Abstract
Introduction
The worldwide prevalence of allergic disorders is steadily increasing, along with the health burden they impose. Patients suffer from allergic symptoms for prolonged periods, a situation that has raised the question of how complementary and alternative medical approaches may be of value in long-term clinical management. Propolis is in current use as an agent that may be administered for prolonged periods and offers both prophylactic and therapeutic benefits to allergy sufferers. 1
Propolis itself is a naturally occurring resin that bees produce. Although propolis has been demonstrated to exhibit antioxidant, anti-microbial, and anti-neoplastic benefits, its value in treating allergic disorders has not been comprehensively investigated so far. There have been a number of preclinical studies undertaken, however, where it was concluded that the extract of propolis decreases inflammation due to allergy and offers potential therapeutic benefits in various allergic disorders, such as allergic rhinitis, asthma, allergic eczema, and food allergies. It appears that these effects are, to some degree, the result of inhibition of mast cell and basophil activation. 1
Propolis forms a hydrophobic, balsam-yielding resin, which is only minimally absorbed by the body and has low bioavailability. The typical components of propolis are resinous and balsamic compounds, beeswax, fatty acids of essential and aromatic types, pollen, and variable amounts of organic matter. There are a multitude of different constituents, and the precise components depend both on where the bees produced them and what species of plants they fed on. Studies have identified hundreds of different compounds typically found in propolis. The bioactive components of propolis are likely to be phenol-containing molecules, such as the flavonols quercetin and galangin and the flavone, chrysin. In addition, compounds related to hydroxycinnamates, such as caffeic, cinnamic, and p-coumaric acid, also likely possess bioactivity. There are also several other classes of molecules typically found in propolis, namely aromatic aldehydes, di- and sesqui-terpenes, esters, lignans, alcohols, amino acids, aliphatic acids, vitamins, and minerals.2-6
There are multiple biological effects of propolis. Currently, the most extensively evaluated aspects are its antimicrobial, anti-inflammatory, and anti-neoplastic effects, as well as its role as an antioxidant and modulator of the immune system. 7 There is a history of varying applications of propolis in ethnomedicine. 8 Since propolis helps to prevent putrefaction, it was employed in Ancient Egypt for mummification. It was also used in the Greco-Roman period for treating trauma since it prevented infection and encouraged scar formation. Texts from Ancient Iran indicate it was employed for the treatment of allergic dermatitis, muscular aches, and joint pains. It was also utilized by the Incas as a way to control fever.2,9
The way in which propolis exerts an antimicrobial effect is still not fully understood. There may be an effect on cell walls, resulting in loss of function and structural deficits for the microbe. The molecules responsible for these effects are likely to be the flavone-like compounds, caffeic, benzoic, or cinnamic acid. 10 A study by Arslan et al. 11 noted an antimicrobial effect of propolis against a fungal organism (Candida albicans) and a bacterial pathogen (Enterococcus faecalis). It has greater potency against the fungus than the bacterium at lower concentrations. When propolis was extracted using ethanol, the extract was able to powerfully inhibit biofilm formation by bacteria of the Pseudomonas and Staphylococcus genera, even when the concentration was a mere 0.05 mg/ml, as shown by Helaly et al. 12
Currently, propolis has achieved popularity as a dietary supplement. At the time of the COVID-19 pandemic, sales of dietary supplements thought to reduce infection increased notably. Anecdotal evidence suggests that allergic symptoms also diminished in patients using a propolis supplement.
This study has the objective of evaluating how the use of the propolis-containing Rhinapi nasal spray impacts the severity of symptoms related to allergic rhinitis.
Materials and Methods
The present study was undertaken as a prospective, multicenter, observational study in accordance with the principles outlined in the Declaration of Helsinki. Ethical approval was obtained from Istanbul Medipol University GETAT Clinical Research Ethics Committee on July 12, 2021, by decree number 25. Verbal consent was obtained from the patients.
