Abstract
Introduction
Adenoid cystic carcinoma (ACC) is a rare malignancy that primarily originates in the salivary glands, accounts for about 1% of head and neck cancers. 1 It can manifest in various regions of the head and neck, including the major and minor salivary glands, oral cavity, paranasal sinuses, nasopharynx, oropharynx, trachea, and other regions. 2 The propensity of ACC for perineural invasion as well as the limited space and complex anatomy of the head and neck, can lead to skull base involvement and intracranial extension, thus posing a significant challenge for treatment. 3
While radiotherapy may be included in the standard care plans, radical surgery is the primary treatment selection for nonmetastatic ACC patients.1,2 However, ACC often displays a paradoxical behavioral combination of an indolent growth pattern but an aggressive progression, resulting in local recurrence and distant metastasis (DM). 4 The rate of DM varies between 8% and 60%, 5 most of them developed in the first 5 years after presentation. Notably, some patients suffer from DM during their initial visit, and a previous study demonstrated that patients with pre-therapy DM have a significantly poorer prognosis than those who develop DM during the observation period. 6 Thus, greater attention to synchronous DM of ACC patients is warranted.
Traditionally, chemotherapy, radiotherapy, and supportive care are the standard treatment options for patients with DM. However, recent studies have demonstrated the importance of primary tumor surgery (PTS) in prolonging overall survival (OS) in multiple cancers with M1 disease at diagnosis, such as head and neck squamous cell, 7 salivary gland carcinoma, 8 and breast cancer. 9 Nevertheless, no relevant studies have explored the role of PTS in the context of synchronous metastases in ACC patients to date.
Against this backdrop, this study aimed to evaluate the prognostic significance of PTS and identify the optimal candidates for ACC patients with synchronous metastases at initial presentation. This analysis utilized population-based data from the surveillance, epidemiology, and end results (SEER) database.
Materials and Methods
Study Population
We retrieved data from the SEER database, which is a highly representative tumor database in the United States. 10 We used SEER*Stat software (version 8.3.6) to extract the data from the sub-database designated as “Incidence-SEER Research Plus Data, 17 registries, Nov 2021 sub (2000-2019).” The inclusion criteria were as follows: (1) primary ACC [histology code C82000 according to the Third Edition (ICD-O-3)] as the first primary cancer; (2) DM at initial presentation (bone, brain, liver, or lung); (3) primary tumor site located in the head and neck region; and (4) diagnosis between 2010 and 2019, as detailed information on metastatic site was only available after 2010. We did not require Institutional Review Board’s approval for this study since SEER data are publicly available.
Covariates
In our study, we retrieved the following information for all cases: sex, age at diagnosis, race, AJCC T staging, primary site, histopathologic type, metastatic organs, and treatment parameters such as PTS, radiotherapy, and chemotherapy. PTS refers to the total or partial removal of the primary tumor, with or without adjacent sites. We excluded surgeries for metastatic lesions due to insufficient information. Patients were grouped according to the primary tumor’s anatomical site: (1) salivary gland; (2) oral cavity; and (3) ENT group, including the larynx, hypopharynx, nasal cavity, middle ear, trachea, and mediastinum. We used the 7th and 8th editions of the AJCC (American Joint Committee on Cancer) T staging, depending on the year of diagnosis. Our study’s endpoints were OS and cancer-specific survival (CSS).
Statistical Analysis
We conducted statistical analyses using SPSS version 25 (IBM, Armonk, NY, USA). Categorical baseline characteristics were compared using Pearson’s chi-square test or Fisher’s exact test, while survival estimates were calculated using Kaplan–Meier analysis. Kaplan–Meier curves were generated and compared using the log-rank test. We performed multivariate analyses using the Cox proportional hazards regression model to assess the impact of independent factors on OS and CSS, and calculated HR and 95% CI. Statistical significance was defined as P < .05, with two-sided tests applied.
Results
Baseline Characteristics and Distribution of Metastatic Sites
A flowchart of patient selection and exclusions in this study is presented in Figure 1. A total of 192 patients with synchronous metastatic ACC who underwent PTS were included in the study cohort. The number of patients who received PTS was approximately equal to those who did not (n = 91, 47.4% vs n = 101, 52.6%). The median age at diagnosis was 63 years (range: 22-94), with a population of 99 (51.6%) male and 93 (48.4%) female patients. The most common site of primary lesions was the salivary gland (n = 95, 49.5%), and almost one-third of the patients (n = 56, 29.2%) had T4 tumors.
