Abstract
Introduction
External auditory canal (EAC) packing after myringoplasty remains a common practice among otolaryngologists, primarily to prevent graft displacement and control bleeding. 1 However, EAC packing has several potential drawbacks, including temporary conductive hearing loss, ear discomfort, risk of graft disturbance during insertion and removal, postoperative local pain, and increased health care costs.1–3 Although several studies have demonstrated the feasibility of myringoplasty without EAC packing, this approach has not gained widespread acceptance. 4–6 Recently, Lou et al.3,6,7 have reported on perichondrium-cartilage graft myringoplasty performed without EAC packing. Their findings suggest that cartilage grafting in the absence of EAC packing does not adversely affect graft success rates and may reduce the number of follow-up visits. However, the follow-up periods in these studies were relatively short.3,6,7 Consequently, the long-term outcomes and potential complications associated with myringoplasty performed without EAC packing remain unclear.
Myringitis is the most common postoperative complication after myringoplasty, characterized by prolonged or recurrent otorrhea. Endoscopic examination reveals desquamation or granulation tissue on the tympanic membrane (TM). 8 Although the precise etiology remains uncertain, trauma, infection, and prior otological interventions are considered potential contributing factors.8,9
Cartilage butterfly myringoplasty has been associated with a relatively-high incidence of postoperative myringitis; reported rates range from 5% to 14%.10,11 Recurrent myringitis can significantly diminish a patient’s quality of life and may contribute to graft failure. However, the impact of EAC packing on postoperative myringitis remains uncertain. We compared the long-term efficacy and incidence of myringitis after cartilage graft underlay myringoplasty for chronic TM perforations with and without EAC packing.
Materials and Methods
Ethical Considerations
The Institutional Ethical Review Board of the First Affiliated Hospital of Hebei North University, Hebei, China, approved the study protocol. Informed written consent was obtained from all participants.
Patient Selection
Patients diagnosed with chronic TM perforation were included in the study. Inclusion criteria were a large TM perforation (25%-50% of the TM surface area) with a remnant TM of ≥1 mm, a dry ear for ≥3 months prior to surgery, and an air-bone gap (ABG) < 35 decibels (dB). Exclusion criteria comprised marginal perforations, revision surgeries, cholesteatoma, ossicular chain abnormalities, middle ear inflammation, and fungal otitis externa. All patients underwent preoperative computed tomography imaging. Preoperative and postoperative audiometric assessments were conducted in accordance with the standards established by the Hearing Committee of the American Academy of Otolaryngology–Head and Neck Surgery. 12 Baseline data included age, sex, affected ear, duration of symptoms, presence of myringosclerosis, smoking status, and history of diabetes. Myringitis was defined as protracted or recurrent otorrhea accompanied by desquamation or granulation tissue on the TM, as observed via endoscopy.
Grouping and Surgical Technique
Consecutive patients who met the inclusion criteria were allocated to either the EAC packing (packing) group or the no EAC packing (no-packing) group based on the order of submission of written informed consent (semi-random allocation). All patients underwent endoscopic cartilage underlay myringoplasty without tympanomeatal flap elevation. This surgical technique has been detailed in previous reports.3,6 Briefly, perforation margins were de-epithelialized, and the epithelium was removed from the distal malleus handle. A single-layer perichondrium-cartilage composite graft was harvested from the ipsilateral tragus. The graft was shaped to be 1–2 mm larger than the perforation margins; a notch was created in the cartilage to accommodate the malleus handle, if present. The middle ear cavity was packed with biodegradable synthetic polyurethane foam (NasoPore; Stryker Canada, Hamilton, ON, Canada). The perichondrium-cartilage composite graft was positioned via transperforation and placed medial to the remnant TM and the annulus. Notably, EAC packing was applied using biodegradable NasoPore in the packing group, whereas no packing was used in the no-packing group.
