Abstract
Chondroid syringoma, a mixed tumor of the skin, is an acquired hamartoma that differentiates into hair follicles, sebaceous glands, and apocrine sweat glands. Chondroid syringoma in the ear region is exceptionally rare. However, we present 2 cases of ear chondroid syringoma: 1 involving multiple lesions in the external auditory canal and the other including a single lesion behind the auricle. We reviewed relevant literature to offer insights into the diagnosis and treatment of ear tumors in the future.
Clinical Data
Case Introduction
We chose 2 patients diagnosed with chondroid syringoma who received surgical treatment in the Department of Otolaryngology at Shandong First Medical University Provincial Hospital between October 1, 2017, and September 30, 2024.
Case 1
The patient was a 56-year-old Han male with a tumor in the right external auditory canal, measuring approximately 0.3 × 0.3 cm, present for over 10 years. Initially, he did not seek treatment, and he experienced no symptoms such as pain or itching. The tumor gradually increased in size until it obstructed the right external auditory canal. The patient had a 20 year history of chronic otitis media in the right ear. Upon physical examination, a round tumor was identified in the right external auditory canal, fully occluding the opening. The tumor was similarly colored to the surroundings, was hard, and had limited mobility (Figure 1). Preoperative computed tomography imaging revealed multiple round, soft tissue density shadows around the right external auditory canal, with some partially fused. The canal was significantly narrowed and blocked, and adjacent bone exhibited partial absorption and destruction (Figure 2).
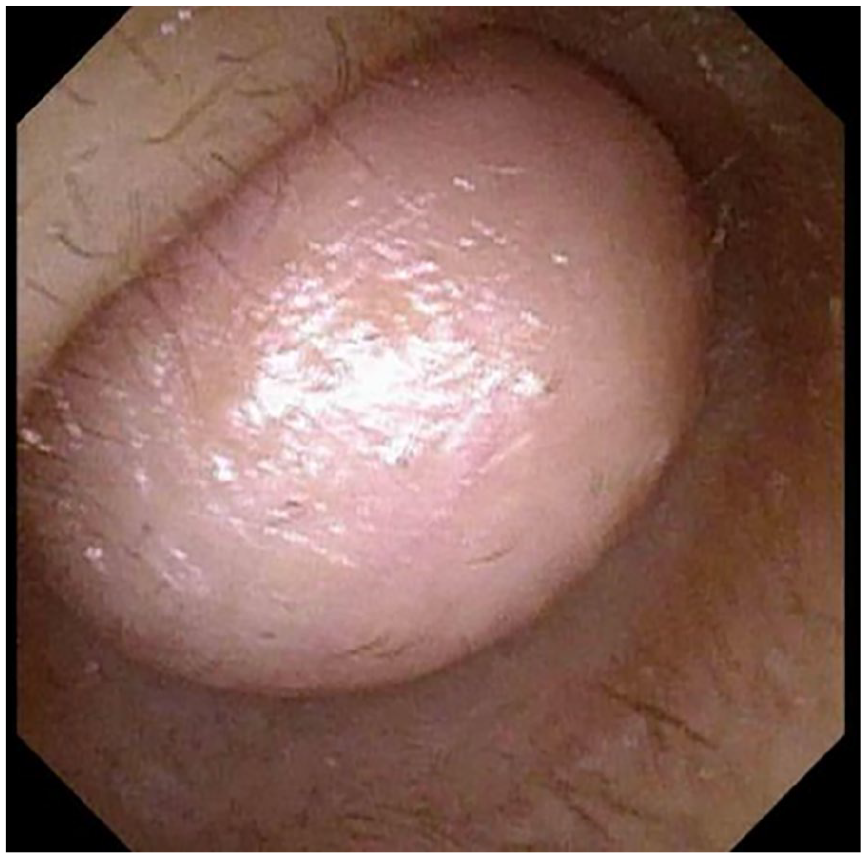
Photograph of fiberoptic otoendoscopy of case 1. A round neoplasm in the right external auditory canal is observed, the same color as the surrounding skin, and it completely blocks the external auditory canal opening.