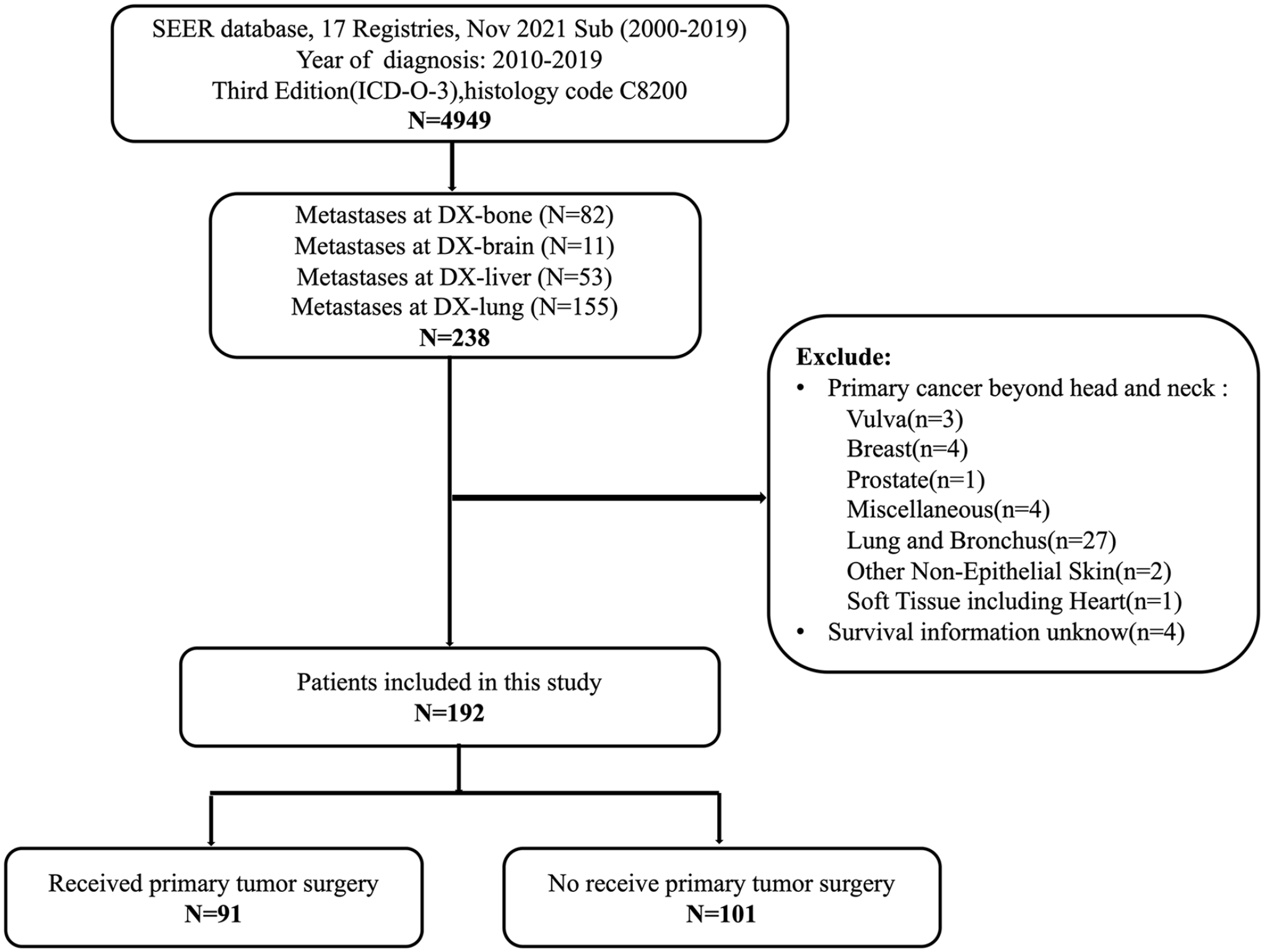
Flowchart illustrating the patient selection and exclusion process from the surveillance, epidemiology, and end results (SEER) database (2010-2019) in this study.
The most frequent metastatic site was the lung, with 136 patients (70.8%) experiencing metastases, followed by the bone with 63 patients (32.8%). Among the patients, 146 (76%) presented with single-organ metastasis, while 46 patients experienced multiple organ metastases. In terms of treatment, adjuvant radiation was administered to 114 patients (59.4%), while only 64 patients (33.3%) received chemotherapy as adjuvant therapy, which was relatively low in our study cohort.
Table 1 shows the baseline characteristics of the PTS and non-PTS groups. Significant differences were observed in the status of bone metastases (P = .035) and brain metastases (P = .03), the number of involved sites (P = .049), T stage (P = .028), and the administration of chemotherapy (P < .001).
Demographic Characteristics of the Study Population.
