Abstract
Introduction
Due to the high rate of nasal deformity and septum deviation in society, septoplasty and septorhinoplasty are among the most common surgical procedures performed by ear-nose-throat surgeons worldwide. Maintaining the form and mass of cartilage grafts is crucial for the long-term desired results. Cartilage grafts tend to reabsorb and lose volume over time. As a result of the decrease in the viability of cartilage grafts, loss of support and deformation occur in the nasal septum.1-5
Polydioxanone (PDS) plates are biodegradable aliphatic polyester-type materials produced by the polymerization of monomeric p-dioxanone. 6 PDS is a resorbable material that can provide a structural platform in nasal cartilage reconstruction procedures. PDS plate has the same structure as PDS suture and is frequently used in septorhinoplasty. It is available in various thicknesses. They are suitable for restructuring according to the required shape and form. After the PDS plate remains intact for approximately 10 weeks, it takes about 5 months to be absorbed entirely.7,8 PDS has been used successfully to repair bone defects and septal reconstruction procedures. No foreign body reaction or long-term complications have been reported due to using a PDS plate.9-11
Platelet-rich plasma (PRP) is part of a small volume of blood plasma separated from the plasma after centrifugation and contains a high concentration of platelets. Once activated, growth factors are released by degranulating the platelets, and wound healing begins. Growth factors such as platelet-derived growth factor, insulin-like growth factor, and vascular endothelial growth factor are some of these factors. 12 PRP is obtained cheaply and easily from the patient’s blood, making its usage easier. 12 Studies report that PRP increases cell proliferation, angiogenesis, collagen synthesis, and cartilage repair and stimulates chondrocyte proliferation and matrix biosynthesis.13,14
This study aimed to investigate the effectiveness of PDS plate and PRP on the regeneration of cartilage grafts, which are frequently used in nasal and septal surgery. The graft volume change, the histopathological quality, and the physical change of the new cartilage formed due to cartilage regeneration after using PDS plate and PRP in cartilage grafts were evaluated.15-19
Materials and Methods
This study was carried out with the approval of Eskişehir Osmangazi University Animal Experiments Local Ethics Committee (HADYEK) dated June 20, 2017 and registration number 583-2/2017. Surgical interventions, follow-up processes, and euthanasia procedures were performed at Eskisehir Osmangazi University Medical and Surgical Experimental Research Center (TICAM). Fifteen healthy white New Zealand Albino-type female rabbits were used for the experimental study. The average weight of white New Zealand Albino-type female rabbits was 2500 to 4000 g, and their age ranged between 15 and 18 months. The rabbits were housed in “Radon brand rabbit cages” at a temperature of 22°C ± 2°C and an environment of 12 hours of darkness and 12 hours of light. The rabbits were fed with commercially available bait, fresh vegetables, and water throughout the experiment.
PRP Production from Rabbits
Ten milliliters of blood was taken from the central auricular artery of the rabbits. The bleeding was stopped with pressure. A special PRP tube (PRP Vacuum blood collection tube; CENCE Company, Istanbul, Turkey) was used for PRP production, and they were placed in the centrifuge device (CENCE Company) in a mutually balanced manner. The tubes were centrifuged at 2000 rpm (644g) for 10 minutes. The plasma obtained after the first centrifugation was transferred to the second tube. The second tube was centrifuged at 2600 rpm (1089g) for 15 minutes. The upper 60% of the resulting serum was removed with a syringe. The remaining 40% was mixed approximately 5 to 10 times with a syringe by pulling and releasing. After this process, PRP is ready to be applied. Material obtained from rabbits’ own blood was administered autologously to each rabbit.
Our study was carried out in 4 different applications on each animal and lasted 3 periods: first month, second month, and third month. These 3 distinct periods were selected, considering the onset of cartilage regeneration and monitoring the clinical recovery process of cartilage regeneration.
All 4 different applications of implants were planted separately on the back of each rabbit apart from each other. The implants in the first randomly selected 5 rabbits were removed at the end of the first month. The implants in the second randomly selected 5 rabbits were removed at the end of the second month, and the implants in the last 5 rabbits were removed at the end of the third month.
Injection of 35 mg/kg Ketamine HCL (Ketalar, Eczacıbaşı, Turkey) and 8 mg/kg Xylazine HCl (Rompun; Bayer, Leverkusen, Germany) for general anesthesia. Depth of anesthesia of rabbits was measured by limb withdrawal response. If necessary, anesthesia was maintained during the experiment by re-administering one-third of the first dose. After the depth of anesthesia was achieved, sufficient surgical area was shaved on the ventral side of the ear and in the dorsal paravertebral region, where cartilage transplantation was planned to receive cartilage graft material. The surgical field was covered sterile after the entire surgical area was stained with Povidone-Iodine (Batticon Antiseptic Solution; Adeka, Istanbul, Turkey) solution. After shaving, the local anesthetic Prilocaine (Citanest 2%; Astra Zeneca, Istanbul, Turkey) was applied superficially to the incision area to reduce pain in the postoperative period in the surgical field.
To obtain cartilage graft materials, a skin-subcutaneous incision was made on the ventral side of the auricula, where the cartilage folds were less. Skin flaps were carefully elevated. The cartilage was accessed by subperiosteal dissection. Four pieces of ear cartilage, 1 × 1 cm large enough to form cartilage graft material, were removed en bloc (Figure 1). The incision site was sutured with 4-0 Polyglactin 910 (Vicryl, Ethicon, Inc.; J&J MedTech, New Brunswick, NJ, USA). Then, the perichondrium of the resulting block cartilage on both sides was dissected and peeled. The block cartilage was cleaned with physiological saline.
