Abstract
Keywords
Introduction
Twenty to thirty percent of the population in developed nations suffers from an allergic illness. 1 Food and medication allergies, as well as asthma, rhinitis, atopic dermatitis, and anaphylaxis, are on the rise globally, particularly in countries with low- or medium-income levels. 2 Allergic disorders cause a wide range of symptoms, from relatively-minor ones to more severe and sometimes fatal anaphylactic reactions. 3
Natural and herbal remedies have long been used in human respiratory disorders because of their widespread availability and relative ease of use. Many have a history of safe and effective use, sometimes dating back thousands of years. 4 Antimicrobial, antioxidant, anti-inflammatory, and anti-proliferative activities have been demonstrated in propolis, one of the beehive products. Ancient peoples from all over the world, including the ancient Egyptians, Romans, Greeks, ancient Chinese, and many more in South and Central America, where beekeeping was commonplace, have relied on it for medicinal purposes.5-7
Propolis has shown promise as a treatment for respiratory viruses in in vitro investigations and animal studies. The most effective flavonoid derived from propolis, quercetin, displayed antiviral activity against human coronavirus OC43, according to one of the first studies conducted by Debiaggi et al. 8 In vitro, Refaat et al. 9 discovered that propolis neutralized Sars Cov-2, and the antiviral activity was enhanced when it was encapsulated in liposomes. Additionally, various compounds derived from propolis have been predicted in silico studies to bind and inhibit Sars-Cov2 proteins: Mpro, RNA-dependent RNA polymerase, and the spike protein.10-16 These compounds include withanone, withaferin-N, caffeic acid, CAPE, chrysin, galangin, myricetin, rutin, hesperetin, pinocembrin, luteolin, quercetin, kaempferol, brousssoflavonol F, and p-coumaric acid.
Khayyal et al. 17 showed that supplementation with propolis-enriched milk formula significantly reduced the severity and frequency of nocturnal asthma attacks. The reduction in asthma attacks correlated with decreased inflammatory markers such as TNFα, ICAM-1, IL-6, IL-8, leukotrienes, prostaglandins F2a, and E2.
Moreover, by reducing the expression of TGF-1β, ICAM-1, α-SMA, MMP-9, IgE, and IgG1, propolis reduces inflammations of neutrophils and eosinophils as well as epithelial-mesenchymal transition.16,17
This trial aimed to determine whether Rhinapi, a hypertonic saline nasal spray with propolis, alleviated allergic rhinitis (AR) symptoms.
Materials and Methods
We set out to conduct this investigation as a prospective, multicenter, observational study, following the guidelines in the Declaration of Helsinki. It extends our previous Turkish study to a varied European population and was conducted in a similar fashion. 18
The University of Oradea, Romania, obtained ethics committee approval (date: April 30, 2024; number: CEFMF/3). The patients were asked to verbalize their agreement.
Subjects
Over 400 participants, including 251 men and 189 females, with previously-diagnosed AR, were recruited from various centers throughout European nations between April 2024 and July 2024 for this study.
Symptoms of AR, including runny nose, sneezing, itching, and nasal blockage, were present in every participant in the study. The males had an average age of 32.84 ± 5.58 years, while the females had an average age of 32.72 ± 4.67. The otolaryngologists examined each patient. After that, for 3 weeks, they were given Rhinapi, Hypertonic Saline Nasal Spray with Propolis (Bee&You, Istanbul, Turkey)—one puff into each nostril, 3 times daily. Deionized water (98%), glycerin (0.1%), eucalyptus oil (0.1%), propolis extract (0.1%), and sodium chloride (1.7%) were the components of the nasal spray.19,20
Before and 3 weeks following Rhinapi treatment, patients’ total symptom scores, quality of life (QoL) scores, and results from the otolaryngology examination were assessed.
The Inclusion Criteria
Patients with previously-diagnosed AR who exhibited AR symptoms such as stuffy nose, runny nose, itching nose, or blocked nose.
