Abstract
Objective:
This study investigated whether attention-deficit hyperactivity disorder (ADHD) has an olfactory dysfunction and, if so, the effect of methylphenidate on this condition.
Methods:
The “Sniffin” Sticks Test and a questionnaire were applied to 50 children with ADHD and 50 healthy children as the control group before and after being treated with methylphenidate.
Results:
There was a statistical difference between ADHD and control groups in odor sensitivity, odor discrimination skills, and odor identification tests (P < .001). General odor scores decreased in the ADHD group (P < .001). A statistically significant score increase was also observed after treatment (P < .001). There was no statistically significant difference between the groups in the taste questionnaire test.
Conclusions:
Although the sense of smell test in our study suggests that it may be a biological marker for diagnosing ADHD, treatment efficacy, and follow-up, further studies need to be conducted in more extensive series and with similar equipment.
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a neuropsychiatric condition characterized by cognitive dysfunction, hyperactivity, and impulsivity. 1 ADHD has been reported in 5.3% to 9.5% of children and 2.5% to 5% of adults, with an increased predisposition and incidence in the male gender. 2 Although several imaging studies have explored the possibility of using this as a diagnostic marker for ADHD, 3 this potential has not yet been realized, possibly due to the complexity of the intrinsic etiology of ADHD. 4 Therefore, clinicians in the United States recommend following the guidelines outlined in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) for the diagnosis of ADHD. 5
ADHD is a disorder diagnosed using the DSM criteria, neuropsychological assessments, and parent/teacher/self-report scales. The diagnosis is made only by clinical evaluation; other methods are auxiliary. There is no biological marker that biological, electrophysiological, or neuroimaging methods can diagnose. Studies that will help diagnose ADHD have been conducted many times. In these studies, smell function has been addressed in a limited way. Therefore, in this study, smell function was examined in children with ADHD. 5
Up to 90% of Parkinson’s disease patients had olfactory deficits, and there is evidence that the cause of these deficits is dysfunction of the dopaminergic system. Since the dopaminergic system is involved in the pathophysiology of ADHD, the olfactory function was investigated in children with ADHD. A questionnaire questioning taste function supported olfactory function. The overlap between neuroanatomical regions and neurotransmitter systems in the olfactory system and the pathophysiology of ADHD allowed us to hypothesize. 5
In the treatment of ADHD, stimulant (methylphenidate, amphetamine. . .) and nonstimulant (guanfacine, atomoxetine. . .) treatments are used to reduce ADHD symptoms by increasing synaptic levels of dopamine and noradrenaline. The first-line pharmacological treatment option for ADHD is psychostimulant drugs such as methylphenidate and amphetamine. 6 Although the short-term results of stimulant treatment are well defined, the long-term results are still unclear. 7 Since methylphenidate is the most commonly prescribed drug for ADHD patients, and this drug also affects dopaminergic regulation. We aimed to evaluate the olfactory and taste function of patients before and after methylphenidate treatment for the first time in the literature. In this direction, we believe that we will help patients who are followed and treated with the diagnosis of ADHD at the diagnostic stage, preserving these social abilities in the treatment stage and seeing and improving the effectiveness of treatment.
Materials and Methods
This study was conducted at Cukurova University’s Faculty of Medicine, Department of Otorhinolaryngology. Fifty children and adolescents aged 8 to 17 years with ADHD who were followed up in the outpatient clinic of Cukurova University Faculty of Medicine, Department of Child and Adolescent Psychiatry, and 50 healthy volunteers were recruited as the control group. The two groups were matched in terms of age and gender. This study included a comparison of two groups of children aged 8 to 17 years in the ADHD group (mean age 10.8, SD = 2.6; 26 males/24 females) and the Control group (mean age 10.2, SD = 2.2) (32 males/18 females). The healthy control group consisted of children of volunteer health workers. Children and adolescents with ADHD and the control group were not allowed to use any medication in the 24 hours before the smell and taste tests. All participants were asked whether they had any problem in the upper respiratory tract or any disease that would affect their smell, and if any, they were included in the study. Acute and chronic otorhinolaryngological diseases and other medical illnesses, those with a history of otorhinolaryngology, surgery or head injury, drug or alcohol abuse, ADHD medications (such as atomoxetine) other than methylphenidate, or smoking habits were determined as exclusion criteria in all groups. Patients were accepted to the study after psychiatric interview, psychiatric examination, and diagnosis of ADHD according to DSM-5 criteria. After the patients were diagnosed, detailed otolaryngological examinations were performed in the Cukurova University Department of Otorhinolaryngology outpatient clinic. Informed consent was obtained from all participant parents and children. Then, smell and taste tests were performed on all participants. The patients included in the study were questioned about their demographic characteristics, educational status, and past and current complaints related to the sense of smell and taste; detailed examinations were performed, and Sniffin’ Stick’s test for smell and general taste questionnaire for taste was performed. All volunteers were examined in detail and evaluated similarly for smell and taste. The process was performed in two sessions. The tests were performed on 50 patients when the patients were diagnosed with ADHD (when no medication was started) and on 40 patients in the 1st month after treatment with methylphenidate. Ten patients were excluded from the study because the tests could not be performed after methylphenidate treatment for different reasons (4 of 10 patients had COVID-19, two could not be reached, and four could not be reached because methylphenidate treatment was not started), and 2nd tests were performed. In terms of time, the smell tests took approximately 1 hour, and the taste test took 5 minutes. The study was carried out following the Declaration of Helsinki with the approval of the Ethics Committee of Cukurova University Faculty of Medicine Ethics Committee with decision number 101/22.
