Abstract
Objective
The objective of this study was to compare the postoperative infection and graft success rates, and the hearing improvement, after endoscopic cartilage underlay myringoplasty with versus without antibiotic ointment coating.
Materials and methods
This was a retrospective case-control study. The clinical records of patients who underwent endoscopic cartilage underlay myringoplasty and who met the selection criteria were retrieved and divided based on middle ear packing status into groups with antibiotic ointment packing (AOP group) and with no antibiotic ointment packing (no-AOP group). The operation time, postoperative infection, graft success status, and hearing improvement were compared between the 2 groups.
Results
Patients with 166 perforations constituted the AOP group, and patients with 141 perforations comprised the no-AOP group. At 3 months postoperatively, middle ear infections had occurred in 24 (14.5%) ears in the AOP group and 4 (2.8%) ears in the no-AOP group (P < .01). At 12 months postoperatively, the graft success rate was 81.3% in the AOP group and 97.9% in the no-AOP group (P < .01). No significant group differences were observed, preoperatively (P = .657) or postoperatively (P = .578), in the air-bone gap (ABG) values or mean ABG gains (P = .758).
Conclusion
Middle ear packing without antibiotic ointment coating does not increase the postoperative infection rate or reduce the graft success rate after endoscopic cartilage underlay myringoplasty compared to antibiotic ointment coating. On the contrary, coating with antibiotic ointment increases the risk of postoperative infection given the complexity of middle ear manipulation.

Introduction
Middle ear packing is a basic procedure during myringoplasty; packing is used to support the graft in situ and prevent graft medialization.1-3 Middle ear packing material that contains an antibiotic ointment is often placed during myringoplasty by some otolaryngologists.3-5 However, no consensus has been reached in terms of whether middle ear packing should contain antibiotic ointment or not. The exact role played by topical prophylactic antibiotics (PAs) in reducing postoperative infection is not fully understood. Most scholars believe that a tympanic membrane (TM) perforation developed after chronic suppurative otitis media becomes colonized by various microbes and that topical PAs reduce the postoperative infection rate and improve the graft success rate.6,7 However, others found that the flora associated with chronic ear disease is resistant to most antibiotics that can be safely prescribed; topical PAs did not reduce the risk of postoperative infection.8-11
In our department, biodegradable NasoPore material containing 2% mupirocin ointment was previously routinely used to pack the middle ear during otologic surgery. Mupirocin (pseudomonic acid A; Bactroban®) is a new topical antimicrobial agent with a broad spectrum of antimicrobial activity against many Gram-positive organisms, including Staphylococcus aureus, methicillin-resistant S. aureus (MRSA), and vancomycin-resistant S. aureus. The mechanism of action is unique; the drug binds to isoleucyl transfer RNA synthetase, thereby blocking bacterial protein synthesis. 12 Mupirocin has been widely used to treat post-tympanostomy tube otorrhea (PTTO) and is efficacious and not ototoxic.13-15 In recent years, we have constantly sought to optimize the surgical procedure, including by not raising a tympanomeatal flap, not trimming the perforation margins, not adding an antibiotic ointment, and not packing the external auditory canal (EAC).3-5,16,17 The objective of this study was to retrospectively compare the operation times, postoperative infection, graft success rates, and hearing improvements after endoscopic cartilage underlay myringoplasty with and without antibiotic ointment coating.
Materials and Methods
Ethical Considerations
Ethical approval for the present study was obtained from the Medical Ethical Committee of Yiwu Central Hospital. Informed consent was obtained from all participants.