Exclusion Criteria
The following exclusion criteria were considered: those who were pregnant, known AR patients who were asymptomatic at the time of the study, those who declined to take part in the research, and patients who were unable to understand or comply with the study.
Methods
Symptoms
All trial participants were asked to score their rhinitis symptoms, including runny nose, sneezing, itching, and nasal blockage, from 1 (very few symptoms) to 5 (very severe).
Total symptom scores, ranging from 1 to 20, were calculated by adding these above-mentioned scores.
Quality of Life
This was scored on a 10-point scale: A score of 1 meant the most excellent quality of life, while a score of 10 meant the worst.
In addition, the outcomes of an otolaryngological examination were used to rate the patients. Each turbinate’s color and the extent of edema were given a score between 1 and 5, with “natural,” “moderate,” “severe,” and “extremely” being the possible outcomes.
The Statistical Analysis
The analysis was conducted using the statistical software package SPSS for Windows 21.0, developed and owned by IBM (Chicago, IL, USA). We used the Kolmogorov-Smirnov test to see how well the data fit the normal distribution. Parametric and nonparametric tests were used to compare the groups. When comparing means according to distribution forms, the Student t-test was utilized, and when comparing scores before and after the intervention, the Wilcoxon signed-rank test was used. The Marginal Homogeneity test examined the cross tables that compared symptoms before and after therapy. These symptoms included nasal discharge, sneezing, itching, blockage, concha color, and congestion. Quantitative data were summarized using mean ± SD or median (Q1; Q3) statistics, while qualitative data were summarized using counts (%). For statistical purposes, a P-value below .05 was deemed significant.