The power analysis performed to show that the odor status was different in the control and attention-deficit patient groups found the effect size to be 0.75. At 5% error and 90% power, at least 39 patients per group would be sufficient. Our study included 50 ADHD patients and had a statistically enough sample size.
Data Collection Tools
One of the functional smell tests, the Sniffin’ Sticks test, was applied to the individuals who met the inclusion criteria. 8
Functional Odor Assessment
Equipment: Objective odor measurements were made using the Sniffin’ Sticks Extended Test’s thresholding, discrimination, and recognition subtests (Threshold n-Butanol, Discrimination, Identification).
Each individual included in the study was shown what to do before administering the test by the clinician. For the threshold identification test, 16 different dilutions of felt-tip pens were presented as solutions in gradually increasing concentrations starting from 4% concentration. At each test step, there were three pens, two containing solvents and one containing odor. Each pen was held 2 cm from the nose of a blindfolded participant. The three 46 pens were presented at 20 to 30-second intervals. The clinician wore odorless gloves. The subject was asked which pen contained the odor. The test was carried out on pen number 16 with low and 1 with high concentrations. The first threshold point was determined when the subject could smell the same concentration of odor two times. In total, seven threshold points were chosen, and threshold test scores were calculated by taking the arithmetic mean of the last four threshold points. In the odor discrimination test, 16 sets of three pencils were presented, two of which were the same, and one contained a different odor. The blindfolded participant was asked to smell these three pens and tell which pen smelled different. There was a 20 to 30-second pause between each set of three presented. In the odor recognition test, the participant was blindfolded and asked to smell 16 pencils individually and choose the most appropriate one from the four options written for that odor. A 20 to 30-second wait was allowed between each presented pen.
The administration of the odor test took approximately 1 hour. It was ensured that the participants had consumed food or drank only water 15 minutes before the start of the odor test. This rule also applies to cigarettes, chewing gum, and drops. In the Burghart Sniffin’s’ Sticks Extended Test scoring, a maximum of 48 points can be obtained, 16 points in total from each subtest. The Turkish validity study determined percentage curves according to age groups 17 to 18. According to these scores, participants were classified as normosmia, hyposmia, and anosmia. According to the combined score, participants with a score below 15 were classified as anosmia, participants with a score between 16 and 30 were classified as hyposmia, and participants with a score between 31 and 48 were classified as normosmia. 9
Taste Test
Subjective Evaluation
The Taste Alteration Scale (TADO) questionnaire, developed by Taro Kano and Kiyoko Kanda and validated in Turkish, was used to collect the data based on the literature. 10 The purpose of the questionnaire was to evaluate the taste state at the time of diagnosis before and after treatment. Our questionnaire is presented in Appendix 1.
Statistical Analysis
Categorical measurements were summarized as numbers and percentages, and numerical measurements were summarized as mean and standard deviation (median and minimum–maximum where necessary). Chi-square statistics was used to compare categorical measurements between groups. McNemar test statistics were used to compare odor status before and after treatment. The Kolmogorov–Smirnov test was used to test whether the numerical measurements met the assumption of normal distribution. Mann–Whitney U test was used to compare the numerical measurements that did not show normal distribution between the two groups. Wilcoxon Signed Rank test compared two dependent numerical measurements that did not show normal distribution. Since some of the numerical measurements did not meet the assumption of normal distribution, the correlation between these continuous measurements was analyzed with the Spearman’s correlation coefficient. IBM SPSS (New York, USA) Statistics Version 20.0 package program was used for the statistical analysis of the data. The statistical significance level was taken as 0.05 in all tests.