Postoperative Follow-Up
Outpatient endoscopic examinations were scheduled at the 2nd and 4th week and at the 3rd, 6th, and 12th month postoperatively. Audiometric evaluations were conducted in all patients at the 6th month. Graft success was defined as a completely-intact TM with no evidence of residual or recurrent perforation.
Statistical Analyses
Data are presented as means ± standard deviations for quantitative variables and as frequencies (percentages) for categorical variables. Between-group comparisons were conducted using independent-samples t-tests for quantitative variables and the chi-squared test for categorical variables. Paired t-tests were used to evaluate differences in ABGs. Statistical analyses were performed using SPSS, version 25 (IBM Corp., Armonk, NY, USA). P-values < .05 were considered statistically significant.
Results
Demographic Data
In total, 129 patients diagnosed with chronic large TM perforations were included in the study. Table 1 presents the baseline demographic data of the participants. Baseline variables, including sex, mean age, affected side, perforation duration, myringosclerosis, smoking status, and diabetes status, were comparable between the groups (P > .05). All patients completed the 12 month follow-up period.
Select Characteristics of Patient Sample.
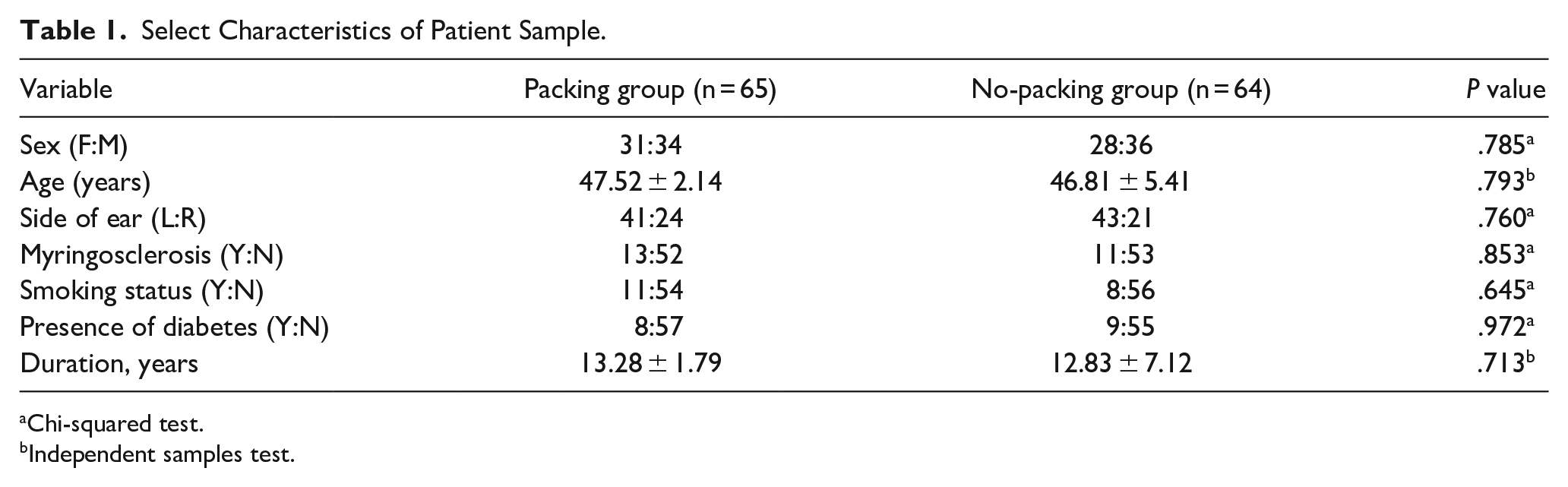
Chi-squared test.
Independent samples test.
Graft Success Rate
Table 2 presents the graft success rates. At the 3rd month postoperatively, no residual perforations were observed in the packing group, whereas 1.6% (1/64) of patients in the no-packing group exhibited residual perforations. Overall graft success rates were 100.0% in the packing group and 98.4% in the no-packing group, with no statistically-significant difference between the groups (P = .993).