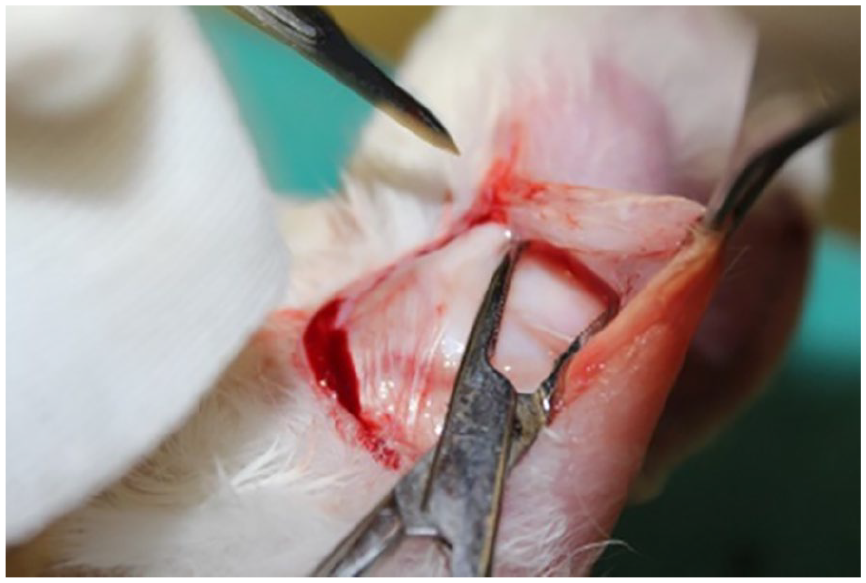
Cartilage graft removal from the ventral region of the auricle.
Four round cartilage grafts 10 mm in diameter were created from the en bloc removed cartilage graft with the help of a 10 mm “Acu-Punch punch biopsy” (Acuderm, Inc., Waltham, MA, USA) tool (Figure 2). A hole was made in the middle of each 10 × 10 mm round cartilage graft with the help of a 3 mm punch biopsy (Kai Medical; Kai Industries Co. Ltd., Seki, Japan) tool, and the cartilage in this area was removed (Figure 3). As a result, an equal-sized 3 mm space was created in the middle of each cartilage as standardized implants (Figure 4).
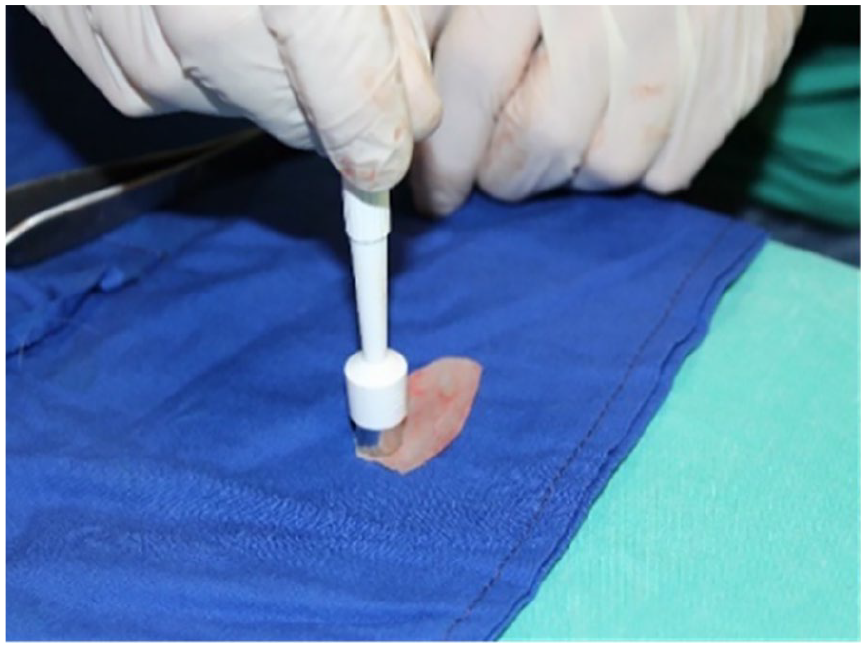
Cartilage graft 10 mm in diameter was created from the en bloc removed cartilage graft with the help of a 10 mm “Acu-Punch punch biopsy.”
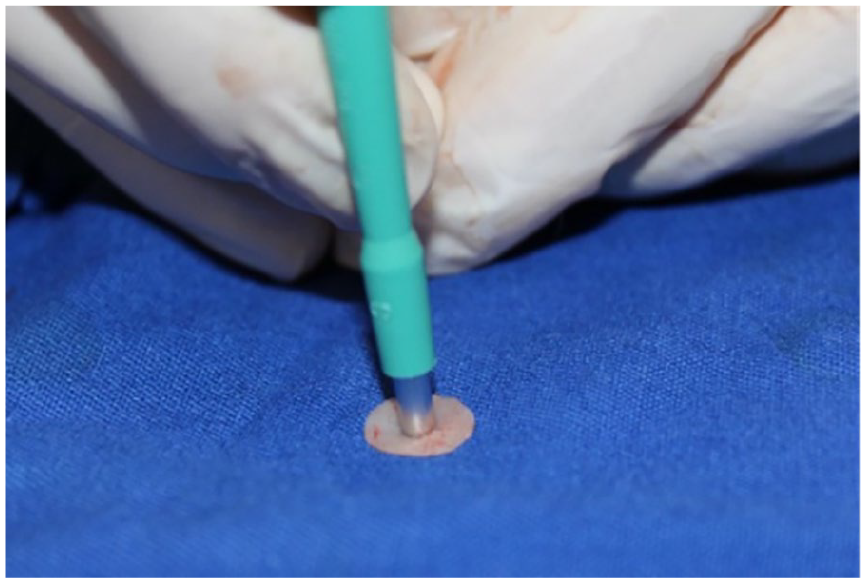
A 3 mm hole was made in the middle of each cartilage graft with the help of a 3 mm punch biopsy.
