Abstract
Introduction
Labyrinthitis is an inner ear (labyrinth) inflammation. Labyrinthitis common causes include infections (upper airway infections, otitis media infection), inflammatory processes, autoimmune processes, temporal bone trauma, hemorrhage, and tumors. In most cases, labyrinthitis is caused by a viral infection. 1 In the rare case of bacterial labyrinthitis, the most often incriminated bacteria are Streptococcus pneumoniae and Haemophilus influenzae.1,2
Herein, we present an exceptional case of pseudomonas labyrinthitis and discuss the clinical aspects, the value of imaging, the treatment, and the prognosis of this pathology.
Case Report
In the ear-nose-throat (ENT) department of Mohamed Taher Maâmouri Hospital, a generally-healthy 18-year-old female presented with left sudden hearing loss. Moreover, the patient complained of intense, day-long, “room spinning” vertigo, recurrent vomiting and severe tinnitus evolving for 24 hours. According to the patient, she had never had any history of otitis during childhood. However, 10 days ago, she had developed otalgia, middle ear fullness, and fever and had been self-medicated with antibiotics (amoxicillin–clavulanic acid 2 g/day) with no improvement. General examination showed a temperature of 38.2°, normal blood pressure, and normal consciousness. Otological examination: retroauricular and ear pinna inspection showed no abnormalities. Otoendoscopic examination revealed a punctiform antero-inferior tympanic perforation with purulent otorrhea. The external auditory canal was normal. Vestibular examination revealed a harmonious peripheral vestibular