code 8743. Follow-up information showed patients at diagnosis aged ≥18 years old, sex, race, and primary site (the primary sites included were skin of the lip (C44.0), eyelid (C44.1), external ear (C44.2), skin of other and unspecified parts of face (C44.3), and skin of scalp and neck (C44.4)). All cases had complete AJCC TNM staging and melanoma mitotic rate records, and all underwent auxiliary examinations, such as SLNB. Patients with unknown or unspecified TNM stage, survival time, survival status, and surgical status were excluded from this study. We also excluded N1-N3, NX in AJCC TNM staging and distant metastasis in TNM staging M1. The survival status and survival time of each patient were confirmed. And narrow margin excision (NME) standard codes were 30, 31, 32, 33 and wide margin excision (WME) codes were 45, 46,47.
Statistical analysis
Hazard ratios (HR) and 95% confidence intervals (CI) were estimated using Cox regression models. Cancer-specific mortality (CSS) was used as the event. Univariate at P < .05 was incorporated into multivariate analysis to predict independent prognostic factors, and survival curves were drawn using the Kaplan–Meier method. Then, all significant variables in the univariate analysis were extracted, and patients treated with WME and NME were randomly matched according to the propensity matching score (PSM) principle, and their survival outcomes were compared. R (version 3.6.1, https://www.r-project.org/) and IBM SPSS (version 22.0) were used for statistical analysis.
Results
Population statistics
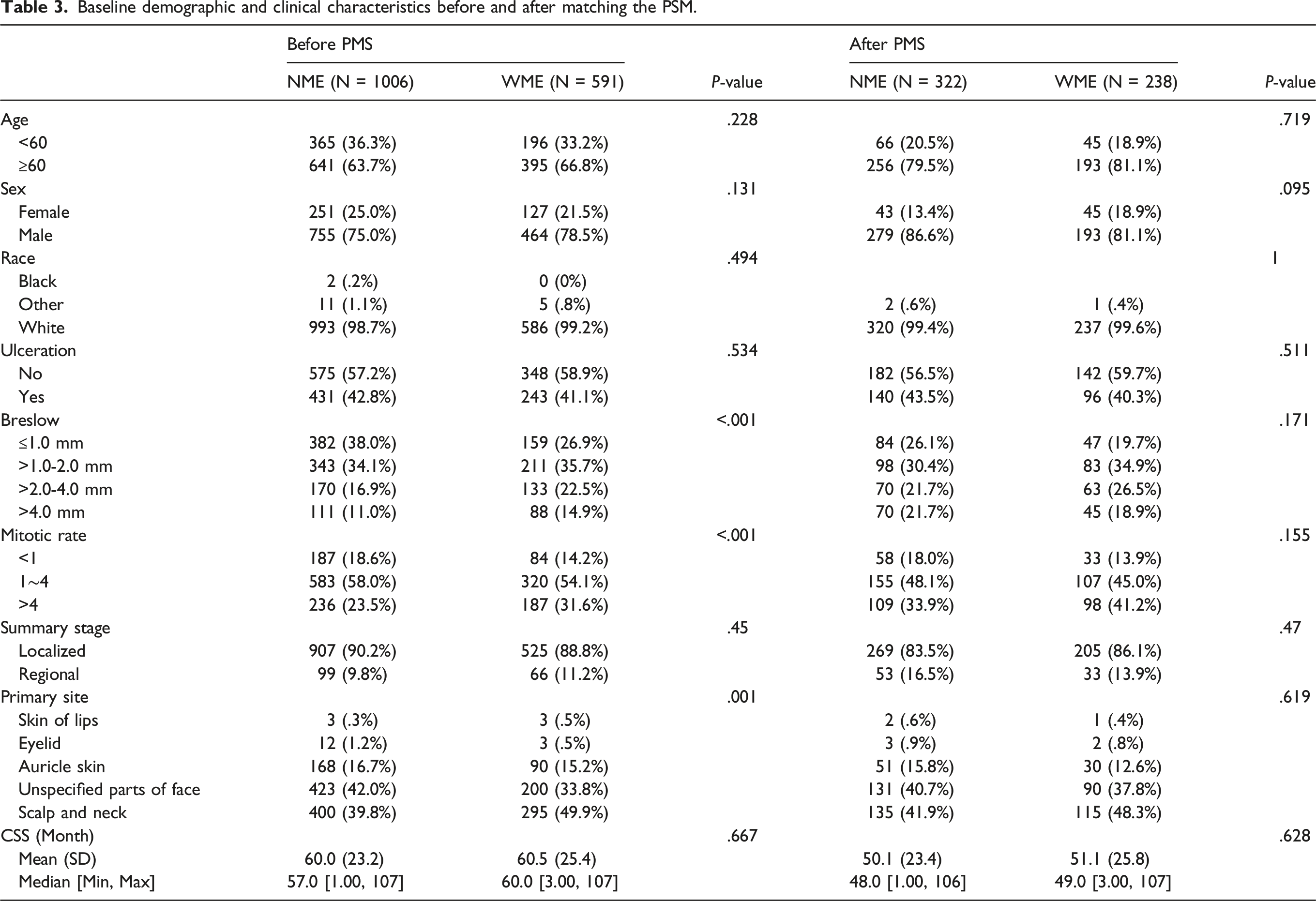
Baseline demographic and clinical characteristics.
