Abstract
Keywords
Introduction
Numerous factors, including the patient’s age, the frequency and intensity of their symptoms, and the existence of any co-occurring disorders, determine the course of allergic rhinitis (AR) treatment.1-3 Successful therapy hinges on avoiding allergies. The use of intranasal corticosteroids is a safe and efficient therapy option. The predominant treatments for severe and refractory AR include combinations of nasal or oral antihistamines, decongestants, cromolyn, or leukotriene-receptor antagonists.4-6 Sublingual or subcutaneous immunotherapy may be tried if the symptoms persist despite standard treatment and are not satisfactorily managed. 7 Transforming the normal progression of AR and alleviating symptoms by the induction of allergen-specific tolerance through immunotherapy is a promising possibility. Despite the efficacy and safety of these medications, non-pharmacological approaches have been developed to treat AR nasal symptoms, such as saline irrigation of the nostrils, which may help decrease the intensity of the disease as stated by patients.8,9
Minimal side effects are associated with saline irrigation, making it a promising supplementary treatment for AR. The full impact mechanism of saline irrigation is still a mystery. The removal of airborne allergens and inflammation mediators, as well as the cleansing of mucus, is thought to play a role in its therapeutic benefits. 10 Patients who undergo saline irrigation also show signs of a return to normal mucociliary transport function. Because of its efficacy and ease of administration, saline irrigation has the potential to replace conventional AR treatments. 9
Saline and seawater have traditionally been used for nasal irrigation and decongestion in cases of nasal congestion. In 2017, Di Berardino et al stated that nasal hypertonic and isotonic sprays increase the effectiveness of other agents used in treatment by clearing airborne allergens and particles from the nasal passages. 11 Studies have also shown that isotonic irrigation improves impaired nasal mucociliary clearance in patients with AR.12,13 Similarly, hypertonic sprays reduce nasal congestion by reducing nasal mucosa and conchae edema.
Many guidelines recommend nasal irrigation with saline as a safe, simple, and treatment-enhancing method before using corticosteroid nasal sprays. 14 In a 2012 meta-analysis, Hermelingmeier et al showed that nasal saline irrigation reduced AR symptoms by 27.66%, medication use by 66%, mucociliary clearance by 31.19%, and quality of life (QoL) by 27.88%. 15
This study aimed to investigate different types of nasal irrigation sprays in treating adults with AR by comparing their effectiveness in reducing nasal congestion, patient comfort, and allergy symptoms’ overall manifestations.
Materials and Methods
The present study was a prospective, multicenter, observational study following the principles of the Declaration of Helsinki. Initially, ethical approval was obtained from Istanbul Medipol University, Traditional and Complementary Medicine Applications Ethics Committee, file number 07, on March 27, 2024. The approval application was made to the Ministry of Health General Directorate of Health Services Traditional and Complementary Medicine Applications on July 18, 2024, with application number 248980922.
Subjects
A total of 1700 patients with AR (866 males and 834 females) meeting the inclusion criteria from different centers in different regions of Turkey were enrolled in the study between April 2024 and July 2024. All subjects in the study showed signs of AR, including runny nose, sneezing, itching, and congestion. After routine otolaryngological evaluation, they started getting the usual AR; each patient also received the prescribed nasal spray 3 times daily for 3 weeks.
Patients were assigned to 1 of 3 groups according to the attending order in ear, nose, and throat (ENT) clinics. The first arriving patient was included in group 1, the next arriving patient in group 2, and the next arriving patient in group 3. Subsequent patients were included in groups according to this order, and the spray treatments specified below were administered to the patients. Geographical area was not considered in the inclusion of patients in groups:
Group 1: Hypertonic nasal spray group (n = 600) (Sinomarin®), containing a 2.3% NaCl hypertonic sea water solution. 16
Group 2: Algae-containing hypertonic nasal spray group (n = 600) (Sinomarin Plus Algae ENT), containing a 2.3% NaCl hypertonic sea water solution and sea algae extracts, namely from Undaria pinnatifida and Spirulina platensis. 17
Group 3: Isotonic saline nasal spray group (n = 500) (Berko-fiz® nasal spray)
Inclusion Criteria for Volunteers
Those who applied to routine ENT outpatient clinics at the specified centers starting from April 2024 were diagnosed with AR during the routine examination process through history, symptoms, and examination findings.