syndrome characterized by a spontaneous right-beating horizontal rotatory nystagmus, which became more intense when the patient looked to the right and diminished when she looked to the left. Segmental tests (Romberg, Fuckuda) were deviated to the left. The neurological examination was normal, especially no involvement of the cranial nerves and no meningeal signs. The rest of the examination was normal. In view of this clinical presentation, the diagnosis of left labyrinthitis complicating acute otitis media was made. Thus, the patient was immediately hospitalized in the ENT department. A tonal audiogram revealed perceptive deafness at 65 dB in the left ear. A temporal computed tomography (CT) scan showed a left opacity of the middle ear without bone lysis (Figure 1). Magnetic resonance imaging (MRI) was performed, showing a left antro-epitympanic filling characterized by a T2 hypersignal, T1 hyposignal, intense enhancement post-gadolinium injection (Figure 2). There was no hypersignal observed on diffusion sequences. Additionally, the CISS sequence revealed attenuation of the T2 fluid hypersignal with intense labyrinthine enhancement affecting the cochlea, vestibule, and semicircular canals (Figure 3).
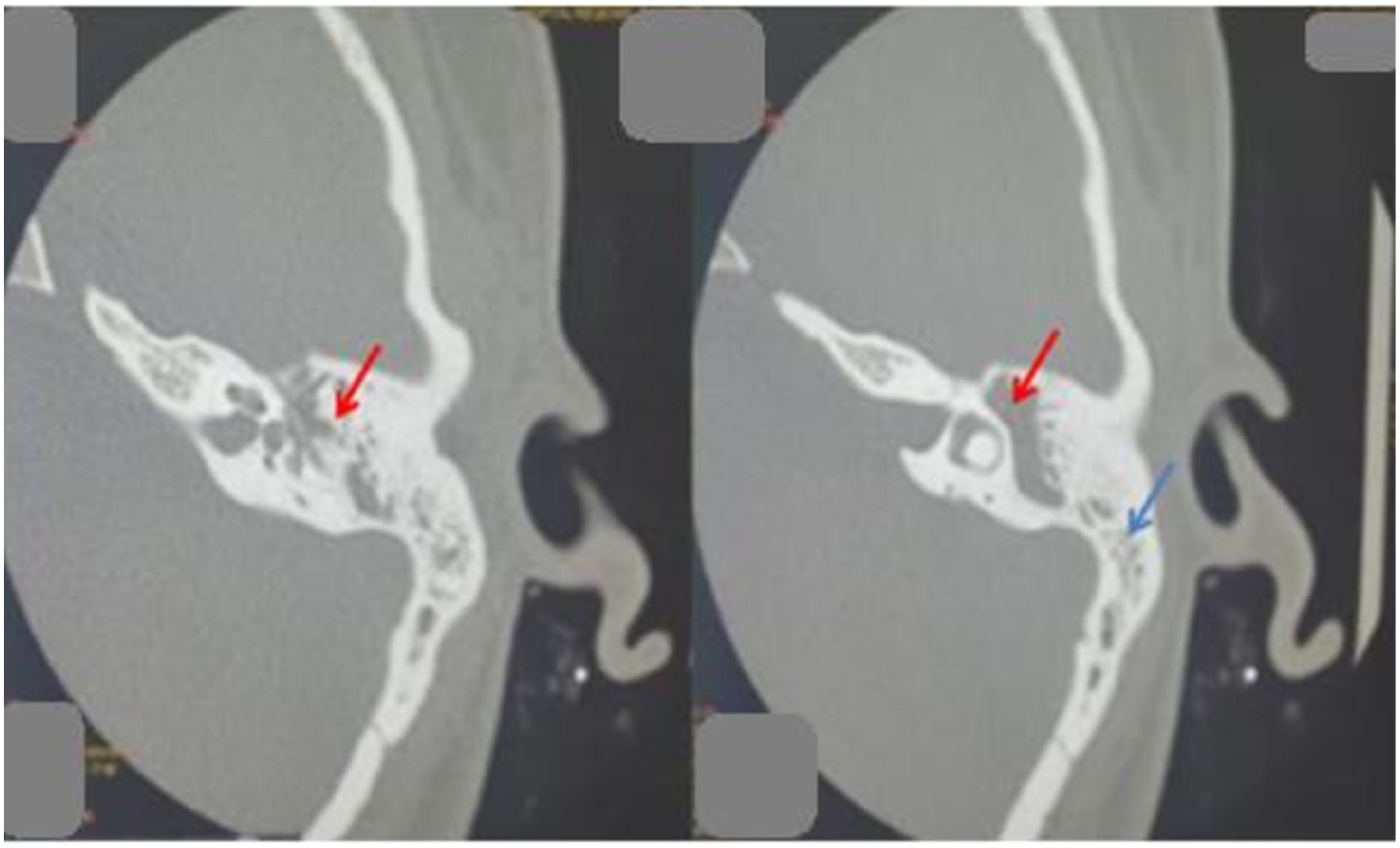
Temporal CT scan sequences showing left middle ear (red arrow) and mastoid (blue arrow) opacities with no bone destruction.
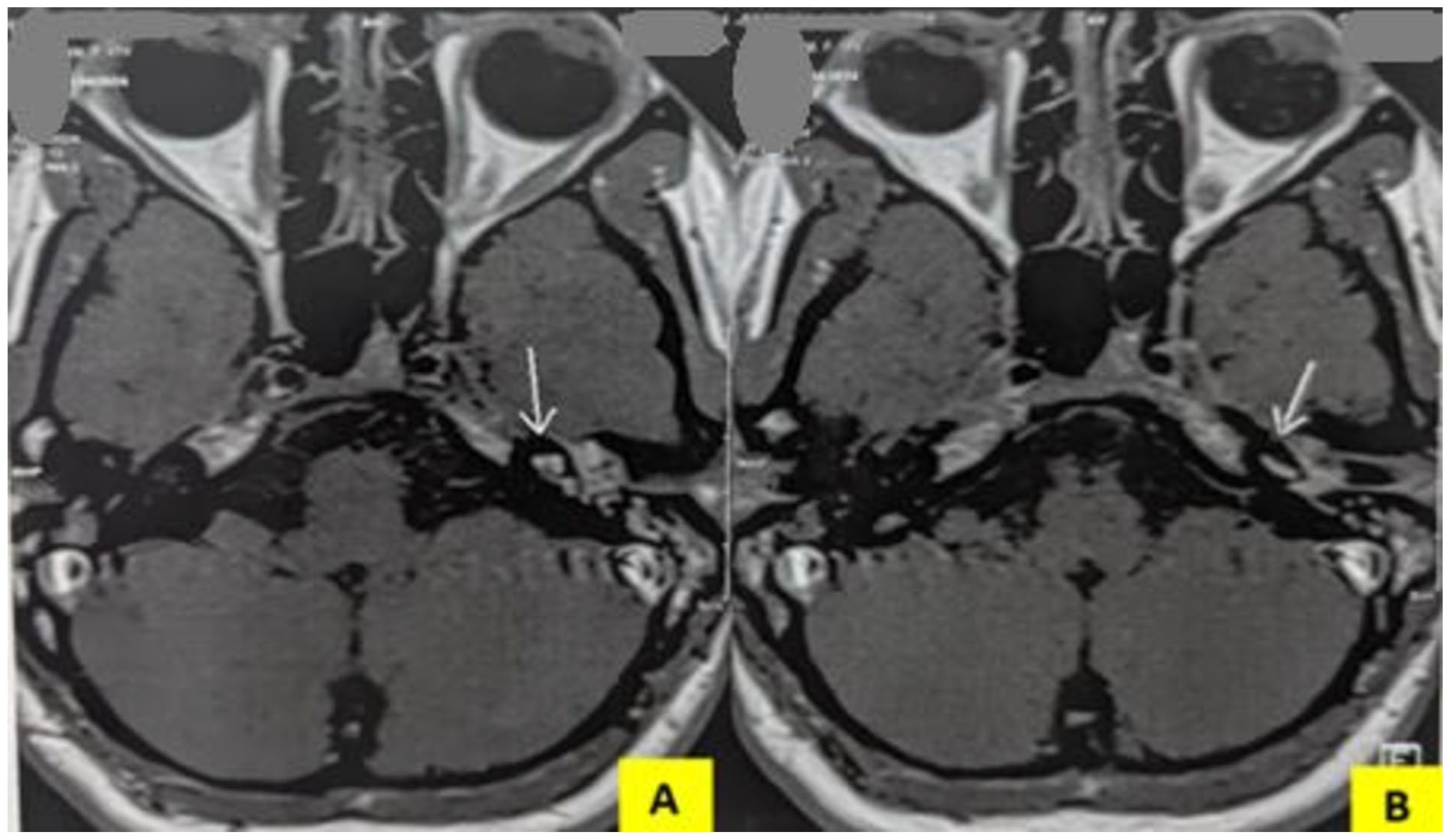
MRI sequences: (A) Intense left labyrinth enhancement post-gadolinium injection (white arrow). (B) Intense cochlear enhancement post-gadolinium injection (white arrow).
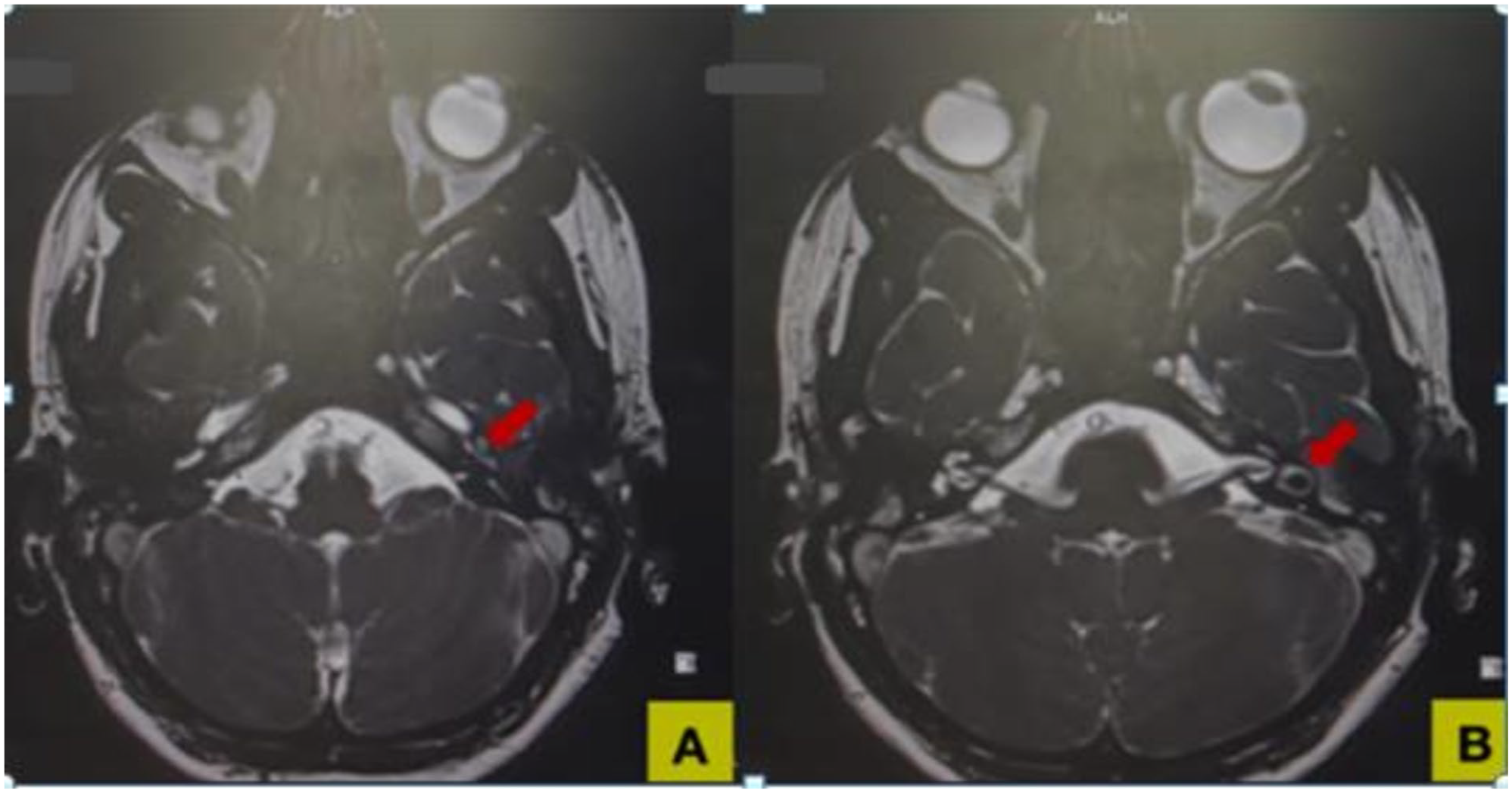