Graft Success Rate and Complications in Both Groups.
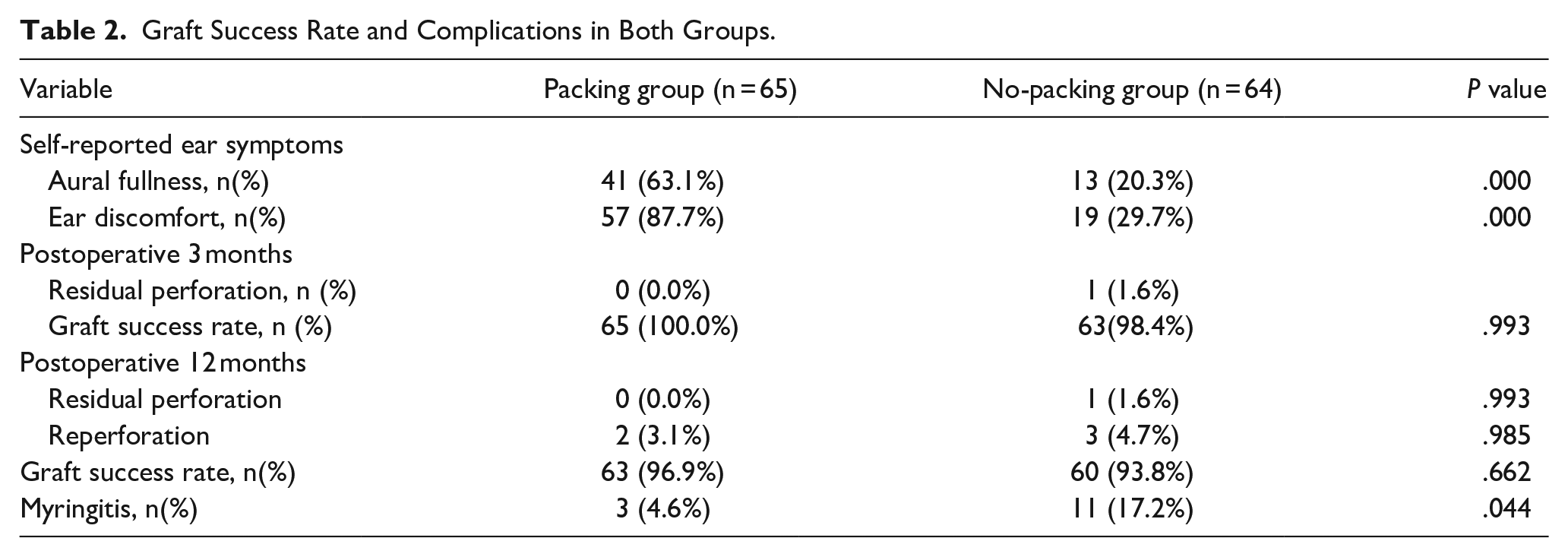
At the 12 month postoperatively, reperforation occurred in 3.1% (2/65) of patients in the packing group and 4.7% (3/64) of patients in the no-packing group. This difference was not statistically significant (P = .985). Graft success rates were 96.9% in the packing group and 93.8% in the no-packing group, with no statistically-significant difference (P = .662).
One of the two reperforations (50.0%) in the packing group and two of the three reperforations (66.7%) in the no-packing group were attributed to myringitis.
Hearing Improvement
All patients in both groups underwent audiological testing at the 6th month postoperatively, revealing no sensorineural threshold shift. Preoperatively, no significant differences in ABGs were observed between the packing and no-packing groups (30.1 ± 2.8 dB vs. 29.7 ± 5.4 dB, P = .831). At the 6th month postoperatively, no significant differences in ABGs were observed between the packing and no-packing groups (16.2 ± 3.1 dB vs. 16.7 ± 1.8 dB, P = .927). Both groups demonstrated significant improvements in hearing between preoperative and 6 month postoperative assessments. Mean ABGs improved by 14.17 ± 3.29 dB in the packing group and 13.85 ± 5.46 dB in the no-packing group (P < .001).