Methods
We retrospectively studied the clinical data and operative records of patients with chronic perforations who underwent endoscopic cartilage underlay myringoplasty in the Department of Otolaryngology. The inclusion criteria were as follows: a chronic perforation with chronic otitis media; a dry ear for at least 3 months prior to surgery based on patient history; and good pneumatization of the middle ear and mastoid region, as shown by high-resolution computed tomography. The exclusion criteria were as follows: active inflammation, revision surgery, a cholesteatoma, and a need for ossiculoplasty. The perforation size was categorized as medium (≤12.5% but <25% of the TM area), large (>25% but ≤50% of the TM area), or subtotal (>50% but ≤75% of the TM area). The perforation size was measured using the ImageJ software (NIH, Bethesda, MD, USA) by 2 surgeons in the outpatient setting.18,19
The patients were classified into an antibiotic ointment packing group (treated using the AOP technique) and a non-antibiotic ointment packing group (treated using the no-AOP technique). In our department, the AOP technique was performed principally from July 2016 to November 2020. This involved the removal of the perforation margins and epithelium of the malleus handle, middle ear packing with biodegradable material (NasoPore) containing 2% mupirocin, and EAC packing. However, subsequently, the surgical technique was optimized, and the no-AOP technique has been employed from December 2020 to the present. The no-AOP technique does not include trimming of the perforation margins, the use of 2% mupirocin ointment, or EAC packing; only removal of the epithelium of the malleus handle and middle ear packing with NasoPore are implemented. All surgeries were performed by the same surgeon. Drops were provided to patients before surgery, but PAs were not administered during surgery in either group. In addition, tympanomeatal flaps were not raised in either group. Patient age, sex, affected side, perforation size, operative duration, myringosclerosis, and smoking status were recorded.
Audiometric data were obtained preoperatively and at 6 months postoperatively. Pure-tone averages (PTAs) were calculated for both air conduction (AC) and bone conduction (BC) by averaging the thresholds at 500, 1000, 2000, and 3000 Hz; in most cases, a threshold of 4000 Hz was used to interpolate the threshold of 3000 Hz as per the standards of the Hearing Committee of the American Academy of Otolaryngology—Head and Neck Surgery. Preoperative and postoperative air-bone gaps (ABGs) were calculated by subtracting the AC PTA from the BC PTA. The extent of ABG closure was calculated as the preoperative ABG minus the postoperative ABG.
Surgical Technique
All patients underwent endoscopic perichondrium-cartilage underlay myringoplasty without tympanomeatal flap elevation. A single-layer perichondrial composite cartilage graft was harvested from the ipsilateral tragus. The lateral perichondrium was peeled circumferentially, with the pedicle remaining attached to the cartilage graft. The cartilage graft was shaped to be 1 mm larger than the perforation margins, with the perichondrium >1 to 2 mm larger than the cartilage graft. Any myringosclerosis of the remnant TM was not treated.
The AOP Technique
The perforation margins were de-epithelialized. The epithelium (if any) was removed from the distal malleus handle.
The middle ear was packed with NasoPore containing 2% mupirocin ointment (Figure 1). The perichondrium-cartilage composite graft was placed across the perforation, and the cartilage graft was placed medial to the remnant TM and the annulus. The perichondrium was placed medial to the remnant TM and the annulus but lateral to the malleus handle (if any). The EAC was packed.

AOP group. (A) Preoperative perforation. (B) Trimming the perforation margins. (C) Middle ear packing with material containing mupirocin ointment. (D) Graft placement. (E) EAC packing, no-AOP group. (F) Preoperative perforation. (G-I) Middle ear packing. (J) Graft placement. AOP, antibiotic ointment packing; EAC, external auditory canal.
The No-AOP Technique
The perforation margins were not de-epithelialized. However, the epithelium (if any) was removed from the distal malleus handle.
The middle ear was packed only with NasoPore (Figure 1). The perichondrium-cartilage composite graft was placed across the perforation, and the cartilage graft was placed medial to the remnant TM and the annulus. The perichondrium was placed medial to the remnant TM and the annulus but lateral to the malleus handle (if any). The EAC was not packed.3,4
Postoperative Follow-Up
Patients were discharged on the day after surgery. Oral amoxicillin and clavulanate potassium were prescribed for 1 week for all patients. The patients were not given eardrops postoperatively. Postoperative follow-up was scheduled for weeks 1, 2, and 4 and months 3, 6, and 12. At each follow-up, the graft was endoscopically examined, and the patients were asked whether they experienced altered taste, vertigo, or tinnitus. Audiometric evaluations were performed at 6 months postoperatively. All postoperative complications were recorded, including graft infection, graft lateralization, and significant graft blunting or medialization.