MRI: CISS sequences. (A) Intense left labyrinthine enhancement of the cochlea (red arrow). (B) Intense left labyrinthine enhancement of the vestibule and semicircular canals (red arrow). CISS, constructive interference steady state.
Laboratory studies found a biological inflammatory syndrome. The direct microbiological examination of the otorrhea sample was negative. However, otorrhea sample culture demonstrated the presence of Pseudomonas aeruginosa. As soon as she was hospitalized, we started probabilistic intravenous antibiotherapy with cefotaxime (200 mg/kg/24 hours) and vancomycin (20 mg/kg/24 hours) for 17 days. Three days after starting antibiotics, the patient achieved apyrexia, but tympanic inflammation persisted and C-reactive protein remained high. After that, in order to adapt to antibiogramm, we switched antibiothrapy to ceftazidime (6 g/24 hours in 3 doses) + vancomycin. Three days later, the patient developed allergic reaction (skin rush) to that association followed by allergy to imipenem and even meropenem. Finally, in collaboration with specialists in infectious diseases and pharmacovigilance, we prescribed 15 mg/kg/24 hours of amikacin during 5 days. Alongside antibiotic therapy, the patient benefited from daily local ear care, and received an anti-dizziness treatment (Acétylleucine 3 g/day during 7 days) and B vitamin therapy. A 14-day steroid treatment 5 mg/kg/24 hours of hydrocortisone had been prescribed after apyrexia. The patient began vestibular rehabilitation 7 days after hospitalization. After 25 days of treatment, we did not note hearing loss improvement, so patient benefited of hyperbaric oxygen therapy (10 sessions) with no significant improvement (hearing loss of 50 dB). Six months later, a left hearing aid was proposed to the patient, and she regained satisfactory static and dynamic balance.
