Abstract
Objective:
We analyzed ear-related factors contributing to postoperative infections after myringoplasty. We also examined the association between the timing of postoperative infections and graft success.
Materials and methods:
This retrospective study reviewed clinical data and operative records of patients with chronic tympanic membrane (TM) perforations who underwent myringoplasty. Factors influencing postoperative infection, including surgical techniques, perforation size, external auditory canal (EAC) packing, prophylactic antibiotic type, and the application of topical antibiotic cream, were assessed. In addition, the relationship between the timing of postoperative infection and graft failure was evaluated. Postoperative infection time was categorized as 1, 2, 3, or 4 weeks postoperatively.
Results:
In total, 263 patients with chronic TM perforations underwent myringoplasty. The overall postoperative infection rate was 12.2% (32/263). The modified over-underlay approach was associated with a postoperative infection rate of 19.0%, in contrast to 11.1% for the conventional over-underlay technique and 8.9% for the underlay technique (P = .081). No significant differences in postoperative infection rates were identified among the NasoPore packing group (10.9%), the erythromycin cream group (19.4%), and the no-packing group (11.9%) (P = .418). Similarly, no significant differences were observed between prophylactic antibiotic type (11.2% vs 12.2% vs 25.0%, P = .373) or between the patients with and without topical antibiotic ointment (12.4% vs 10.3%, P = .986). Among the 32 patients with postoperative infections, 21.9% achieved graft success, whereas 78.1% experienced graft failure. Graft success rates were 71.4% (5/7), 9.1% (2/22), and 0.0% (0/3) at 1, 2, and 3 weeks postoperatively, respectively (P < .01).
Conclusions:
Our findings suggest that prophylactic antibiotic type, perforation size, the use of topical erythromycin cream, and EAC packing do not significantly influence postoperative infection. However, the surgical technique may affect the risk of postoperative infection. In addition, the timing of postoperative infection may influence graft failure.
Introduction
Myringoplasty, a fundamental procedure for tympanic membrane (TM) reconstruction, exhibits graft success rates strongly influenced by surgical technique, graft size, graft rejection, middle ear mucosal disease, and eustachian tube dysfunction.1-4 However, postoperative infection remains a critical risk factor for graft failure, leading to partial or complete graft necrosis.5,6 Biodegradable NasoPore, a US Food and Drug Administration 510(k)-approved biocompatible and biodegradable/fragmentable synthetic polyurethane foam, has demonstrated high biocompatibility and effectively supports grafts or prostheses after middle ear surgery.1,3,4 Early identification and management of postoperative infections are essential for optimizing graft success rates. Although numerous studies have investigated the role of prophylactic antibiotics in preventing postoperative infections, limited research has evaluated the ear-related factors that influence postoperative infections and their impact on graft survival after myringoplasty.7,8 This study analyzed ear-related factors contributing to postoperative infections after myringoplasty; it also investigated the association between the timing of postoperative infections and graft success.
Materials and Methods
Ethical Considerations
The Medical Ethical Committee of Yiwu Central Hospital approved the study protocol. Written informed consent was obtained from all participants.
Methods
This retrospective study analyzed clinical data and operative records of patients with chronic TM perforations who underwent endoscopic perichondrium-cartilage myringoplasty in the Department of Otolaryngology between January 1, 2019, and December 31, 2023. Clinical data were extracted by inpatient and outpatient electronic medical record systems, including surgical technique, date of surgery, patient age and sex, affected ear, perforation size, graft healing status, presence of residual or re-perforations, perforation margin trimming, postoperative infection and the timing of infections, ear discharge (amount and color of discharge, etc), presence or absence of external auditory canal (EAC) packing, and use of topical erythromycin cream. The inclusion criteria were age more than 16 years, perforation size more than 1/8 of TM and less than 3/4 of TM, chronic TM perforations associated with chronic otitis media, a dry ear for ≥3 months prior to surgery, and regular medical follow-up with at least 6 months. Exclusion criteria included incomplete data, revision surgery, cholesteatoma, total perforation, lost follow-up, and ossiculoplasty. The perforation size was categorized as medium (1/8-1/4 of TM), large (1/4-1/2 of TM), and subtotal (1/2-3/4 of TM).