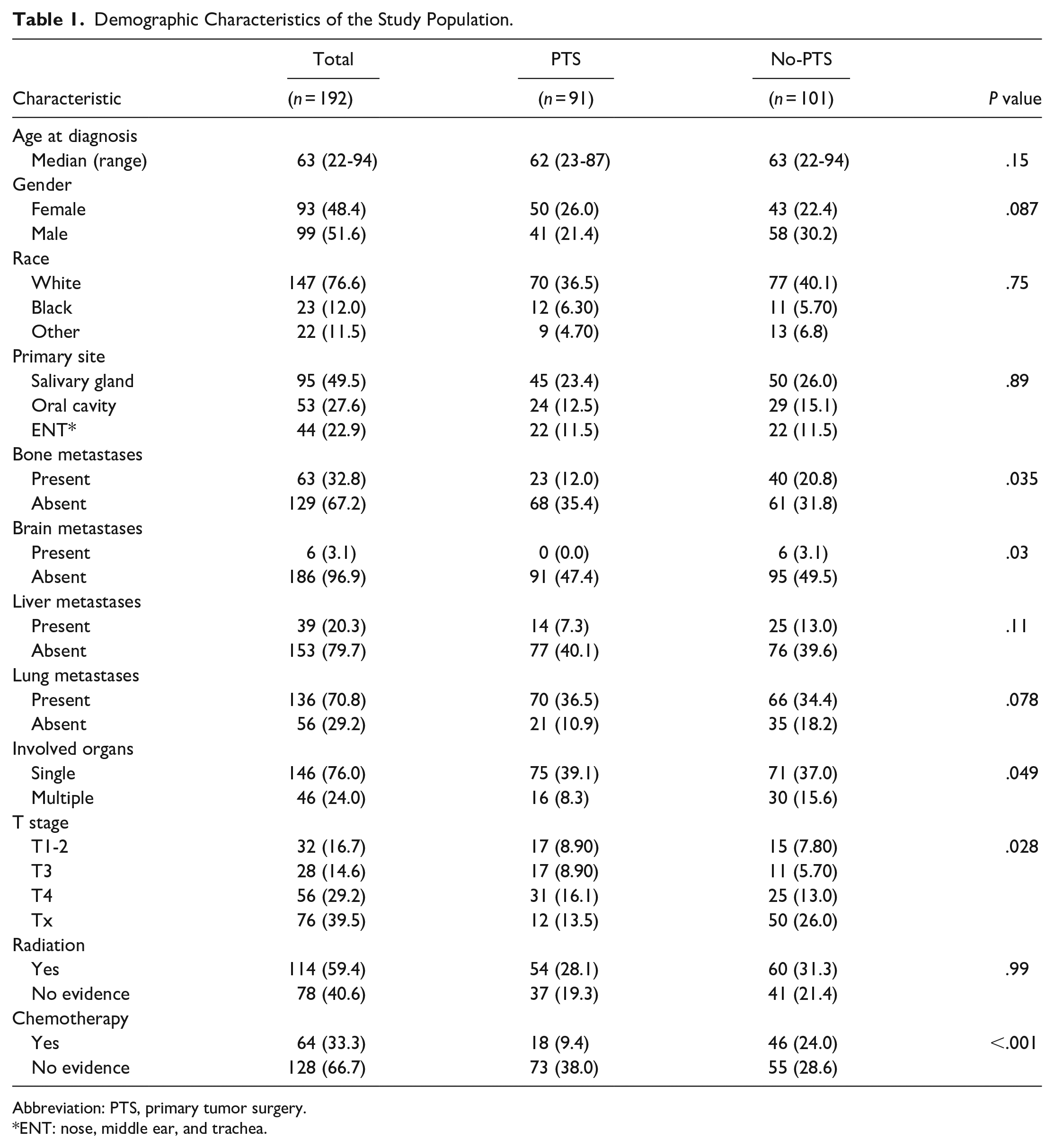
Abbreviation: PTS, primary tumor surgery.
ENT: nose, middle ear, and trachea.
Prognostic Significance of PTS in the Overall Cohort
The effect of PTS on OS and CSS was assessed in the entire cohort. Figure 2 shows the Kaplan–Meier curves of OS and CSS for the PTS and no-PTS groups. In the entire cohort of 192 patients, survival analysis using the log-rank test demonstrated a significant association between PTS and improved OS and CSS (log-rank P = .006 and .014, respectively). Table 2 indicates that PTS acted as a predictor of a favorable outcome for OS and CSS, even after adjusting for other covariates [PTS vs no-PTS, hazard ratio (HR): 0.46, 95% confidence interval (CI): 0.24-0.88, P = .02 for OS; HR: 0.46, 95% CI: 0.23-0.92, P = .028 for CSS].
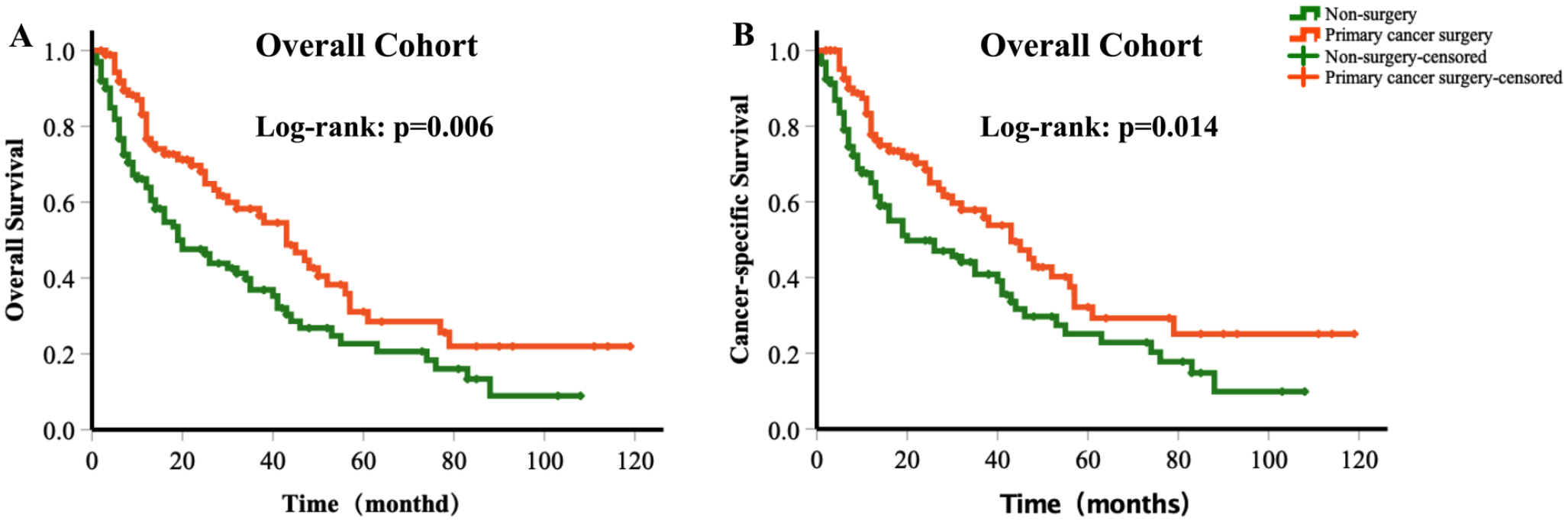
Kaplan–Meier curves depicting overall (A) and cancer-specific survival (B) based on whether PTS was performed in the overall cohort or not (n = 192).