Myringitis
The incidence of myringitis was significantly higher in the no-packing group (17.2%, 11/64) than in the packing group (4.6%, 3/65) (P = .044). All patients with myringitis received topical ofloxacin ear drops and dexamethasone injections. In the packing group, two of the three patients (66.7%) achieved complete epithelialization without recurrence, whereas one patient (33.3%) experienced recurrent myringitis, leading to reperforation (Figure 1). Among the 11 patients with myringitis in the no-packing group, three (27.3%) achieved complete epithelialization without recurrence, two (18.2%) experienced recurrent myringitis leading to reperforation (Figures 2 and 3), and six (54.5%) experienced recurrent myringitis without reperforation during the follow-up period.
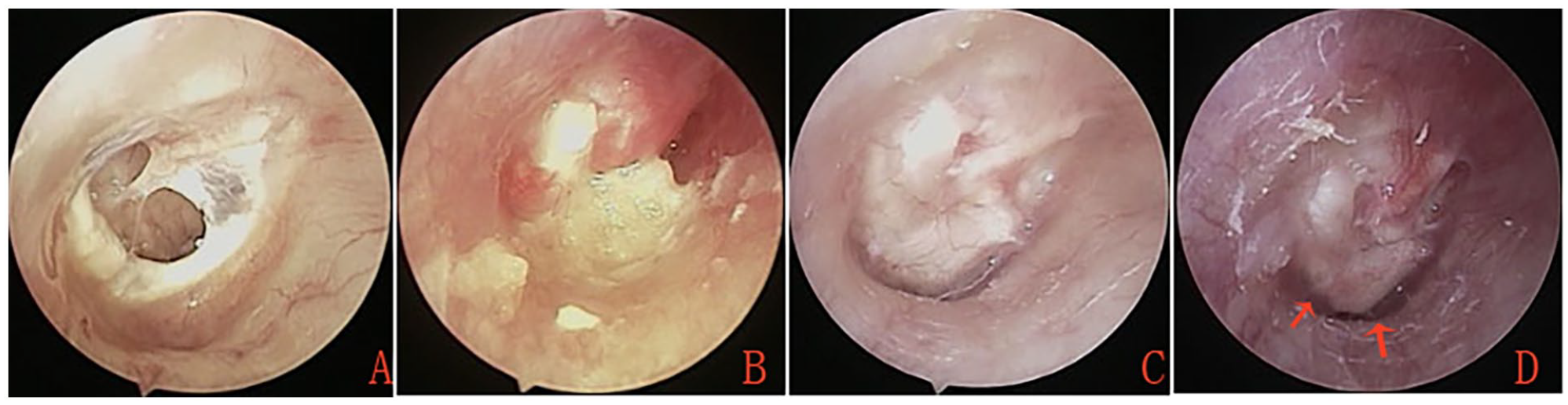
External auditory canal (EAC) packing group. (A) Preoperative tympanic membrane perforation. (B) Postoperative appearance at the 2nd week. (C) Postoperative appearance at the 6th month. (D) Postoperative appearance at the 2nd year. Red arrows indicate areas of myringitis.