Surgical Technique
All patients underwent endoscopic perichondrium-cartilage graft myringoplasty under total intravenous anesthesia performed by the same otolaryngologist. The perichondrium-cartilage graft was harvested from the ipsilateral tragus in all the patients. The perichondrium-cartilage graft was placed medial to the TM remnant and annulus in an underlay manner in the underlay technique. 4 The conventional over-underlay technique involved the removal of the epithelial layer of the TM remnant, the cartilage graft was placed medial to the TM remnant in an underlay manner, but the lateral perichondrium was elevated and placed lateral to the fibrous layer of the TM remnant. However, the modified over-underlay technique did not involve the removal of the epithelial layer of the TM remnant but the lateral perichondrium was directly placed lateral to the epithelial layer of the TM remnant. 9 The middle ear cavity was packed with biodegradable synthetic polyurethane foam (NasoPore, Stryker Canada, Hamilton, ON, Canada), with or without erythromycin cream. EAC packing was performed using biodegradable NasoPore with erythromycin cream, erythromycin cream alone, or no packing. The use of biodegradable NasoPore as a packing material for the middle ear and EAC is standard practice in our department.
Postoperative Care and Follow-up
Patients were discharged the day after surgery and prescribed oral systemic prophylactic antibiotics for 1 week. In addition, the patients were advised to keep their ears clean, avoid water entering the EAC, prevent upper respiratory tract infections, etc.
For prophylactic antibiotics use, a standardized antibiotic protocol was designed as follows: Amoxicillin and clavulanate potassium should be considered as a choice of first-line therapy, Cefuroxime Axetil was selected for the patients with penicillin allergy; however, Levofloxacin tablet was used for the patients with both penicillin and cephalosporin allergy.
Postoperative follow-up visits were scheduled at 1, 2, 3, and 4 weeks and 2, 3, and 6 months. All patients completed the 6-month follow-up period. At each follow-up visit, graft status and the presence of postoperative infection were assessed through endoscopic evaluation. NasoPore fragments were aspirated at the 2-week follow-up visit as needed. Postoperative wound site infection was defined as the presence of yellow sticky purulent discharge in the EAC and middle ear within 1 month after surgery, with local pain, with or without fever. Watery color otorrhea was not considered indicative of postoperative infection.
Assessment of Ear-Related Factors Influencing Postoperative Infection
We evaluated potential ear-related factors influencing postoperative infection, including surgical techniques (underlay technique, conventional over-underlay technique, or modified over-underlay technique), perforation size (medium, large, or subtotal), EAC packing method (erythromycin cream, NasoPore packing, or no packing), the use of topical antibiotic cream, and prophylactic antibiotic type (including amoxicillin and clavulanate potassium, or Cefuroxime Axetil, or Levofloxacin tablet). The application of topical erythromycin cream was defined as its use in packing the middle ear and EAC. In addition, the correlation between the timing of postoperative infection and graft failure was evaluated. Postoperative infection time was categorized as 1, 2, 3, or 4 weeks postoperatively. Graft survival was defined as the presence of partial or complete inosculation between the graft and TM remnant, as well as graft neovascularization. Graft success was defined as an intact graft without residual or recurrent perforations during the follow-up period. Patients displaying postoperative midline septic foci in the nose or throat or those exhibiting upper respiratory tract infections within 1 month after surgery were excluded.
Statistical Analysis
Statistical analysis was performed using SPSS version 21 (IBM, Armonk, NY, USA). Non-parametric data are presented as numbers and percentages. The Chi-square test or Fisher’s Exact Test was used to compare postoperative infection rates among groups. P-values <.05 were considered statistically significant.
Results
Demographic Characteristics
In total, 263 patients with chronic TM perforations were included. Of these, 141 (53.6%) were female and 122 (46.4%) were male. The left ear was affected in 158 patients (60.1%), whereas the right ear was affected in 105 patients (39.9%). The mean age of the patients was 48.1 ± 12.6 years, and the mean duration of perforation was 13.8 ± 6.4 years.
Postoperative Infection, Treatment Protocol, and Graft Survival
Of the 263 patients, 32 (12.2%) experienced postoperative infections. Among these patients, 13 (40.6%) presented with purulent discharge in the EAC; 19 (59.4%) exhibited discharge in both the middle ear and the EAC. All patients with postoperative infections received oral antibiotics as prophylactic antibiotics before infection; however, 28 patients received ceftriaxone 2 to 3 g + dexamethasone 5 mg intravenous injection daily for 3 days after infection, then, ceftriaxone alone intravenous injection was administered until the infection was completely controlled.