Results
A total of 100 people were included in the study. The mean age of the control group was 10.8 ± 2.6 years, and the mean age of the ADHD group was 10.2 ± 2.2 years. There was no significant difference in age distribution (P = .276). There were 26 males and 24 females in the control group and 32 males and 18 females in the ADHD group, and there was no significant difference between the two groups in terms of gender (P = .311). It has a statistically sufficient sample size (Table 1).
Comparison of the Groups in Terms of Demographic and Odor Status Before Treatment.

Data are summarized as mean ± standard deviation, median (min–max). TDI: Threshold-Discrimination Identification Score.
A statistically significant difference was found in the mean values of pretreatment olfactory threshold, odor discrimination, odor identification tests, total TDI scores, and olfactory status between the control group and the attention deficit hyperactivity group (P < .001). In the ADHD group, the scores were generally lower than in the control group, and the odor status of the patients was hyposmia except for two patients (1 patient was normosmia, and one patient was anosmia). One anosmic patient was included in the hyposmic group and evaluated. No significant difference between the two groups was found for age and gender (P = .276 and P = .311; Table 1).
In the comparison of the mean values of olfactory threshold, odor discrimination, odor identification tests, total TDI scores, and odor status between the control group and the posttreatment “Attention-Deficit Hyperactivity Disorder” group, a statistically significant difference was found between the values before and after treatment (P < .001). There was an increase in each of the scores after the treatment. The change in odor status was statistically significant (P < .001). After the treatment, the number of normosmic patients increased from 1 to 8, and a substantial increase in scores was observed in hyposmic patients, although they remained in the hyposmic group (Table 2).
Comparison of the Control and Treatment Groups in Terms of Odor Discrimination Score and Status.
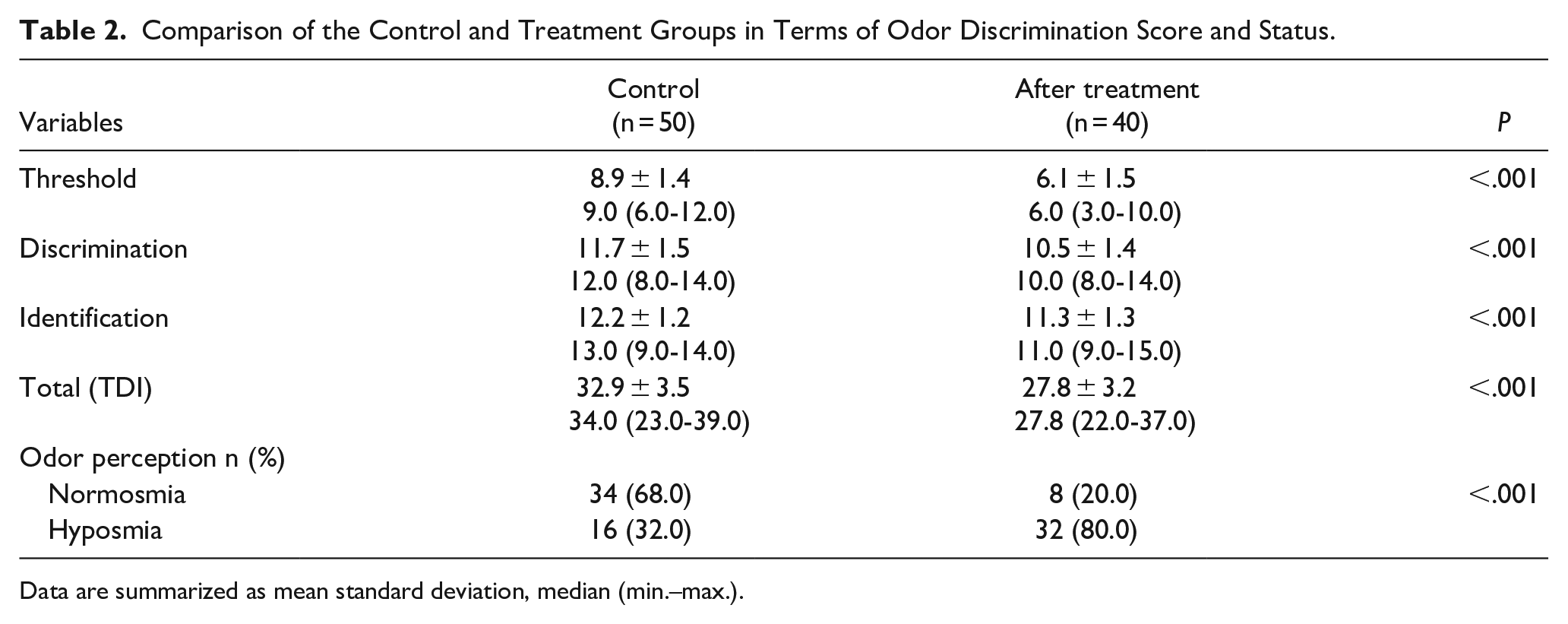
Data are summarized as mean standard deviation, median (min.–max.).