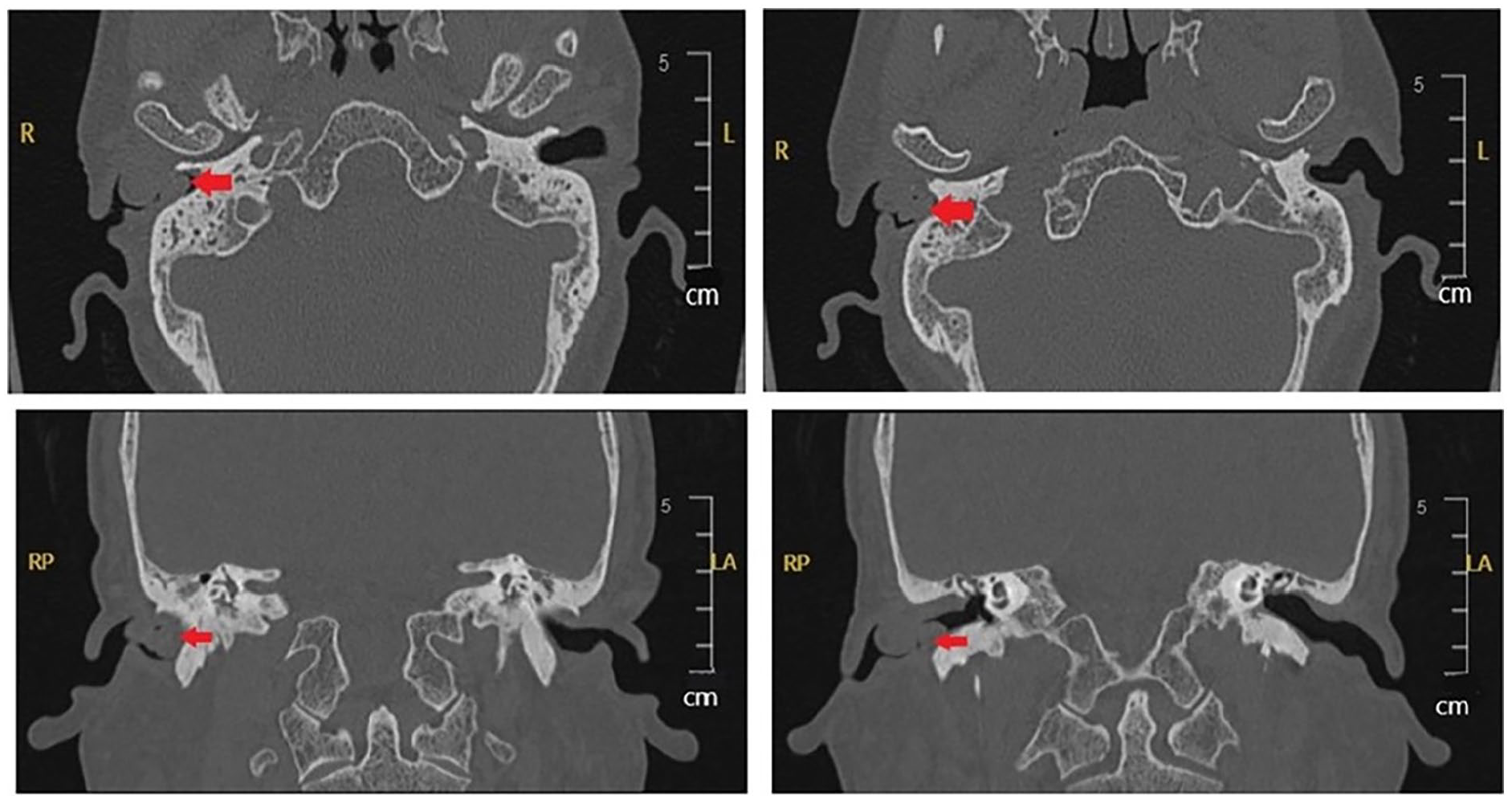
Horizontal CT scan of the temporal bone of case 1. Multiple round soft tissue densities of partially-fused shadows were identified in the right external auditory canal. The right external auditory canal was narrowed and blocked, and the adjacent bone was partially absorbed and destroyed (the red arrow indicates the lesion). CT, computed tomography.
Case 2
The patient was a 35-year-old Han male with a tumor approximately 0.5 × 0.3 cm behind the left auricle for over 10 years. While there was no obvious cause, he experienced intermittent pain but did not seek treatment. The tumor gradually increased in size, and pain intensified.
Physical examination revealed a neoplasm behind the left auricle matching the color of the surrounding skin, with a hard texture, and limited mobility. Preoperative color Doppler ultrasound, during the auxiliary examination, exhibited a cystic nodule in the subcutaneous soft tissue of the left earlobe, approximately 2.4 × 1.7 × 1.3 cm. The nodule had a regular shape, clear boundaries, a thick and rough cyst wall, and poor sound permeability. It displayed low echoes with dot-like strong echoes. Color Doppler flow imaging indicated blood flow signals in the cyst wall (Figure 3).