Subjects
For this study, some 10,000 individuals (consisting of 5028 male and 4972 female patients) were enrolled from different centers in different regions of Turkey between March 2022 and March 2023. All trial participants exhibited symptoms of allergic rhinitis, such as nasal discharge, sneezing, nasal itching, or nasal obstruction. The mean age for participants was 36.83 ± 12.26 years (range: 17.00-73.00 years). All patients underwent an otolaryngological examination. They were then administered Rhinapi, Propolis-Added Hypertonic Saline Nasal Drops (Bee&You, Istanbul, Turkey) one puff to each nostril, three times a day, for a period of 3 weeks. The nasal spray contained the following ingredients: deionized water (98%), glycerin (0.1%), eucalyptus oil (0.1%), propolis extract (0.1%), and sodium chloride (1.7%).13,14
Total symptom scores, quality of life (QoL) scores, and otolaryngology examination scores were evaluated before and 3 weeks after treatment with Rhinapi.
Data were also gathered on the occupational status of the trial participants. They were then classified into one of three groups based on this information: Group 1, consisting of homemakers or hybrid office workers; Group 2, consisting of office-based workers or students; and Group 3, representing gardeners, manual laborers, or technicians.
Inclusion Criteria
Patients with symptoms of nasal discharge, sneezing, nasal itching, or nasal obstruction
Exclusion Criteria
The subjects were without symptoms of nasal discharge, sneezing, nasal itching, or nasal obstruction.
The subjects who did not want to participate in the study.
Methods
All trial participants filled in surveys that enquired about symptoms of allergic rhinitis (nasal discharge, sneezing, nasal itching, nasal obstruction). A score of 1 indicated minimally symptomatic, ranging up to the maximum severity, represented as 5. In the same manner, total symptom scores were also obtained, giving a score of between 1 and 15.
For the QoL score, 1 indicated the lowest quality, whereas 10 indicated the highest quality.