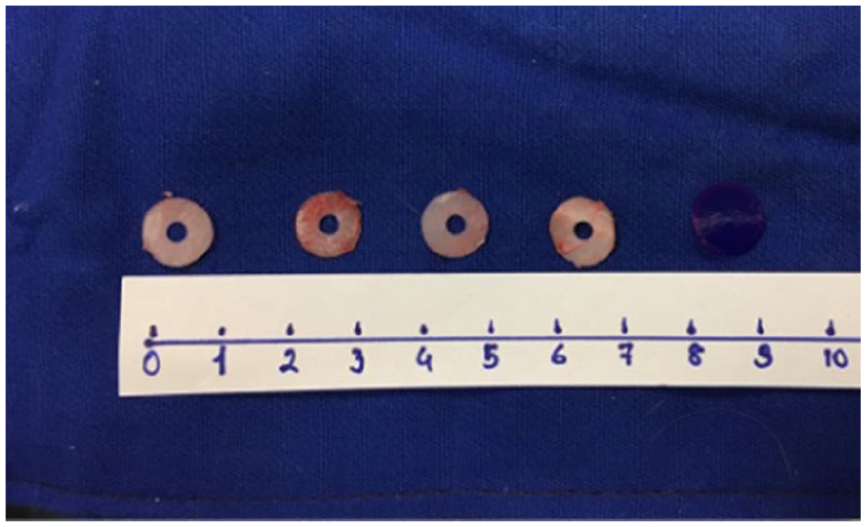
Standardized cartilage in the same size and shape before implantation.
Experiment groups
Study Group 1: The cartilage graft alone was implanted in the created pocket in this group without any other procedure
(first month: n = 5, second month: n = 5, third month: n = 5)
Study Group 2: The cartilage graft was implanted in the created pocket, and PRP was applied together with the cartilage graft in this group
(first month: n = 5, second month: n = 5, third month: n = 5)
Study Group 3: The PDS plate and cartilage graft were implanted in the created pocket. PDS Plate (0.25 mm, Ethicon, Inc.; J&J MedTech) of the same size and shape as the cartilage was prepared, and both were fixed on top of each other with 4.0 PDS suture (Ethicon, Inc.; J&J MedTech) to fit snugly. The created graft material was then placed in the pocket as a whole unit (Figure 5)
(first month: n = 5, second month: n = 5, third month: n = 5)
Study Group 4: The PDS plate and cartilage graft combination was prepared and implanted as in study group 3. Additionally, PRP was applied over the combined implant
(first month: n = 5, second month: n = 5, third month: n = 5)
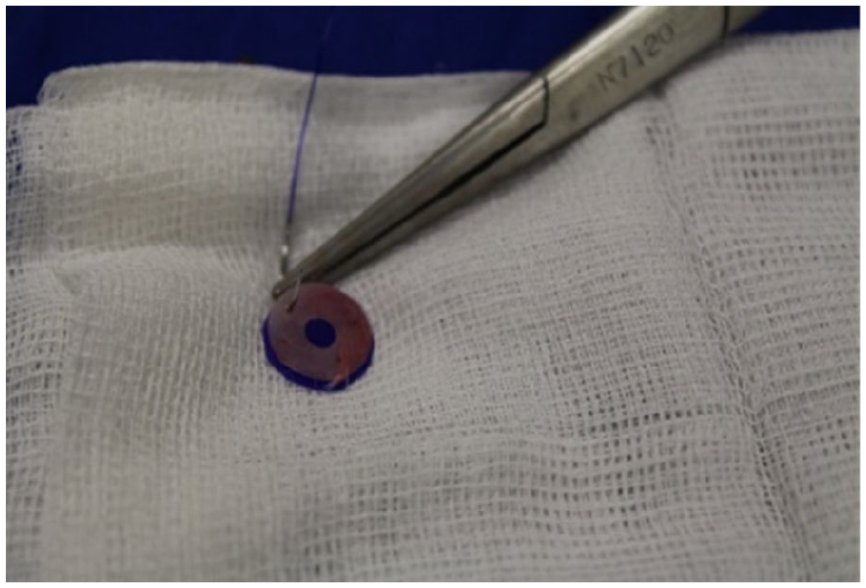
Suturing the cartilage piece and PDS plate before implantation.
Implantation
Four approximately 1.5 cm long skin incisions were made on the back of the rabbits. Using forceps and clamps, pockets of suitable size were opened under the skin through which previously prepared implants could be placed separately.
The pockets in the cranial part are numbered 1 and 2 from left to right; those in the caudal part are numbered 3 and 4 from left to right. The same numbering system was used for all animals. A distance of at least 2 cm was left between the created pockets, cranial/caudal and left/right. Graft materials were placed into the designed pockets respectively. After the implantation, the opened pockets were sutured using 4-0 Polyglactin 910 (Vicryl, Ethicon, Inc.; J&J MedTech), and the operation was terminated (Figures 6 and 7).
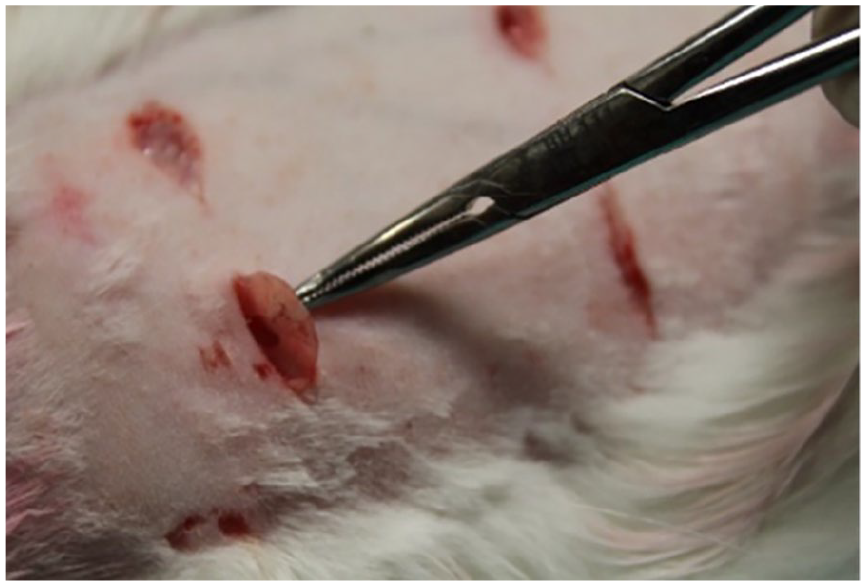
Placing the cartilage into previously opened pockets.
