Abstract
Introduction
Head trauma is one of the most common causes of olfactory loss. 1 The mechanisms of traumatic olfactory loss included shearing of the olfactory nerves at the cribriform plate and injury to the central olfactory system. 2 While approximately one-third of patients with traumatic olfactory loss may recover spontaneously, the prognosis of traumatic olfactory loss is generally poor.3,4
The treatment of traumatic olfactory loss does not have a consensus. 5 Several drugs, including steroids and zinc, have been tried to treat traumatic olfactory loss; however, their effects are still controversial.4,6 Treatment involving a high-dose course of oral prednisolone has resulted in improved olfactory thresholds in 16.4% of patients suffering from traumatic olfactory loss. 7 When treated with a high-dose course of oral prednisolone in combination with zinc tablets, the olfactory threshold improved in 28.2% of victims suffering from this condition. 8 Olfactory training has been used to treat various types of olfactory loss.9-11 It has been reported to have a mild effect on improving PEA thresholds when treating traumatic olfactory loss.12-14
Recently, medical personnel have been injecting platelet-rich plasma (PRP) into the olfactory cleft of patients to treat olfactory dysfunction related to coronavirus 2019 (COVID-19), reporting it to be safe with promising results in several small-cohort studies.15,16 PRP is a biological product defined as being a volume of the plasma fraction of autologous blood having a platelet concentration above baseline. 17 It contains growth factors such as TGF-beta, EGF, VEGF, NGF, and IGF, and have been considered as having anti-inflammatory and pro-regenerative properties.18,19 Hyaluronic acid (HA) is a high molecular weight glycosaminoglycan that serves as a backbone for proteoglycans in the extracellular matrix. 20 It has a viscosupplementation effect and can possibly induce the PRP release of growth factors.21,22 Therefore, it has been reported that PRP and HA combination therapy holds the potential to produce superior outcomes when compared to PRP therapy alone. 20 The purpose of this study was to investigate the effect of PRP and HA nasal injections in the treatment of traumatic olfactory loss.
Patients and Methods
Patients
In this study, we collected patients who lost olfactory function after an episode of head injury and whose brain magnetic resonance imaging (MRI) results showed damage to their olfactory bulbs and adjacent regions. These patients who received nasal injections of PRP and HA for their treatment between August and December of 2023 were enrolled.
Prior to the nasal injection, each patient’s olfactory function was evaluated using both birhinal, right as well as left, and unirhinal phenyl ethyl alcohol (PEA) odor detection threshold tests. The mechanisms, procedures, and possible adverse effects of the PRP and HA nasal injections were explained to the patients. After injection, patients were followed up 1 and 3 months later to evaluate both the effects and side effects of PRP and HA nasal injections. At 1 and 3 months after nasal injection, patients were then asked whether they felt their olfactory function had improved, with both birhinal, right as well as left, and unirhinal PEA odor detection threshold tests being performed once again to measure the change in PEA thresholds. At 3 months after nasal injection, a traditional Chinese version of the University of Pennsylvania Smell Identification Test (UPSIT-TC) was employed to assess the effect of PRP and HA nasal injections on the identification ability. The Institutional Review Board (III) of Taichung Veterans General Hospital approved this retrospective work on May 15, 2024 (Protocol code CE24227C).
PRP and HA Nasal Injections
Preparation of the PRP and HA injection fluid
PRP and HA injection fluids were prepared using a Cellular Matrix® A-CP-HA Kit (Regen Lab SA, Le Mont-sur-Lausanne, Switzerland). A Cellular Matrix® A-CP-HA tube contains 2 ml of natural, non-crosslinked HA, at a concentration of 20 mg/ml (40 mg total), in addition to a thixotropic cell-separation gel and sodium citrate anticoagulant solution. This tube device isolates RegenPRP® with a high platelet, low granulocyte count, and minimal red blood cells in a closed-circuit system. Initially, 6 cc of whole blood was drawn from the patients being injected into a centrifuge tube. The centrifuge tube was inverted 5 times to mix the whole blood with the sodium citrate anticoagulant solution. The tube was then placed in a centrifuge and centrifuged at 3430 rpm for 5 minutes. During centrifugation, the separator gel moved to form a barrier that separated the red blood cells from other blood components, with the HA migrating to the top of the gradient (Figure 1). The tube was subsequently inverted 20 more times to mix the PRP and HA before finally 5 cc of the mixed fluid was drawn out using an empty syringe (Figure 2). The combination of PRP and HA fluids was now ready for injection.

