Abstract
Herein described is an eight-year-old female patient presenting with bilateral external ear canal duplication and a history of recurrent postauricular abscesses, otorrhea, and moderate left-sided conductive hearing loss. Previous incision and drainage of the postauricular abscesses yielded little long-term success. Medical history was otherwise unremarkable. Physical examination revealed a cystic mass in the right ear and postauricular fibrosis, likely a result of previous surgical interventions. An audiogram revealed asymmetric hearing function, with mild-to-moderate conductive hearing loss in the left ear contrasting normal hearing in the right ear, while tympanometry confirmed an intact right tympanic membrane but was precluded on the left due to purulent otorrhea. A head CT scan revealed postauricular soft tissue swelling and well-pneumatized mastoid air cells. Both ears were explored intraoperatively via postauricular incisions. Surgical excision included removal of a cyst, cartilaginous remnants, and fibrotic tissue. The patient was followed up to assess for postsurgical complications. The surgical site healed uneventfully; no new symptoms or recurrences of past symptoms of otorrhea, cyst formation, or abscesses had developed within the twelve weeks post-surgery. Although external ear canal duplications are rare, it is imperative that early, accurate diagnoses be made to ensure optimal patient outcomes.
Keywords
Introduction
Branchial cleft anomalies are quite common, constituting approximately 30% of all congenital neck masses, most often presenting in early adulthood or childhood with no partiality to either sex.1-3 First branchial cleft anomalies, however, are much rarer and constitute 1% to 8% of all branchial cleft anomalies, with an estimated 1 in 100,000 being affected.4,5
First branchial cleft anomalies are subdivided into type I (ectoderm derived) and type II (ectoderm and mesoderm) according to a classification system put forth by Arnot and Worth.6,7 The two can be distinguished by the presence or absence of ectodermal cartilage. Notably, cartilage is absent in type II. Additionally, type I first branchial cleft anomalies typically pass posterior to the pinna lateral to the facial nerve, while type II anomalies traverse through the parotid gland medial to the facial nerve.2,8,9 Type II anomalies tend to involve a more complex course, often requiring more careful surgical intervention due to their relationship with the facial nerve.3,4 These anomalies can result in a variety of cysts, sinuses, cartilaginous remnants, and fistulae in the preauricular, postauricular, parotid, and upper neck areas. They are clinically significant given their propensity to become infected. 8 Type II first branchial cleft anomalies are described much more frequently in current literature than type I, which are quite rare.10-12 First branchial cleft anomalies may present as duplications of the external ear canal. Literature is characterized by few examples of unilateral external ear canal duplication, yet there have not been any reported cases of bilateral external ear canal duplication, to the best of our knowledge.9,13-24 Herein, we report a type I first branchial cleft anomaly, characterized by bilateral external ear canal duplication.
Case Presentation
The patient, an eight-year-old female, presented to a tertiary care center with bilateral recurrent postauricular abscesses with occasional bilateral otorrhea (Figure 1). While previous incision and drainage procedures provided short term relief, the abscesses recurred both unilaterally and bilaterally. Otorrhea occurred independently as well as in association with the postauricular abscesses. Family history was negative for recurrent otorrhea or abscesses; medical history aside from this presentation is unremarkable.