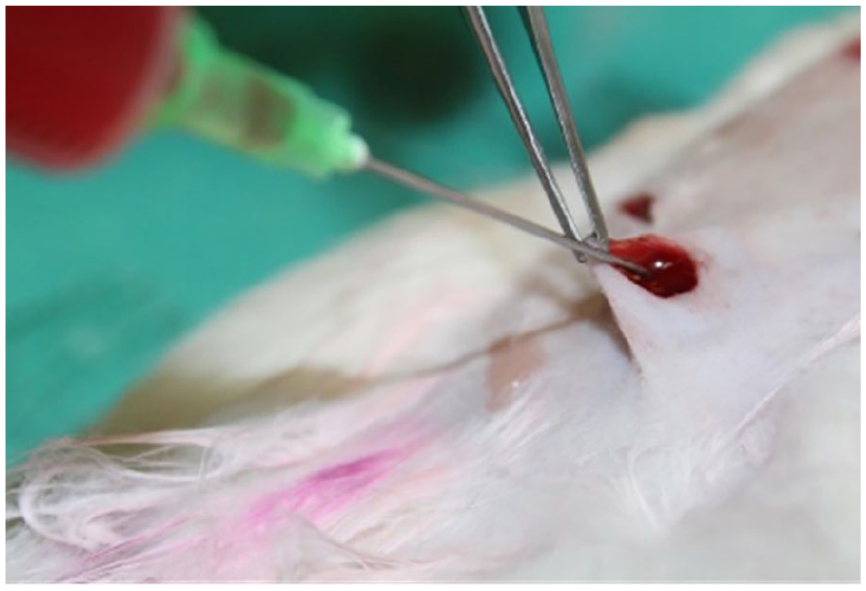
Application of PRP on the cartilage implant.
For antibiotic prophylaxis, a single dose of ceftriaxone (Rocephin Im 1 gr; Roche, Istanbul, Turkey) was administered to the rabbits intramuscularly at a dose of 50 mg/kg.
In the postoperative period, no complications such as separation of the wound lips, wound site infection, or hematoma were observed.
Five of the experimental animals were sacrificed at the end of the first month, 5 at the end of the second month, and 5 at the end of the third month by giving a lethal dose of thiopental sodium (150 mg/kg). Graft cartilages were removed from their pockets and numbered immediately after the animals were sacrificed (Figures 8 to 11).
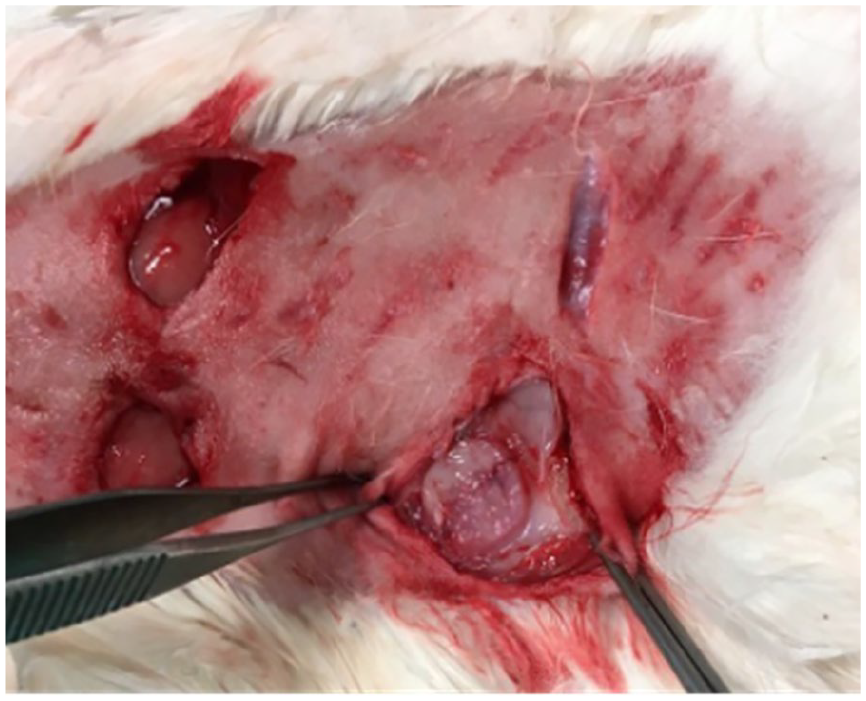
Removal of the cartilage graft from the pockets.
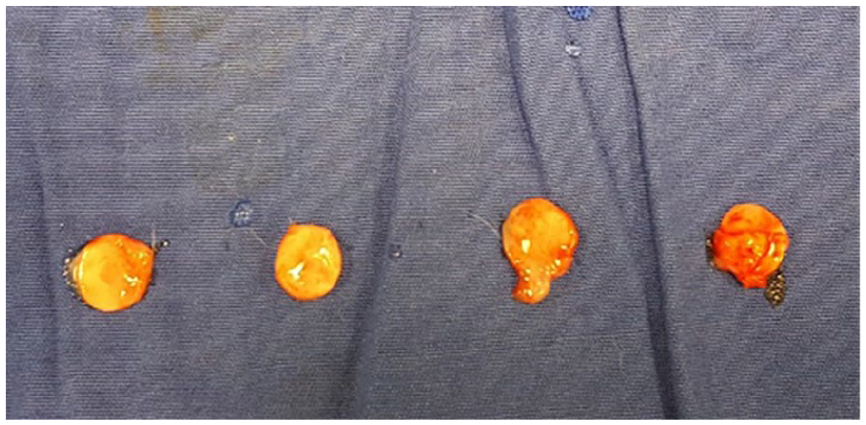
The view of the 4 cartilage grafts removed at the end of the first month.