Outcome Measures
The primary outcome measures were postoperative infection status; the secondary outcome measures were the graft success rate and audiometric outcomes. Graft success was defined as an intact graft without a residual perforation or any re-perforation. Graft infection was defined as a purulent discharge in the middle ear or EAC.
Statistical Analyses
Statistical analyses were performed with SPSS ver. 21 (SPSS Inc, Chicago, IL, USA). Data are expressed as means with standard deviations (SDs) or as percentages (%). The Chi-squared test was used to compare categorical data. The Wilcoxon and Mann-Whitney U tests were employed to compare nonparametric variables, and the independent and paired samples t-tests were used to compare parametric variables. A P-value < .05 was considered to indicate statistical significance.
Results
Demographic Characteristics
A total of 307 chronic perforations were included. Of the 307 ears, 166 perforations were in the AOP group and 141 were in the no-AOP group. Demographic data are shown in Table 1. Sex, mean age, the affected side, operative duration, perforation size, myringosclerosis, and smoking status were matched between the 2 groups.
Demographic Characteristics of Patients.

Abbreviations: AOP, antibiotic ointment packing; F, female; L, left; M, male; R, right.
Chi-square test.
Independent samples t-test.
Postoperative Infection and Graft Success Rates
All patients completed 12 months of follow-up. At 3 months postoperatively, middle ear infections had occurred in 24 (14.5%) ears in the AOP group and 4 (2.8%) in the no-AOP group (P < .01). All infections were treated via topical application of ofloxacin ear drops combined with systemic oral antibiotics (Table 2). However, in the AOP group, middle ear infections developed in 2 ears within 1 week postoperatively. The perforations closed after infection control. Middle ear infections developed in the remaining 22 ears from 2 to 3 weeks postoperatively, causing partial or complete graft necrosis and residual perforations (Figures 2 and 3). In the no-AOP group, middle ear infections developed in all 4 ears within 1 week postoperatively; the perforations closed after infection control in 3 ears, but the graft became partially or completely necrotic and caused a residual perforation in 1 ear. In addition, residual perforations were noted in 6 ears in the AOP group, perhaps attributable to imperfect operative technique or a graft that was too small, though this was seen in only 2 ears in the no-AOP group. The graft success rate was 83.1% (138/166) in the AOP group and 97.9% (138/141) in the no-AOP group (P < .01).