Those who agreed to participate in the study.
All patients over 18 years of age.
Exclusion Criteria for Volunteers
Patients with infection detected in the field of ENT.
Patients with psychiatric disorders.
Pregnancy.
Patients with sinonasal or other malignancies.
Heart failure.
Patients with a history of cardiac pathology, such as hypertension.
Patients receiving immunosuppressive treatment.
Patients that lacked mental competence.
Before and 3 weeks following therapy, people were asked to rate their overall symptoms, QoL, and results from an otolaryngology examination.
The trial participants’ occupational position was also recorded, categorizing them as either low-level, moderate-level, or severe allergen exposed.
Methods
People who took part in the study were asked to rate the severity of AR symptoms, including stuffy nose, sneezing, itching, and nasal blockage. The scale ran the gamut from 1 (very mild symptoms) to 5 (very severe). Similarly, overall symptom scores ranging from 1 to 20 were collected.
The QoL scale ran from 1 (the worst possible quality) to 10 (the best possible quality).
On top of that, an otolaryngological examination was used to score the patients. There was a sliding scale from 1 to 3 for the turbinates’ color (natural, pale pink or slightly reddish, red or pale) and edema severity (none, mild, or moderate).
Statistical Analysis
The analysis was undertaken using the IBM SPSS for Windows 21.0 statistical software application (SPSS, Inc., an IBM Company, Chicago, IL, USA). The Kolmogorov–Smirnov test was utilized to determine whether the variables conformed to a normal distribution. Parametric and non-parametric tests were used for group comparisons. The Student’s t-test was used to compare means based on distribution forms, and the Wilcoxon signed-rank test was used to compare pre-and post-scores. Chi-square tests were employed for the analysis of cross-tables created for independent variables. McNemar–Bowker tests were used to analyze cross-tables (crosstabs) designed to compare pre-and post-treatment symptoms (discharge, sneezing, itching, congestion, turbinate color, and turbinate edema).
The Kruskal–Wallis test was used to compare symptom scores and QoL values among the groups (3 groups) based on their distribution forms, and a post hoc test was conducted for multiple comparisons.
In summarizing the data, count (%) was used for qualitative data, and mean ± standard deviation or median (Q1; Q3) statistics were used for quantitative data. P < .05 was considered statistically significant.
Results
Group 1 (Hypertonic Nasal Spray Group)
In group 1, there were 307 (51.2%) males and 293 (48.8%) females (n = 600). The mean age of group 1 was 37.06 ± 12.16 years.
Exposure to the allergens
The difference in male and female exposure to allergens was statistically significant (P = .044, χ2 = 6.260). Men were exposed to severe allergens more frequently (58.2%), while this rate was 41.8% in women.
Symptom scores
Before treatment, the discharge scores were mild (40.7%), moderate (20.0%), and severe (27.2%), whereas after treatment, they were none (39.7%) and mild (43.0%). A significant difference was found between pre-treatment and post-treatment discharge scores (P < .001).
Before treatment, the sneezing scores were mild (25.7%), moderate (21.8%), severe (13.8%), and very severe (20.7%), whereas after treatment, they were none (60.0%) and mild (31.5%). A significant difference was found between pre-treatment and post-treatment sneezing scores (P < .001) (McNemar–Bowker test).
Before treatment, the itching scores were mild (25.5%), moderate (30.3%), severe (10.5%), and very severe (17.0%), whereas after treatment, they were none (56.7%) and mild (27.2%). A significant difference was found between pre-treatment and post-treatment itching scores (P < .001).