For the Pre–Posttreatment comparison group sample:
A total of 40 patients were evaluated. The mean age of the patients was 10.4 ± 2.4 years (median: 9.5, min: 8, max: 17). 25 (62.5%) of the patients were male and 15 (37.5%) were female. Odor test and taste questionnaire were performed at the time of diagnosis, that is, before treatment was started, and control tests were performed 1 month after treatment was started. The change between these two groups was analyzed (Table 3).
Comparison of odor scores in the pre- and posttreatment group.
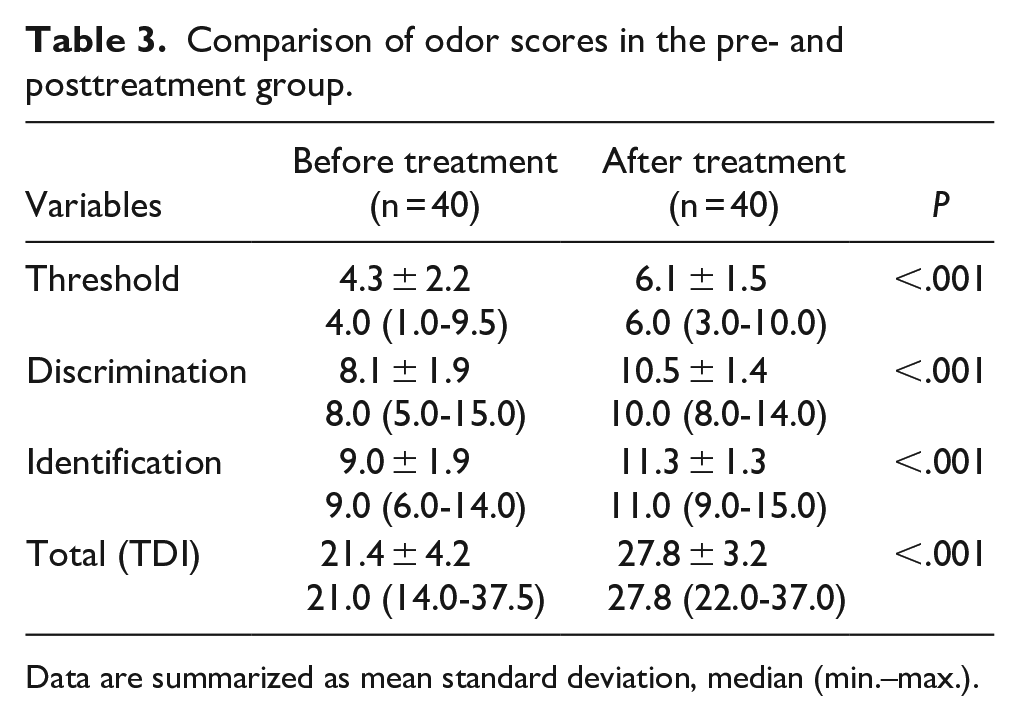
Data are summarized as mean standard deviation, median (min.–max.).
A statistically significant difference was found between the mean values of odor threshold, odor discrimination, odor identification tests, and TDI scores before and after treatment (P < .001). There was a general increase in the scores after treatment. A significant increase was observed in the total scores even if the odor status of the patients was hyposmotic (Table 3).
The number of hyposmic patients increased from 1 to 8 after treatment. There were 32 hyposmic patients, but their scores increased (Table 4).
Odor Threshold Situation After Treatment.

Change after treatment was found to be statistically significant (P < .05).
A significant correlation was found between age and odor scores in the control group (P < .001). There was a considerable improvement in odor scores with increasing age. In the ADHD group, an increase in identification scores and a corresponding increase in total (TDI) scores were observed with increasing age and found to be statistically significant (P < .05).
No significant difference was found between odor scores and gender in both the control group and the ADHD group (P = .460 and P = .109).
Taste Survey Results
The results of the taste questionnaire showed that all patients and the control group had no taste-related problems, and typical taste scores of 6 points (total score) were obtained. No taste-related disorder was observed in the patients and the control group.