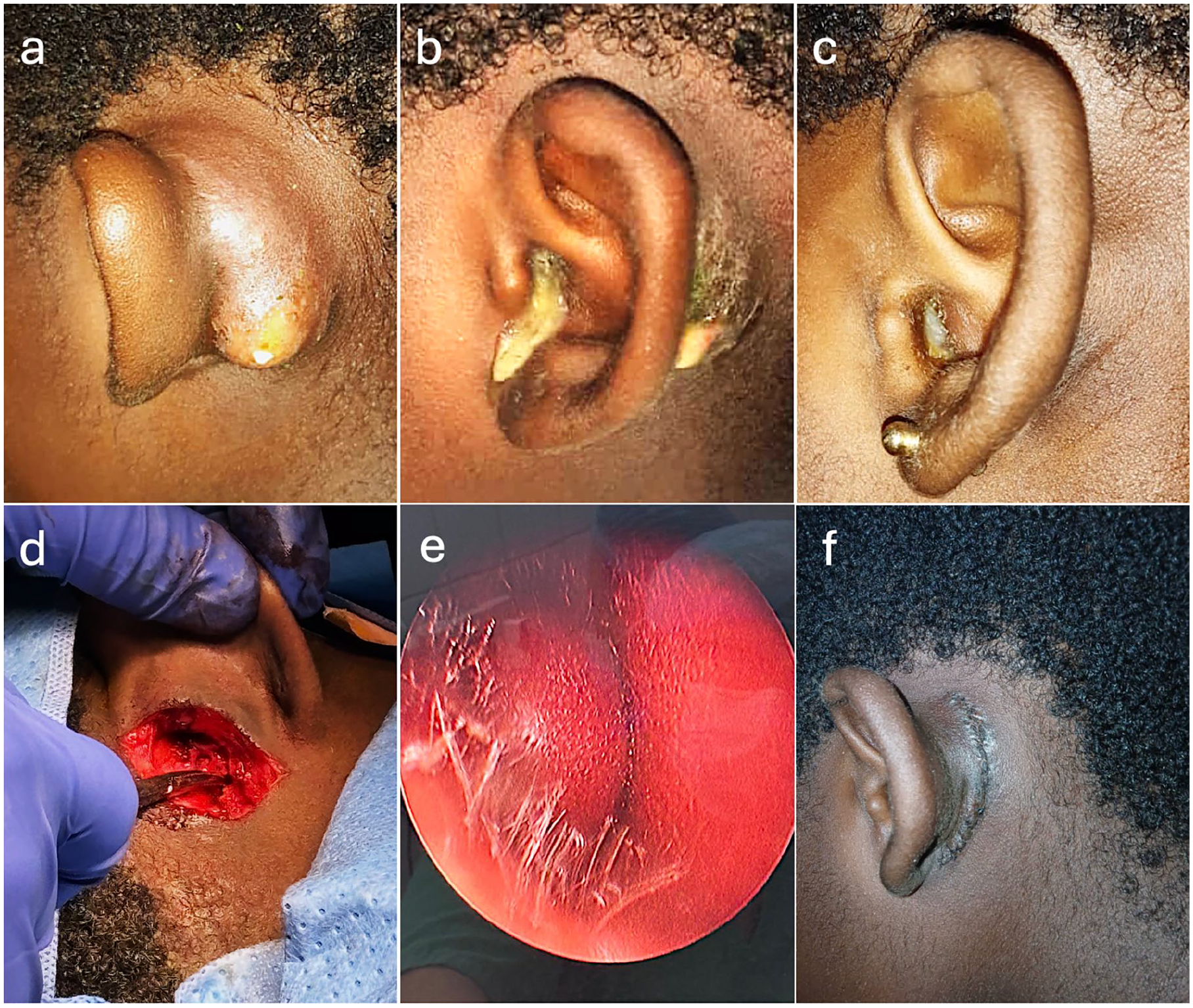
(a and b) Postauricular bilateral abscesses. (c) Left ear with otorrhea with no abscess. (b) Left ear with both otorrhea and an abscess. (d) Intraoperative picture showing the postauricular surgical approach. (e) Cystic mass present in the anterior wall of the right ear canal. (f) Postoperative photograph of the postauricular region.
An audiogram was performed, revealing a mild-to-moderate left-sided conductive hearing loss, but the right side was found to be within the normal range. Tympanometry was performed on the right side, revealing an intact tympanic membrane. Left-sided tympanometry could not be performed due to the presence of purulent discharge. Physical examination revealed the presence of a cystic mass in the right ear (Figure 1f). The left ear was devoid of any apparent cysts. Examination further revealed postauricular fibrosis and scarring, likely artifacts from previous surgical attempts.
A non-contrast CT scan of the head was performed, revealing postauricular soft tissue swelling and well-pneumatized mastoid air cells (Figure 2). The association of the squamous lined postauricular tract and its communication with the ear canal, in the context of the recurrent postauricular cystic infections, provided the basis for a bilateral ear canal duplication diagnosis. No other imaging was performed.