Before treatment, the congestion scores were mild (31.7%), moderate (22.7%), and very severe (14.7%), whereas after treatment, they were none (62.3%) and mild (28.3%). A significant difference was found between pre-treatment and post-treatment congestion scores (P < .001).
Evaluation of turbinates
The color of the turbinates was pale pink and slightly red (34.7%) and red or pale (13.0%) before treatment, whereas after treatment, it was natural (75.5%) and somewhat red (14.7%). A significant difference was found between pre-treatment and post-treatment turbinate color scores (P < .001).
The turbinate edema was mild (49.0%) and moderate (20.7%) before treatment, whereas after treatment, it was none (63.3%) and mild (9.5%). A significant difference was found between pre-treatment and post-treatment turbinate edema scores (P < .001).
The total symptom and QoL scores before and after treatment are shown in Table 1.
Total Symptom Score and QoL Scores at Pre- and Post-Medication Period.
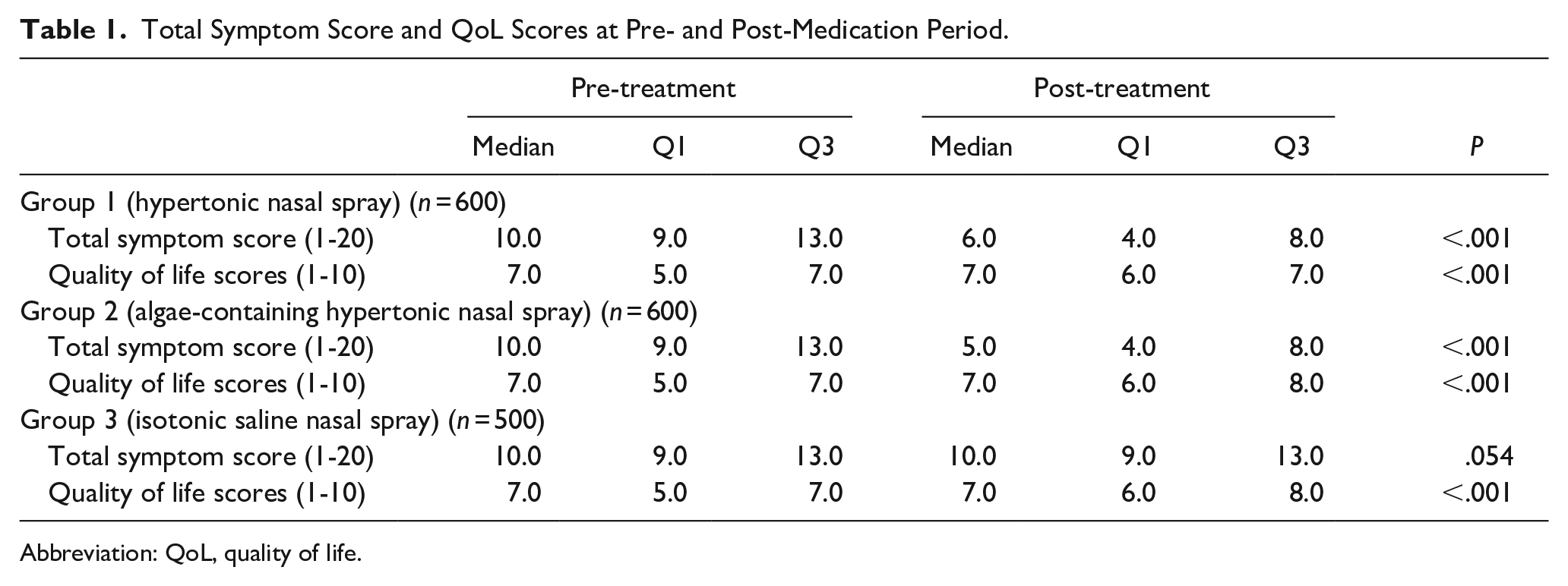
Abbreviation: QoL, quality of life.
The median total symptom scores before and after treatment were found to be different (P < .001). The total symptom scores decreased after treatment.
The median QoL scores before and after treatment were found to be different (P < .001). The median QoL scores increased after treatment.