Discussion
Attention-deficit hyperactivity disorder (ADHD) is a disorder characterized by attention deficit, hyperactivity, and impulsivity. This disorder, which has a prevalence rate of 10% in the population, is more common in boys. Although the disorder’s etiology is not known precisely, genetic, biochemical, and environmental factors are emphasized. ADHD can be diagnosed by the clinician based on a clinical interview and psychiatric examination under the Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria. However, neuropsychological assessments and parent/teacher/self-report scales help make the diagnosis. There is no known biological, electrophysiological, or neuroimaging method to diagnose ADHD11. Many studies have tried to find such a biological marker. In the etiological studies conducted by Polanczyk et al., 11 a relationship between dopamine regulation and orbitofrontal cortex dysfunction and ADHD was revealed.11 These are also the mechanisms that affect olfactory processes. The relationship between ADHD and olfactory function has been demonstrated in previous studies.
When the studies investigating the relationship between olfactory processes and ADHD are examined, it is seen that the results are contradictory. The survey conducted by Romanos et al. 12 (the sample size was 20, with ages ranging from 5 to 17) found that odor sensitivity was higher in children with ADHD compared to the healthy control group.12 No difference was observed in odor identification and discrimination.12 The study conducted by Schecklmann et al. 13 (the sample size was 27, with ages ranging from 10 to 16) found that odor discrimination ability was better in children with ADHD compared to the control group. No difference was observed in odor sensitivity and odor identification tests. At the same time, the group receiving medication had the same level of odor discrimination ability as the control group. 13 However, the study conducted by Karsz et al. 14 (the sample size was 44, with ages ranging from 7 to 16) reported that odor identification was lower in the group with ADHD than in the control group. 14 Similar to the Karsz et al.’s 14 study, the study conducted by Ghanizadeh et al. 15 (the sample size was 50, with ages ranging from 8 to 15) reported that odor identification and sensitivity were lower in children with ADHD than in the control group. Still, this lower level was not statistically significant. 15 In the study conducted by MercanIsık et al. 16 (the sample size was 109, with ages ranging from 6 to 16), it was reported that the mean odor discrimination test, the mean odor identification test, and the mean TDI scores of the unmedicated ADHD group were significantly lower than those of the other two groups and that the mean odor threshold test scores of the medicated ADHD group were significantly lower than those of the control and unmedicated groups. Although MercanIsık et al. 16 compared two groups, with and without medication, their results supported us. In our study, similar to these, odor sensitivity discrimination and identification were found to be low in patients with ADHD and found to be statistically significant. The lack of information about our patients’ intelligence status was one of our study’s shortcomings.
In light of the findings obtained in our clinical study, we show that children with ADHD have significant impairments in odor threshold, odor discrimination, and odor identification tasks compared to healthy control participants. In another study aiming to reveal the reason for the relationship between odor and ADHD, although no statistically significant difference was found, it was found that there was a decrease in cortical activity during olfaction in the ADHD group compared to the control group by neuroimaging methods. 17 Such findings support previous theoretical predictions that ADHD should be associated with olfactory dysfunction due to neuro/anatomical and neurochemical similarities. The prefrontal lobe, long hypothesized to be dysfunctional in ADHD, contains essential primary and secondary areas for olfactory processing. These olfactory deficits are unrelated to age, gender, and intelligence level and are not a consequence of olfactory task difficulty. Another explanation for these deficits may be the inattention of children with ADHD during the olfactory task. In a study conducted with adults, no difference was found in all olfactory areas in the drug-taking and nontaking groups. In contrast, neuroimaging studies conducted at the same time showed that there was a problem in cortical activity in the temporal and frontal regions of the brain during olfaction in the nontaking group and that the oxygen levels of these regions were higher; in other words, the activity in these regions was less.17,18
The activity in these regions increased in drug users. This study also indirectly supports our study. While odor sensitivity, odor discrimination, and odor identification scores were low in the ADHD group, these scores increased to approach normal levels with 1-month methylphenidate treatment. It sometimes took too long to keep the patients focused during the tests. In the groups that were thought to benefit significantly from the treatment (methylphenidate), if the patient’s family also said that the patient’s attention and focusing were better, school achievement increased, the 2nd test was performed in a shorter time, and the scores were significantly higher. This shows that when the focus increases, the score increases, and the effectiveness of the treatment can be evaluated in this respect. In the studies conducted, groups receiving and not receiving medication were compared. Our study was the first in the literature to assess the same patient group with and without prescription. Such a prospective study had yet to be conducted before.