After centrifugation, the red blood cells were separated from other blood components, and the hyaluronic acid migrated to the top of the gradient.
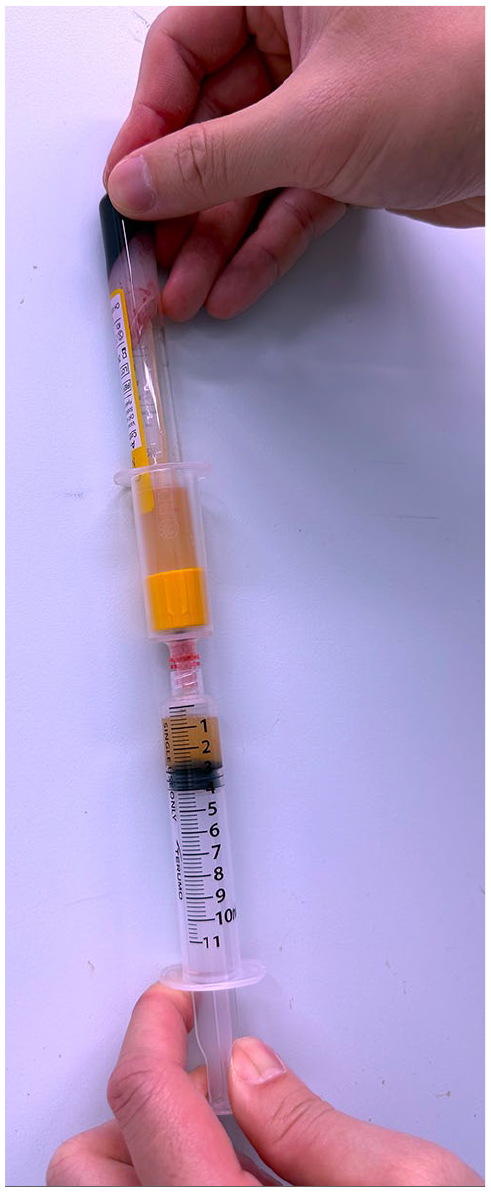
The mixture of platelet-rich plasma and hyaluronic acid fluid was drawn out using an empty syringe.
Injection procedures
The PRP and HA nasal injections were performed under local anesthesia with the patient being placed in the supine position. Under nasal endoscopy, the nasal cavities were packed with cotton pledgets soaked with a 2% xylocaine/epinephrine solution. After removing the pledgets, a 2% xylocaine/epinephrine solution was injected into both the upper middle margin of the nasal septum and the medial surface of the middle turbinate. Then, an elevator was used to lateralize the middle meatus to expose the olfactory cleft. Finally, about 1 cc of the PRP and HA fluid mix was injected into the right and left upper middle region of the nasal septum (Figure 3), and about 1 cc of the mix was injected into the medial surface of the right and left middle turbinates (Figure 4). After nasal injection, the patient was put also on olfactory training.
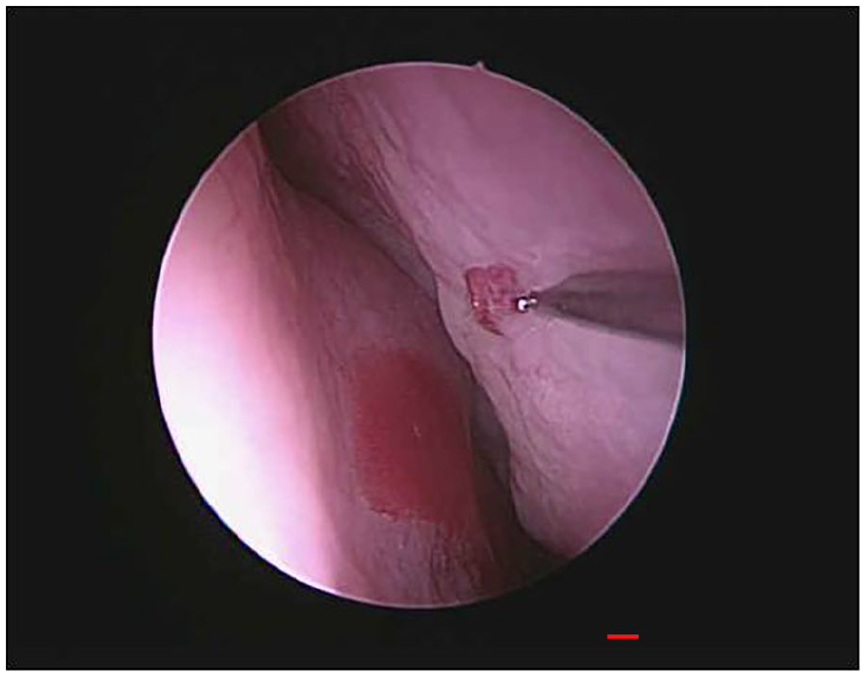
The mixture of platelet-rich plasma and hyaluronic acid fluid was injected into the upper middle region of the nasal septum.