However, 4 patients received Levofloxacin sodium chloride inj 0.3 g bid intravenous injection + dexamethasone 5 mg intravenous injection daily for 3 days, then, Levofloxacin sodium chloride inj alone intravenous injection was administered.
In addition, topical Levofloxacin hydrochloride ear drops were applied twice daily. All 32 patients with postoperative infection were completely controlled within 7 to 11 days.
Regarding graft survival, 7 patients (21.9%) achieved complete graft survival without residual perforation, 16 patients (50.0%) experienced partial graft necrosis with partial graft survival and residual perforation, and 9 patients (28.1%) experienced complete graft necrosis with total perforation.
Graft Success Rate
At 6 months postoperatively, graft failure was observed in 36 patients (10.3%), resulting in an overall graft success rate of 86.3% (n = 227). Among the 36 graft failures, 9 (25.0%) were attributed to insufficient graft size or other factors, 2 (5.6%) were due to graft collapse, and 25 (69.4%) were the result of postoperative infection.
Ear Factors Affecting Postoperative Infection
Table 1 summarizes potential ear-related factors influencing postoperative infection rates. Regarding surgical techniques, the modified over-underlay approach was associated with a postoperative infection rate of 19.0%, in contrast to 11.1% for the conventional over-underlay technique and 8.9% for the underlay technique (P = .081). No statistically significant differences in postoperative infection rates were identified based on EAC packing, perforation size, prophylactic antibiotic type, or the application of topical erythromycin cream.
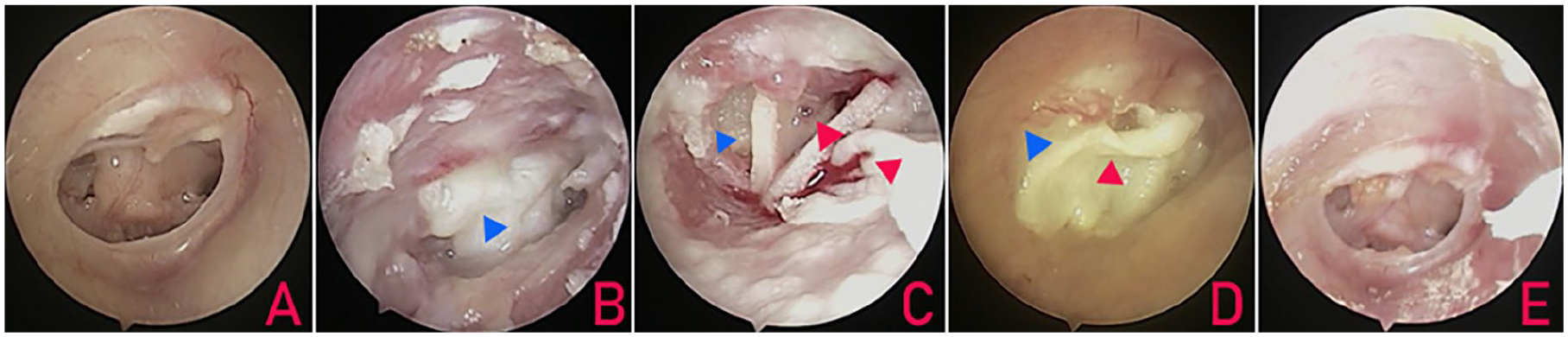
The Difference Comparison of Factors Related to Postoperative Infection.
The categorical data were presented as the number (percentage). Differences were examined using the Chi-square test or Fisher’s Exact test.
Abbreviation: EAC, external auditory canal.
Postoperative Infection Time and Graft Success
All patients who developed postoperative infections were successfully treated with a combination of topical and systemic antibiotics. Among patients without EAC packing, 7 (21.9%) experienced postoperative infections within 1 week, whereas 1 (3.1%) presented with an infection at 2 weeks postoperatively. The duration of infection in this group was <1 week. Conversely, among patients with EAC packing, 21 (65.6%) experienced infections at 2 weeks postoperatively, and 3 (9.4%) developed infections at 3 weeks postoperatively due to delayed follow-up. The duration of infection in this group exceeded 1 week.