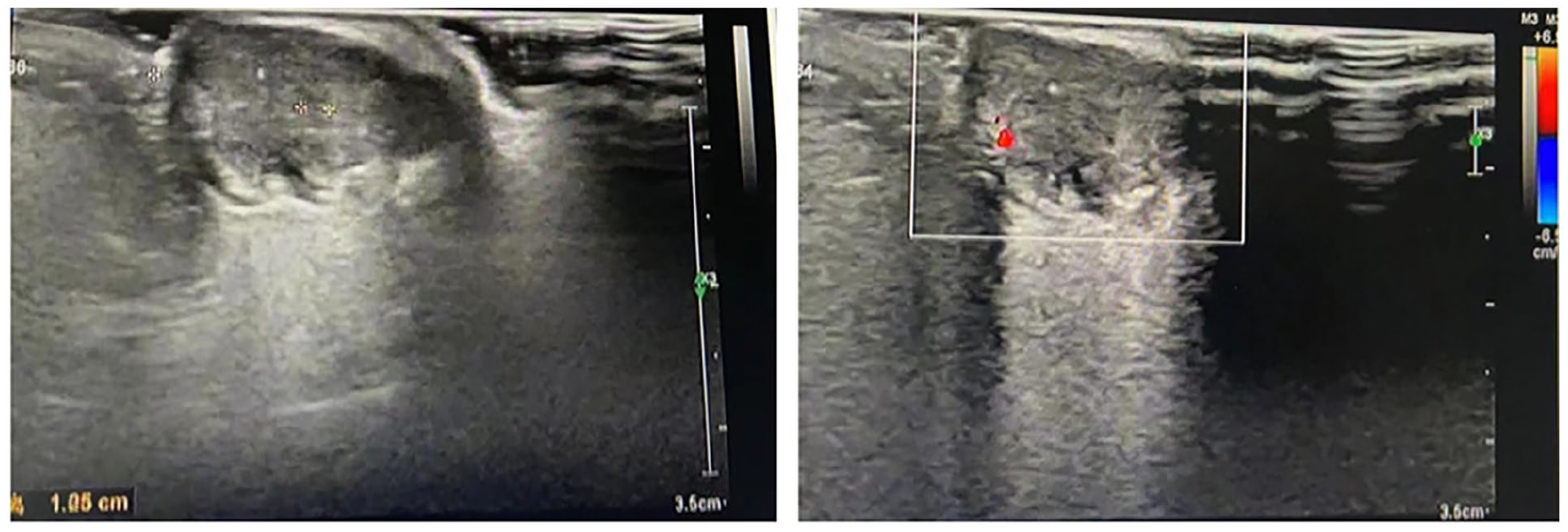
Color Doppler ultrasound examination of a tumor behind the ear of case 2. A cystic nodule approximately 2.4 × 1.7 × 1.3 cm in size was found in the soft subcutaneous tissue of the left earlobe. It had a regular shape, clear boundaries, and a slightly–thick-and-rough cyst wall. Poor sound transmission was observed inside of the cyst, which was filled with low echoes and dot-like strong echoes. Blood flow signals were identified on the cyst wall.
Surgical Procedure
Case 1
The patient was placed in a supine position, with the right ear facing upward after general anesthesia. We performed routine disinfection and placed sterile towels around the surgical site. An incision was made in the right ear, followed by dissection of the skin and subcutaneous tissue. Using a microscopic examination, we identified a spherical tumor blocking the external auditory canal, with a beaded appearance. The surface skin remained intact, and we preserved as much surrounding skin as possible. We excised the tumor at its base (Figure 3). The tumor was solid and tough, with a well-defined margin from the surrounding tissue. Its base was situated in the anterior and upper walls of the external auditory canal.
After completely removing the tumor, we confirmed that the external auditory canal was unobstructed. The tumor was flash-frozen and sent for pathology, indicating that it was likely a benign tumor. However, paraffin embedding and immunohistochemistry were undertaken for definitive diagnosis. We repositioned the residual skin of the external auditory canal and employed a free flap from behind the auricle for repair. We noted that the patient had a perforated tympanic membrane from previous otitis media. Consequently, we repaired the perforated tympanic membrane and filled the external auditory canal with gelatin and iodoform gauze (Figure 4).