Discussion
Infectious labyrinthitis is most often viral in origin. The viruses most often implicated include herpes, measles, mumps, and rubella. 1 When labyrinthitis is bacterial in origin, Streptococcus and H. influenzae are the most often incriminating bacteria. 2 In our case, the incriminating bacteria was P. aeruginosa, which is extremely rare. A similar case of pseudomonas tympoanogenic labyrinthitis was reported in 1985 by Tanaka et al. 3 In tympanogenic bacterial labyrinthitis, inflammation may be caused either by bacterial toxins and/or host cytokines and inflammatory mediators (serous labyrinthitis) or directly by bacterial invasion (suppurative labyrinthitis). 1 In fact, both bacteria and toxins can spread from the middle ear to the inner ear through bone lysis or dehiscence, or through oval or round windows. 1 Suppurative labyrinthitis is aggressive, causes irreversible cophosis, and may rapidly progress to meningitis. 4 Labyrinthitis with incomplete reversible hearing loss, resulting from acute otitis media, is usually serous, and isolated without other complications. 4 Thus, the distinction between serous and suppurative labyrinthitis should be made retrospectively, after cochlear and vestibular function have returned. 5 In this case, the diagnosis of serous labyrinthitis was retrospectively established given the incomplete hearing loss and the absence of complications.