Furthermore, the patients were scored according to the results of an otolaryngological examination. The color of the turbinates was scored from 1 to 3, as was the degree of edema present in the turbinates.
Statistical Analysis
The analysis was undertaken using the IBM SPSS for Windows 21.0 statistical software application (SPSS Inc., an IBM Company, Chicago, IL, USA). The degree to which the data obtained corresponded to a normal distribution was assessed using the Kolmogorov-Smirnov test. For quantitative data, the mean ± standard deviation (SD) was calculated. Qualitative data items were expressed as percentages (%).
In comparing groups using data with a normal distribution, the independent samples t-test was utilized.
One-way ANOVA (Analysis of variance) was utilized in group comparisons and Tukey’s post hoc test was employed for multiple comparisons.
To assess cross-tabulated results for the symptom scores for nasal discharge, sneezing, itching, and nasal obstruction, before and after medication, the Marginal Homogeneity test was used. Any relationship between age and symptomatic score was determined via the Pearson correlation coefficient.
The level taken to indicate statistical significance was set for the study as a P-value below .05.
Results
The symptom rating scores for nasal discharge, sneezing, nasal itching, and nasal obstruction both pre- and post-medication are listed in Table 1.
Nasal Discharge, Sneezing, Nasal Itching, and Nasal Obstruction Scores at Pre-Medication and Post-Medication Period.
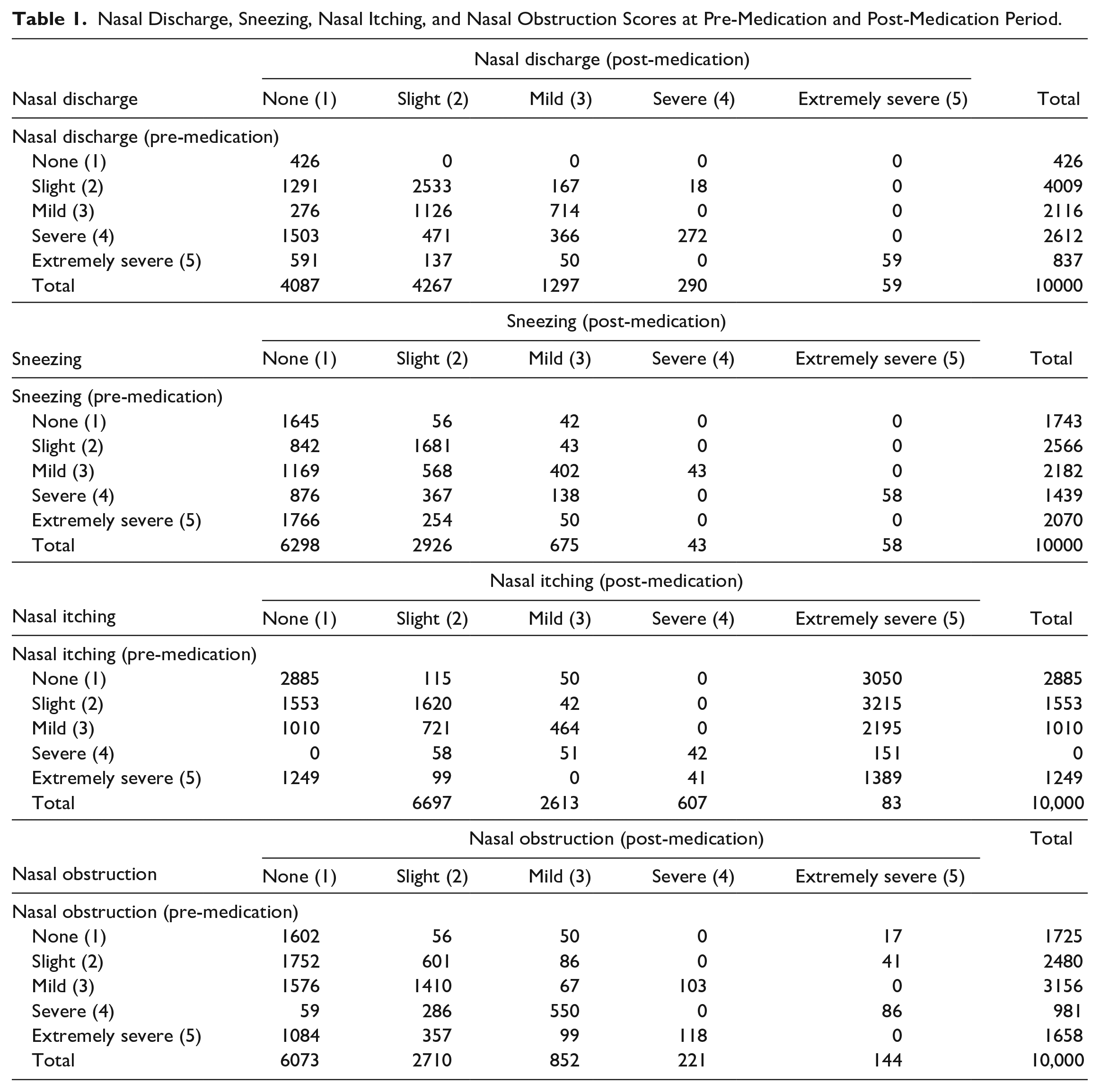
The scores for nasal discharge (P < .001), sneezing (P < .001), nasal itching (P < .001), and nasal obstruction (P < .001) all demonstrated statistically significant improvement following the use of Rhinapi for the study period, as indicated by the Marginal Homogeneity Test (Table 1).
Total symptom score and QoL scores both pre- and post-medication periods are shown in Table 2.
Total Symptom Score and QoL Scores at Pre- and Post-Medication Period.
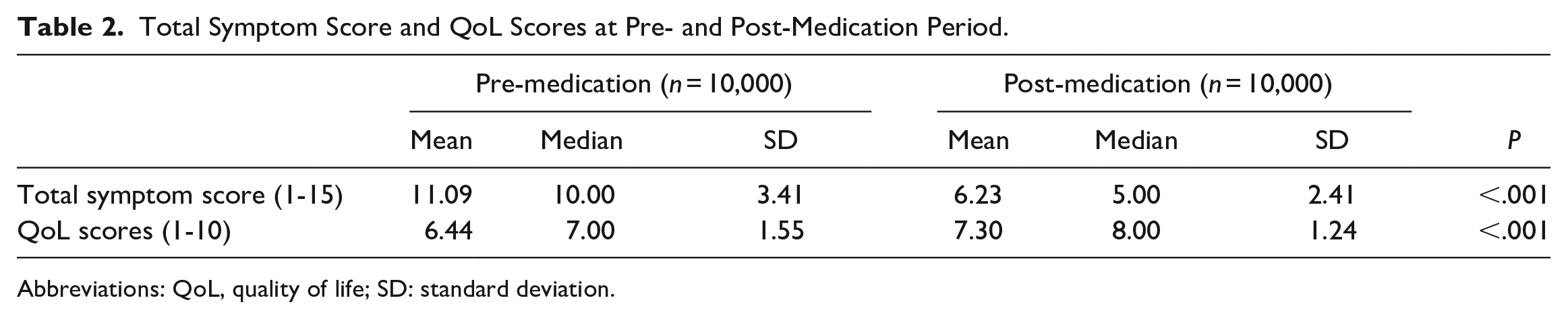
Abbreviations: QoL, quality of life; SD: standard deviation.