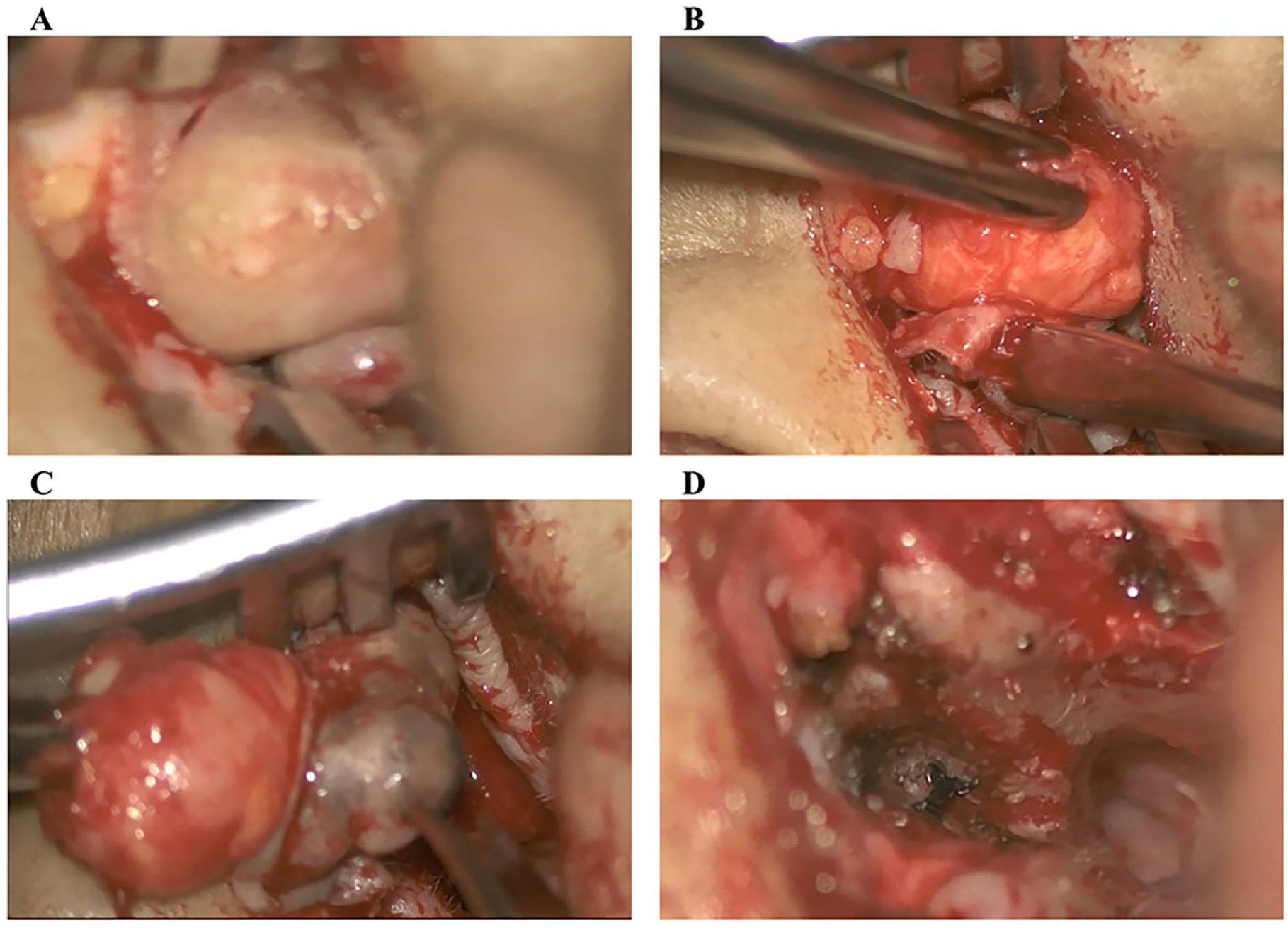
Images of the surgical process of case 1. (A)The tumor appeared beaded. (B) Prior to resection, the tumor was found to be subcutaneous and round, with clear boundaries. (C) During removal, the tumor appeared beaded. (D) After resection, the skin of the external auditory canal was repositioned.
Case 2
The patient was placed in a supine position, with the head turned to the right, exposing the left ear upward. After routine disinfection and the application of a sterile drape, we identified a hard, round mass approximately 2 cm in diameter behind the patient’s left earlobe. Following local infiltration anesthesia, we made an incision on the skin surface over the mass, dissecting the skin and subcutaneous tissue. A subcutaneous cystic-solid mass was revealed, including a small amount of cottage-cheese-like discharge. The mass had clear boundaries and was removed entirely. We loosened the surrounding skin, corrected the wound surface, sutured it, and applied a pressure bandage.
Postoperative Pathology
Case 1
Postoperative routine pathology revealed that the tumor tissue, measuring 1.5 × 1.0 × 1.0 cm, primarily consisted of epithelial cells and polymorphic stromal components (Figure 5). The epithelial components formed branched glandular ducts and cysts. The cyst wall of the glandular epithelium was lined with 2 cell layers: the inner layer comprised single-layer cuboidal and columnar epithelial cells, mostly arranged in layers, possessing rich cytoplasm and medium-sized nuclei at the base, with red cytoplasm. Apical secretion was evident. The outer layer consisted of spindle-shaped or flat myoepithelial cells. Eosinophilic amorphous substances were present in the expanded cyst cavity. The tumor’s stromal components exhibited diversity, including fibrosis, mucinous degeneration, stromal cartilage, and a significant prevalence of adipose tissue. Neither epithelial nor stromal cells displayed atypia. Immunohistochemistry results revealed the presence of CK7 (+), P40 (+), Ki-67 (+2%), P53 (−), CEA (−), GCDFP-15 (−), AR (+), and CK5/6 (+) (Figure 6). Based on these findings, we determined that the tumor in the right external auditory canal was consistent with a chondroid syringoma.