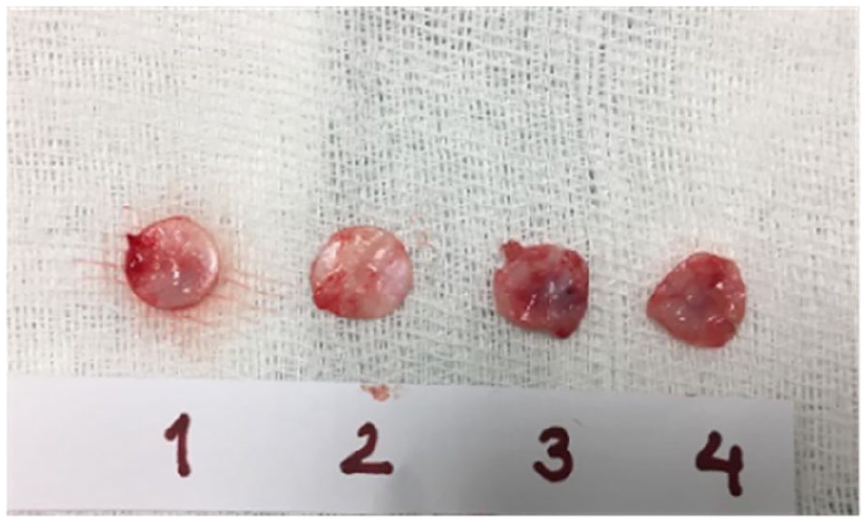
The view of the 4 cartilage grafts removed at the end of the second month.
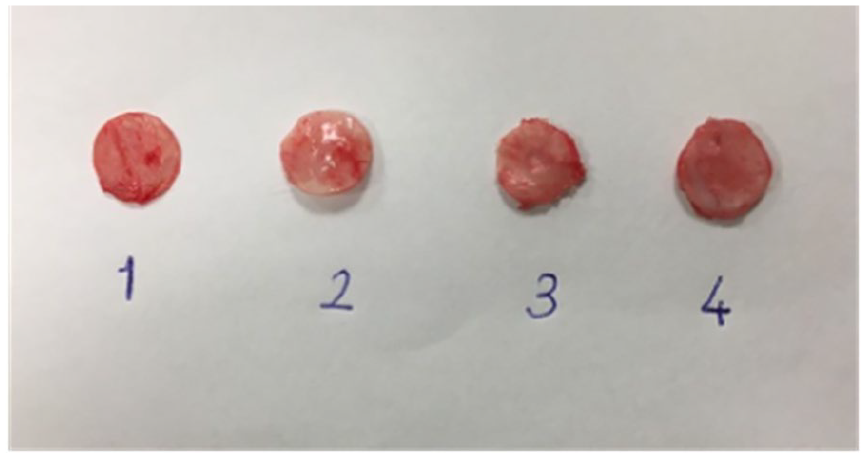
The view of the 4 cartilage grafts removed at the end of the third month.
The removed specimens were fixed with 10% formaldehyde and preserved appropriately for pathological examination.
Histopathological Examination
The materials were passed through graded alcohols in a fixed vacuum tissue tracking device and a paraffin station and embedded in paraffin using the routine method. Three to 4 μm thick sections were obtained from paraffin blocks with slide microtome knives.
The study’s histopathological preparation and evaluation procedures were conducted in ESOGÜ Faculty of Medicine, Department of Pathology. Two pathologists performed all histopathological evaluations. Prepared sections were stained with hematoxylin, eosin, and Masson Trichrome. The preparations were coded by writing various numbers and letters instead of group names. Pathologists were not informed which group the practices belonged to. The trials were randomly distributed to pathologists. It was planned to prevent subjective evaluation during pathological examination by coding group information before histopathological examination.
All tissue samples were evaluated under a light microscope. Cartilage graft microstructure, viability, and tissue reaction were considered after PRP and PDS plates were applied together. The samples were examined on 7 parameters:
Histopathological cartilage viability score
Presence and severity of fibrosis
Chondrocyte nucleus loss
Peripheral chondrocyte proliferation
Physical measurement of material
Presence, type, and severity of inflammation
Evaluation of foreign body reaction
Histopathological cartilage viability score was previously determined by Cakmak et al. 20 It was done with the scoring system used in his study (Table 1).
Histopathological Cartilage Viability Scoring.

The presence and severity of fibrosis were evaluated as minimal: 1, moderate: 2, and severe: 3.
The presence of inflammation was evaluated as acute and chronic, while the severity of inflammation was assessed as mild, moderate, and severe.
Chondrocyte nucleus loss and peripheral chondrocyte proliferation scoring were evaluated according to the following parameters.
Those between 0% and 25%: 1 (+),
Those between 25% and 50%: 2 (++),
Those between 50% and 75%: 3 (+++),
Those who are 75% and above: 4 (++++)
The physical properties of the cartilage grafts were evaluated by measuring the 3 mm hole drilled in the middle of each cartilage. The resulting changes were noted and then statistically analyzed.
Evaluation of foreign body reaction was noted as present or absent.
Histopathological cartilage viability score in the specimens, presence, type, and severity of inflammation, presence and severity of fibrosis, chondrocyte nucleus loss, peripheral chondrocyte proliferation, and physical measurement parameters of the material were used to compare all groups. Two-way analysis of variance was used to compare groups in terms of parameters. Tukey test was used as a multiple comparison test when determining the difference between groups. To reach the groups according to categorical parameters, exact chi-square analysis was employed using the Pearson chi-square and Monte Carlo methods. All analyses were performed using the SPSS 21.0 package program (IBM, Armonk, USA). In the study, a probability value of P < .05 was considered significant.
Results
Our experimental study was concluded with 15 animals as planned. No animals died or became ill during the 1, 2, and 3-month follow-up.
In the study, 5 rabbits studied in the first month, 5 rabbits studied in the second month, and 5 rabbits studied in the third month were compared. The values in the cartilage, cartilage+PRP, cartilage+PDS, and cartilage+PRP+PDS groups in each month were compared with each other.
As a result of the histopathological cartilage viability score statistical analysis, a significant difference was obtained between the groups using cartilage+PRP and cartilage+PRP+PDS only in the first month. When both groups were compared, a statistically significant decrease was found in the histopathological cartilage viability score after PDS use. The same result was not obtained when the second and third months were examined. There was no statistically significant difference between the groups in the second and third months. Although it was not statistically significant, the histopathological cartilage viability score was determined to increase in the first and third-month groups where PRP was used along with cartilage. In the second month, contrary to expectations, a decrease in the histopathological cartilage viability score was detected in the other groups compared to the group in which only cartilage was used. Additionally, it was observed that the histopathological cartilage viability score decreased in all groups in the third month, and the score decreased more in the groups containing PDS.