Group 2 (Algae-Containing Hypertonic Nasal Spray Group)
In group 2, there were 307 (51.2%) males and 293 (48.8%) females (n = 600). The mean age of group 1 was 37.06 ± 12.16 years.
Exposure to the allergens
The difference in male and female exposure to allergens was statistically significant (P = .044, χ2 = 6.260). Men were exposed to severe allergens more frequently (58.2%), while this rate was 41.8% in women.
Symptom scores
Before treatment, the discharge scores were mild (40.7%), moderate (20.0%), and severe (27.2%), whereas after treatment, they were none (41.2%) and mild (41.5%). A significant difference was found between pre-treatment and post-treatment discharge scores (P < .001).
Before treatment, the sneezing scores were mild (25.7%), moderate (21.8%), severe (13.8%), and very severe (20.7%), whereas after treatment, they were none (62.7%) and mild (29.0%). A significant difference was found between pre-treatment and post-treatment sneezing scores (P < .001).
Before treatment, the itching scores were mild (31.7%), moderate (22.7%), and very severe (14.7%), whereas after treatment, they were none (66.3%) and mild (26.0%). A significant difference was found between pre-treatment and post-treatment itching scores (P < .001).
Before treatment, the congestion scores were mild (25.3%), moderate (30.5%), severe (10.5%), and very severe (17.0%), whereas after treatment, they were none (59.7%) and mild (27.0%). A significant difference was found between pre-treatment and post-treatment congestion scores (P < .001).
Evaluation of turbinates
The color of the turbinates was pale pink and slightly red (34.7%) and red or pale (13.0%) before treatment, whereas after treatment, it was natural (86.8%) and somewhat red (12.2%). A significant difference was found between pre-treatment and post-treatment turbinate color scores (P < .001).
The turbinate edema was mild (49.0%) and moderate (21.0%) before treatment, whereas after treatment, it was none (74.3%) and mild (25.2%). A significant difference was found between pre-treatment and post-treatment turbinate edema scores (P < .001).
The total symptom and QoL scores before and after treatment are shown in Table 1.
The median total symptom scores before and after treatment were found to be different (P < .001). The total symptom scores decreased after treatment.
The median QoL scores before and after treatment were found to be different (P < .001). The median QoL scores increased after treatment.
Group 3 (Isotonic Saline Nasal Spray Group)
In group 3, there were 252 (50.4%) males and 248 (49.6%) females (n = 500). The mean age of group 1 was 36.75 ± 11.75 years.
Exposure to the allergens
The difference in male and female exposure to allergens was statistically significant (P = .023, χ2 = 7.553). Men were exposed to severe allergens more frequently (58.4%), while this rate was 41.6% in women.
Symptom scores
Before treatment, the discharge scores were mild (40.0%), moderate (20.4%), and severe (29.6%), whereas after treatment, they were none (4.2%), mild (40.6%), moderate (20.4%), and severe (29.2%). There were no significant differences between pre-treatment and post-treatment discharge scores (P = .135).
Before treatment, the sneezing scores were mild (25.6%), moderate (24.2%), severe (15.4%), and very severe (18.6%), whereas after treatment, they were none (16.2%), mild (26.2%), moderate (24.6%), severe (15.6%) and very severe (17.4%). There were no significant differences between pre-treatment and post-treatment discharge scores (P = .147).
Before treatment, the itching scores were mild (32.4%), moderate (25.6%), and very severe (14.2%), whereas after treatment, they were none (25.4%), mild (33.4%), moderate (26.2%), and very severe (13.2%). There were no significant differences between pre-treatment and post-treatment discharge scores (P = .051).
Before treatment, the congestion scores were mild (24.0%), moderate (31.6%), severe (10.4%), and very severe (16.6%), whereas after treatment, they were none (17.8%), mild (23.4%), moderate (32.4%), severe (11.0%) and very severe (15.4%). There were no significant differences between pre-treatment and post-treatment discharge scores (P = .062).