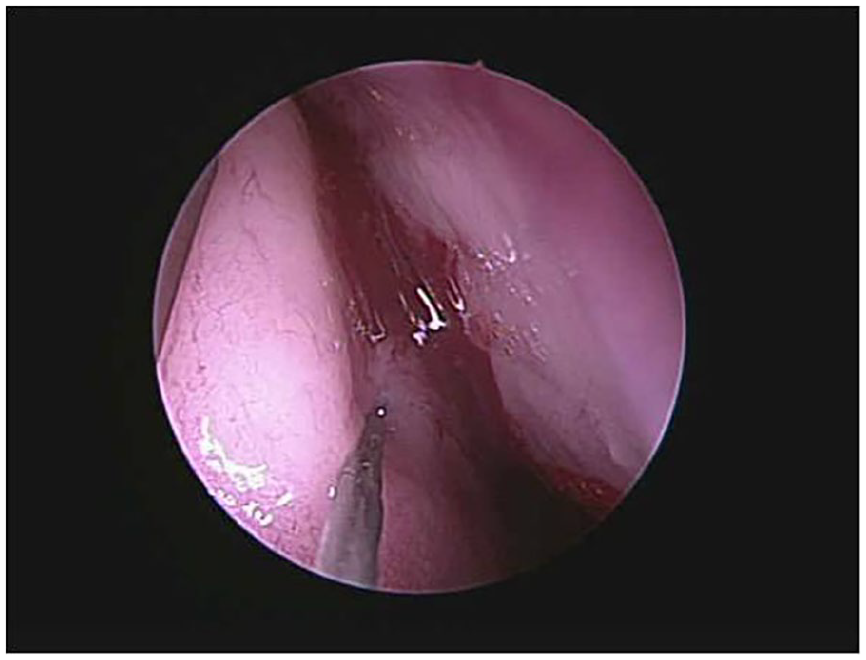
The mixture of platelet-rich plasma and hyaluronic acid fluid was injected into the medial surface of the middle turbinate.
Smell Tests
The testing procedures of the PEA odor detection threshold test and UPSIT-TC have been described in our previous article.14,23 The PEA concentrations ranging from 10−1 to 10−9 log vol/vol in half-log concentration steps were used in this study, and the PEA thresholds of the bilateral nostril, and right as well as left unilateral nose, were measured separately. The UPSIT-TC score ranged from 0 to 40. In this study, only the UPSIT-TC score of the bilateral nostril was acquired at 3 months after the mixed PRP and HA nasal injections.
MRI
Structural MRI with an olfactory bulb (OB) sequence has been suggested to be included in the diagnostic evaluation of traumatic olfactory loss. 24 In this study, MRI was done using a 1.5-Tesla Exite MRI system (GEMS, Milwaukee, WI, USA) with a quadrature head coil. Routine imaging pulse sequences consisted of axial T1-weighted images and FLAIR images, along with axial and coronal T2-weighted fast spin-echo images. Contrast-enhanced T1-weighted images with axial and coronal sections were acquired. After a sagittal localizing scan, 2- to 2.5-mm-thick T2-weighted coronal and sagittal (both TR = 5000 ms, TE = 106 ms, NEX = 2, Matrix = 256 × 256) images without an interslice gap were obtained with a 12-cm field of view. The pictures of MRI in 2 patients are shown in Figure 5. OB volumes were calculated using Mimics Medical 21.0 (Materialise, Leuven, Belgium).