Multivariate Cox Regression Models of Prognostic Factors for Overall and Cancer-Specific Mortality.
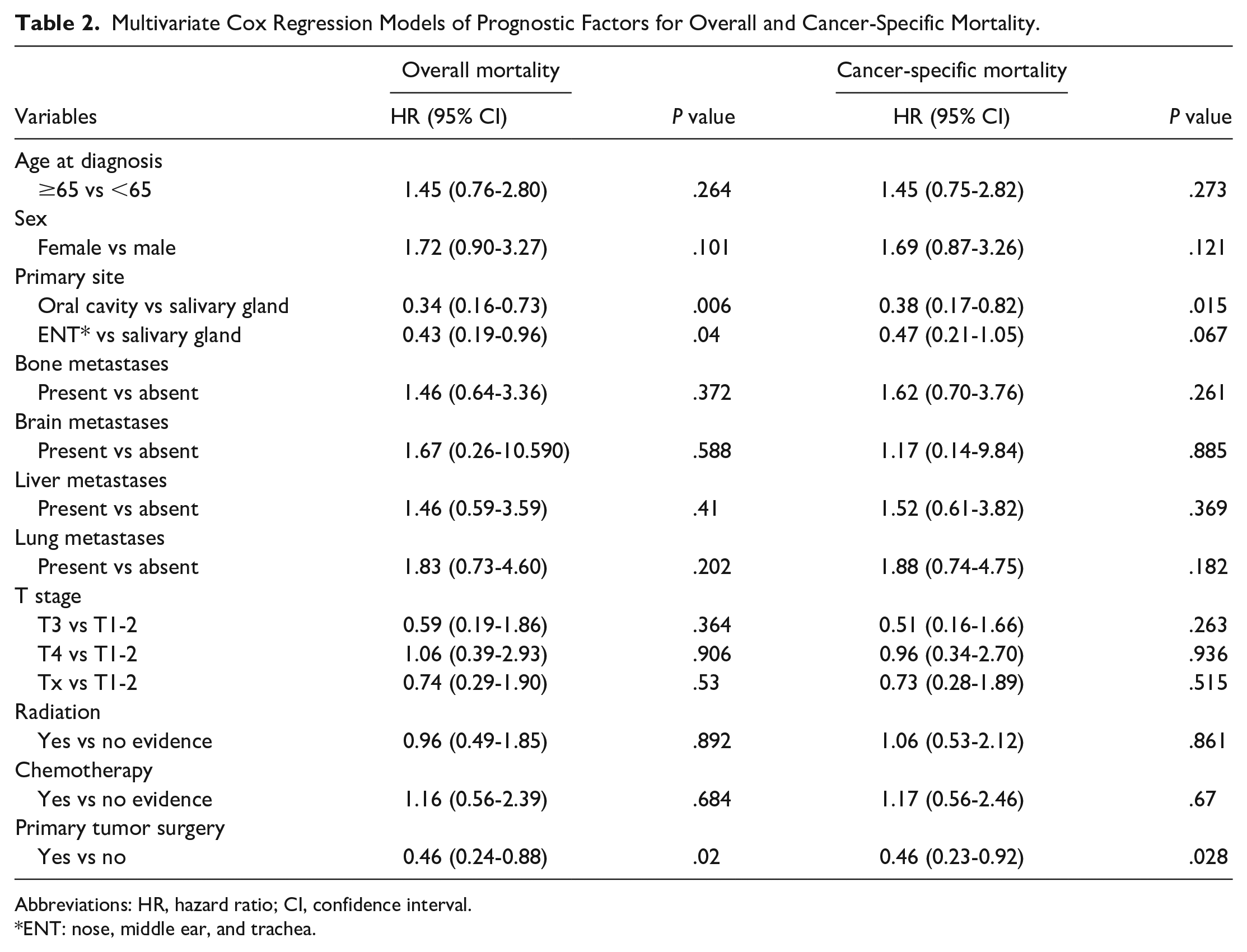
Abbreviations: HR, hazard ratio; CI, confidence interval.