Results
Patients
Of the 460 enrolled patients, 440 took the sprays as directed and came to the final control, but 20 were unavailable for the second assessment, so they were excluded from the study.
Symptom Scores
Table 1 shows the nasal discharge, sneezing, itching, and obstruction symptom ratings before and after medication.
Nasal Discharge, Sneezing, Nasal Itching, and Nasal Obstruction Scores at PreMedication and PostMedication Periods.
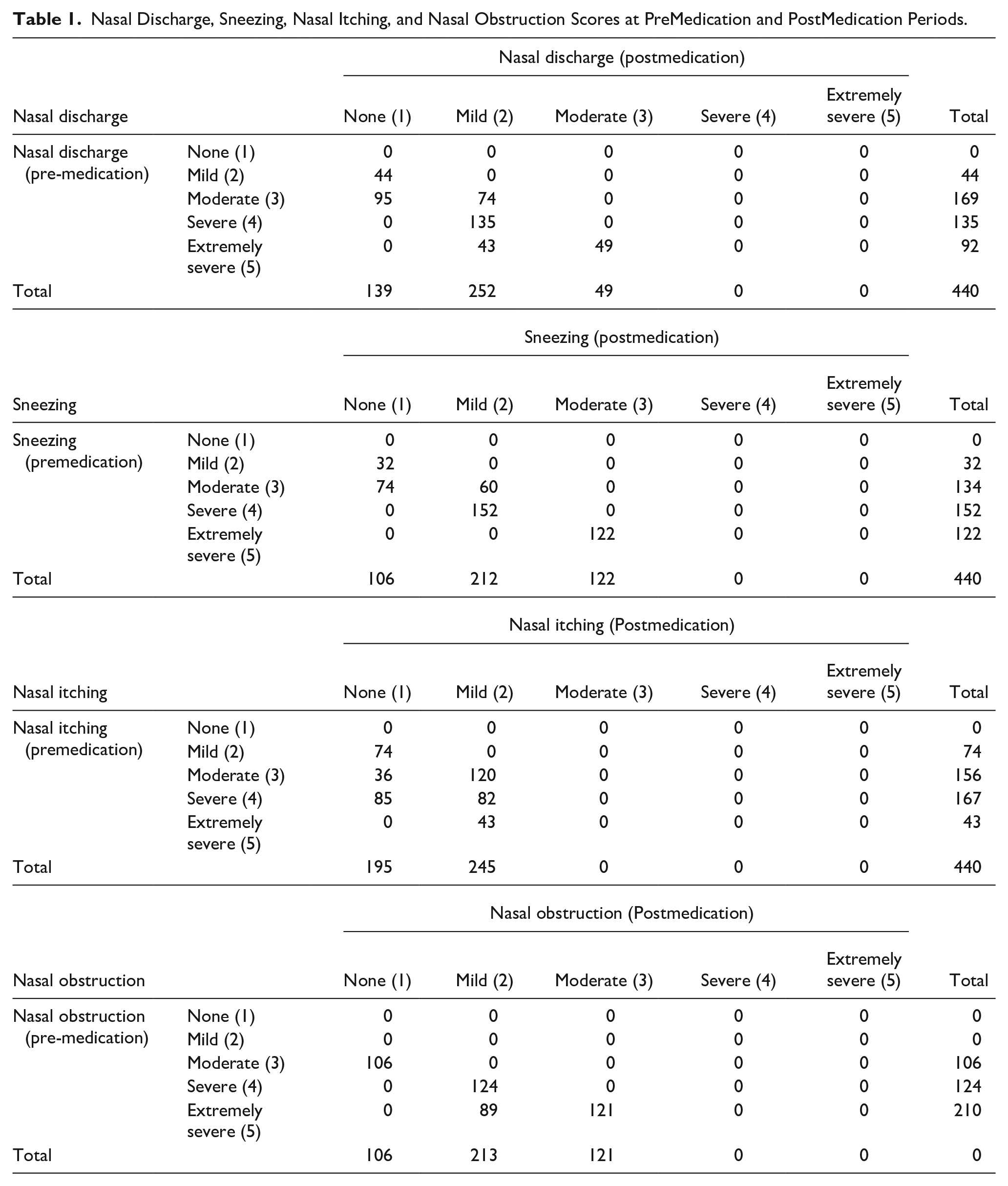
Using Rhinapi throughout the trial resulted in statistically-significant improvements in the following symptoms: nasal discharge (P < .001), sneezing (P < .001), nasal itching (P < .001), and nasal obstruction (P < .001), as shown in Table 1 from the Marginal Homogeneity test.
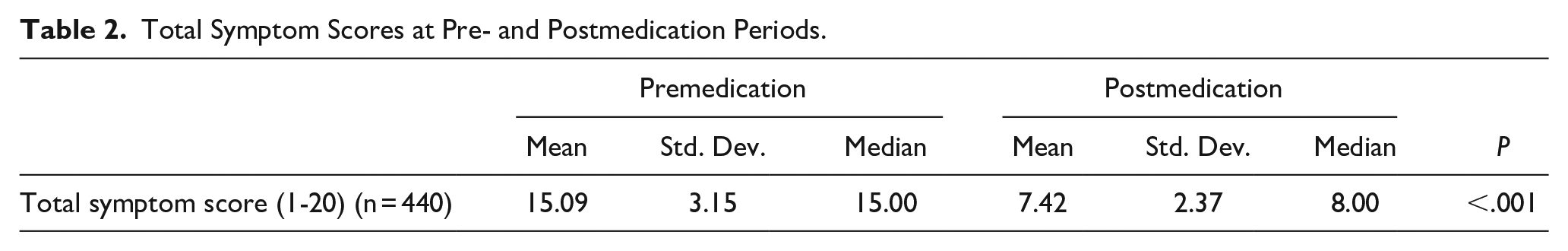
The total symptom and quality-of-life (QoL) scores before and after the medication are given in Tables 2 and 3.
Total Symptom Scores at Pre- and Postmedication Periods.
QoL Scores at Pre- and Postmedication Period.

Note. 1 is the worst, and 10 is the highest.
Total Symptom Score
A statistically-significant change (P < .001) was seen between the mean total symptom scores before and after medication. The average symptom score before treatment was 15.09 ± 3.15, while the mean postmedication score was 7.42 ± 2.37 (Table 2).
Quality of Life Scores
The mean QoL scores change between before and after medication was statistically significant (P < .001). The QoL score before medication was 4.97 ± 0.72; after medication, it increased to 8.04 ± 0.76 (Table 3).