Previous research findings regarding the olfactory function of children with ADHD were quite inconsistent. The difference in the studies examining the relationship between ADHD and olfactory functions may be explained by the methods used.12-14 In these studies, techniques such as Sniffin’Sticks test, the Pennsylvania University Odor Identification test, phenylethylalcohol, and propyleneglycol were used to measure olfactory functions. 14 The use of different methods may have led to different results between studies. Therefore, it can be said that our study is compatible with the literature. It has also been suggested in previous studies that the difference between the results of the studies may be due to methodological differences, including the heterogeneity of study settings, methodologies used, age groups, and case definitions, and the source of cases, gender, or sociodemographic status of the population. 19 Schecklmann et al. 17 claimed that the duration of drug use may permanently improve olfactory functions and reported that the difference between the studies may also be related to the duration of drug use. However, it is stated that the chronic effects of methylphenidate use on olfactory functions are not fully understood. In our study, the sample group was not a sample group of patients who were still using the drug and those who had not started to use the drug, as in the study of Romanos et al., 12 but a sample group of patients who were still using the drug and those who had not started to use the drug. Therefore, our study can be evaluated differently in this respect. This increases the importance of our study even more. However, the results of the two studies do not precisely overlap. It is thought that the reason for this may be that the etiology of ADHD in the sample may be different. Since many gene regions cause ADHD, the gene regions associated with odor functions and ADHD may not have overlapped in two other studies.
Determining whether there is olfactory dysfunction in children with ADHD has an essential place in making inferences for the recommendation of rehabilitation treatment approaches. The suggestion that olfactory dysfunction may predict other behavioral problems or psychopathy in ADHD warranted further investigation. A larger sample size may confirm that two distinct subgroups of olfactory functioning exist in the ADHD population, and these subgroups may be related to the traditional three subgroups noted for ADHD. Mercan Isık et al. found that the predominantly inattentive type had lower odor sensitivity than the other groups. 16 Another study (Weiland et al.) comparing the inattentive subtype with the combined subtype revealed a decrease in identification in the inattentive subtype. 20 In our research, the subgroups were not evaluated separately. Romanos (combined type), Karsz (combined and Attention University Olfactory deficit in the foreground), Schecklmann (combined and attention bars deficit in the foreground), Ghanizadeh (undifferentiated), and our study was also undifferentiated.
Previous studies on olfactory dysfunction have generally investigated adult patients. 18 Another study evaluating odor identification in adult ADHD patients conducted by Sari and Taşkıntuna 21 (the sample size was 65, ages 6 to 15) found no significant difference from the control group subjects. Our study is the first to prospectively show changes in odor recognition and taste in children and adolescents with ADHD with treatment in the same patient group, and in general, studies report that odor identification, discrimination, and sensitivity are increased in ADHD, contrary to our research. As shown in previous reports on neurodegenerative diseases and psychiatric disorders describing deficits in olfactory perception (Marin et al. 12 ), our results show that odor sensitivity is significantly reduced (lower odor threshold, ie, lower Sniffin’ Sticks score) in unmedicated ADHD patients. This significant specific effect on the olfactory field indicates that the neurophysiological mechanisms underlying olfactory sensitivity differ from those underlying discrimination and identification.
In patients receiving methylphenidate treatment, Kevin et al. 22 and MercanIsık et al. 16 found that the reduced olfactory sensitivity seen at baseline normalized with methylphenidate. In a well-controlled study, Karsz et al. 14 found selectively superior sensitivity in previously unmedicated children. Murphy et al. 18 found higher discrimination in children after discontinuation of methylphenidate treatment. Ghanizadeh et al. 15 suggest that an enhanced sense of smell may be a characteristic feature of childhood ADHD. Continuous drug treatment with the dopaminergic-stimulant methylphenidate by Romanos et al. 12 resulted in normalized olfaction, further supporting the idea that dopamine is highly involved in olfactory processes. Still, our study noted a positive normalization of the reduced sense of smell. There are also indications that olfactory function in ADHD improves and normalizes with stimulants and dopaminergic drugs, as Andrew et al. 23 Methylphenidate is an indirect dopamine receptor agonist due to presynaptic inhibition of dopamine and noradrenaline reuptake. Therefore, it is a logical inference to suggest that dopaminergic dysregulation is also involved in the amelioration of olfactory impairment in ADHD. 24 In a recent study, improvement in odor threshold was observed in ADHD patients not taking medication compared to those taking medication and healthy controls. 16 Our analysis also showed that methylphenidate treatment significantly increased odor scores even in the early period. Statistical significance was found. Whether methylphenidate directly affects the dopaminergic interneurons of the olfactory bulbs or indirectly affects through the dopaminergic striatal system mediate, this observation remains to be resolved. Methylphenidate is an indirect dopamine receptor agonist due to presynaptic inhibition of dopamine and noradrenaline reuptake. Therefore, it is reasonable to suggest that dopaminergic dysregulation is vital in improving odor sensitivity in ADHD.