Histopathological Cartilage Viability-Score Statistical Analysis.
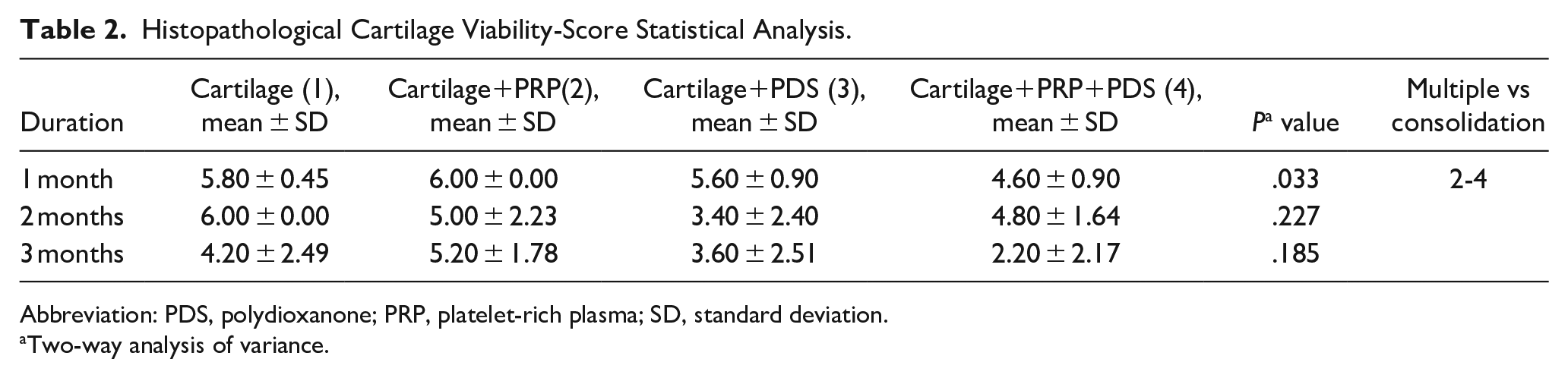
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma; SD, standard deviation.
Two-way analysis of variance.
A statistically significant difference was found between the groups in the first and third months when fibrosis was examined. In the comparison between the groups, it was determined that there was an increase in fibrosis in the PDS plate-used groups in the first and third months compared to the other groups. No statistically significant value was detected regarding the effectiveness of PRP on fibrosis. Additionally, a high fibrosis rate was noted in all groups at 1 month.
Statistical Analysis of the Presence of Fibrosis.
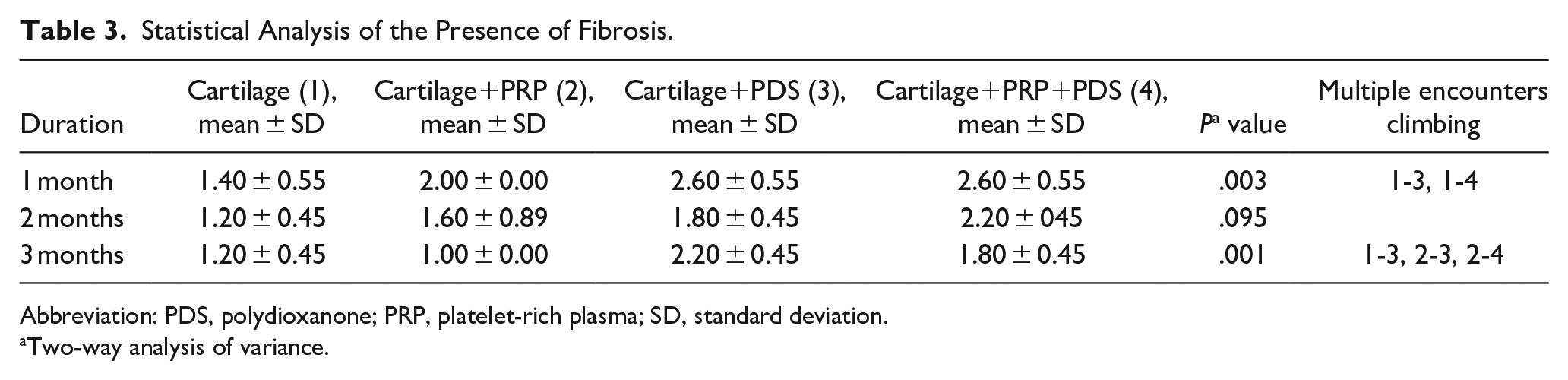
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma; SD, standard deviation.
Two-way analysis of variance.
No statistically significant difference was detected between the groups when examining the amount of chondrocyte nucleus loss.
Statistical Analysis of Chondrocyte Nucleus Loss.
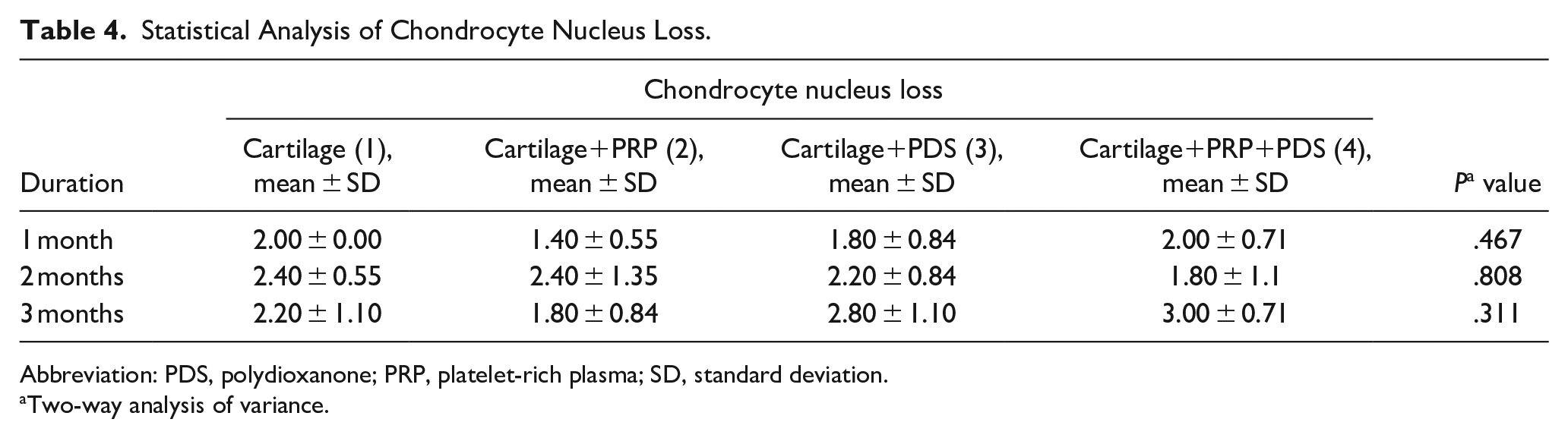
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma; SD, standard deviation.