ENT Examination
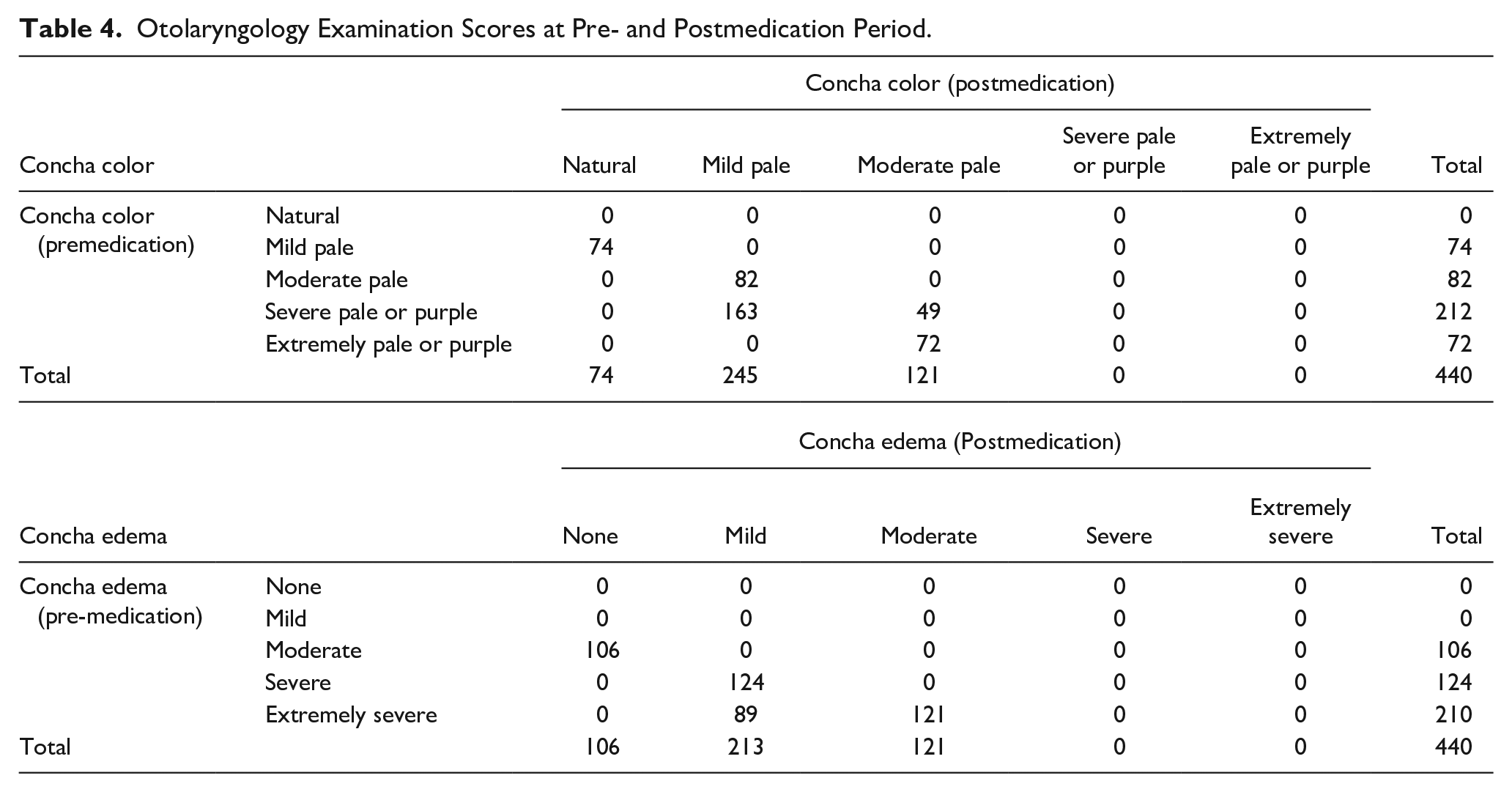
The results of the otolaryngological examination, both before and after the treatment, are displayed in Table 4 (P < .001).