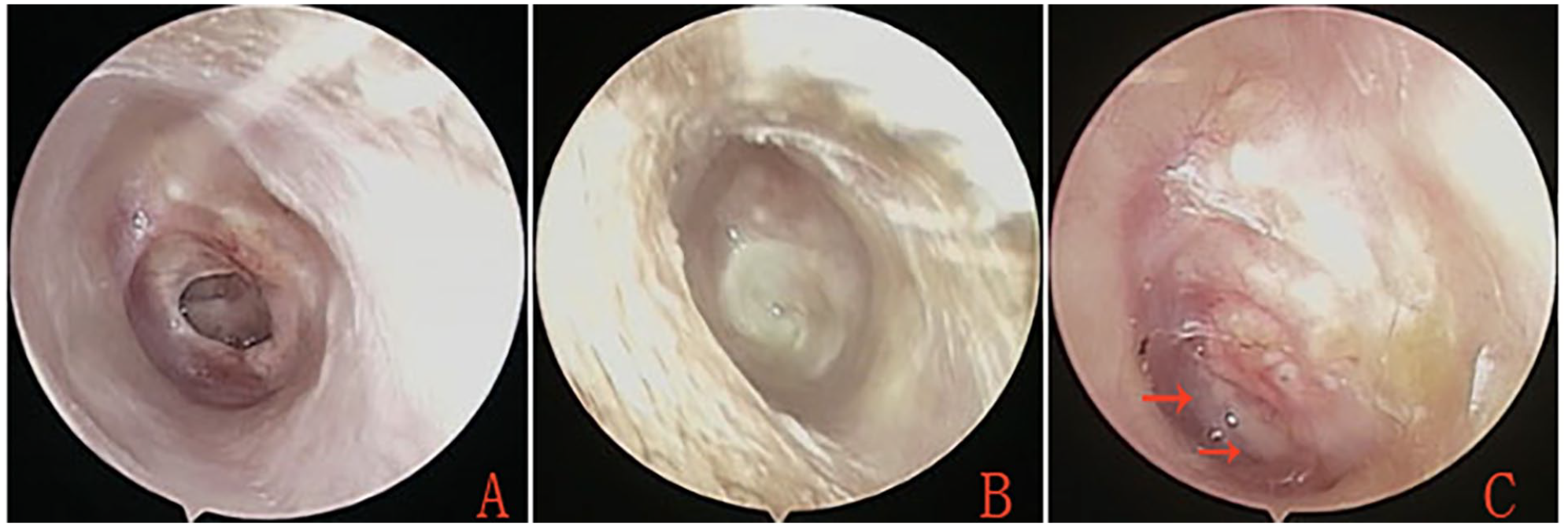
No external auditory canal (EAC) packing group. (A) Preoperative tympanic membrane perforation. (B) Postoperative appearance at the 4th week. (C) Postoperative appearance at the 3rd month. Red arrows indicate areas of myringitis.
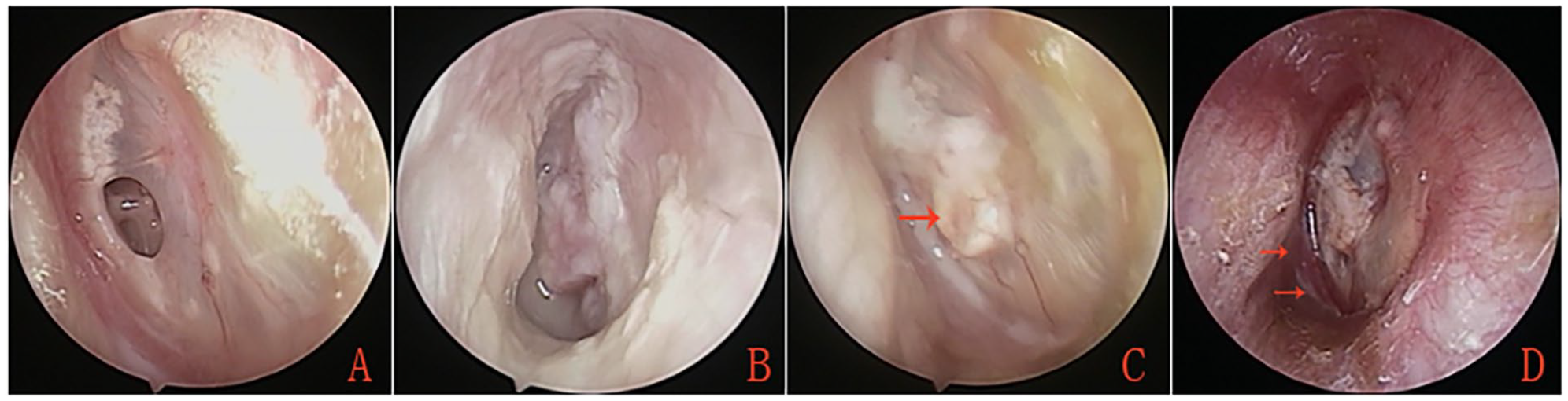
No external auditory canal (EAC) packing group. (A) Preoperative tympanic membrane perforation. (B) Postoperative appearance at the 4th week. (C) Postoperative appearance at the 2nd month. (D) Postoperative appearance at the 3rd year. Red arrows indicate areas of myringitis.