The Graft Success Rate, Operation Time, and Complications.
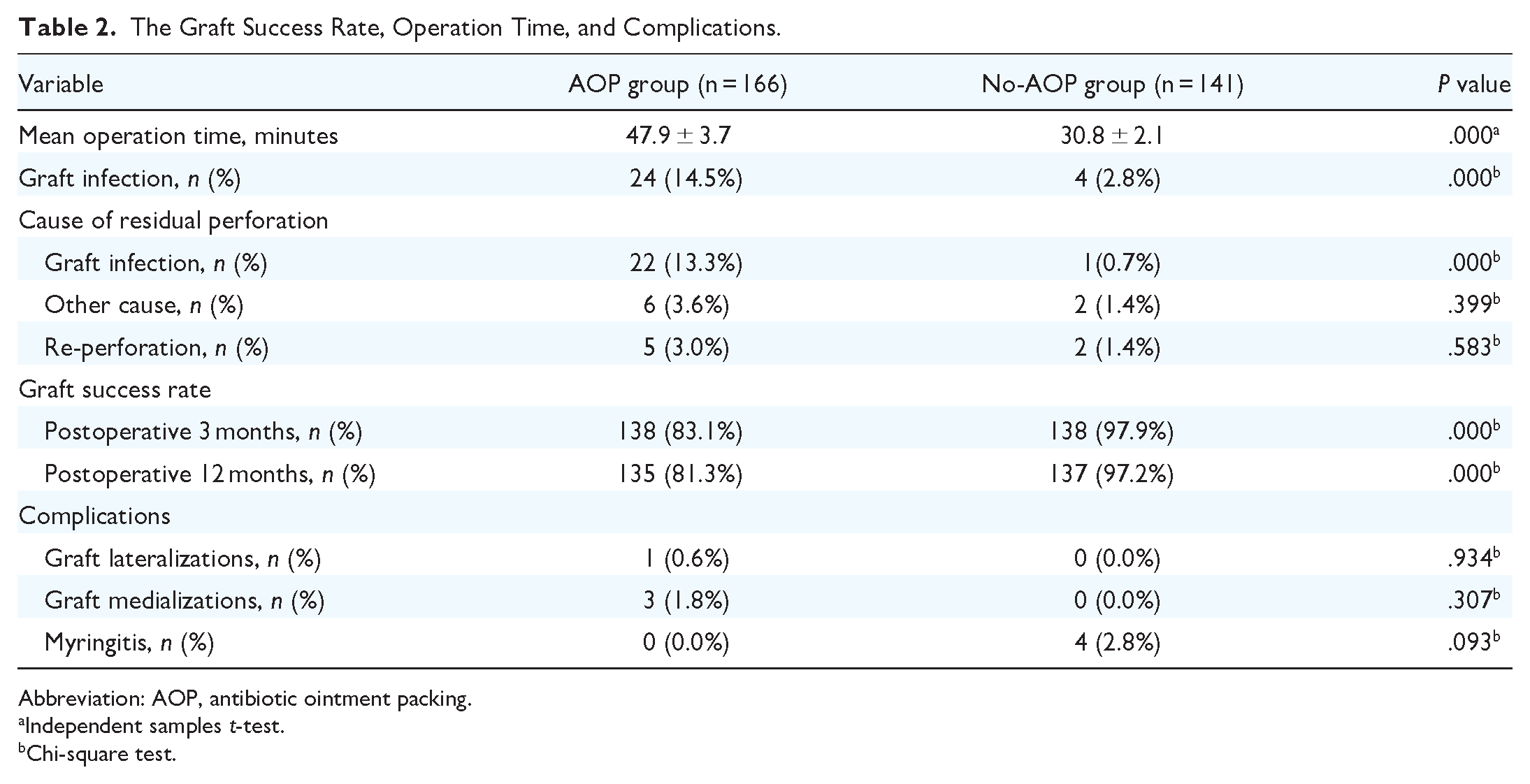
Abbreviation: AOP, antibiotic ointment packing.
Independent samples t-test.
Chi-square test.

AOP group. (A) Preoperative perforation. (B) Image acquired at 14 days postoperatively. (C) Image acquired at 16 days postoperatively. (D) Image acquired at 3 weeks postoperatively. AOP, antibiotic ointment packing.

AOP group. (A) Preoperative perforation. (B) Image acquired at 3 weeks postoperatively. (C) Image acquired at 25 days postoperatively. (D) Image acquired at 4 weeks postoperatively. (E) Image acquired at 5 weeks postoperatively. (F) Image acquired at 12 months postoperatively. AOP, antibiotic ointment packing.
At 12 months postoperatively, spontaneous closure was achieved by 2 of the 6 residual perforations in the AOP group and by 1 of 2 residual perforations in the no-AOP group. In addition, re-perforations were noted in 5 ears in the AOP group and 2 ears in the no-AOP group. The graft success rate was 81.3% (135/166) in the AOP group and 97.9% (137/141) in the no-AOP group (P < .01) (Table 2).
Hearing Outcomes
The postoperative ABGs exhibited significant improvements compared to the preoperative ones in both groups, but no significant difference was observed between the groups in terms of the pre- or postoperative average BC PTAs (Table 3). In addition, no significant group differences were observed preoperatively (P = .657) or postoperatively (P = .578) in terms of the ABG values or mean ABG gain (P = .758). Finally, no significant group differences were observed in terms of the pre- or postoperative average BC PTAs (P = .594 and P = .841, respectively).
Comparison of Hearing Gains, the ABG, and BC.

Abbreviations: ABG, air-bone gap; AOP, antibiotic ointment packing; BC, bone conduction; dB, decibel; PTA, pure-tone average.
Paired samples t-test; comparison between the same groups in regard to ABG or BC, pre-, and postoperatively.
Mann-Whitney U test; comparison between 2 groups in terms of gain, pre-, or postoperatively.