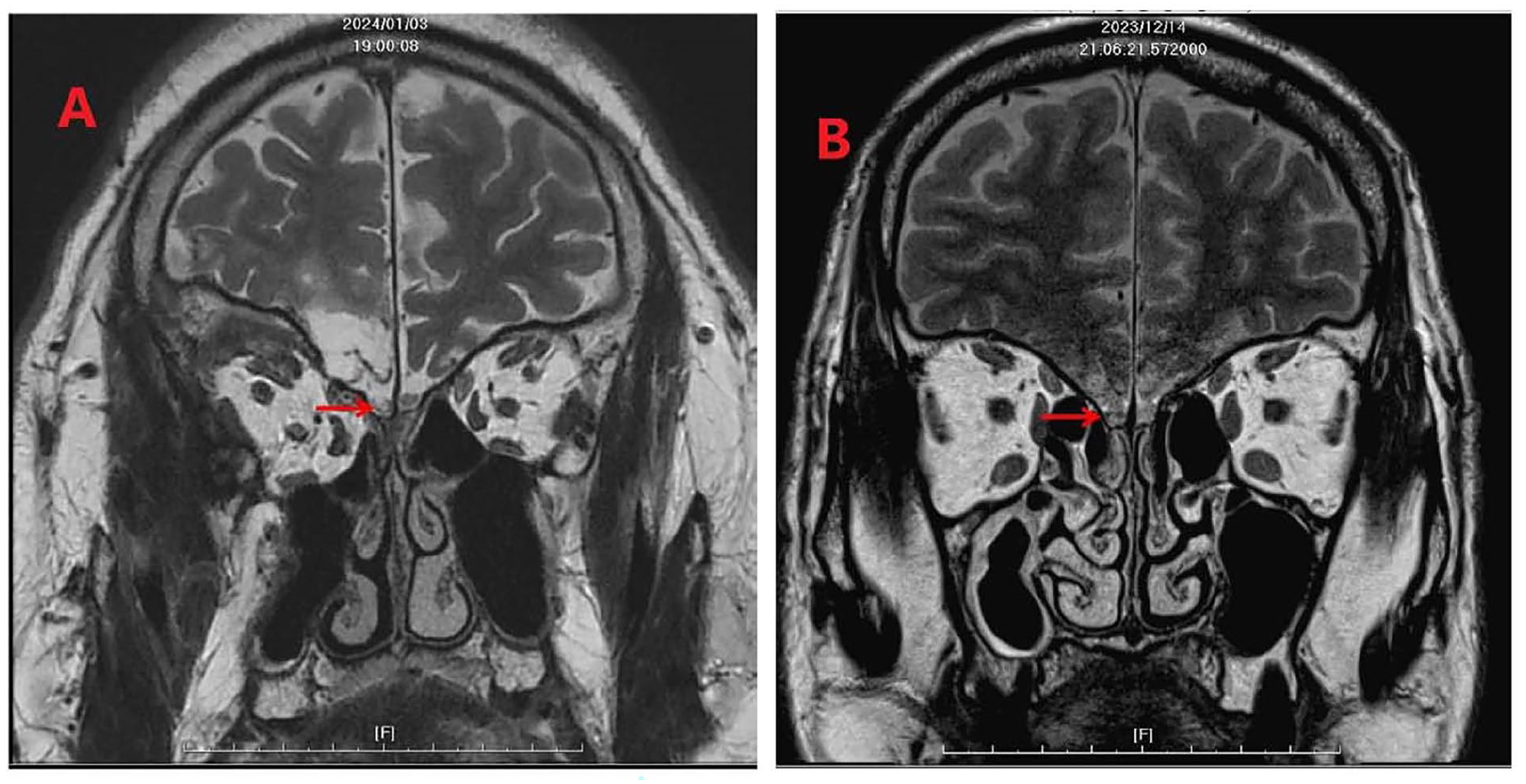
(A) A red arrow indicated the injured olfactory bulb in patient 9. (B) A red arrow indicated the injured olfactory bulb in patient 10.
Statistical Analysis
The OB volume was presented as mean ± standard deviation. The Mann-Whitney U test was used to compare the OB volumes between the nostrils with improved PEA thresholds at 3 months after the PRP and HA nasal injections and those with the same or increased PEA thresholds at 3 months after PRP and HA nasal injections. All analyses were performed using IBM SPSS for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Two-tailed P-values <.05 were considered statistically significant.