Complications
Significant differences were observed between the packing and no-packing groups in self-reported rates of aural fullness (P < .001) and ear discomfort (P < .001). Notably, 23 patients (35.4%) reported mild ear pain during the removal of the packing material (Table 2). No patients experienced sensorineural hearing loss, intractable tinnitus, significant graft lateralization, medialization, significant blunting, or graft cholesteatoma during the follow-up period.
Discussion
Our findings demonstrate comparable graft success rates between the packing and no-packing groups at the 3rd and 12th month postoperatively, consistent with prior studies.2,3,6 Similarly, Lou et al. reported graft success rates of 93.18% and 100.00% in their respective studies.3,6 Significant hearing improvement was observed in both the packing and no-packing groups, consistent with previous studies.2,3,6 Although both groups exhibited a significant hearing improvement before and after surgery, no statistically-significant differences in hearing outcomes were observed between the groups. These findings indicate that EAC packing does not significantly influence graft success rates or hearing outcomes in underlay myringoplasty for repairing large chronic perforations.
Although the absence of EAC packing offers several advantages, such as saving time and reducing postoperative aural fullness and conductive hearing loss,1–3,6 our study revealed a higher incidence of myringitis in the no-packing group than in the packing group. Myringitis is a recognized complication of tympanoplasty, but it has been more frequently reported with cartilage butterfly and lateral tympanoplasty techniques.11,13,14 Postoperative myringitis rates of 5% to 14% after cartilage graft butterfly tympanoplasty have been reported in several studies.10,11 We observed myringitis in both groups using cartilage graft techniques. Although the precise pathogenesis of myringitis remains unclear, the current consensus suggests that infection or local trauma disrupts epithelial migration, resulting in granulation tissue formation and inhibiting reepithelialization.8,15 Additionally, residual mucous glands at the perforation margins may contribute to graft mucosalization and granulation. 9 Factors contributing to the higher incidence of myringitis associated with cartilage grafts relative to temporal fascia or perichondrium grafts remain uncertain. We hypothesize that the loose connective tissue structure of the temporal fascia and perichondrium, which resembles the fibrous layer of the TM, facilitates better graft integration and reduces the risk of myringitis. Graft epithelialization primarily depends on the migration of epithelial cells from the remnant TM, a process strongly influenced by epithelium adhesion to the fibrous layer scaffold. This scaffold enables migration through the undulating movement of the fibrous layer. Conversely, cartilage—a dense connective tissue with a hard and uneven surface—is less favorable for epithelial adhesion. Thus, epithelial migration on cartilage primarily relies on epithelial crawling.
The epithelialization of cartilage grafts may progress more slowly than perichondrium or temporalis fascia grafts in myringoplasty. Several factors, such as infection and trauma, can readily impede or disrupt epithelialization of the cartilage graft. Therefore, we recommend preserving the perichondrium on at least one side of the cartilage graft and orienting it toward the EAC.