Cox univariate and multivariate survival analysis
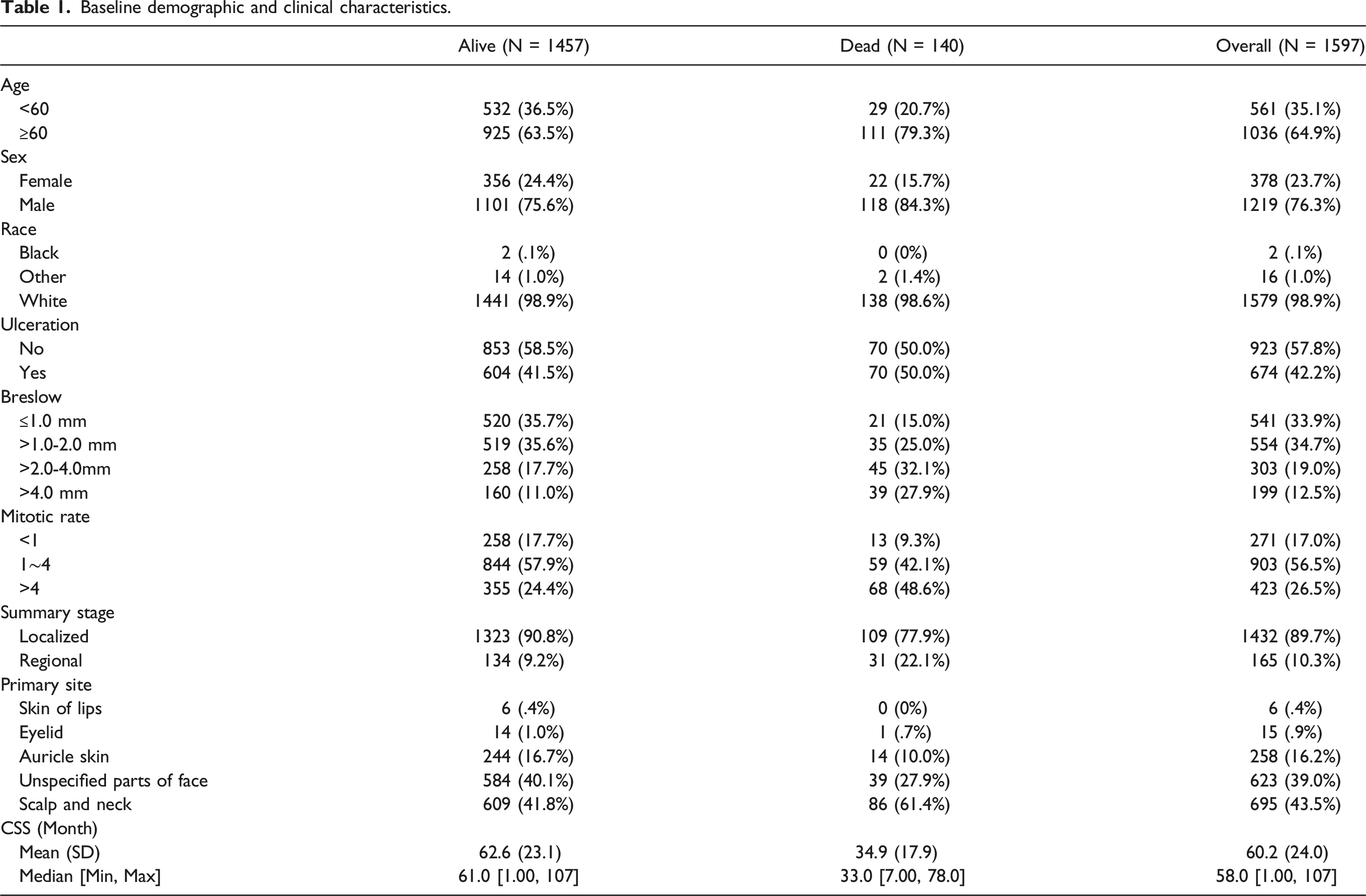
Univariate analysis for patients with head and neck CMM after negative SLNB.