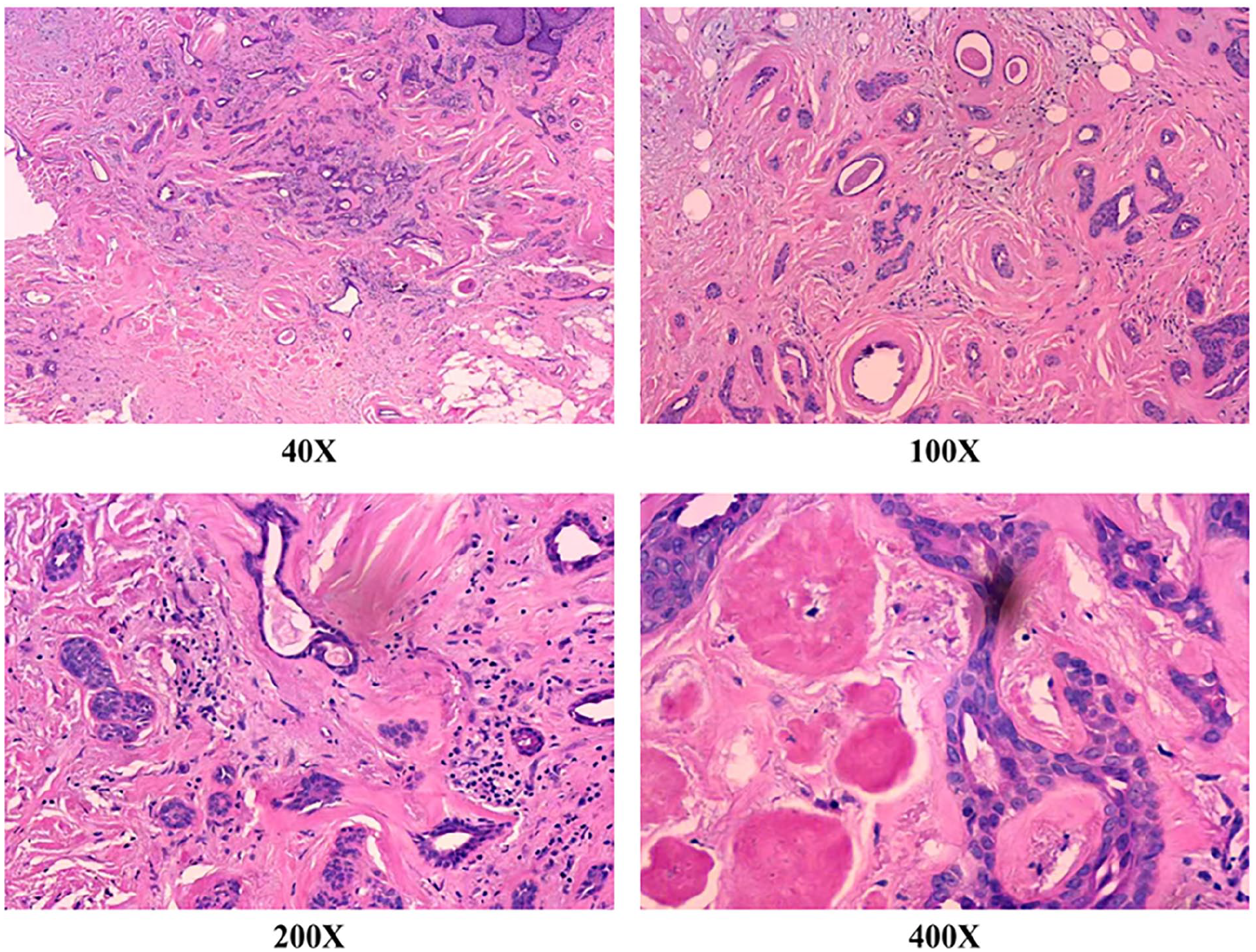
Images of H&E staining of case 1.
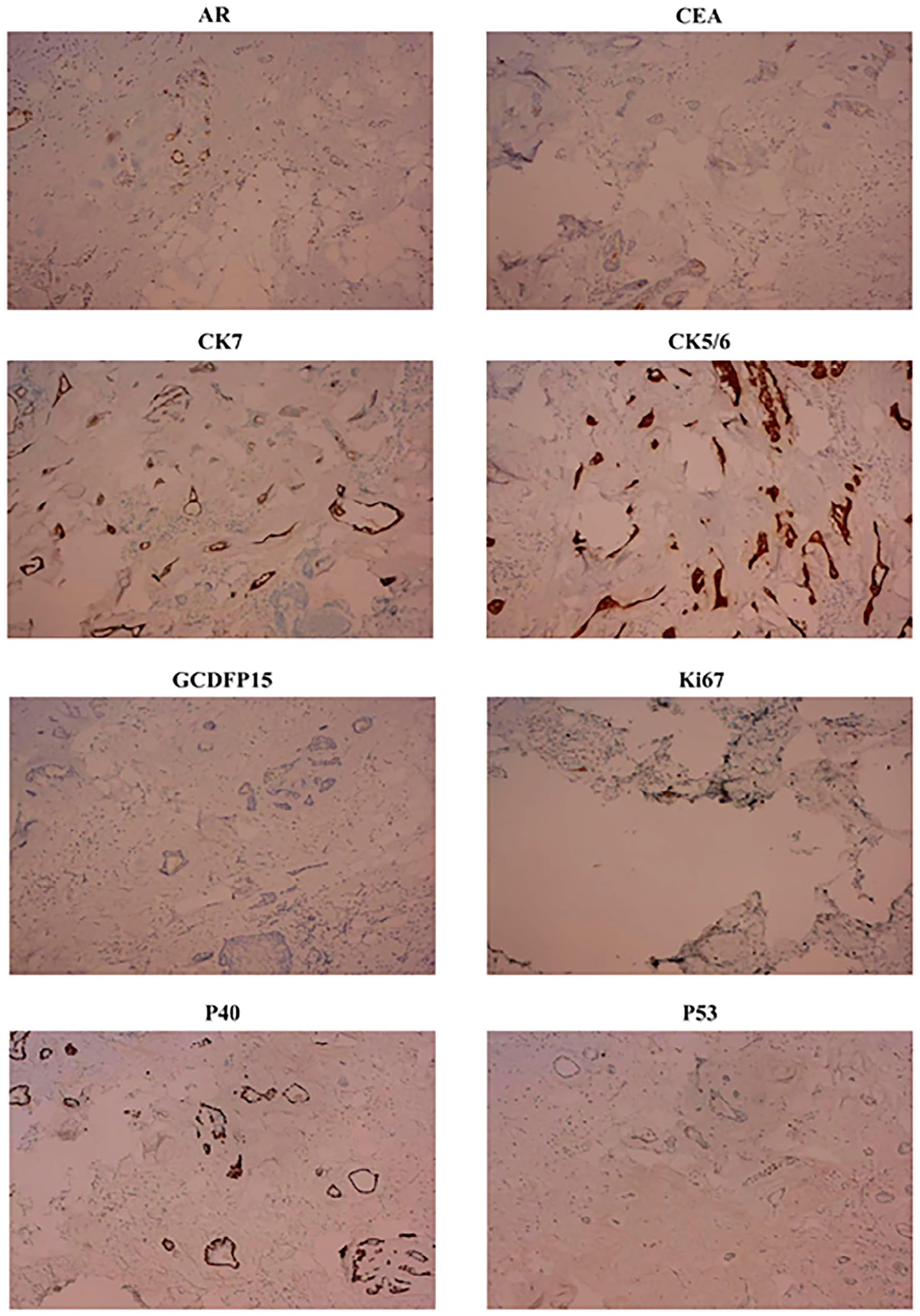
Immunohistochemistry images of case 1. AR (+), CEA (−), CK7 (+), CK5/6 (+), GCDFP-15 (−), Ki-67 (+2%), P40(+), P53 (−).
Case 2
Routine postoperative pathology revealed a chondroid syringoma measuring 2 cm in diameter surrounding the left ear. The tumor tissue mostly consisted of epithelial cells and polymorphic stroma. The epithelial components exhibited tubular and cystic growth patterns, while the stroma appeared fibrotic and myxoid. The tumor manifested as small island-like tubules with cystic structures of various shapes and sizes. Eosinophilic amorphous substances were identified in the expanded cystic cavity (Figure 7).
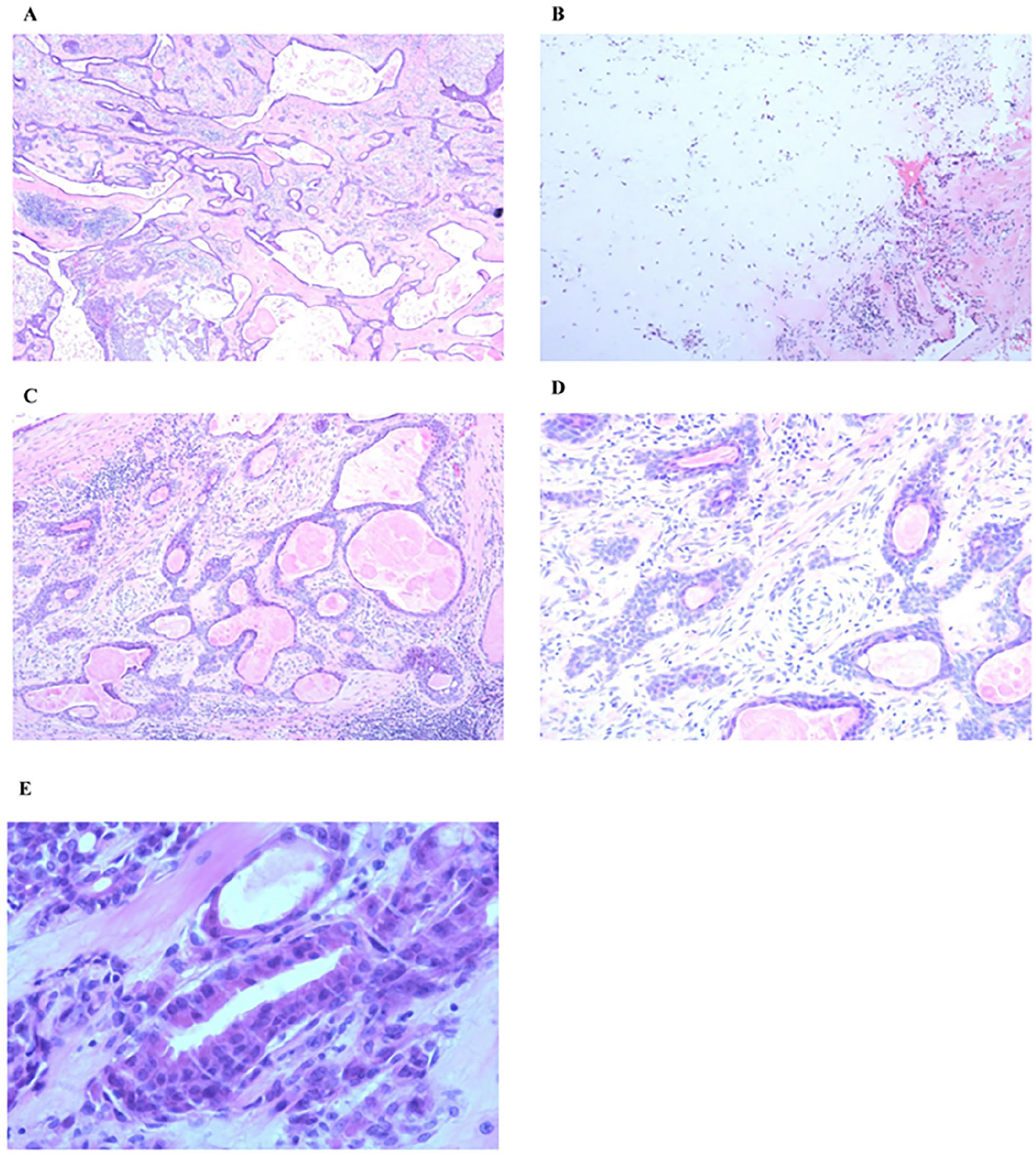
Images of the H&E staining of case 2. (A) Tumor composition. Epithelial and interstitial components are present. The epithelial components are arranged into glandular ducts, cystic and solid epithelial islands, and interstitial fibrosis (magnification, 40×). (B) Myxochondroid stroma (magnification, 40×). (C) Eosinophilic membrane in the glandular duct lumen (magnification, 100×). (D) Glandular duct epithelium lined with double-layered cells. The inner layer consists of cuboidal and columnar epithelium, whereas the outer layer has flat or cuboidal myoepithelial cells (magnification, 200×). (E) Apocrine secretion observed in the inner layer of cells (magnification, 400×). H&E, hematoxylin-eosin.