This research on olfaction in children and adolescents has identified significantly reduced odor sensitivity due to ADHD. There is considerable evidence that dopaminergic dysregulation is functionally involved in this phenomenon. Our results suggest that stimulant treatment (methylphenidate) normalizes the diminished sense of smell, and we recommend that olfactory sensitivity may serve as a biologically important marker for ADHD, with potential relevance for assessing treatment response as well. Despite certain limitations, particularly in disorders with dopaminergic pathology (eg, dopamine modulates odor in the olfactory bulb), our systematic review provides the foundation for future supportive and confirmatory studies investigating the sense of smell as a potential association in child and adolescent psychiatric disorders. Smell dysfunction has various underlying causes and may not be sensitive or specific enough to function as a disease biomarker.
The impairment in olfactory functioning in ADHD patients does not appear to be a consequence of difficulty with the olfactory task, as some participants with high intelligence tests also performed worse on both the identification and threshold tasks than some of those with low intelligence. Furthermore, participants’ intelligence test was not correlated with either. Thus, odor dysfunction was unrelated to age, gender, or intelligence test. 23
In this study, they did not assess odor thresholds, and due to the small sample size for girls, gender effects on odor identification ability could not be evaluated. Two other studies also reported no differences in odor-sensory functioning among ADHD patients compared to typical controls. The gender difference in olfactory discrimination reported in previous studies has never been explained, and its relevance was unclear. In our study, no significant relationship was found between genders. Regarding age, no significant relationship was observed between odor threshold and odor discrimination. As age increased, odor identification scores and, thus, total scores increased.
Further studies are needed to explore this phenomenon. These studies did not support a diagnosis-specific gender difference in the olfactory test. There were also no differences in odor identification and threshold performance between boys and girls in both patient and control groups. This is in line with the recent ADHD study, which reported that gender was not a significant predictor for odor detection ability in ADHD patients, according to the parental reports 15 by Murphy et al. 18 Women with ADHD performed better on odor recognition tasks than men, but this study tested adult-aged ADHD patients, and the current study also involved a small sample size. However, the sample size was adjusted with age- and sex-matched groups, and we had a more compatible and sufficient sample size compared to the literature. 20
Although sensitivity to bitter taste has been previously evaluated in adult women with ADHD, our study is the first to investigate the association of odor and taste with ADHD in children and adolescents in the same patient group. Weiland et al. 19 found that PROP bitter sensitivity was also higher in the ADHD group compared to the control group. It was suggested that the relationship between ADHD and bitter taste sensitivity may be related to the hormone dopamine. According to their hypothesis, the TAS2R38 receptor gene on the 7th chromosome, which also determines the sensitivity to bitter taste, and genetic factors that play a role in the etiology of ADHD are related. 25 In our study, the ADHD group did not differ from the control group regarding taste sensation. If the methods of the studies are analyzed to reveal the differences between the two studies, it is seen that the samples in the two groups are different from each other. Weiland et al. 19 used a female middle-aged sample in their study, whereas children and adolescents, including males, were included in our study. Therefore, it was thought that the difference between the studies might be because the sample in our study included males, whereas the study of Weiland et al. 19 consisted of females. To determine whether the difference in the results between the sample groups was related to age, the statistical analysis was repeated by fixing the age factor in our study, and it was found that the results were not affected by age in the child and adolescent groups. In a study by Mennela et al., 25 the sensitivity to bitter taste differed in adults and in children. Their study found that adults had less sensitivity to bitter taste exceed. The results of this study are contrary to Weiland et al. 19 Therefore, it can be suggested that sensitivity to bitter taste decreases with age in healthy individuals, whereas it increases with age in individuals with ADHD. The drug use status of the sample group in the study conducted by Weiland et al. 19 was not similar to our study but consisted of both drug-taking and nonmedication-taking participants. Among the studies investigating ADHD and olfactory dysfunction, Weiland et al. 19 examined taste and smell functions together in adult female ADHD patients. This was the first study in which taste function was investigated for the first time in the ADHD population. At the end of this study, an impairment in smell and taste sensitivity was found in women with ADHD, with a higher perception of bitter taste compared to the control group. 25 In that study, it was stated that the relationship between bitter taste and ADHD may be genetic, and chromosome 7 may act as an intermediary in this relationship. 25 As a result, an impression was obtained from the studies that the PROP bitter test may be a biological marker in the diagnosis of ADHD. However, further studies are needed due to the differences in the results.
The relationship between ADHD and smell and taste functions has not been evaluated. This study is critical because it is the first time that smell and taste functions were examined in children with ADHD in the same patient group compared to a control group of the same age and gender. The study’s limitations are that our participants’ intelligence levels needed to be evaluated, and the sample group was sufficient but small in number. Since there is no study in which both smell and taste sensitivity were examined together in children and in adolescents with ADHD, our study aimed to examine smell and taste functions together in children and adolescents with ADHD and to investigate the relationship. Although there is no objective data for taste, taste-related disorder was not observed in ADHD patients in our survey. Further studies are needed to obtain more evidence on the diagnostic specificity, sensitivity, and validity of determining taste sensitivity as a biological marker of ADHD.