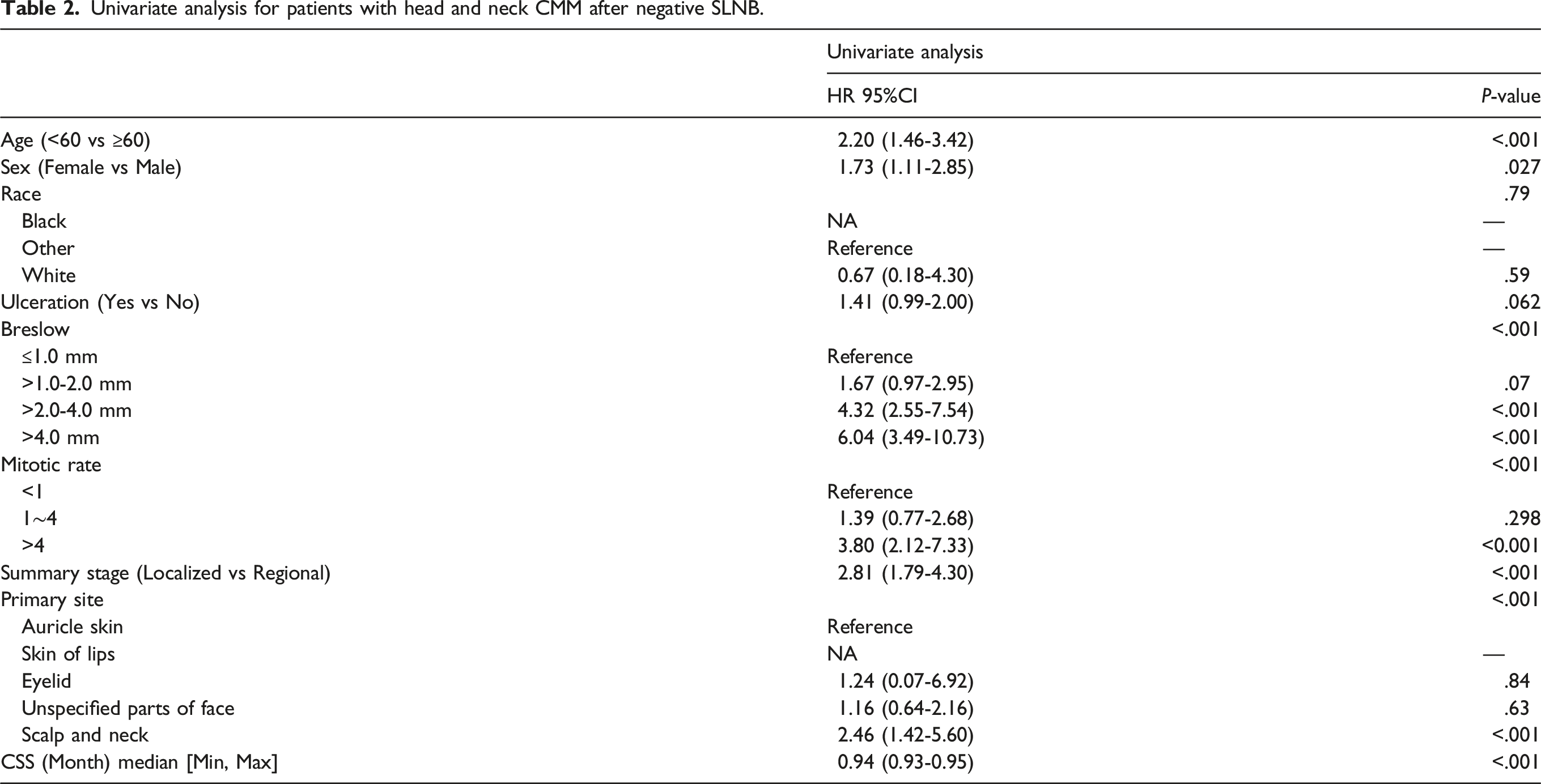
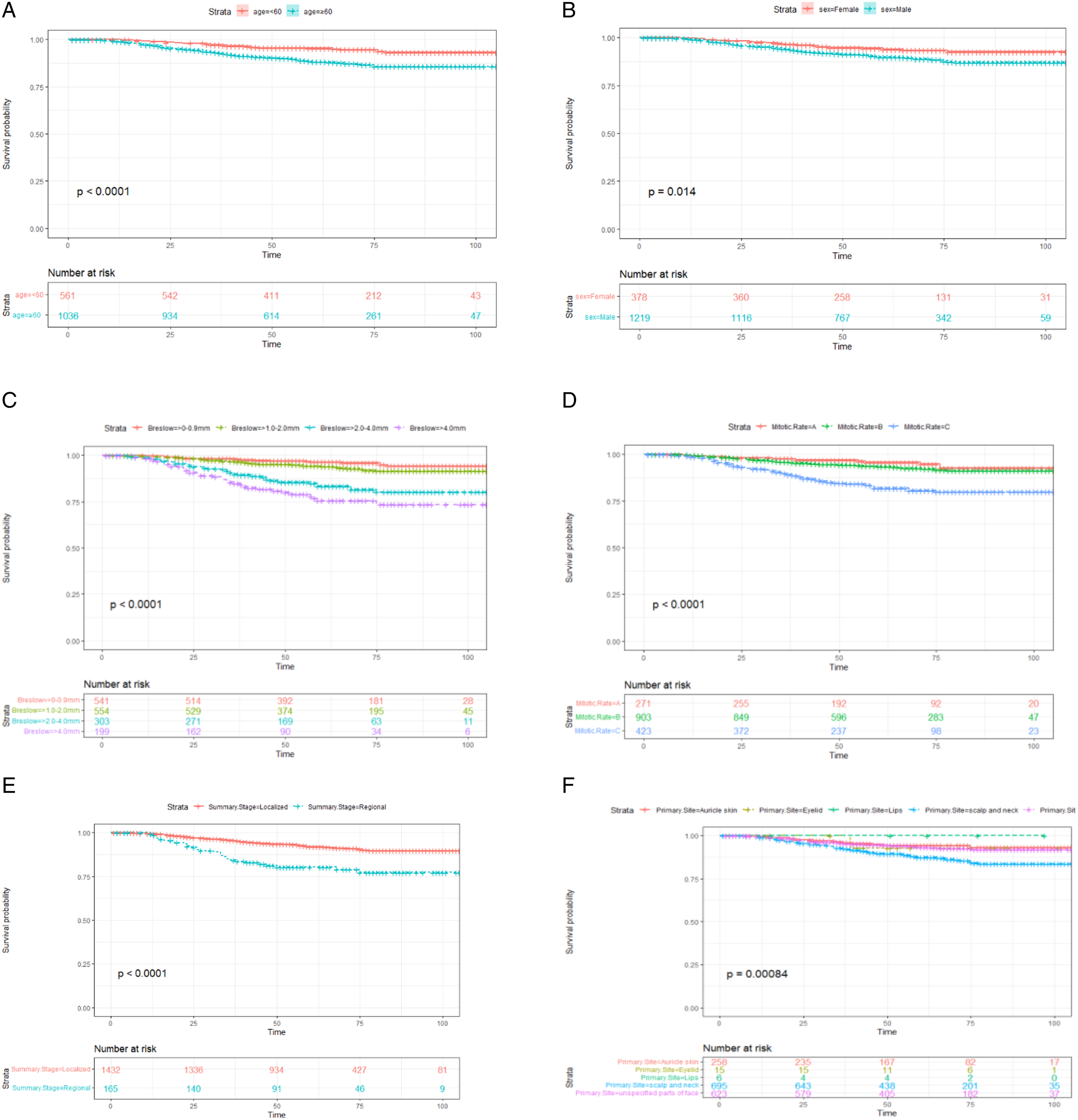
(A–F) Cancer specific survival curve for patients with head and neck CMM after negative SLNB based on age (A), sex (B), Breslow (C), Mitotic rate (D), summary stage (E) and Primary Site (F). Mitotic rate A: <1, Mitotic rate B: 1∼4, Mitotic rate C: >4.