Two-way analysis of variance.
Statistical Analysis of Peripheral Chondrocyte Proliferation.
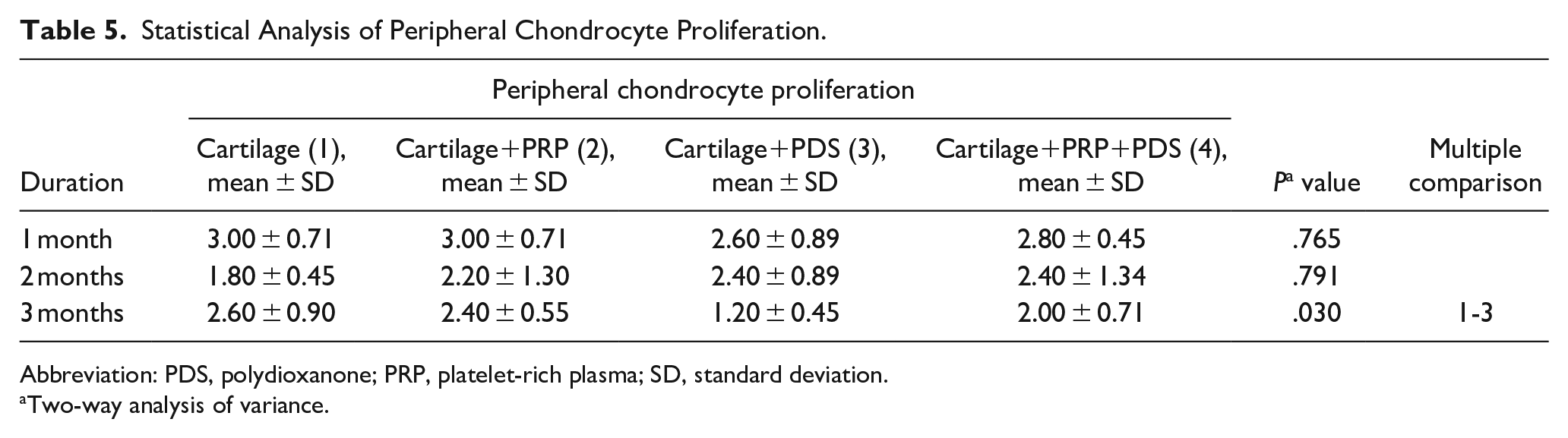
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma; SD, standard deviation.
Two-way analysis of variance.
In examining peripheral chondrocyte proliferation, a statistically significant difference was found only in the third-month comparison. When the group using only cartilage was compared with the group using cartilage+PDS, it was determined that peripheral chondrocyte proliferation was significantly reduced at the end of the third month with PDS. It was also noted that in all groups, there was maximum peripheral chondrocyte proliferation in the first month, and this value decreased in the following months.
All removed implants were initially assessed macroscopically to determine whether or not the space in the middle of the cartilage grafts was filled. It was noted that the gap in the middle was closed entirely in many cartilage grafts. It was determined that the space in the middle of the cartilage grafts was filled more in the group where cartilage+PRP was used in all months compared to the other groups. As a result of our macroscopic observation, the space in the middle of 5 out of 5 preparations in the second -month group using cartilage+PRP was filled.
In the Macroscopic Analysis of Materials, the Gap in the Preparations Evaluation of the Occupancy Rate.
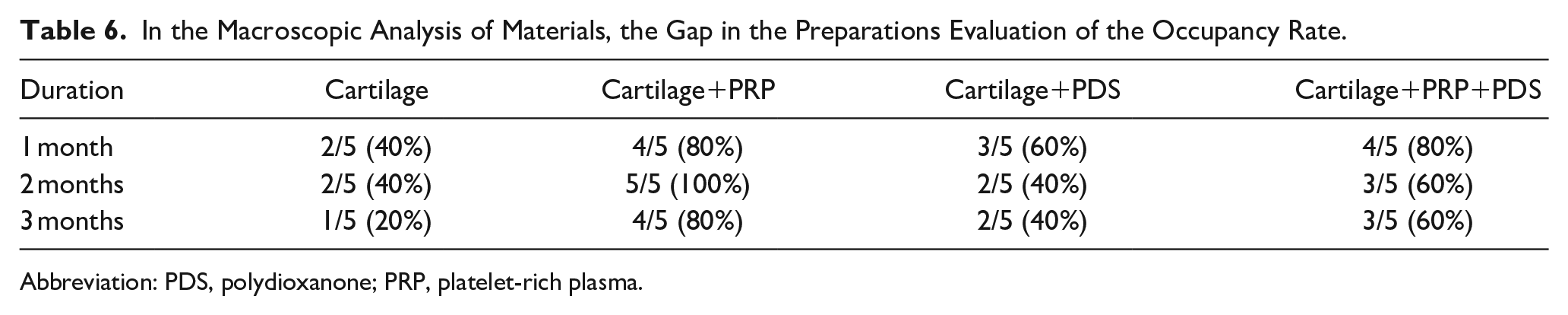
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
In physical measurement of the material, It was measured how much of the 3 mm (3000 µm) gap opened in the middle of each cartilage was closed. A total of 3 specimens, 1 of 20 models in the first month, 1 of 20 samples in the second month, and 1 of 20 specimens in the third month, could not be evaluated by pathology due to deformation and shape change of the cartilage. These non-evaluable cartilages were not included in the statistical analysis of physical measurement data.