In this study, we also compared the taste and smell functions of children and adolescents with ADHD with those of healthy children and adolescents. Although odor sensitivity, odor discrimination, and identification were low in the ADHD group, there was a statistically significant difference between healthy children and adolescents. In all the relevant studies of olfactory sensory function in ADHD patients, the irrelevant results reported may be due to differences in the measurements and procedures used. This research on olfaction in children found significantly reduced odor sensitivity with ADHD. We have reason to believe that dopaminergic dysregulation is also functionally involved in this phenomenon. Our results suggest that stimulant treatment (methylphenidate) exerts a normalizing effect on altered olfactory perception and that olfactory sensitivity may be helpful as a biologically important marker for ADHD. Therefore, it seems warranted to establish an easy-to-administer, cost-effective, and standardized test of olfactory function worldwide. Studies should be supported in the hope that accurate assessment of olfactory function in inpatients with ADHD will allow us to use such assessments in the early diagnosis, treatment, and prognosis of the disease. Further studies with larger sample sizes are required to confidently declare differential deficit or intact ability in these olfactory functions. Future studies are needed to obtain more evidence regarding the diagnostic sensitivity, specificity, and validity of olfactory function as an important biological marker of ADHD.
Our study had some limitations, such as the fact that it was prospective, and there was a declaration of the COVID-19 pandemic immediately after we started working. Fewer patients could be included in the survey than planned. Four patients initially included in the study had to be excluded because they had Covid 19. Therefore, we could report the early results (after one month) of methylphenidate. The test collaboration of the Sniffin’ Stick (children) was quite challenging. In our study, ADHD was not divided into subtypes, and the intelligence of the patients was not evaluated. It has been shown subjectively that no taste disorder exists, but this will be supported with more objective tests in future studies. It will be more helpful.
Our study could have been further detailed by comparing the early and late periods with long-term drug use by performing ADHD subtyping, intelligence testing in patients, and objective taste analysis. However, this was impossible due to the COVID-19 epidemic and funding limitations. When we looked at the literature, we could not find similar studies with or without medication in the same patient group. In this respect, our study constitutes the first prospective example.
Conclusion
ADHD is a behavioral condition characterized by the difficulty of persisting in one activity, as well as focusing and controlling impulses. Currently, the neurobiological aspects of ADHD have received growing attention. Part of these studies suggests an essential role of the dopaminergic system, which has also been described as the central nervous system’s “attentional hub,” connecting all major sensory systems. The inclusion of ADHD in the sensory processing disorder approximately two decades ago mediated the link between inattention and misdirected sensory processing in the human brain.
Sniffin’ Sticks test is meaningful in assessing ADHD patients because it was developed to assume they could have a problem with the olfactory sensation. The existing disease does not affect the results of the psychophysical semiobjective test, which can be used without correction (the olfactory examination results reflect the state of the receptor and are not the result of attention changes with ADHD).
The olfactory deficiencies in ADHD in all areas of odor identification, discrimination, and odor perception threshold. These deficits are unrelated to age, gender, and intelligence level. Significant improvement was observed in the early period with methylphenidate treatment. It is predicted to be a handy marker in ADHD diagnosis and treatment follow-up. More extensive, longitudinal studies are needed to confirm these findings. Of course, future studies are required to obtain more evidence regarding diagnostic sensitivity, specificity, and validity to determine olfactory function as a biological marker of ADHD.
Footnotes
Appendix 1: Taste Alteration Scale Questionnaire
The column on the left below contains items describing various symptoms and problems related to changes in taste, and the rows on the right contain options related to your taste buds.
Please tick (*) the option that best fits your situation.
Acknowledgements
We thank SevinçPurenYucel from the Biostatistics Department for her statistical evaluation.
Author Contributions
Taylan Bilici: Planning, designing, literature survey, and data collection and writing.
Suleyman Ozdemir: Planned, designed, and edited literature surveys and gave active intellectual support.
Gonca Gül Çelik: Planning, designing, literature survey, and data collection and gave active intellectual support.
All authors contributed to planning, designing, conducting literature surveys, and collecting data, giving active intellectual support.
Availability of Data and Materials
All data for this study are presented in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethics committee approval was taken from Cukurova University. (Date: 3rd of July, 2020 Number: 110)
Informed Consent
There is no need for informed consent because the data were evaluated retrospectively.