ENT: nose, middle ear, and trachea.
Prognostic Significance of PTS in Patients with Site-Specific Metastases
To gain further insights into the prognostic significance of PTS in ACC patients with DM at initial presentation, we analyzed the associations between different metastatic sites and surgical outcomes. We conducted survival analyses in populations with site-specific metastases. The results showed that the prognostic effect of PTS varied depending on the involved sites. Patients with lung (log-rank P = .016 for OS and P = .027 for CSS) or bone (log-rank P = .041 for OS and P = .065 for CSS) metastases who received PTS had a significant survival benefit compared to those who did not undergo PTS. In contrast, there was no significant difference between the 2 groups in patients with liver metastases (log-rank P = .107 for OS; P = .153 for CSS; Figure 3). It is noteworthy that we did not perform subgroup Kaplan–Meier analysis in patients with brain metastases due to the limited number of cases (n = 6).
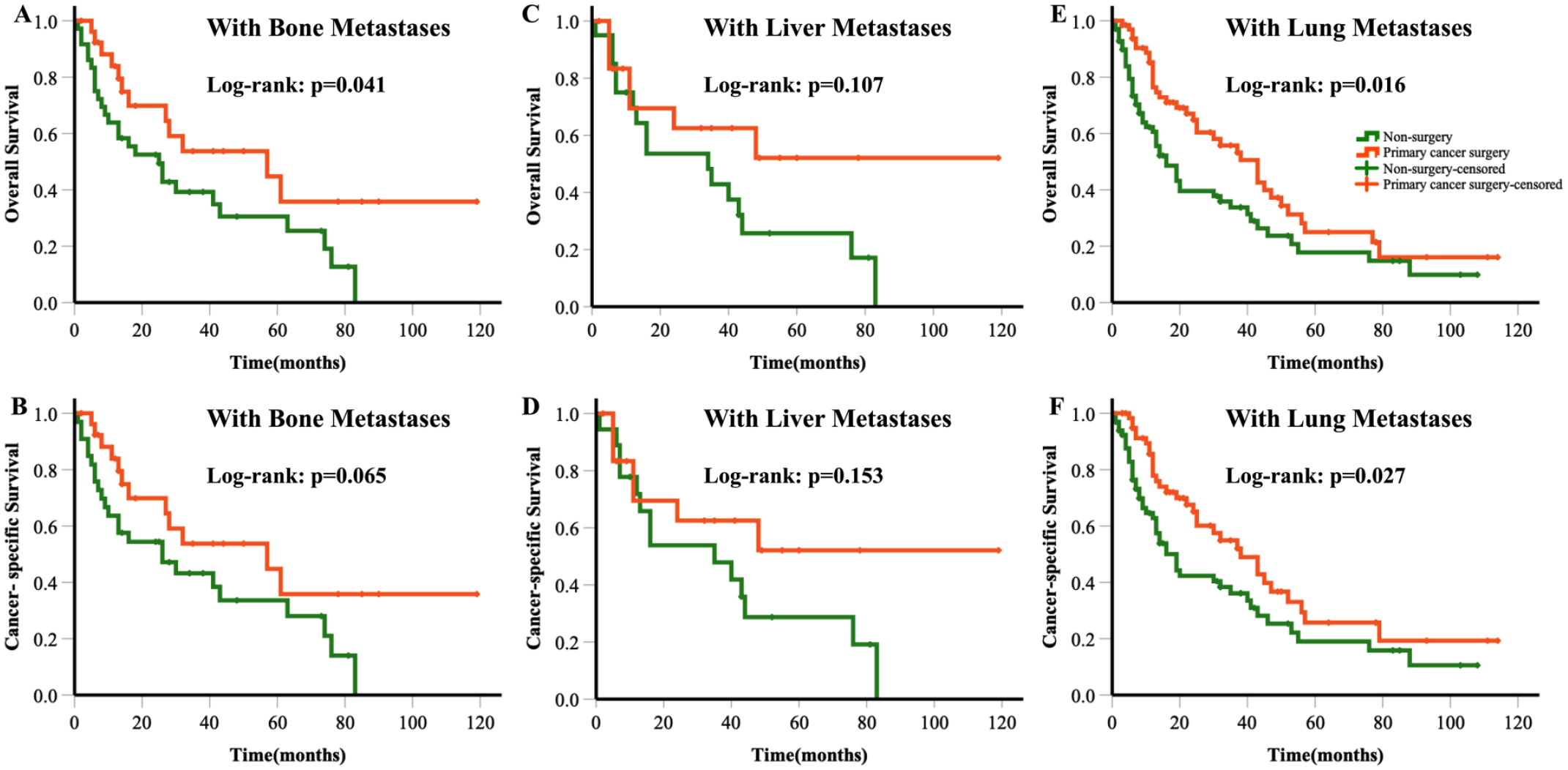
Kaplan–Meier curves illustrating overall and cancer-specific survival based on whether or not PTS was performed in patients with lung (A and B), bone (C and D), and liver (E and F) metastases.