Postoperative infection resulted in partial graft necrosis in 16 patients (50.0%) and complete graft necrosis in 9 patients (28.1%), leading to residual perforation and graft failure (Figures 1 and 2). However, 7 patients (21.9%) with postoperative infections within 1 or 2 weeks achieved complete graft survival. Of the 7 patients, the graft was re-intervened in 2 patients with postoperative graft expulsion. Finally, all 7 patients achieved successful closure without residual perforation.
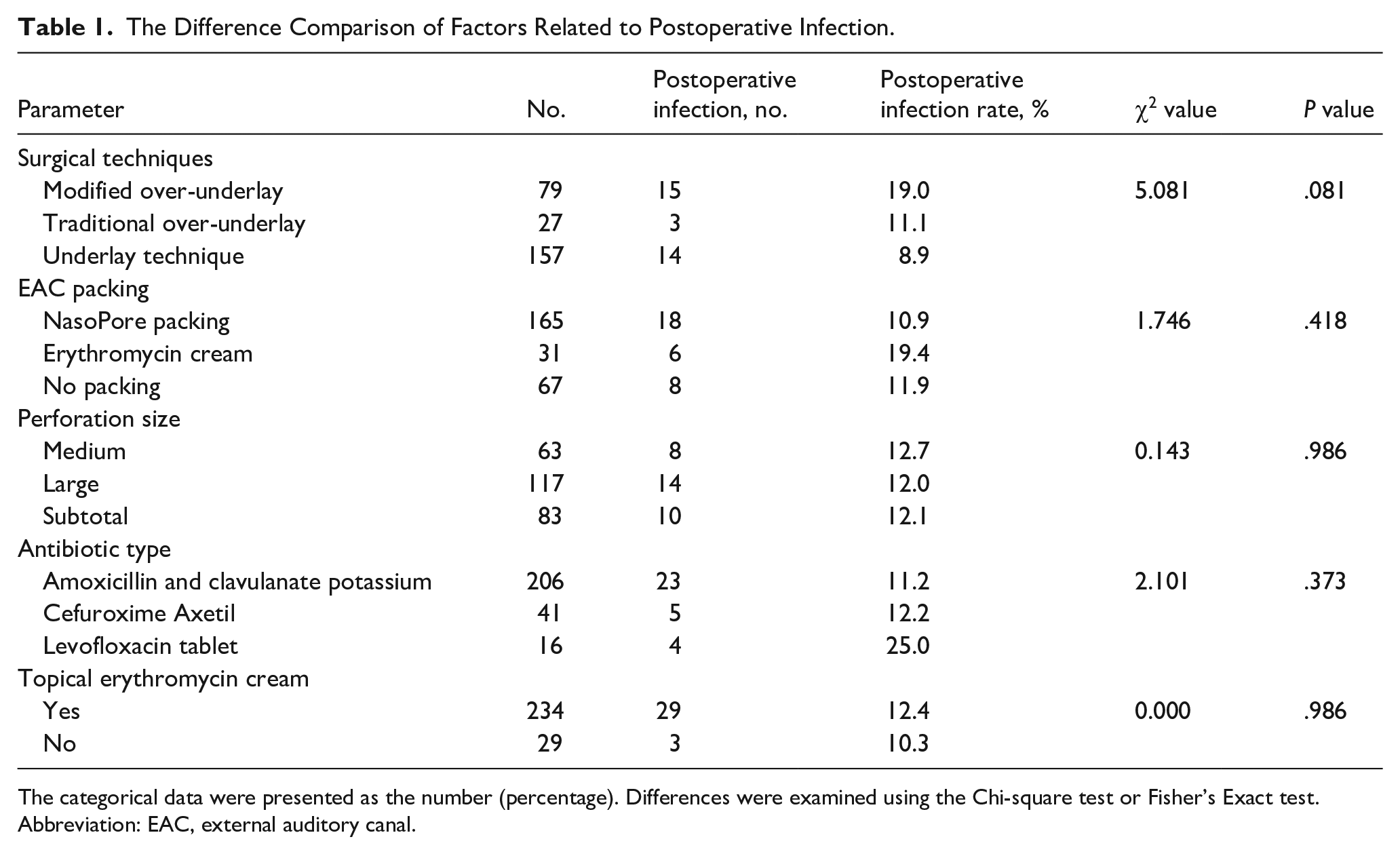
Right large perforation. (A) Preoperative perforations; (B) postoperative 2 weeks; (C) 4 weeks; (D) 3 months; and (E) 24 months. The blue triangle indicates the purulence discharge and the red triangle indicates the graft.