To further investigate the prognostic factors after negative SLNB, we constructed a multivariate Cox regression model of CSS (Figure 2), and the results showed that Breslow>4.0 mm was the highest independent risk predictor for patients (HR 3.82, 95% CI 2.04-7.16, P < .001). Significant independent risk predictors also included Breslow > 2.0-4.0 mm (HR 3.04, 95% CI 1.74-5.31, P < .001) and mitotic rate > 4 (HR 1.54, 95% CI 1.06-2.25, P = .023). Age< 60 years old was a significant survival predictor (HR .56, 95% CI 0.37-0.85, P = .007), and not scalp and neck CMM were also important factors for longer survival (auricle skin: HR .51, 95% CI .29-.90, P = 0.02; unspecified parts of face: HR .59, 95% CI .40-.87, P = .007). Cox multivariate analysis for patients with head and neck CMM after negative SLNB.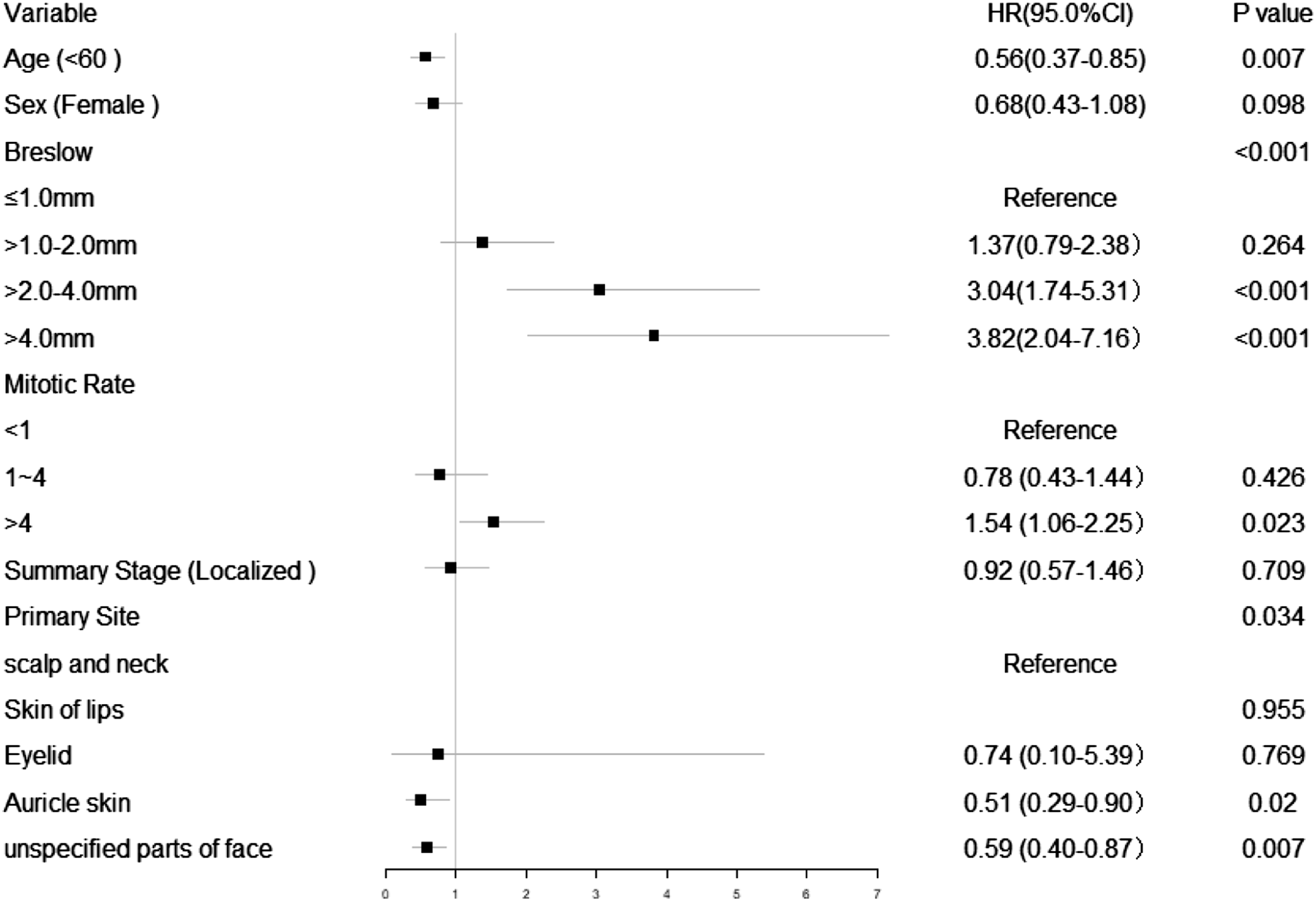
Surgical survival analysis
Baseline demographic and clinical characteristics before and after matching the PSM.