Total Symptom Score
The mean total symptom scores pre- and post-medication were different and this difference was at the level of statistical significance (P < .001). The mean pre-medication symptom score was 11.09 ± 3.41, while the mean post-medication score was 6.23 ± 2.41 (see Table 2).
QoL Scores
The mean QoL scores pre- and post-medication were different and this difference was at the level of statistical significance (P < .001). The mean pre-medication QoL score was 6.44 ± 1.55, whereas the mean post-medication score was 7.30 ± 1.24 (Table 2).
The scores obtained on otolaryngological examination, both pre- and post-medication, are shown in Table 3.
Otolaryngology Examination Scores at Pre- and Post-Medication Period.
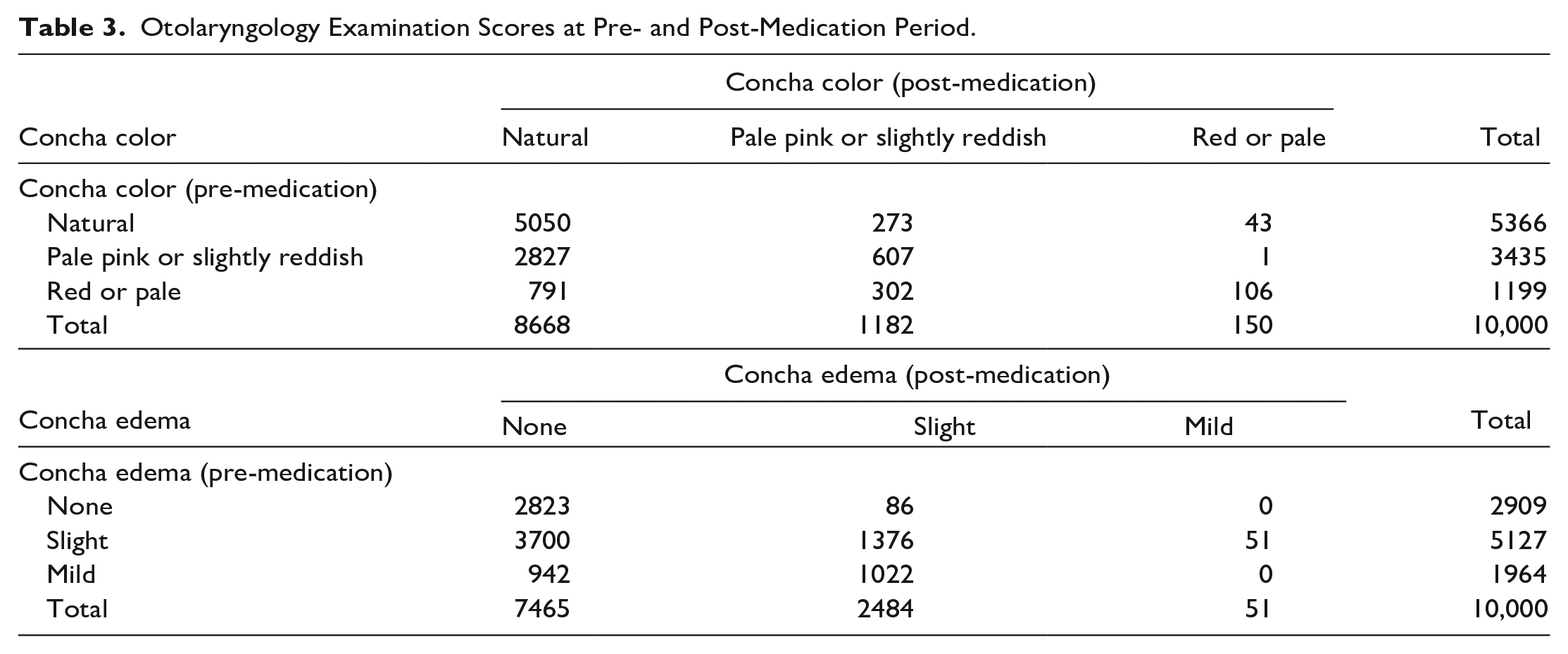
The scores obtained for conchal coloration pre- and post-medication were different and this difference was at the level of statistical significance (P < .001) (see Table 3).