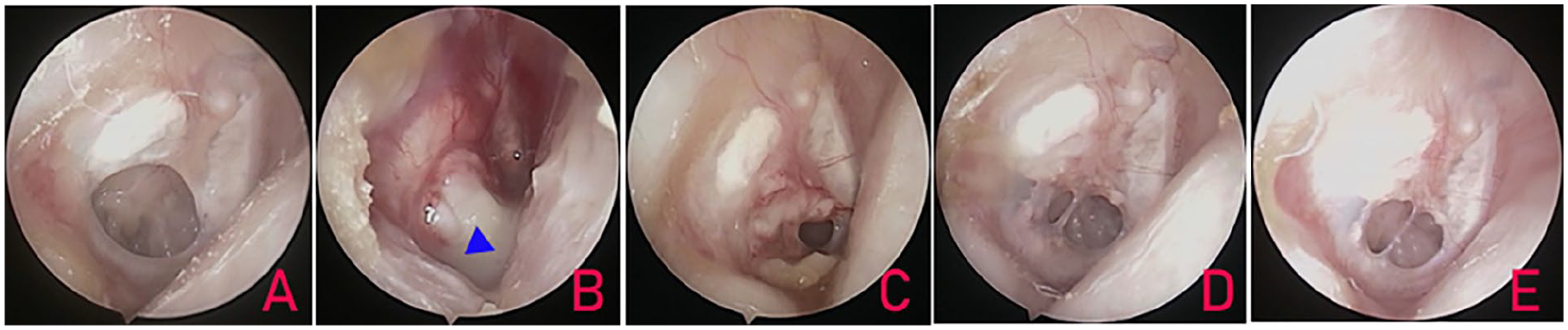
Right subtotal perforation. (A) Preoperative perforations; (B) postoperative 3 weeks; (C) graft extrusion and restoration 4 weeks; (D) 6 weeks; (E) 12 months. The blue triangle indicates the purulence discharge and the red triangle indicates the graft.
Table 2 shows the association between postoperative infection time and graft success. Graft success rates were 71.4% (5/7), 9.1% (2/22), and 0.0% (0/3) at 1, 2, and 3 weeks postoperatively, respectively (Figures 2 and 3; P < .01). Notably, among the 7 patients who exhibited infections within 1 week of surgery, 5 with graft survival experienced infection onset on postoperative days 3 to 4.
The Postoperative Infection Time and Graft Success.
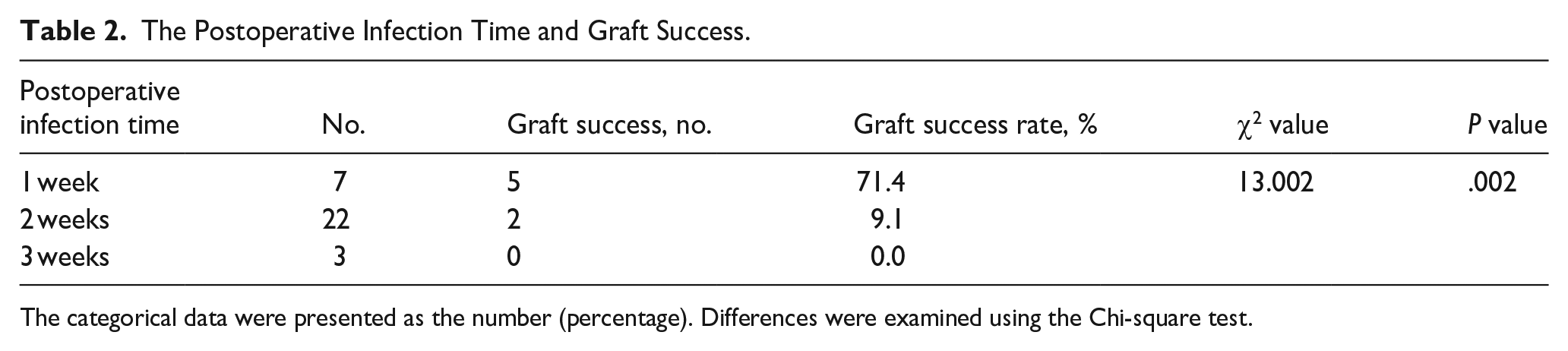
The categorical data were presented as the number (percentage). Differences were examined using the Chi-square test.