Prognostic Significance of PTS in Patients with the Number of Involved Organs
The number of organs involved in metastasis could potentially influence treatment decisions and imply a different prognosis. Therefore, we analyzed the effect of PTS on patients with single-organ metastasis and multiple organ metastasis. Kaplan–Meier curves and corresponding log-rank tests showed significantly lower OS in the no-PTS group between the single-organ metastasis and multiple organ metastasis populations (log-rank P = .039 and P = .047, respectively). However, there was no statistical significance between the 2 groups in CSS (log-rank P = .066 and .071, respectively). The results of Kaplan–Meier analyses are presented in Figure 4.
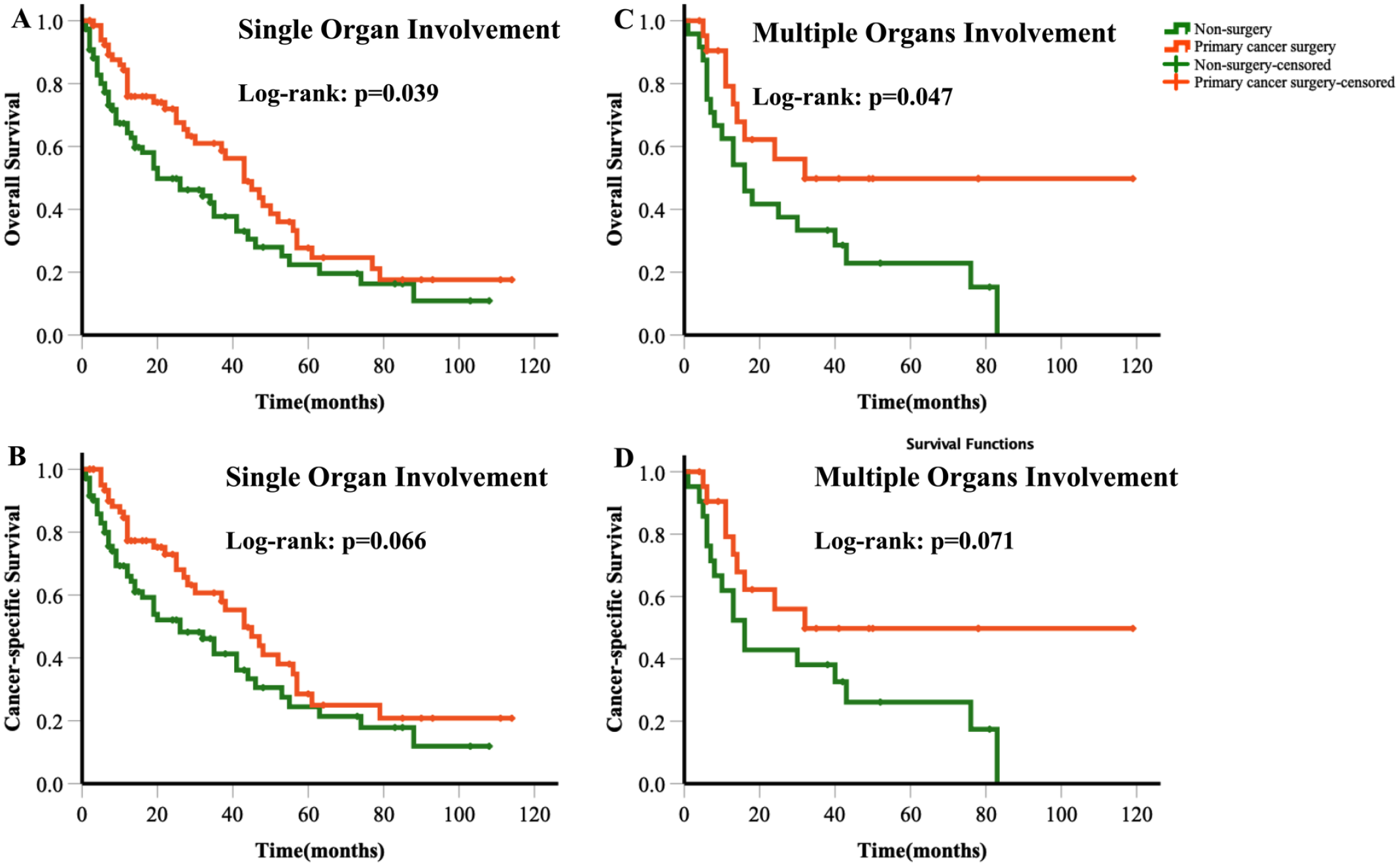
Kaplan–Meier curves showing overall and cancer-specific survival based on whether or not PTS was performed in patients with single-organ involvement (A and B) and multiple organ involvement (C and D).
Prognostic Significance of PTS in Patients with Radiotherapy
ACC has a high neurotropic potential, and non-R0 resection rate is high in the head and neck region. Therefore, adjuvant radiotherapy is often necessary to improve local control. We compared the survival benefit between patients who received radiotherapy and those who did not (Figure 5). In the non-radiotherapy cohort, we observed a significant survival benefit of OS and CSS in the PTS group (OS, P = .007; CSS, P = .008). However, for the radiotherapy group, PTS did not seem to provide a survival advantage in univariate log-rank tests (OS, P = .2378; CSS, P = .423). Although no significant statistical differences were found among patients in the radiotherapy-only group, surgery combined with radiotherapy group, or surgery-only group, PTS potentially offered the best results.