Our findings revealed a significantly-higher incidence of myringitis in the no-packing group than in the packing group. Despite the use of a similar surgical approach in both groups, the underlying mechanism for the increased susceptibility to myringitis in the absence of EAC packing remains unclear. Although previous studies have shown that raising the tympanomeatal flap can alter epithelial migration and contribute to myringitis, 16 this surgical step was not performed in our study. The sole factor distinguishing the groups was the use of biodegradable NasoPore packing in the packing group. We hypothesize that the absence of EAC packing contributed to the increased incidence of myringitis. EAC packing helps maintain the graft in alignment with the remnant TM, prevents outward epithelial migration, and facilitates the centripetal migration of proliferative epithelium. Outward epithelial migration can result in graft failure and delayed healing. 17 Furthermore, Lou 18 demonstrated that biodegradable NasoPore can accelerate epithelialization of the graft. Our findings revealed an enhanced inflammatory response and increased exudation in the no-packing group, resulting in EAC otorrhea. This inflammatory state may increase the risk of infection in the middle ear and EAC, disrupt graft epithelialization, and impair the healing process. In contrast, biodegradable NasoPore packing facilitates the absorption of exudates and blood, mitigating the risk of middle ear infection. Additionally, EAC packing provides structural support to the graft and serves as a scaffold for epithelial migration, promoting graft epithelialization. Consistent with our findings, a previous study regarding endoscopic sinus surgery demonstrated significantly-better reepithelialization with biodegradable bone substitute particles relative to nonbiodegradable particles. 19 The causal relationship between myringitis and TM perforation remains uncertain. However, several studies have indicated that postoperative myringitis may contribute to graft failure.8,15,16 Some scholar reported that TM perforation occurred in 30.7% of patients with myringitis. 8 In our study, one patient in the packing group and two patients in the no-packing group experienced recurrent myringitis, resulting in graft reperforation. These findings support the association between recurrent myringitis and the risk of graft reperforation, emphasizing the importance of early identification and treatment. In our cohort, all patients with myringitis were treated with a combination of topical ofloxacin ear drops and dexamethasone. Notably, the recurrence rate of myringitis was significantly higher in the no-packing group (72.7%) than in the packing group (33.3%). This disparity may be attributed to the extent of graft epithelial loss and incomplete epithelialization after initial treatment. Our results indicated that patients in the no-packing group exhibited a larger area of graft epithelial loss relative to those in the packing group.
Consistent with previous studies, our findings demonstrated a significant reduction in self-reported postoperative ear symptoms in the no-packing group compared with the packing group.3,7 Notably, no graft cholesteatoma was observed in either group during the follow-up period. Although the packing group experienced transient ear symptoms, we hypothesize that recurrent myringitis in the absence of EAC packing is more bothersome for patients. Therefore, we recommend EAC packing after cartilage myringoplasty. Our study had some limitations, including a small sample size and short follow-up period. Future studies with larger sample sizes and extended follow-up periods are needed to investigate long-term outcomes and refine treatment recommendations.
Conclusions
Although the graft success rate and hearing improvement after cartilage graft underlay myringoplasty without EAC packing were comparable to the outcomes with packing, the no-packing approach was associated with a higher incidence of myringitis. Therefore, we recommend EAC packing after cartilage myringoplasty.
Footnotes
CRediT Authorship Contribution Statement
Dong Li: Interpretation of data for the work, design of the work, analysis of data for the work, drafting the work, agreement to be accountable for all aspects of the work, and final approval of the version to be published; Yanping Li, Yongqing Ding, and Yunchao Xin: Analysis of data for the work, agreement to be accountable for all aspects of the work, and final approval of the version to be published; Hongyan Zhang, Lixin Wu, and Yuying Yang: Analysis of data for the work and agreement to be accountable for all aspects of the work; Yantao Lin: Interpretation of data for the work, design of the work, and final approval of the version to be published; Yachao Liu: Interpretation of data for the work, design of the work, and final approval of the version to be published.
Data Availability Statement
All data generated or analyzed during this study are included in the published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Technology Agency of Zhangjiakou City, Hebei Province, China (Grants#2221164D and 1821053D).