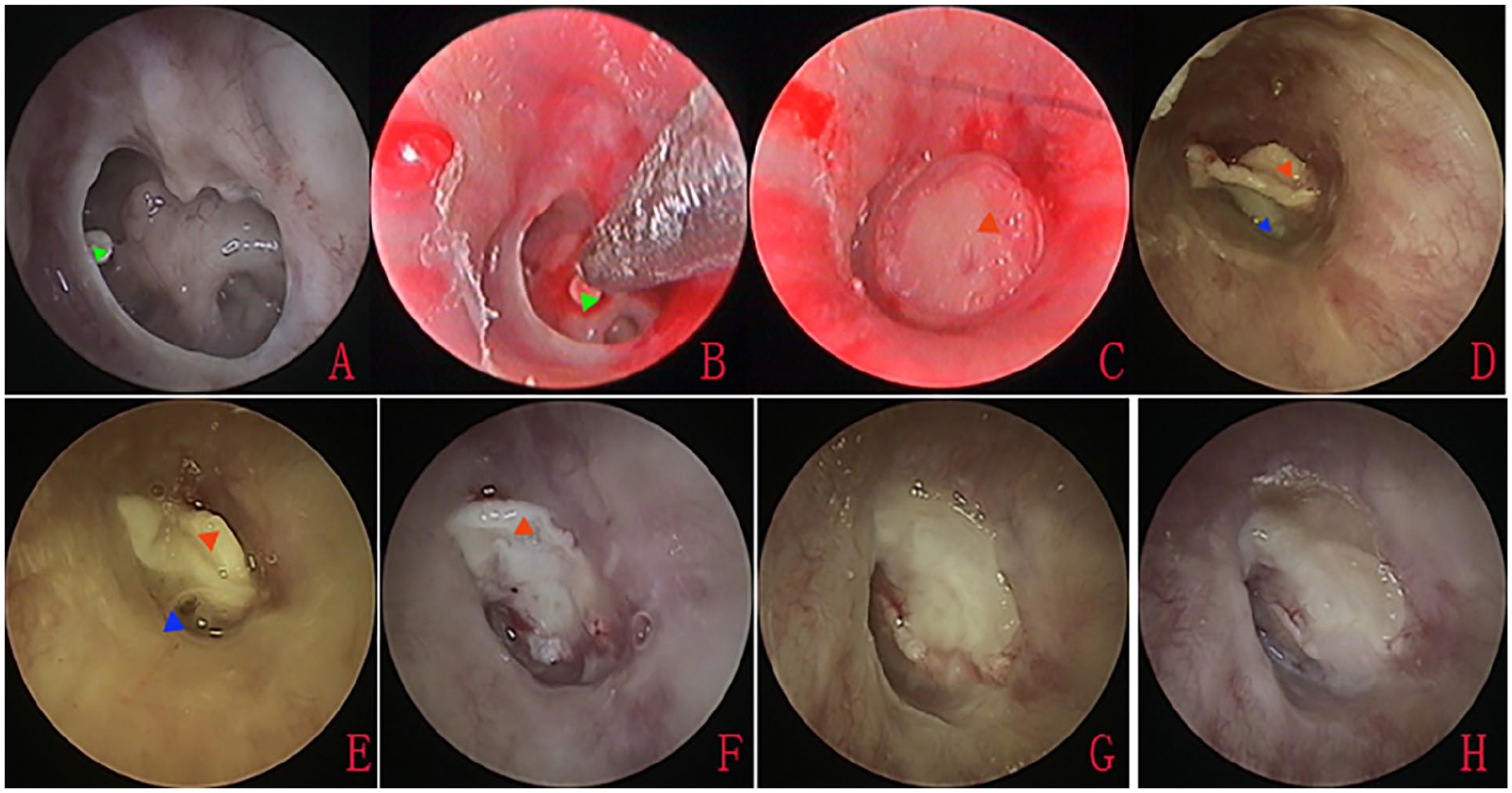
Left subtotal perforation. (A) Preoperative perforations with crust-like substance of anterior margin; (B) removal of crust-like substance of anterior margin; (C) perforation closure using graft; (D) graft extrusion at postoperative 1 week; (E) graft expulsion and restoration 9 days; (F) 2 weeks; (G) 18 days; (H) 6 weeks. The green triangle indicates the crust-like substance, the blue triangle indicates the purulence discharge, and the red triangle indicates the graft.