Discussion
The international incidence of chondroid syringoma ranges from 0.01% to 0.098%. 1 While it can occur at any age, it is more prevalent in men, with a male-to-female ratio of 5:1 to 2:1. 2 This disease typically occurs in the nose, cheek, upper lip, scalp, forehead, and mandible, occasionally affecting the axilla, trunk, and limbs. Scrotal involvement is rare. 3 Case 1 presented multiple chondroid syringomas in the external auditory canal, which is even rarer, and no similar case reports exist in the literature. Case 2 involved a tumor in the external ear.
Theodor Billroth first described chondroid syringoma in 1859 as a rare benign tumor of the skin appendages. Clinically, it manifests as solitary, firm, painless, well-defined, intradermal, or subcutaneous nodules 0.5 to 3.0 cm in diameter, although a report exists of a giant mixed skin tumor on the face measuring approximately 6.5 × 10.5 cm. 4 Patients with chondroid syringoma typically have no symptoms, with tumors growing slowly, rarely rupturing, and seldom recurring after resection. 5 Malignant transformation is uncommon. Nguyen and Cassarino 6 reported a case of benign chondroid syringoma on the right thumb relapsing 20 years later, leading to a pathological diagnosis of malignant chondroid syringoma (MCS) with peripheral nerve invasion. However, after comprehensive surgical resection, there was no recurrence or metastasis during the 5 year follow-up.
The cause of chondroid syringoma remains unclear, but it may be associated with trauma. Clinically, this condition presents as a single, solid, and well-demarcated nodule. The surface skin color is typically normal, although it can occasionally appear purple-red, remaining smooth, with rare instances of ulceration. 7 Due to its nonspecific clinical features, chondroid syringoma is commonly misdiagnosed as other conditions, including epidermal cysts, dermatofibromas, neurofibromas, neurothecomas, intradermal nevi, pilomatricomas, steatocystomas, chondromatosis, soft tissue myoepitheliomas, and adnexal tumors. Accurate diagnosis relies on histopathological identification. 8 In this study, case 1 had a history of chronic otitis media over 20 years, while case 2 exhibited a small amount of slag in the tumor, suggesting a potential inflammatory reaction. This indicates that the development of this type of tumor may be associated with inflammation. During surgery, we repaired the eardrum of case 1 to alleviate any inflammatory stimulation. However, the relationship between inflammation and chondroid syringoma requires further investigation in a larger sample size.