The scores obtained for the degree of conchal edema pre- and post-medication were different and this difference was at the level of statistical significance (P < .001) (see Table 3).
Comparisons by gender
The mean total symptom score prior to Rhinapi was calculated as 10.99 ± 3.37 for men and 11.18 ± 3.44 for women. The average score was thus found to be higher for women than for men (P < .01) (Table 4).
Total Symptom Score and QoL Scores at Pre- and Post-Medication Period in Males and Females.
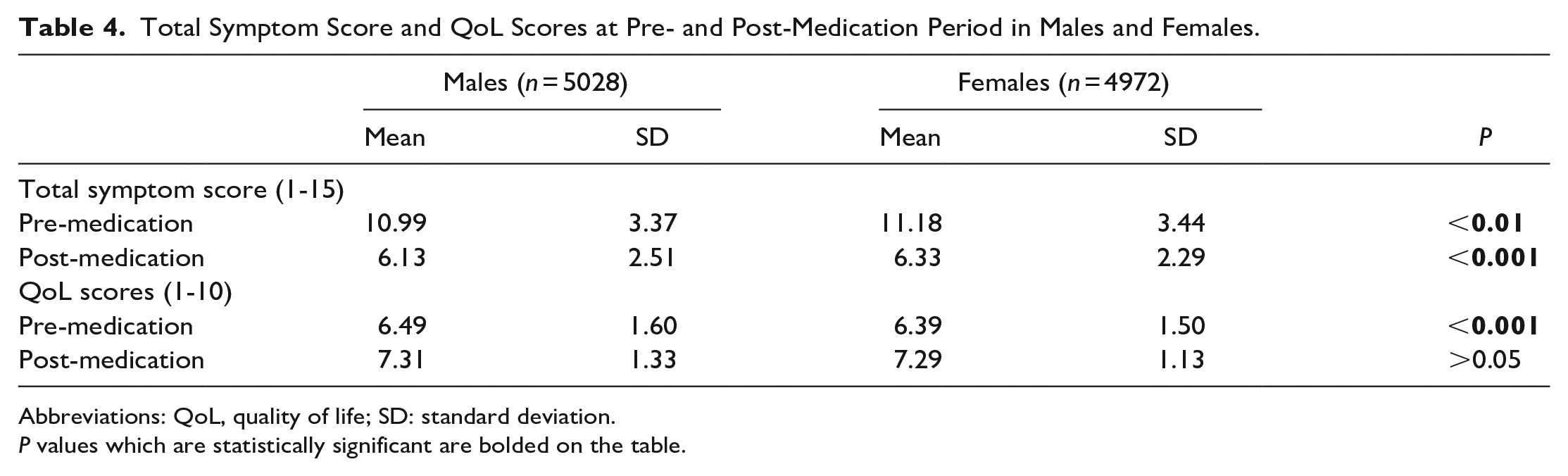
Abbreviations: QoL, quality of life; SD: standard deviation.
P values which are statistically significant are bolded on the table.
The mean total symptom score after Rhinapi was calculated as 6.13 ± 2.51 for men and 6.33 ± 2.29 for women. The average score was found to be higher for women than for men (P < .001) (Table 4).
The mean QoL score prior to Rhinapi use was calculated as 6.49 ± 1.60 for men and 6.39 ± 1.50 for women. The average score was found to be lower for women than for men (P < .01) (Table 4).