Results
Patients
Thirty-one patients were collected in this study, although 3 did not return for follow-up. Among the remaining 28 patients, 14 were males and 14 were females, with ages ranging from 20 to 55 years at a mean of 31.5 years.
The interval from the date of head trauma to the date of receiving PRP and HA nasal injections ranged from 4 to 167 months with a mean of 26.9 months. All patients had taken a course of prednisolone with a zinc gluconate tablet and performed olfactory training. The PEA thresholds measured just prior to their PRP and HA nasal injections are shown in Tables 1 to 3.
Birhinal PEA Threshold and Post-NI TC-UPSIT Score.

Abbreviations: NI, nasal injection; PEA, phenyl ethyl alcohol; UPSIT-TC, traditional version of University of Pennsylvania Smell Identification Test.
Right Unirhinal PEA Threshold.

Abbreviations: NI, nasal injection; PEA, phenyl ethyl alcohol.
Left Unirhinal PEA Threshold.

Abbreviations: NI, nasal injection; PEA, phenyl ethyl alcohol.
Olfactory Function 1 Month After PRP and HA Nasal Injections
One month after being given the PRP and HA nasal injections, 20 of the 28 (71.4%) patients felt that their olfactory function had improved. PEA tests revealed that the threshold of the bilateral nostrils decreased in 9 patients, with that of the right nostril decreasing in 11 and that of the left nostril decreasing in 9 (Tables 1-3). In total, either the bilateral or unilateral PEA threshold decreased in 22 (78.6%) patients.
Olfactory Function 3 Months After PRP and HA Nasal Injections
Three months after the patients had received PRP and HA nasal injections, another 4 patients reported that their olfactory function had improved. Therefore, in total, 24 of 28 (85.7%) patients felt that their olfactory function had improved at 3 months after receiving nasal injections. The PEA test results show that the threshold of bilateral nostrils decreased in 6 patients as compared with their thresholds pre-injection, that of the right nostril decreased in 9, and that of the left nostril decreased in 7 (Tables 1-3). In total, bilateral or unilateral PEA thresholds decreased in 15 (53.6%) patients. In addition, the bilateral UPSIT-TC scores at 3 months after the nasal injections revealed that 1 patient was normosmic, 11 were hyposmic, and 16 were anosmic based upon our olfactory diagnosis for UPSIT-TC (Table 1). 25
Adverse Effects and Safety Profile
Since the injection was performed under local anesthesia, patients were asked about the pain severity of the procedure. Among 28 patients, 20 considered that the procedures were painful but tolerable, and the remaining 8 answered that the procedures were mildly painful. After completing nasal injections, the patient was observed for possible adverse effects over a half-hour period. Mild epistaxis was observed in all patients but it stopped within half an hour. No patient complained of headaches or had visual problems. At 2 follow-ups after the nasal injection, no adverse effect of the PRP and HA nasal injection was reported in any patient.
OB Volumes
At 3 months after receiving their PRP and HA nasal injections, the PEA threshold in patients decreased in 9 right nostrils and 7 left nostrils as compared with the thresholds pre-injection. The OB volumes in these 16 nostrils ranged from 5.58 cm3 to 77.82 cm3 with a mean (standard deviation) of 32.98 (21.329) cm3. The OB volumes in the remaining 40 nostrils which did not experience decreased PEA thresholds ranged from 4.81 cm3 to 68.97 cm3 with a mean (standard deviation) of 26.46 (20.297) cm3. There were no significant differences in OB volumes between those in the 16 nostrils with decreased PEA thresholds and those in the 40 nostrils without decreased PEA thresholds (P = .246).
Discussion
Recently, PRP injections into olfactory clefts have been used to treat COVID-19-related olfactory dysfunction. 16 A comprehensive review which included 6 studies involving adult patients who had been treated with PRP for olfactory dysfunction due to COVID-19 concluded that PRP was a promising and safe therapeutic option for olfactory dysfunction. 18 Head trauma is a common etiology of sensorineural olfactory dysfunction. To date, there has been no study involving the use of PRP to treat traumatic olfactory dysfunction. The combined PRP and HA injections have been considered as producing better outcomes in treating knee osteoarthritis when compared to PRP alone therapy. 20 HA offers the viscosupplementation effect which can most likely help injection fluid stay in the submucosa better. 21 Therefore, we tried to investigate the effect of PRP and HA nasal injections in the treatment of traumatic olfactory loss.