Ultrastructural analysis reveals that myoepithelial cells surround glandular epithelial cells in mixed tumor of the skin (MTS) pathological tissues. The matrix components of chondroid syringoma include collagen (types I and IV), fibronectin, and laminin. 9 Electron microscopy and immunohistochemical analyses suggest that these cells originate from either epithelial or mesenchymal tissue. There is ongoing debate regarding whether most chondroid syringoma derive from eccrine or apocrine sweat glands. Recent data suggest that they can differentiate into either type. 10 Most chondroid syringoma are apocrine sweat glands, typically multilobed, located deep in the dermis, presenting as well-defined masses with a cartilaginous appearance. The lobules are separated by fibrous septa. The epithelial component consists of nests or cords of cuboidal or diagonal cells, characterized by abundant eosinophilic cytoplasm and basophilic nuclei. 10 These cells may appear isolated in nests or cords or within irregular tubulo-alveolar or duct-like structures. The tubulo-alveolar structure suggests differentiation into the secretory portion of the sweat gland, lined with 2 or more layers of epithelial cells. The outer cells are flatter and originate from myoepithelial cells. Apparent apocrine secretion can occasionally be observed. 11
Occasionally, epithelial cells arrange themselves in a lace-like pattern. Glycogen-rich clear cells, positive for periodic acid-schiff (PAS) staining, are identified, and the ductal structure consists of 1 or 2 layers of cells, indicating differentiation into dermal sweat gland ducts. 10 There is no atypia or necrosis, and mitotic figures are rare. Spindle-shaped myoepithelial cells appear within the chondroid matrix. Rarely, calcification foci occur in the bone marrow cavity. 12 Hyaline cells are present in about 40% of chondroid syringomas, exhibiting oval shapes, darkly-stained ground glass or glassy cytoplasm, and eccentric nuclei, resembling plasma cells. Therefore, they are termed plasmacytoid cells. In instances where most lesions consist of hyaline cells, they are called chondroid syringomas rich in hyaline cells. 13 A few eccrine-type MTSs contain relatively-uniform, small, round lumens formed by a single layer of cuboidal epithelial cells, lacking apocrine secretion. 3
MCS is extremely rare and often presents as malignant from the outset, while some cases arise from benign transformations. MCS is more frequent in women and appears on the trunk and limbs. 4 The presence of atypical cells, pathological mitotic figures, satellite lesions, tumor necrosis, marginal invasive growth, and metastasis in MTS histology suggests malignant transformation. Additionally, local recurrence following benign MTS resection should raise concerns about malignant transformation. 14 The local recurrence rate of MCS after surgery is approximately 49%, with a mortality rate of approximately 25%. Recurrence can occur within 1.5 months to 6 years after surgery, and the metastasis rate is about 60%. Lymph node and distant metastasis rates are around 42% and 40%, respectively. The most common site for distant metastasis is the lung, followed by the bone and brain. 15 Histologically, MCS must be differentiated from myxoma, myxoid chondrosarcoma, basal cell carcinoma, malignant melanoma, alveolar soft tissue sarcoma, and adenoid carcinoma. 16
Surgical resection is the primary approach for treatment, as the disease often presents as isolated nodules. Most chondroid syringomas in the head and neck are benign, and malignant cases are rare. However, even benign chondroid syringomas can recur and become malignant if not completely removed. Therefore, complete resection and regular follow-up are required for all chondroid syringomas. 17 Pathological results such as cell atypia, mitotic figures, marginal infiltrative growth, satellite lesions, and tumor necrosis indicate malignant transformation. The patient in case 1 underwent complete surgical resection and has been monitored for 3 months without any signs of recurrence. The patient in case 2 has been followed up for 6 years without recurrence or malignant transformation.
In conclusion, because the skin lesions of chondroid syringoma are nonspecific and patients often report no subjective symptoms, clinicians should consider this disease when observing a subcutaneous tumor with a smooth surface, hard texture, and clear boundaries on the head, face, or neck. The primary treatment involves complete resection, followed by a regular follow-up after pathological diagnosis.
Footnotes
Acknowledgements
X.F.Y. and H.J. provided direction and guidance throughout the preparation of the manuscript. Q.W.W. and X.S. performed the literature search and wrote the original manuscript. Z.Y.A. and Z.S.J. provided constructive suggestions and significantly revised the manuscript. L.C.L., Z.H.Y., and H.J. collected clinical samples. All authors read and approved the final manuscript.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (#82401329), the National Natural Science Foundation of Shandong Province (#ZR2024QH372), and China Postdoctoral Science Foundation (#2024M751886).
Patient consent and Ethical Statement
The studies involving human participants were reviewed and approved by the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (No. 2024-443). The patients/participants provided written informed consent to participate in this study. Pathological samples were acquired during the operation.