The mean QoL Score post-medication was 7.31 ± 1.33 for men and 7.29 ± 1.13 for women. Any difference in the QoL scores between men and women was statistically insignificant (P > .05)
Comparison by occupational group
The occupational groups differed significantly in terms of pre- and post-medication total symptom scores (P < .001):
- Prior to receiving Rhinapi, the total symptom scores in Group 2 (10.65 ± 3.20) were significantly lower than those in Group 1 (11.19 ± 3.50) or Group 3 (11.22 ± 3.34) (P < .001).
- After taking Rhinapi for the study period, symptom scores in Group 3 (5.79 ± 2.15) were lower than those of either Group 1 (6.29 ± 2.36) or Group 2 (6.59 ± 2.74). Moreover, the symptom score in Group 1 (6.29 ± 2.36) was lower than that in Group 2 (6.59 ± 2.74) (P < .001).
The occupational groups also differed significantly in terms of QoL scores (P < .001):
- Prior to receiving Rhinapi, the QoL score in Group 3 (6.57 ± 1.45) was higher than Group 1 (6.40 ± 1.60) or Group 2 (6.38 ± 1.52) and this difference achieved statistical significance (P < .001).
- Following administration of Rhinapi, the QoL scores in Group 2 (7.40 ± 1.19) were significantly higher than those in Group 1 (7.27 ± 1.29) or Group 3 (7.29 ± 1.16) (P < .001).
Results of testing for correlation
The age of patients and the total symptom score following the use of Rhinapi were negatively correlated (r = −0.164; P < .001). As age increased, the value of the total symptom score diminished.
There was a negative correlation between age and the pre- and post-medication QoL values (r = −0.033; P < .010 and r = −0.40; P < .001, respectively). QoL score values decreased as patients grew older.
Discussion
Propolis is a naturally occurring resin produced by bees. It serves a protective function within hives and lessens the bees’ risk of infections. This substance is sometimes also referred to as “bee glue.” Propolis contains an excess of 500 different molecules, including flavonoids, terpenes, aromatic alcohols, and esterified compounds. 5 Propolis has been used in ethnomedicine as a treatment for diabetes mellitus, asthma, ulceration, and burns. 15 Furthermore, scientific investigations have shown that propolis has useful clinical functions, such as reducing inflammation and acting as an anti-neoplastic and anti-microbial agent. There have been numerous studies addressing these properties of propolis.8,16,17
This study was undertaken to evaluate any benefit of Rhinapi nasal spray on the symptoms of allergic rhinitis. Some 10,000 individuals (consisting of 5028 male and 4972 female patients) were enrolled from different centers. All trial participants exhibited symptoms of allergic rhinitis, such as nasal discharge, sneezing, nasal itching, or nasal obstruction. The treatment duration was 3 weeks. Following the end of treatment, statistically significant improvement was detected in each nasal discharge, sneezing, nasal itching, and nasal obstruction (P < .001) scores, when compared to those obtained prior to treatment. The mean total symptom scores improved after administration of Rhinapi. The mean symptom score prior to treatment was 11.09 ± 3.41, while after treatment it became 6.23 ± 2.41.
The mean QoL scores also improved after medication. The mean pre-medication QoL score was 6.44 ± 1.55, whereas the mean post-medication score rose to 7.31 ± 1.24. Following the administration of Rhinapi, there was a significant improvement in the scores for conchal coloration and edema. Total symptom scores were higher in female patients both before and after the treatment period. The QoL scores were lower in female participants prior to medication use, but, following medication, this difference between men and women disappeared. Overall, the total symptom score and QoL values were observed to decrease as patient age increased.