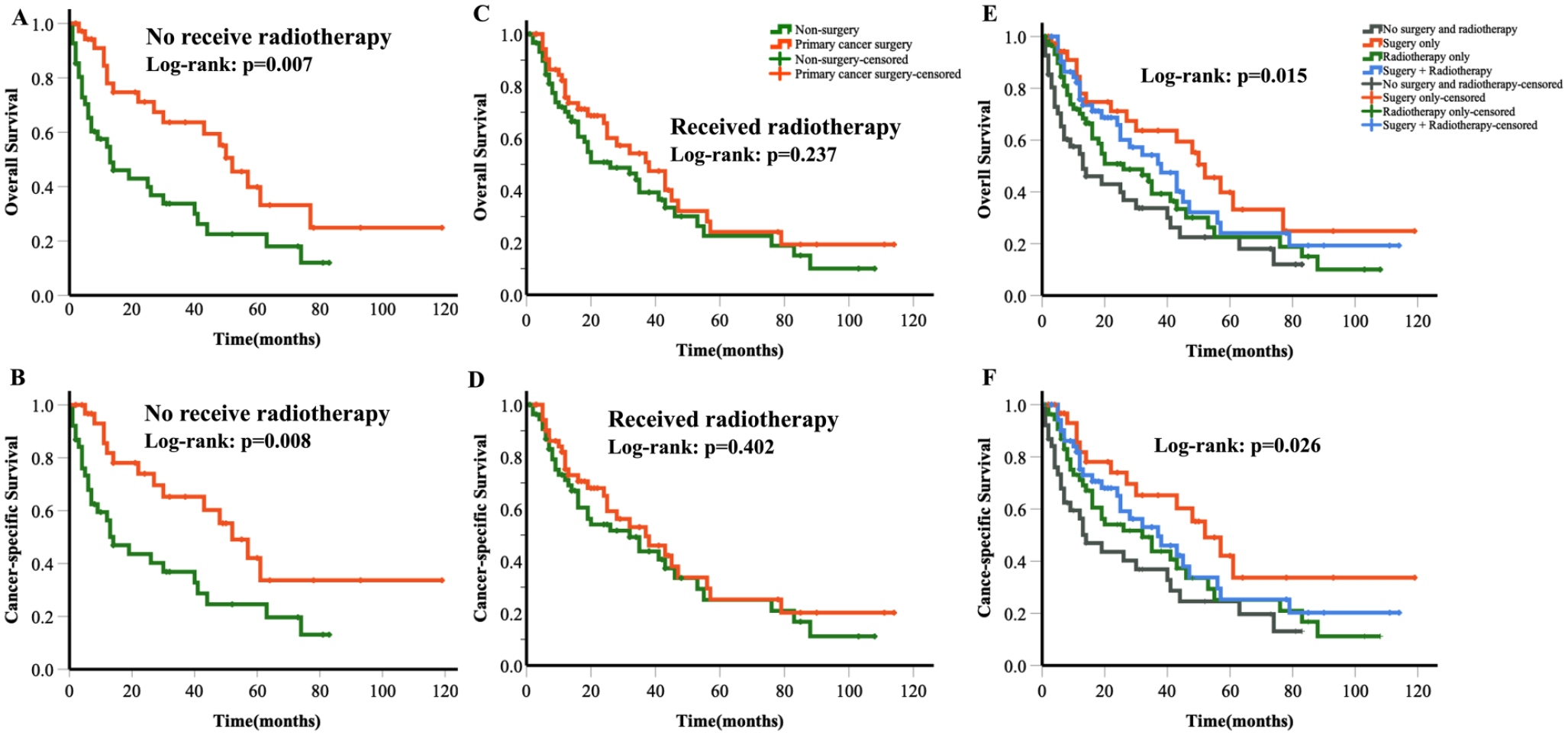
Kaplan–Meier curves demonstrating overall and cancer-specific survival based on whether PTS was performed in patients who did not receive radiotherapy (A and B), received radiotherapy (C and D), and divided into 4 groups (E and F).
Discussion
Currently, treatment options for metastatic ACC of head and neck are limited, and recommended modalities include radiotherapy, chemotherapy, and supportive care. The benefit of PTS for metastatic ACC patients remains a matter of debate. 11 Our study suggests that PTS may confer a survival advantage for ACC patients with bone or lung metastases. In the non-radiotherapy cohort, we observed a significant survival benefit in the PTS group, both in terms of OS and CSS. Although no significant statistical differences were found among the patients in the radiotherapy-only group, the surgery combined with radiotherapy group, or the surgery-only group, PTS potentially offered the best results.
In a previous study by Spiro et al., it was suggested that even when pulmonary metastases occur, excision of the primary tumor should be performed whenever feasible, as prolonged survival has been observed in 20% of patients with distant spread. However, a recent study showed that PTS did not confer a survival advantage in ACC patients. 12 We believe that the primary tumor site and the small number of cases included in the study may have contributed to this result. The suboptimal endpoint and lack of cost-effective systemic therapies have led to a shift toward a multimodal approach in treating metastatic head and neck ACC. 13 While surgery is the preferred treatment approach for early ACC, its potential benefits for metastatic ACC, particularly for patients with DM at initial presentation, remain to be confirmed.