The prognosis surrounding traumatic olfactory loss is generally poor, with several modalities having been used to treat the condition. 4 In our previous studies, a 16.4% improvement rate in PEA thresholds was observed at 3 to 21.5 months (mean: 5.5 months) after treatment using a course of oral prednisolone, while a 28.2% improvement rate was acquired at 3 to 15 months (mean: 6.1 months) after combining zinc tablets with oral prednisolone.7,8 When olfactory training was used to treat these patients for 6 months, a 22.2% improvement rate in PEA thresholds was found. 14 In this study, 71.4% of patients felt that their olfactory function improved 1 month after receiving the combined PRP and HA nasal injections, with 85.7%of patients feeling that their olfactory function had improved 3 months after the injections. PEA threshold decreased in 78.6% of patients at 1 month after PRP and HA nasal injections, and in 53.6% of patients at 3 months after receiving injections. These results indicate that the PRP and HA nasal injections seemed to have both a fast and stronger effect in treating traumatic olfactory loss.
The side effects of PRP nasal injections have been reported in several studies in which PRP was injected into the olfactory clefts of patients experiencing COVID-19-related olfactory dysfunction. Studies from both Steffens et al and Yan et al reported no adverse reactions were elicited by PRP nasal injection.19,26 With regard to side effects, the study performed by Lechien et al mentioned several, including transient epistaxis and pain. 27 In this study, the most common complaint was pain caused by the procedure of lateralization of the middle turbinate to expose the olfactory cleft. Due to the multiple injections we performed, mild but transient epistaxis occurred in most patients. However, for the most part, the combination of PRP and HA nasal injections was well tolerated, with minimal adverse effects being felt in our patients.
Although our results show that the PRP and HA nasal injections were safe and effective in the treatment of traumatic olfactory loss, there were several limitations in our study. First, our study was not a randomized trial with a control group. Second, the olfactory function of our patients was only evaluated by the PEA odor detection threshold test. The UPSIT-TC was not performed before nasal injection and unilaterally because of the cost of the UPSIT-TC. Therefore, its effectiveness in treating traumatic olfactory loss was not conclusive. In addition, our patients were only followed up for 3 months after receiving their nasal injections; therefore, the long-term effect and safety of PRP and HA nasal injections in treating traumatic olfactory loss could not be assessed in this study. Moreover, there are several issues regarding PRP injections for the treatment of olfactory dysfunction. First, the techniques of injection have not yet been standardized. The injection sites and doses have been different among the different studies. Steffens et al injected 1 cc of PRP into the olfactory cleft. 19 Yan et al injected 1 cc of PRP at 2 sites within the olfactory cleft along the superior septum, posterior to the head of the middle turbinate. 26 Lechien et al injected 0.2 to 0.5 cc of PRP at several points in the middle turbinate and in the nasal septum. 15 The timing of the injection has also been considered an important factor influencing the effectiveness of PRP nasal injection in the treatment of COVID-19-related olfactory dysfunction. 18 However, it is still not known whether an earlier injection provided a better effect on the treatment of traumatic olfactory dysfunction. Treatment duration was also different between the different studies. The effect of a single injection was reported in most studies, although El Naga et al injected PRP every 3 weeks for a total of 3 injections. 28
Conclusions
Our results show that 71.4% of patients diagnosed with traumatic olfactory loss felt that their olfactory function had improved 1 month after receiving PRP and HA nasal injections, with 85.7% of patients feeling that their olfactory function improved 3 months after the injections. The PEA threshold decreased in 78.6% of patients at 1 month after the PRP and HA nasal injections, and in 53.6% of patients at 3 months after injections. This indicates that the PRP and HA nasal injections obtained favorable results and were well tolerated. However, randomized controlled trials having a longer follow-up period are still needed to confirm the effect and safety of PRP and HA nasal injections in the treatment of traumatic olfactory loss.
Footnotes
Consent for Publication
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Patient Consent
Not applicable.
Research Ethics and Patient Consent
This study was approved by the Institutional Review Board (III) of Taichung Veterans General Hospital (protocol code CE24227C, date of approval 2024/5/15).