Khosravi et al. 18 investigated how an ethanolic extract of propolis affected mouse pulmonary epithelium in cell culture. The cells were of the TC-1 JHU-1 lineage and had been activated by exposure to the conidia of Aspergillus fumigatus, a potent allergen. The propolis came from the south of Iran, where it was produced by members of the Apis mellifera species. 18 An ethanolic extract of propolis appears to inhibit the expression of interleukins −13 and −17, which foster inflammation, and favor the expression of interleukin-12. Exposure to A. fumigatus may provoke an allergic inflammatory response in cases of bronchopulmonary aspergillosis, allergy-related sinusitis, and asthma. The fact that propolis has been shown to inhibit allergic inflammation provides a rationale for its use in the clinical management of allergic conditions. 1
An ethanolic extract of green propolis sourced from Brazil was also investigated for potential benefit in allergic disorders. This study, by Tani et al., 19 utilized white cells and mononucleocytes harvested from the peripheral circulation of individuals with a known allergy to pollen, specifically the Japanese cedar pollen, Cry j1/2. 19 The release of CysLTs by white cells sensitized to Cry j1 was reduced when the propolis extract was administered and showed a dose-response relationship. The IC50 for this agent was 5.8 μg/ml. However, suppression of histamine degranulation was noted only when the propolis extract was present at raised levels, that is, 100 μg/ml, which suggests that the constituents of propolis that interfere with histamine signaling have a low concentration in the extract of propolis used for the experiment. 1
Furthermore, basophilic responses also appear to be altered in response to propolis. Kashiwakura et al. 20 looked at the effects of propolis on basophils obtained from the bone marrow and activated by DNP23-human serum albumin. Propolis at a level of 100 μg/ml significantly inhibited the expression of interleukins -4, -6, and 13, which act as cytokines stimulating inflammation. This inhibition occurred because the extract prevented Lyn, protein kinase B (Akt), and Erk (extracellular signal-regulated kinase) from becoming phosphorylated. 20
Shinmei and colleagues looked at how effective Brazilian propolis granular was in preventing symptoms of allergic rhinitis in a murine model. 21 The researchers obtained the propolis from the Yamada Apiculture Centre Inc. in Okayama, Japan. Single medicinal use of propolis administered orally to the animals did not cause any change in sneezing or rubbing of the nose, regardless of dose (200, 500, or 1000 mg/kg). The allergic rhinitis in these animals was initially induced using exposure to ovalbumin. When the propolis was administered repeatedly, however, there was a slow but definite reduction in sneezing and rubbing of the nose by the experimental animals, when the dose was 1000 mg/kg. This effect was observed between weeks 2 and 4 21 and was statistically significant.
There do not appear to be any safety or toxicity concerns about propolis, either from animal or clinical trials.22,23 Indeed, where mice were exposed to mitomycin C, the degree of DNA damage was less in animals administered propolis, according to Kumari et al. 24
Overall, propolis seems to regulate the immune response in several ways, both through inhibition and stimulation of different parts of the immune response, notably the way neutrophils adhere to the endothelium and pass into the tissues, as well as altering the levels of cytokines, chemokines, CRP, prostaglandin E2, immunoglobulin expression, and a variety of pathways involved in signaling. Other effects may also be involved.25-30
Abdelhafeez 31 investigated the effect of intra-nasal corticosteroids on QoL of the patients with perennial allergic rhinitis. Patients used budesonide topical aqueous nasal spray for 8 weeks. Their results showed that intranasal corticosteroids improved nasal symptoms of sleep quality, somnolence, and daytime fatigue, and had positive impacts on the QoL of the patients. 31 In Phinyo et al.’s 32 meta-analysis, they reported that 50% or more intranasal corticosteroid doses showed a similar efficacy as regular intranasal corticosteroids in improving QoL and nasal symptoms.
In the present study, we did not compare propolis spray with intranasal corticosteroids. We did not recommend the use of propolis spray as the only treatment modality. We suggested propolis spray may be recommended alongside other treatments such as intranasal corticosteroids.31,32
Conclusion
It is concluded that Rhinapi (Anatolian Propolis and Hypertonic Saline Combination Nasal Spray, marketed by Bee&You) decreases total symptom scores, improves QoL, and reduces the severity of otolaryngological examination scores. Propolis spray may be recommended for allergic rhinitis 33 patients, alongside other treatments.
Footnotes
Author Contributions
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Aslı Elif Tanugur Samanci MD is the Scientific Director of Bee&You, the company that developed the product tested in this study. Reşat Kubilay Irkan is the Medical & Marketing Director of Bee&You, the company that developed the product tested in this study. All other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Bee&You. However, Bee&You did not solicit this research project or protocols with the investigators or the institution. Bee&You was not responsible for the management of the study, data processing, and reporting.
Ethics Committee
Ethics committee approval was obtained from Istanbul Medipol University GETAT Clinical Research Ethics Committee (Date: 12.07.2021, Number: 25).
Informed Consent
Verbal consent was obtained from the patients.