Although the favorable prognostic impact of PTS has been confirmed in some cancer types,9,14 the underlying biological mechanism for this association remains unclear. The “seed and soil” hypothesis for metastasis postulates that early interventions targeting both the disseminating tumor cells and the microenvironment of distant sites are essential to improving the prognosis of advanced malignancies. 15 This supports the hypothesis that treating the primary tumor could retard the progression of existing metastases, potentially prolonging survival.16-18 Some scholars propose that reducing tumor burden can impair pro-oncogenic inflammation and tumor signaling, boosting both immunity and systemic therapy efficacy. 19 However, these hypotheses have not been validated in ACC. For head and neck ACC, due to their relatively inaccessible anatomical location, advanced tumors can cause severe symptoms and fatal complications, including asphyxia, hemorrhage, and inanition. PTS may largely delay or even avoid the occurrence of these lethal events. 12
Due to the confined space of the head and neck regions and the high propensity for perineural invasion of ACC, radiotherapy is often indicated to improve local control of ACC. In cases of surgical contraindications or non-respectable tumors, radiotherapy may be considered. However, there are limited studies comparing the effectiveness of radiotherapy with surgery treatment. 20 A retrospective study demonstrated that adjuvant radiation resulted in locoregional control of 36% and 10-year OS rates of 37% for ACC of the head and neck, which are inferior to the combination of surgery and radiotherapy. 21 Our results align with these findings. In univariate log-rank tests comparing patients who received PTS with those who underwent surgery combined with radiotherapy or radiotherapy alone, PTS did not appear to confer a survival advantage in patients with distant metastatic ACC of the head and neck. Furthermore, the OS benefit of postoperative radiotherapy varies among different studies.22,23 Notably, these studies are retrospective, and the heterogeneity of the study cohorts cannot be ignored. Additionally, given the specific location of ACC and its treatment involving the skull base and cranial nerves, radiotherapy-induced morbidity, such as blindness, hearing loss, and cranial neuropathies, is a significant concern for patients.24,25
Chemotherapy is generally reserved as a palliative treatment for ACC patients who are not surgical candidates. The majority of regimens are platinum based. 26 Despite several prospective and retrospective studies, the overall Objective Response Rate (ORR) does not typically exceed 20%. 27
Targeted therapy is currently the primary treatment option for advanced head and neck ACC, but effective targeted drugs are scarce in clinical practice. 28 Multi-targeted tyrosine kinase inhibitors (TKIs) have shown some antitumor activity in this population. In certain circumstances for ACC patients with DM, vascular endothelial growth factor receptor targeting TKIs, including axitinib, sorafenib, and lenvatinib, have been recommended.29,30 Early clinical trials of multi-kinase inhibitors targeting VEGFR have shown modest yet encouraging clinical benefits in metastatic ACC. These targeted treatments may be preferred over cytotoxic chemotherapy, particularly due to their favorable toxicity profile.
While our study has yielded insightful results, it is important to acknowledge its limitations. One of the major limitations is the use of a SEER database, which carries inherent biases and limitations, including selection bias, access bias, and data completeness challenges. Therefore, the generalizability of our findings to other populations or settings may be limited. Furthermore, it is important to note that perineural invasion, a significant histopathological feature in ACC associated with adverse outcomes such as OS, local control, and DM, was not included in the SEER database. This limitation restricted our ability to analyze the impact of perineural invasion on our study outcomes.
Moreover, we only focused on the effect of surgical resection on patients with ACC with synchronous metastases and could not obtain further information on recurrences or DM after surgery. This narrow focus and exclusion of other factors that may affect survival outcomes such as tumor stage or histology are further limitations of our study. Therefore, future studies with a more comprehensive and prospective approach are necessary to validate our findings and explore other aspects of ACC treatment.
Conclusion
In conclusion, our study provides important evidence supporting the favorable impact of PTS on survival outcomes in patients with bone and lung metastatic ACC at initial presentation. However, we acknowledge that not all patients may be suitable for surgery, and we suggest that a multidisciplinary team with both medical and surgical oncologists should carefully evaluate ACC patients with DM for PTS. Furthermore, larger prospective clinical studies are needed to validate our findings and explore the role of surgery and radiotherapy in the management of ACC. Our study highlights the need for further research in this area and provides a foundation for future investigations into the optimal treatment strategies for ACC patients with metastases.
Footnotes
Acknowledgements
The authors would like to thank the SEER program for providing open access to the database.
Authors’ Contributions
Conception and design: Shaoyan Liu and Song Ni; Administrative support: Shaoyan Liu and Song Ni; Collection and assembly of data: Han Li, Li Zhao, and Yixuan Song; Data analysis and interpretation: Song Ni and Yang Liu; Article writing and final approval: All authors.
Availability of data and materials
The data used and analyzed in this study are included in the article or are available from the corresponding and first authors on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Beijing Hope Run Special Fund of Cancer Foundation of China (Grant No. LC2017L04).