P < .01.
Complications
During the follow-up period, no complications were observed in either group, that is, no sensorineural hearing loss, altered taste, vertigo, or tinnitus. Graft lateralization and graft medialization were observed in 1 (0.6%) and 3 (1.8%) ears in the AOP group but in no ears in the no-AOP group. In addition, no patient in either group developed postoperative otitis media effusion or graft retraction. However, granular myringitis, with minimal moistness and without infection, was noted in 4 (2.8%) patients in the no-AOP group.
Discussion
Packing material coated with antibiotic ointment or soaked in an antibiotic solution is commonly used by a few otologists.3-5 The type of antibiotic ointment varies among departments; individual preferences vary widely based more on tradition than evidence. In 1 American study investigating the use of PA by otolaryngologists, PAs were routinely employed by approximately half. 20 Intraoperative placement of antibiotic ointment is believed to prevent postoperative middle ear infection. However, we have been unable to establish a rationale or an evidence base for this contention. In fact, few publications have focused on intraoperative antibiotic application although some compared various types of packing material. There is no documented evidence that antibiotic ointment reduces graft infection. Previous studies had demonstrated that no trimming of the perforation margins and no EAC packing did not affect graft success and hearing improvement3-5,21; although these could be confounding factors, they have not been incorporated into the statistical analysis in this study.
Mupirocin ointment (2%) is approved in the USA for the treatment of impetigo. In some European and other countries, the ointment is used to treat primary and secondary skin infections 12 and is widely employed to treat chronic otorrhea and PTTO. Both clinical and animal studies revealed no evidence of ototoxicity or subsequent hearing loss.13-15 Thus, 2% mupirocin ointment was used previously during ear surgery in our department. The use of PAs is controversial during myringoplasty. Some scholars believe that PAs reduce the operative infection rate6,7; others have suggested that they do not lower the postoperative infection rate.8-11 The postoperative infection rate in our AOP group was significantly higher than that in the no-AOP group. This suggests that topical antibiotic ointment did not prevent postoperative infection, in line with a previous study. Westman et al 10 found no statistically significant difference in postoperative infection rate between patients who received PA and those who did not (10.2% vs 7.7%). Jackson 11 conducted a prospective controlled study of 4000 patients employing cephalosporin and oxacillin as PAs and found no statistically significant difference in the postoperative otologic infection rates. Therefore, most scholars do not support the use of PAs during otologic surgery.8-11,22,23 Especially, PAs do not protect against the very rare postoperative infections that cause total graft necrosis. 6 Similarly, in surgery in general, spreading of antibiotic ointment on a surgical wound does not effectively prevent postoperative wound infections.24,25
Although some literature has suggested that mupirocin ointment is useful to prevent postoperative infection of the tympanostomy tube,13-15 the present study yielded no evidence that mortifloxacin prevented postoperative infection. This may be related to minimal injury to the TM and middle ear. The middle ear mucosa uses the innate and adaptive immune systems to prevent infections. Trimming of the perforation margins and removal of the epithelium of the malleus handle not only introduce EAC flora into the middle ear but also destroy the defense mechanism of the middle ear, thereby changing the bacterial flora and triggering dysbacteriosis of the surgical site. Thus, some scholars have suggested that the workflow should be optimized to reduce the risk of contamination. 26 In our no-AOP group, we did not perform any middle ear-related procedure except for removal of the epithelium of the malleus handle and middle ear packing using only biological materials. In addition, another drawback of coating with antibiotic ointment is that the ointment can adhere to and contaminate the endoscopic lens.