Discussion
Some scholars evaluated the ear-related risk factors leading to failure in myringoplasty. Ordóñez-Ordóñez et al 10 believed that inflammation at the time of surgery, place, and size of perforation, presence of tympanosclerosis or cholesteatoma, and surgical technique were not associated with graft failure. Dangol et al 2 found that a diseased contralateral ear was the only poor prognostic factor for graft uptake. On the contrary, Dursun et al 11 found that the condition of the contralateral ear did not significantly affect the surgical success, the size of perforation and EAC protrusion were significantly related to graft success. Nevertheless, Fermi et al 1 demonstrated that the graft material was the only statistically significant prognostic factor for graft failure, the type of surgery and the type and size of perforation were not significantly associated with the postoperative result. Kolsi et al 12 found that the only factor significantly influencing graft failure was the history of chronic rhinosinusitis. These findings did not definitively identify the ear-related factors influencing graft failure in myringoplasty.
More scholars have confirmed that postoperative infection is a major risk factor for graft failure after myringoplasty.5,13-15 Vartiainen et al 16 reported that 61.4% (27/44) of graft failure resulted from postoperative infection. Dispenza et al 15 found that all 10 patients contracting an infection of the middle/external ear resulted in surgical failure during the postoperative period. Housley et al 13 demonstrated that postoperative infections were associated with a statistically significant higher failure rate. Therefore, the identification of risk factors for postoperative infection is essential to improve graft success rates.
Several factors contribute to graft infection, including preoperative polyps or granulation tissue, water exposure, smoking, and postoperative mucositis. In addition, midline septic foci such as adenoids, sinusitis, recurrent colds, chronic tonsillitis, and allergic rhinitis can lead to eustachian tube dysfunction and graft failure. Systemic conditions, including inadequate menstrual hygiene, malnutrition, diabetes mellitus, and immunocompromised states, are also known to increase the risk of postoperative infection. However, we investigated potential ear-related factors influencing postoperative infection in this study.
Although myringoplasty is considered a clean surgical procedure, postoperative bacterial changes in the middle ear can result in recontamination and subsequent infection, the host-derived acute inflammatory responses contribute to graft rejection and infection. 14 In addition, prolonged operation time further enhances the risk of infection. 17 In the present study, a higher postoperative infection rate (19.0%) was observed with the modified over-underlay technique relative to other grafting techniques. Unlike the conventional underlay technique, where the epithelial layer is directly exposed to air, the modified over-underlay technique involves placing the single-sided perichondrium graft lateral to the epithelial layer. 9 This placement may alter the bacterial flora.
Westman et al 17 reported middle ear infection rates of 15.4% and EAC infection rates of 34.6% in patients who underwent myringoplasty with a tympanomeatal flap and EAC packing. Nair et al 18 reported postoperative infection rates of 19% in conventional myringoplasty with EAC packing and 4.7% in autologous platelet-rich fibrin graft procedures without EAC packing. Although EAC packing may alter bacterial flora and increase the risk of postoperative infection, our findings did not demonstrate a significant association between EAC packing and increased postoperative infection rates. Similarly, Tabaru et al 19 recently demonstrated that myringoplasty without ear packing did not affect graft take rates and hearing improvements. Although several studies have demonstrated an inverse correlation between perforation size and graft success rates, no associations have been observed between large perforation size and an increased risk of postoperative infection.1,2,11 In addition, the application of topical erythromycin cream and postoperative prophylactic antibiotics have been used to prevent postoperative infection during myringoplasty. However, the present study did not demonstrate a significant reduction in postoperative infection rates with the use of topical erythromycin cream or the type of postoperative prophylactic antibiotics. Consistent with these findings, Westman et al, 17 in a multivariable regression analysis, reported that patients who did not receive prophylactic antibiotics did not experience an increased risk of postoperative infection. On the contrary, prophylactic antibiotics did not reduce the risk of infection.
In our study, patients without EAC packing exhibited earlier infection onset such that 7 of 32 patients developed infections within the first week postoperatively. By contrast, of the 24 infections observed in patients with EAC packing, 21 presented with purulent discharge upon removal of the packing at 2 weeks postoperatively, whereas 3 exhibited purulent discharge and graft necrosis at 3 weeks postoperatively. These findings suggest that most postoperative infections occur within the first 2 weeks after surgery, which agrees with the work of other scholars. Dispenza et al 15 reported that all 10 cases of postoperative infection occurred within the first 2 weeks and before the removal of the EAC dressing. The absence of EAC packing facilitates early detection of postoperative infection. We recommend removing EAC packing material at 1 week postoperatively, regardless of its initial placement.