Otolaryngology Examination Scores at Pre- and Postmedication Period.
Discussion
The rising incidence of allergic disorders is a significant concern for public health around the world. Clinical care of allergic disorders is complicated by issues such as the cost and underuse of effective therapies such as intranasal corticosteroids. Many patients suffering from long-term allergies incorrectly consider complementary and alternative medicine (CAM) options to be safer.2,21
In order to explore the possibilities of this approach, we decided to study propolis, a natural resinous substance made of bees. Bees create propolis, called “bee glue,” to defend their hives and fight off illnesses. More than 500 chemical components have been discovered in propolis, including flavonoids, terpenoids, phenolics, and their esters. 22 Propolis has been traditionally used to treat asthma, diabetes, ulcers, and burns. 21 In addition, multiple reviews have shown that propolis has potent anti-inflammatory, antibacterial, and anticancer effects.23-25 While propolis’ antioxidant, antibacterial, and anticancer properties are well-known,17,26-29 its anti-allergic potential has not been thoroughly investigated.20,30
In systemic and local anaphylactic animal models, propolis-derived chemicals such as CAPE, pinocembrin, and chrysin act against IgE-mediated allergy. The bioactive compounds found in propolis lower histamine levels in the bloodstream, which in turn reduces the activation of NF-κB, the release of platelet-activating factor and cys-leukotrienes, as well as the expression of TNF-α, IL-1β, and Th2-related cytokines such as IL-4, IL-5, IL-6, and IL-13. As a result, the reaction to IgE antibodies is drastically reduced.31-36 Itching, nasal rubbing, sneezing, and watery rhinorrhea are some of the allergic symptoms that can be alleviated in animal models by lowering levels of specific components, pro-inflammatory cytokines, and the IgE antibody response.31,32,36
Over 4 weeks, the in vivo effects of granular Brazilian propolis from Okayama, Japan's Yamada Apiculture Center Inc, on symptoms of allergic rhinitis, including sneezing and nasal rubbing, were studied by Shinmei et al. The study showed that nasal rubbing and sneezing in OVA-sensitized mice were unaffected by a single oral administration of propolis therapy at 200, 500, and 1000 mg/kg doses. Mice treated with 1000 mg/kg of propolis showed a steady improvement in symptoms after 4 weeks, with a marked decrease in sneezing and nasal rubbing. Mice given Tranilast orally at a dose of 300 mg/kg for 3 weeks showed a marked reduction in sneeze and nasal rubbing, and the effects of propolis were comparable to those of a mast cell stabilizer. Propolis does not suppress IgE synthesis since neither a single nor repeated therapy administration reduces blood IgE levels. Also, nonsensitized mice stimulated with histamine to develop allergic rhinitis showed no improvement after receiving propolis treatment, suggesting that propolis did not inhibit histamine’s effect.
Conversely, when mast cells were stimulated with antigen or compound 48/80, an ethanol extract of green Brazilian propolis derived from Brazilian propolis granule reduced histamine release. Hence, rather than inhibiting histamine activity, the therapeutic effects of propolis on allergic rhinitis symptoms are probably attributable to the reduction in histamine release by activated mast cells.37,38 Although Brasilian and Anatolian propolis have similar structures, they also have different compounds, which may alter their efficacy on histamine activity.
Animal studies have shown that propolis and its bioactive components, including tectochrysin and CAPE, significantly lower levels of various immune system markers, including α-SMA, MMP-9, MDA, ROS, IgE, IgG1, TNF-α, and cytokines associated with Th1 and Th2 including IFN-γ, IL-5, IL-6, and IL-10. In addition to downregulating the Th2 immune response, propolis increases the expression of glutathione-S-transferase, Treg differentiation, and polymorphonuclear myeloid-derived suppressor cells (PMN-MDSC).39,40 The active mechanism supporting these effects should be investigated with further animal and clinical studies.