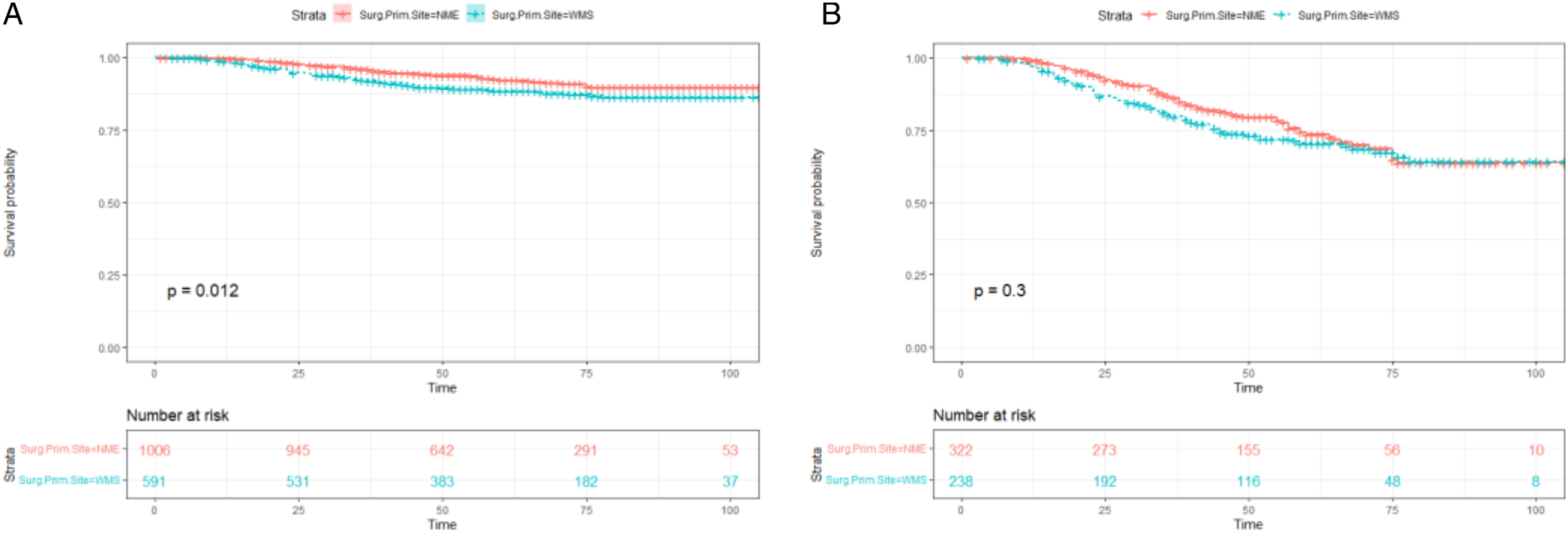
Total survival curves for patients with the WME and NME groups. (A): before matching the PSM; (B): after matching the PSM.
Discussion
For CMM patients without clinically positive lymph node evidence, SLNB is the most important pathological evidence of lymph node metastasis. It can detect occult lymph node metastasis in some lymph node-negative patient groups and has become the most accurate auxiliary test for evaluating the prognosis of CMM patients. The study found that age ≥60 years old, scalp and neck CMM, Breslow >2.0 mm and mitotic rate >4 were associated with the prognosis of patients with head and neck CMM after negative SLNB.