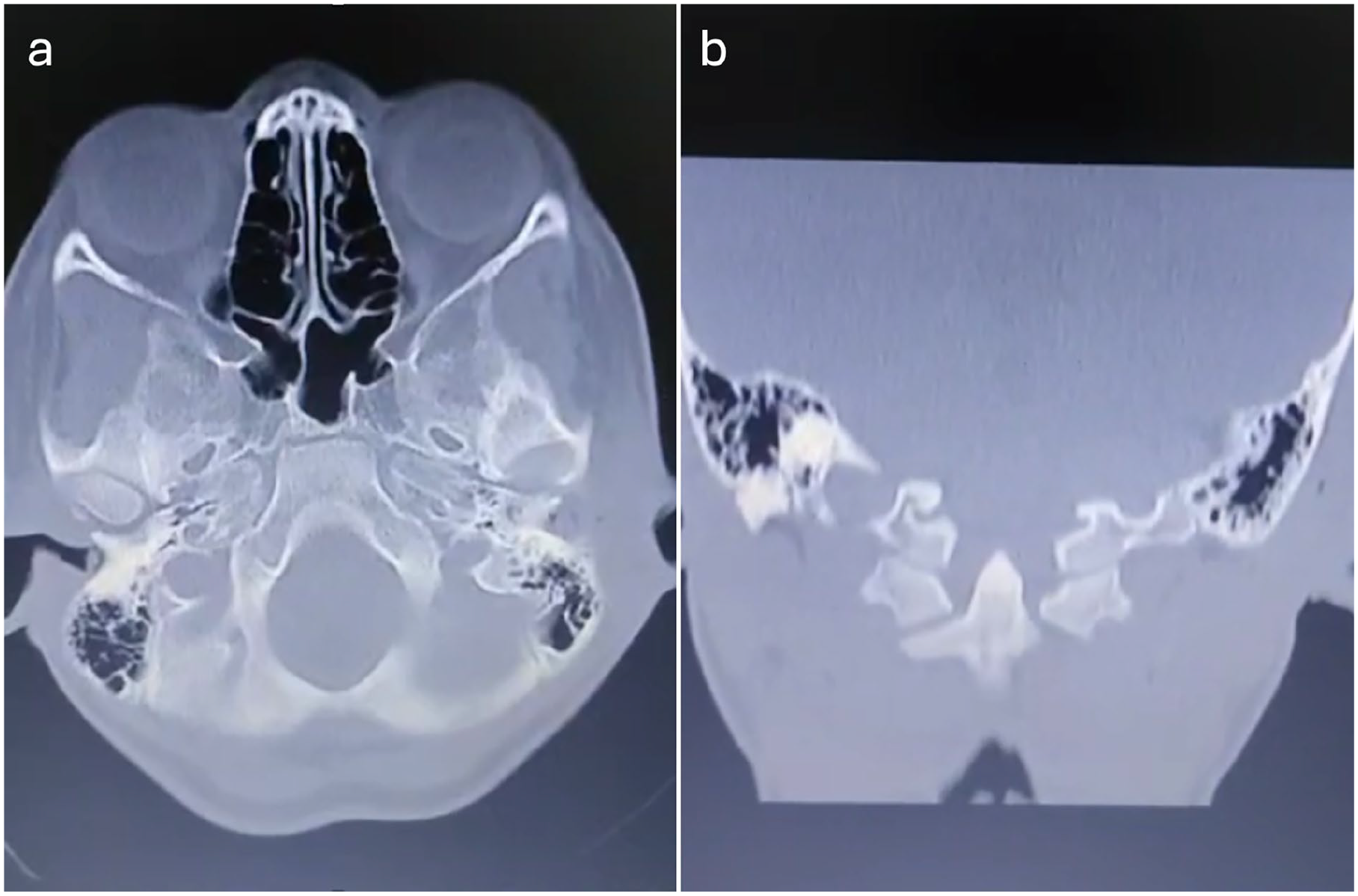
Preoperative axial (a) and coronal (b) head CT scan showing postauricular soft tissue swellings.
The sites of abscesses were bilaterally explored through postauricular incisions. On the right side, surgical excision included the removal of a cystic mass near the ear canal with an associated tract of cartilaginous remnant and skin. The left side excision was notable for the removal of fibrotic tissue, scarring, and small regions of inflammation. Both sides required dissection and soft tissue resection extending along the course of the ear canal. Dissection anteriorly was not required for full excision. Complete excision of the affected areas was achieved on both sides with the postauricular approach.
The surgical sites healed uneventfully without any other complications. At the 6 week follow-up, the patient denied any recurrence of prior symptoms—including otorrhea, cyst formation, or abscesses. No new symptoms developed in the twelve weeks post-surgery. Histopathology revealed the presence of skin fragments containing an intradermal cyst lined by stratified squamous epithelium producing laminated keratin, with no observable associated skin adnexa. These histopathological findings are suggestive of a sinus tract with an epidermal inclusion cyst.
Given the presence of ectodermal cartilage, absence of skin adnexa, and the location of the lesions posterior to the pinna, the patient’s branchial cleft anomalies were likely type I.