Propolis has bactericidal and virucidal effects. It may also prevent viral and bacterial infections from attaching by blocking proteins that pathogens use to adhere to and enter host cells. In addition to blocking pathogens from interacting with host cells, propolis can bind to proteins on the host's surface. On the other hand, according to Corbo et al.’s. 41 experimental results, propolis can be expected to effectively stick to the mucous lining of our air passages, which also has lipophilic properties.
The ability of propolis to induce a layer of external zone water gives propolis-functionalized surfaces particle-repellent properties. This ability stems from the abundance of hydrophilic functional groups in all propolis samples, regardless of specific variations in chemical composition. Due to the physical nature of the exclusion mechanism, propolis’ action should be very generic rather than particle (or pathogen or allergen)-specific. Indeed, external zone water has been recognized to exclude a variety of solutes, starting with simple salt. As a result, any microbial or allergen entry may be prevented by a physical barrier in the form of a propolis-generated external zone water layer. This way, propolis could act as the first line of defense against respiratory pathogens and allergens. 42
In a study looking into the effects of poplar propolis on allergic rhinitis, Yasar et al. 38 used honeybees from Turkey’s Apis mellifera caucasica species. 38 Oral and intranasal administration of propolis ethanol extract was compared for their anti-allergic effects. Oral propolis alleviated symptoms (nasal irritation, sneezing, and volume of nasal discharge) more than intranasal propolis in lowering inflammation, vascular proliferation, and goblet cell counts. 38 Significantly, compared to 50 µg intranasal mometasone furoate (a steroidal drug) and 10 mg/kg oral ketotifen (an antihistamine), the inhibitory effects of 200 mg/kg oral propolis on inflammation, goblet cell counts in nasal mucosa, and symptom scores were more pronounced, indicating that poplar propolis, which is produced by Apis mellifera caucasica honeybees, could be used orally to treat allergic rhinitis. 38
After using the Rhinapi nasal spray, participants in the study by Cingi et al. 18 reported less severe symptoms and improved quality of life scores. Results show that Rhinapi (Bee&You’s Anatolian propolis and hypertonic saline combination nasal spray) lessens overall symptom scores, boosts quality of life, and reduces the severity of observed nasal inflammation. However, the study by Cingi et al. 18 was carried out in various centers of the same country, whereas this study was performed in various European countries from different origins. 18
This trial aimed to determine whether Rhinapi, a hypertonic saline nasal spray with Anatolian propolis, alleviated AR symptoms. According to our data, using Rhinapi nasal spray considerably reduced AR symptom scores, which include nasal discharge, sneezing, itching, and blockage. Rhinapi nasal spray dramatically-improved quality of life and total symptom scores. Nasal spray infused with propolis reduced swelling and enhanced concha color. However, this was an open, unblinded study, and further clinical studies are needed before propolis can be recommended widely for human use in AR. 18
Basista and Filipek. 43 performed a survey study in Poland and found that between 3.5% and 7.6% of beekeepers experienced propolis allergies. Atopic dermatitis, allergic rhinitis, eczema, and other similar conditions were the clinical manifestations of these reactions. Recently, Zhang and Yan. 44 reported a case where a patient with contact stomatitis due to an allergic response was treated with propolis throat candies.
Conclusion
In an open study, QoL and AR symptom scores improved after 3 weeks of treatment with Rhinapi, a propolis-added hypertonic saline nasal spray (Bee&You, Istanbul, Turkey). The concha color and edema also improved. Further studies are needed to define the place of propolis in AR therapy.
Footnotes
Author Contributions
The authors contributed in planning, designing, literature survey, data collection, interpretation of the results, active intellectual support, and English editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bee&You sponsored this study. However, Bee&You did not solicit this research project or protocols with the investigators or the institution and was not responsible for managing the study, data processing, or reporting.
Ethics Committee
Ethics committee approval was obtained from Oradea University, Medical Faculty, Ethics Committee (Date: April 30, 2024; Number: CEFMF/3).
Informed Consent
Verbal consent was obtained from the patients.