Although CMM can occur anywhere on the body, melanoma at the head and neck site is more likely to exhibit aggressive clinicopathological features and has a poorer prognosis and higher recurrence rate than melanoma at other skin sites. 15 However, the head and neck can be aesthetically divided into more than ten subunits: scalp, forehead, orbital area, ear and periauricular area, cheeks, nose and neck. Different aesthetic subunits have certain differences in the prognosis of CMM. 16 The data of this study were analyzed by Cox multivariate regression analysis, suggesting that negative SLNB patients with scalp and neck CMM had a lower CSS rate, while patients with auricular skin and unspecified parts of the face had higher survival rates.
Age is a highly significant and powerful predictor of outcome in patients with cutaneous melanoma; although it lacks a significant predictive role for lymph node involvement, recurrence, and metastasis, increasing age may enhance the aggressive histological features of CMM, leading to poorer survival.17,18 This study also confirmed that age ≥60 years old is an important factor affecting the prognosis of CSS in negative SLNB patients, which is consistent with the data of an earlier study. 19 Although it is known that CSS rates are lower in male CMM patients, this study did not demonstrate an important relationship with survival outcomes in negative SLNB patients, which is different from some studies.
The American Joint Committee on Cancer (AJCC) TNM included tumor thickness, ulceration, mitotic rate and lymph node status in the risk assessment of recurrence and metastasis in patients with localized melanoma. 20 Multiple studies have confirmed that thin and medium-thick tumors (Breslow thickness ≤ 4.0 mm) are favorable prognostic factors for survival in CMM patients. 21 This study confirms that there is a close relationship between tumor thickness and CSS in SLNB-negative patients. The features of the 7th edition AJCC T staging for CMM are related not only to overall tumor thickness and the presence of ulcers but also to the value of mitotic rates in lesions <1 mm thick (T1). 22 The mitosis rate is an important predictor of sentinel lymph node metastasis in T1a melanoma. 18 Increased tumor mitotic rates reduce 5-year overall survival and cancer-specific survival in patients with head and neck melanoma. 23 Although in the 8th edition of the AJCC Handbook, the tumor mitotic rate was deleted as a staging criterion, this study still recorded the data of the specific negative SLNB mitotic rate. Cox multivariate regression analysis found that a mitotic rate>4 was associated with SLNB-negative. A high mitotic rate is also an important CSS risk prediction factor for patients.
Surgery and systemic tumor control are important treatments for prolonging patient survival. In the absence of local organ invasion and tumor metastasis, surgical resection should be the first choice. However, extensive local excision, sentinel lymph node biopsy, and completion of lymph node dissection and reconstruction require special consideration of the location of the head and neck melanoma and the proposed margins and surrounding structures involved. 24 Although there are few controlled prospective studies evaluating WME in CMM, it has been shown that NME may lead to high recurrence rates in CMM patients, but larger WME does not confer oncological benefit. 10 Our study further discussed the influence of tumor surgical resection extent on the prognosis of negative SLNB patients. We first performed a PSM analysis of patients with different resection extents by age, sex, and CMM pathology to reduce the effect of treatment selection bias on outcomes by matching clinical baseline data. The results of the study confirmed that WME and NME have no significant effect on CSS in patients with head and neck CMM after negative SLNB.
Limitations
This study has some limitations that must be addressed. First, treatment factors such as radiotherapy and chemotherapy were not considered in this study; second, as this was a retrospective review, there may be some selection bias and record entry errors; third, this study lacks specific information on patient relapse, so more prospective randomized studies are needed.
Conclusion
This study analyzed the risk factors affecting CSS in negative SLNB patients with CMM in the head and neck regions and observed a statistically significant difference in the prognosis of patients with CMM in different anatomical subunits of the head and neck regions. Close clinical follow-up for this population is necessary, and periodic medical examinations should be carried out.
Footnotes
Author contributions
Shulian Li, MS carried out the development or design of the methodology, statistical analysis, and original draft. Jun Tang MD designed the methodology and wrote original draft. Lanlan Wang. MS helped critically revise the draft for the design of the methodology and data curation. Juxiang Gou MS and Wei You MS participated in the methodology, data curation, and validation. Qiang Ji designed the conceptualization and carried out the article and editing. All authors have checked and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics
We acknowledge the SEER database for providing their platforms and contributors for uploading their meaningful datasets. SEER belongs to public databases. The patients involved in the database obtained ethical approval. Users can download relevant data for free for research and publish relevant articles. Our study is based on open data, so there are no ethical issues or other conflicts of interest.