Most studies reported that preoperative and intraoperative PAs did not affect the graft success rate.10,11 However, the graft success rate in our no-AOP group was significantly higher than that in the AOP group at 3 months postoperatively. Graft success depends on many factors, including placement of a sufficiently large graft, surgical skill, graft displacement, postoperative infection, and any unrecognized middle ear mucosal disease. In this study, we found a significant difference in the rate of residual rate caused by postoperative infection between the 2 groups, but there was no group difference in the residual perforation rate attributable to other causes. Thus, postoperative infection is key in terms of graft failure; the host-mounted acute inflammatory reaction triggers graft rejection and necrosis. This finding is similar to that of another study. Westman et al 10 found a significant difference in the graft success rates of patients with and without postoperative infections [78.2% (n = 104/133) vs 86.9% (n = 1332/1532)]. Jackson 11 reported that successful TM grafting was associated with lower postoperative infection rates; the infection rate of patients with graft failure was 50.0%, and grafting success was associated with a postoperative infection rate of 6.6%. Westman et al 10 found that patients who developed postoperative infections within 6 weeks were at significantly greater risk of a perforated TM than those who did not.
Surprisingly, postoperative infection did not always cause graft failure. In this study, postoperative infections were rapidly detected in the no-AOP group because of the absence of EAC packing. Postoperative infections were found within 1 week after surgery in all 4 patients, but most postoperative infections in the AOP group were not found until the EAC packing was removed 2 to 3 weeks after surgery. In the no-AOP group, systemic and topical antibiotics were applied in a timely manner, which ensured graft survival in 3 of 4 ears. On the contrary, although the postoperative infections were controlled in the AOP group when antibiotics were given at 2 to 3 months postoperatively, the infections resulted in partial or complete graft necrosis in 22 of 24 patients. A previous study suggested that if an infection is not promptly addressed, neovascularization ceases and the graft fails. 4 Jackson 11 emphasized that acute-phase postsurgical infection usually developed within 3 weeks postoperatively. It is crucial to monitor early graft changes and rapidly prescribe antibiotic therapy if even a mildly purulent discharge is observed. We believe that postoperative infection should be identified within 1 week after surgery. Pau 27 suggested that although clinically placed grafts exhibited continued locus minoris resistentiae, decisive therapeutic steps should be taken when the first indication of inflammation occurred. Thus, the absence of EAC packing does not affect the graft success rate; indeed, this helps surgeons to identify early postoperative infections. The intraoperative placement of antibiotic ointment to prevent postoperative infection and improve the graft success rate is not sufficiently effective to justify routine use.
In this study, there was no significant difference in hearing gain between the 2 groups. This finding is consistent with Masalha et al 8 and John et al, 23 who found that PAs did not influence the audiometric results. Yang et al 28 found that preoperative culture of MRSA did not significantly affect the hearing improvement. The present study further confirmed that mupirocin ointment was not ototoxic. Theoretically, ABG improvement depends on TM closure following myringoplasty. The ossicular chain was protected from injury during operations on patients with no ossicular chain abnormalities. Although graft lateralization and graft medialization were observed in 1 (0.6%) and 3 (1.8%) ears in the AOP group but in none in the no-AOP group, the difference was not significant. In this study, we did not encounter deteriorative sensorineural hearing loss, vertigo, or tinnitus.
The limitations of this study include its single-center, retrospective (non-prospective), case-control, randomized controlled design, and unbalanced sample size. We did not assess auditory brainstem responses, and the follow-up was short. Neither postoperative bacterial culture nor antimicrobial susceptibility testing was performed for patients with purulent discharge in either group. Another limitation was no preoperative assessment of Eustachian tube function. In addition, the surgical methods differed between the 2 groups in terms of trimming of the perforation margins, placement of EAC packing, and surgical duration. These factors may have confounded the primary outcomes.
Conclusion
Middle ear packing without antibiotic ointment coating does not increase the postoperative infection rate or reduce the graft success rate of endoscopic cartilage underlay myringoplasty. On the contrary, coating with antibiotic ointment and the associated complexity of middle ear manipulation increase the possibility of postoperative infection.
Footnotes
Authors’s Note
Author Contributions
Zhengcai Lou: Interpretation of data for the work, design of the work, analysis of data for the work, drafting the work, agreement to be accountable for all aspects of the work; final approval of the version to be published; Zihan Lou: Interpretation of data for the work, analysis of data for the work, drafting the work and technical the diagram, agreement to be accountable for all aspects of the work, and final approval of the version to be published; Zhengnong Chen: Interpretation of data for the work, design of the work, final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Technology Agency of Jinhua City, China (Grants # 2022-3-042).