Evaluation of turbinates
The color of the turbinates was pale pink and slightly red (33.8%) and red or pale (15.8%) before treatment, whereas after treatment, it was natural (81.6%) and somewhat red (14.6%). A significant difference was found between pre-treatment and post-treatment turbinate color scores (P < .001).
The turbinate edema was mild (44.6%) and moderate (21.4%) before treatment, whereas after treatment, it was none (69.4%) and mild (25.6%). A significant difference was found between pre-treatment and post-treatment turbinate edema scores (P < .001).
The total symptom and QoL scores before and after treatment are shown in Table 1.
There were no significant differences in the median total symptom scores before and after treatment (P = .054).
The median QoL scores before and after treatment were found to be different (P < .001). The median QoL scores increased after treatment.
The Total Symptom Score and Quality of Life Score Analysis Between 3 Groups
At the pre-treatment period, there were no significant differences between the total symptom scores (P = .950) and QoL scores of the 3 groups (P = .861).
During the post-treatment period, there were statistically significant differences between the total symptom scores of the 3 groups (P < .001).
The total symptom scores of group 1 (P < .001) and group 2 (P < .001) were significantly lower than those in group 3. There were no significant differences between groups 1 and 2 (P = .213).
During the post-treatment period, there were statistically significant differences between the QoL scores of the 3 groups (P < .001). QoL scores of group 1 (P < .001) and group 2 (P < .001) were significantly higher than those in group 3. There were no significant differences between groups 1 and 2 (P = 1.000).
Discussion
Significant roles are played by immune-mediated inflammatory responses in the complex pathophysiology of AR.5,18-20 Patients with AR have been discovered to have elevated cytokines in their serum and nasal fluids. The nasal fluid of patients with seasonal AR has been observed to have higher levels of IL-5, eotaxin, MIP-1α, and IL-17. 21 Some research suggests that saline irrigation can reduce AR inflammatory cascades by removing inflammatory mediators.22-24 In addition, irrigation can potentially restore mucociliary function and eliminate sticky secretions.15,25-31 The advantages of saline irrigation may partly be due to these physiological changes.29,30
There are several approaches to saline irrigation, but research into the best combinations of volume, pressure, concentration, and delivery systems is still in its early stages. The 2 most common solutions for saline irrigation are isotonic saline, which has a sodium chloride concentration of 0.9%, and hypertonic saline, which has a sodium chloride concentration higher than 0.9%. Due to its increased osmotic pressure, hypertonic saline irrigation should be more effective in lowering nasal mucosal edema and eliminating airborne allergens and inflammation mediators. 29
Hypertonic saline nasal irritation (HSNI) was the target of Li et al’s 31 investigation into the effectiveness of oral antihistamine use in children with AR in alleviating nasal symptoms and enhancing QoL. Patients reported fewer nasal symptoms after HSNI. Although the rates of adverse effects and antihistamine use were similar between the groups, studies comparing HSNI with isotonic saline nasal irrigation (ISNI) demonstrated that patients in the HSNI group had better nasal symptom scores (mean difference, 1.22 points; 95% CI 1.01-1.44; I 2 = 0%; P < .001). Children with AR may find HSNI to be a more suitable supplementary treatment option than ISNI.
Patients with poorly managed symptoms may benefit from additional non-pharmacological treatments, including saline irrigation. Although the ideal saline concentration for irrigation remains a mystery, our research proved that HSNI is the most effective method. Both the HSNI and ISNI groups saw comparable side effects. Consequently, it is prudent to supplement intranasal corticosteroids and antihistamines with HSNI therapy for individuals who do not respond well to or are unable to tolerate these medications. In addition, patients who continue to experience moderate to severe symptoms after undergoing the treatments above may benefit from immunotherapy.1-3,7
Marine algae have recently received much attention as they are a valuable source of chemically diverse bioactive compounds with numerous health benefits. 32 Anti-allergic agents derived from marine algae have already been reported. 32
In the present study, we investigated the effects of a hypertonic nasal spray (group 1), a hypertonic nasal spray containing algae (group 2), and an isotonic saline nasal spray on AR. In hypertonic nasal spray and algae-containing hypertonic nasal spray groups, symptom scores of discharge, sneezing, itching, and congestion scores decreased; turbinate color and edema also reduced after treatment. The total symptom scores decreased, and QoL scores increased after treatment. In the saline group, there were no significant differences in symptom scores and total symptom scores after treatment; however, improvement was detected in turbinate color and edema values after treatment. QoL scores increased after treatment.