Discussion
It is imperative that head and neck surgeons be aware of less common otologic presentations such as the one described herein. Earlier accurate diagnosis will result in fewer iatrogenic insults such as ablating the facial nerve, or incomplete resection, leading to probable recurrence of the anomaly and further complications. 3 Literature has indicated that the risk of recurrence is high, with 2.4 procedures needed for complete removal of the abnormality. 3 Additionally, earlier intervention—complete surgical excision—may protect patients from infectious complications, such as cysts and abscesses. Indeed, incision and drainage of the patient’s postauricular abscesses on previous occasions at other healthcare facilities did not prevent recurrence, and thus, it seems that draining the recurrent cysts is an inadequate method of treatment.
Congenital first branchial cleft anomaly diagnosis is often made through the consideration of a combination of relevant clinical details, patient demographics, imaging findings, and intraoperative exploration. While imaging is helpful in making the diagnosis, there are several shortcomings to be aware of. Point-of-care ultrasound is a relatively-affordable option, but it has been reported to have an accuracy of 55.6% in diagnosing congenital first branchial cleft anomalies. 25 Head CT imaging is more accurate (75%) and provides excellent visualization of the first branchial cleft anomaly’s anatomical association with both the middle ear and external ear canal, 17 but is more expensive and puts the patient at risk of significant radiation exposure, which is significant as many presentations of congenital first branchial cleft anomalies are in pediatric populations. 25 MRI avoids the radiation exposure risk but is more costly and may not be accessible in all parts of the world. Auricular endoscopy is a cheaper, less invasive option that may allow for the visualization of the tympanic membrane and external ear canal; it may be helpful in informing the treatment approach.
Imaging may not always be enough to confirm a diagnosis of external ear canal duplication, and thus, further diagnostic measures may be required. Fine-needle aspiration or biopsy for cytological evaluation is particularly useful in evaluating cystic neck masses where clinical and imaging findings are inconclusive. Additionally, it may be helpful in ruling out malignancy or other more serious pathologies. Intraoperative surgical exploration may serve as confirmatory measures in instances where surgical intervention is indicated, and less invasive medical measures have failed. Histopathology from surgical specimens may further provide evidence for the diagnosis, as demonstrated in this patient’s circumstance.
The rarity of the patient pathology provides unique insight into refining differential diagnoses in similar presentations. Differential diagnoses should be weighted with an awareness of etiology likelihood and contrastive indications. Other conditions that may lead to a similar presentation include the following: dermoid cysts, salivary gland neoplasms, lymphadenopathy, cystic hygroma, and thyroglossal duct cysts.26-31 The presence of squamous lined postauricular tract and chronic postauricular cystic infections in association with its communication with the ear canal provided particular indication of ear canal duplication. The histopathological confirmation of present ectodermal cartilage, absence of skin adnexa, and the lesions located behind the pinna provide basis for a type I branchial cleft anomaly. It is worth noting the relative infrequency of first branchial cleft anomalies in comparison with more common etiologies. While instructive, this presentation of bilateral external ear canal duplication is so far limited to the described patient and does not necessitate dramatic alterations in diagnostic decision-making.
Ultimately, diagnostic decisions should be guided by the patient’s demographic characteristics, namely age. While the age of onset of symptoms has not been well characterized in literature, one study identified a cohort of 100 patients with confirmed first or second branchial cleft anomalies and an average age of 4.0 ± 2.7 years. 25 The initial presentation of symptoms during adulthood is much rarer. 32 Additionally, while a genetic component to first branchial cleft anomaly occurrence has not been solidly established, there has been a report of five first branchial cleft anomalies spanning across four familial generations. 32 Thus, a family history of first branchial cleft anomalies may warrant targeted workup in a patient who presents with findings consistent with first branchial cleft anomalies.
The success of the exploratory surgical approach and excision methodology provides a direct contrast to previous unsuccessful interventions in the patient’s care and underscores the necessity of more fully exploring differential diagnoses, including less common ones, in related patient presentations. The application of an approach as outlined above may aid physicians in improving patient outcomes and speed of diagnosis while reducing symptom relapse.
Conclusion
Herein, we presented the case of an eight-year-old female with a presentation of bilateral ear canal duplication with complete resolution of symptoms after surgical excision. First branchial cleft anomalies are uncommon. Of those involving external ear canal duplication, most documented cases are unilateral, and this is the first report of which we are aware of a bilateral external ear canal duplication in literature. This case highlights the importance of thorough intraoperative exploration and complete resection of anomaly remnants in the management of first branchial cleft anomalies with failure in diagnosis and treatment allowing the recurrence of symptoms and poor patient outcomes.