Factors influencing the variability of graft outcomes in patients experiencing postoperative infection remain unclear. Although most studies indicate that a higher graft failure rate is associated with postoperative infection, not all infections result in graft failure.13,15,18 Prescott et al 20 observed graft success in 77% and graft failure in 36% of patients with postoperative infections. Similarly, Khanal et al 6 reported graft success in 1 and graft failure in 2 of 3 patients with postoperative infections.
Despite the use of cartilage grafts in this study, previous works have demonstrated a substantial detrimental effect of middle ear infection on chondrocytes. 21 In our study, 21.9% of patients with postoperative infections achieved graft success, whereas 78.1% experienced graft failure. In addition, the present study revealed a significant decline in the graft success rate over time, with a 71.4% success rate at 1 week postoperatively, decreasing to 9.1% at 2 weeks and 0% at 3 weeks (P < .01). These findings suggest a strong correlation between graft failure and the timing of postoperative infection, a high graft success rate was observed when infection occurred within the first postoperative week.
Consistent with our findings, several studies have demonstrated a significantly elevated risk of graft failure among patients who develop postoperative infections within 6 weeks. 12 Dispenza et al 15 reported that postoperative infection occurred within the first 2 weeks in 10 patients, leading to graft failure. Similarly, Khanal et al 6 observed a graft failure rate of 66.7% (2/3) when infections occurred within 2 to 3 weeks after surgery. Prescott et al. 20 reported a graft perforation rate of approximately 33% when infection occurred during the first 2 weeks postoperatively. Likewise, Housley et al 13 observed that early postoperative infection (<1 month) when the graft-TM complex is still healing was associated with higher failure rates (40%).
The correlation between the timing of postoperative infection and graft survival remains unclear. Graft healing is a complex process characterized by inosculation, neovascularization, and epithelization. 22 Inosculation occurs within the first week after grafting, initiating subsequent neovascularization, which is a crucial factor in graft survival.22-24 During the first week postoperatively, preexisting fibrous connective tissue within the graft and developing neovascularization contribute to an enhanced anti-infection capacity. 25 Prompt administration of topical and systemic antibiotics during early infection can significantly improve graft survival. Conversely, at 2 to 3 weeks postoperatively, neovascularization diminishes; the graft enters a phase of renewed fibrous proliferation and epithelization. This renewed fibrous tissue formation weakens the graft’s anti-infection defenses. Consequently, even with the administration of topical and systemic antibiotics, grafts may become readily absorbed and liquefied. Pau et al 26 emphasized that clinically incorporated grafts exhibit continued vulnerability (locus minoris resistentiae). Therefore, decisive therapeutic interventions are imperative at the first signs of inflammation. Uncontrolled infection can halt neovascularization and ultimately lead to graft failure.
Unfortunately, although postoperative infection within 1 week did not result in graft necrosis, it may lead to graft expulsion in a few patients. In this study, of the 7 patients with complete graft survival, 2 patients had postoperative graft expulsion, the graft was re-intervened to achieve perforation closure, who had been included in the graft success. Therefore, early re-intervention should be considered for patients with graft expulsion without graft necrosis. Nevertheless, the postoperative graft expulsion and restoration did not significantly affect graft success rates because of the small patient’s number.
This study had several limitations. First, wound bacterial cultures provide a more standardized approach to confirming infection. However, unfortunately, wound cultures and drug sensitivity tests were not performed in this study, the absence of bacterial culture and drug sensitivity data precluded a comprehensive understanding of the infecting pathogens and the development of optimal antibiotic treatment strategies. Second, the relatively small sample size limited the statistical power of the multivariate regression analysis. Third, the study did not evaluate the impact of systemic diseases or immune status on patient outcomes. Finally, potential relationships of infection with patient demographics (age, sex, and body mass index), surgical factors (eg, operation time), and contralateral ear were not analyzed.
Conclusions
Our findings suggest that prophylactic antibiotic type, perforation size, the use of topical erythromycin cream, and EAC packing do not significantly influence postoperative infection. However, the surgical technique may affect the risk of postoperative infection. In addition, the timing of postoperative infection may influence graft failure.