When comparing the 3 groups, the total symptom scores of groups 1 and 2 were significantly lower; QoL scores of groups 1 and 2 were considerably higher than those of the saline group. There were no significant differences between groups 1 and 2. These results indicated that adding algae to the hypertonic seawater solution does not offer additional benefits in managing allergic symptoms in patients despite previous preclinical results and literature suggesting potential anti-allergic effects of algae-derived molecules.32-37 The reasons for this lack of algae activity are unclear at this point. One likely explanation is that at the time of evaluation at 3 weeks, differences in symptom scores between groups 1 and 2 may no longer be visible as both hypertonic solutions were highly active during the study. In a clinical study conducted by the same authors in rhinosinusitis patients using the exact solutions, the algae-containing hypertonic solution was superior to both hypertonic and isotonic solutions in terms of improvement of total nasal symptom scores, indicating that the addition of algae to irrigation solutions does offer additional benefits (manuscript in preparation). Therefore, additional research is needed to evaluate algae’s potential effects in AR patients.
Notwithstanding the above considerations, algae-containing hypertonic solutions and standard hypertonic solutions remain highly efficacious and safe in managing symptoms in patients with AR. This is in agreement with previous clinical studies conducted with the same or similar hypertonic solutions in patients with different ENT conditions, including AR, acute rhinosinusitis, or in the post-operative setting.38-43
Conclusion
In our study, we observed that hypertonic nasal sprays with or without algae significantly reduced AR symptom scores and increased QoL scores compared to saline nasal sprays. We concluded that these sprays could be added to the treatment regimen for patients with AR alongside routine treatment.
Footnotes
Acknowledgements
None.
Author Contributions
Ayten Güner Atayoğlu: Planning, designing, literature survey, data collection. Nuray Bayar Muluk: Planning, designing, literature survey, interpretation of the results, active intellectual support, writing, submission. Rahime Koca: Planning, designing, literature survey, data collection. İbrahim Çukurova: Planning, designing, literature survey, data collection. Erdem Atalay Çetinkaya: Planning, designing, literature survey, data collection. Özgür Yörük: Planning, designing, literature survey, data collection. Cengiz Bal: Planning, designing, statistical analysis, interpretation of the results. Arzu Tatar: Planning, designing, literature survey, data collection. Nihat Susaman: Planning, designing, literature survey, data collection. Nagehan Dilşad Erdoğmuş Küçükcan: Planning, designing, literature survey, data collection. Enes Güngör: Planning, designing, literature survey, data collection. Necdet Özçelik: Planning, designing, literature survey, data collection. Elvin Alaskarov: Planning, designing, literature survey, data collection. Zeynel Ozturk: Planning, designing, literature survey, data collection. Oguzhan Oguz: Planning, designing, literature survey. Burak Mustafa Taş: Planning, designing, and literature survey. Cemal Cingi: Planning, designing, literature survey, data collection, English editing.
Availability of Data and Materials
All data for this study is presented in this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Clinical Research Organization, Turkey provided all nasal sprays used in this study. However, this organization did not solicit the investigators or institutions’ research projects or protocols.
Ethics Committee Approval
Initially, ethical approval was obtained from Istanbul Medipol University, Traditional and Complementary Medicine Applications Ethics Committee, file number 07, on March 27, 2024. The approval application was made to the Ministry of Health General Directorate of Health Services Traditional and Complementary Medicine Applications on July 18, 2024, with application number 248980922.
Informed Consent
Verbal consent was obtained from the patients.