Clinically, labyrinthitis is characterized by the abrupt onset of an intense, rotating vertiginous attack lasting up to 72 hours, followed by balance disorders and/or short-lasting positional vertigo. The auditory impairment (partial or total hearing loss, tinnitus) distinguishes it from vestibular neuritis. Examination reveals a harmonious peripheral vestibular syndrome. The absence of neurological signs is the rule.
Audiometry is useful for assessing the severity of hearing loss. 1 Vestibular system investigations are not indicated in the acute phase of the disease. However, at a later stage, they may be useful for assessing residual balance disorders and long-term compensation. 1
In the acute phase of tympanogenic labyrinthitis, high resolution CT (HRCT) scan often reveals middle ear and mastoid opacification. 5 Bone erosion may or may not be visible, depending on the severity of bone lysis. Even if bone lysis is visible, it clearly underestimates the extent of the disease. In our patient, temporal bone CT images showed only middle ear and mastoid opacification. A few weeks after suppurative labyrinthitis, an HRCT scan may show a subtle thickening of the spiral lamina and a slight increase in density within the semicircular canals related to granulation tissue formation in the fibrous stage of labyrinthitis. These bony proliferative changes may end in total obliteration of the cochlea (ossifying labyrinthitis). In the acute phase, the gadolinium-injected MRI shows labyrinth enhancement. This enhancement is explained by accumulated gadolinium in the inflamed labyrinthine membranes as the labyrinthine vasculature is disrupted. 6 This enhancement is particularly noticeable in the subacute phase. Thus, on gadolinium-weighted T1 sequences, labyrinthitis is reflected by a slight diffuse enhancement of the membranous labyrinth. This labyrinth enhancement may be total or segmental. This enhancement is to be distinguished from intralabyrinthine schwannoma, which is segmental and strongly enhanced. 6
Pre-contrast T1 images are acquired to distinguish labyrinthitis enhancement and hemorrhage. 7 In the case of hemorrhage, the low signal intensity is replaced by a spontaneous high intensity due to methemoglobin. 7
Later, in the event of progression to labyrinthitis ossificans, which is not uncommon in suppurative labyrinthitis, T2 fast spin echo images show the loss of normal hyperintensity of fluid in the membranous labyrinth. 8
Treatment of labyrinthitis is based on the administration of intravenous probabilistic antibiotics and effusion drainage. 9 Germ-specific antibiotics are then administered according to culture and sensitivity results. Adjuvant steroids seem to have a statistically-significant reduction in subsequent hearing loss. 1 Benzodiazepines and antihistamines can be used to treat initial vertigo for a short time, so as not to delay compensation. 1 Vestibular rehabilitation is recommended to be started as soon as the severe symptoms disappear.
If treated in time, serous labyrinthitis can be cured with few after-effects. If left untreated, it progresses to purulent labyrinthitis. The functional prognosis of purulent labyrinthitis is disappointing, with definitive cophosis and a classic progression to ossifying labyrinthitis. 10
Conclusion
Labyrinthitis is increasingly rare in the era of antibiotic therapy. P. aeruginosa is an exceptional germ in this context. MRI is an indispensable tool for the positive and differential diagnosis of labyrinthitis and for assessing its extent.
Early and appropriate treatment is the best guarantee to avoid the progression of serous labyrinthitis into purulent labyrinthitis with a poor prognosis.
Footnotes
Acknowledgements
There were no acknowledgments to mention.
Author Contributions
All authors made a substantial contribution to the acquisition, and analysis and interpretation of data in addition to writing—review and editing. All authors approved the final version.
Availability of Data
The data underlying this case report will be shared on request to the corresponding author.
Consent
Written informed consent was obtained from the patient’s family to publish this case report and accompanying images. On request, a copy of the written consent is available for review by the Editor-in-Chief of this journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethics approval for reporting individual cases or case series. We anonymously reported clinical and imaging information concerning our patient’s case.