While histopathological evaluation was carried out during the physical measurement of the materials, vertical sections were taken from the preparations, and how much of the space in the middle was filled was calculated. No statistically significant difference was found in comparing the physical measurement of the material between the groups.
Statistical Analysis of Physical Measurement of the Material.
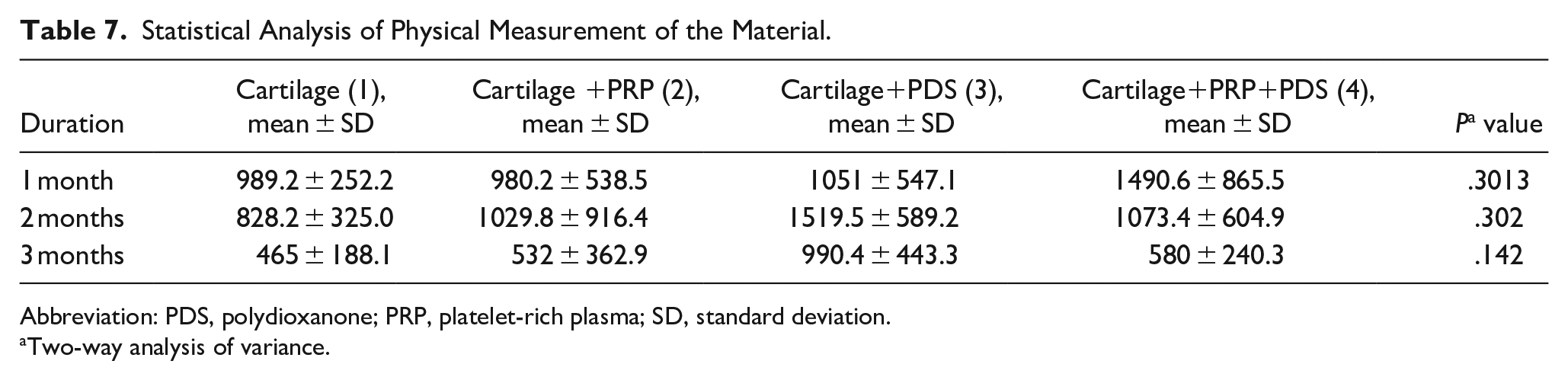
Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma; SD, standard deviation.
Two-way analysis of variance.
Inflammation type was evaluated as chronic in all groups. No statistically significant relationship was observed when the relationship between the severity of inflammation and the groups was assessed. Although no statistically significant difference was observed, it was determined that the severity of inflammation increased in the groups in which the PDS plate was used each month.
Evaluation of Inflammation Severity for the First Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .624.
Evaluation of Inflammation Severity for the Second Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .050.
Evaluation of Inflammation Severity for the Third Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .866.
As a result of the evaluation of foreign body reaction, significant differences were observed between the groups in all months. The data obtained were statistically significant. The rate of foreign body reactions increased in all groups where a PDS plate was used in all months.
Evaluation of Foreign Body Reaction for the First Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .013.
Evaluation of Foreign Body Reaction for the Second Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .000.
Evaluation of Foreign Body Reaction for the Third Month.

Abbreviation: PDS, polydioxanone; PRP, platelet-rich plasma.
Pearson chi-square P-value: .015.
Discussion
Various studies are ongoing to improve cartilage graft viability and clinically estimate long-term graft predictability. Recently, cartilage wrapping the graft with fascia, acellular dermal matrix, or amniotic membrane, treating the graft with adipose-derived stem cells, reducing the crush level, performing subcutaneous growth factor injections, and covering the cartilage graft with hyaluronic acid are the primary studies conducted on this subject.6-12
Many studies in the literature examine the effect of PRP on cartilage.16-19 These studies were conducted mainly by ear-nose-throat, plastic and reconstructive surgery, orthopedics, neurosurgery, and dermatology researchers. As a result of these studies, successful results have been obtained in different subjects, such as bone, wound, tendon, cartilage healing, and skin rejuvenation.16-19
In the literature, the rabbit model was primarily used in studies on cartilage and its survival. 20 It is stated that the 3-month follow-up period for studies using cartilage in rabbits is equivalent to 1 year in human life. 21 Cartilage graft studies in rabbits showed no difference between 2 and 4 months, and a 2-month follow-up period was sufficient if the rabbit’s lifespan was considered. 21 In our study, we chose 1, 2, and 3 months as the longest for the survival of the cartilage we used in rabbits.
The effect of PRP on cartilage regeneration has been investigated by many researchers.17,19 In several recent studies in the literature, the proliferative effects of PRP on articular cartilages, synoviocytes, and meniscus cells were investigated.17,18 An increase in cartilage regeneration and a decrease in graft degeneration have been reported in femoral osteochondral grafts treated with PRP. 19
Significant results were obtained in the study conducted, in which the effect of PRP applied subcutaneously on the ear cartilage was investigated in an animal model. As a result of the study, when the cartilage weight loss between the groups was examined, although it was not statistically significant, the loss of cartilage weight in the groups using PRP was less than in the other groups. They also found an increase in the histopathological cartilage viability score in all groups using PRP, although it was not statistically significant. 17 As a result of our study, contrary to the information in the literature, we did not detect a statistically significant effect of PRP on cartilage regeneration. No statistically significant difference was obtained in the histopathological cartilage viability score when comparing the PRP groups with the other study groups. When the results were examined, an increase in the histopathological cartilage viability score was detected in the groups using PRP in the first and third months. However, it was not statistically significant, while a decrease in the histopathological cartilage viability score was detected in the second month after PRP use. The low number of rabbits in the groups was probably responsible for our failure to obtain statistically significant values. Due to the ethics committee’s application, the number of experimental animals in the groups was determined to be 5. In future studies, the number of experimental animals in the groups can be increased by reducing the number of groups and periods. Different and statistically significant values can be obtained in new studies by increasing the number of experimental animals in the study groups.
Many studies have been conducted on PDS plates in the literature.6,7,10,11 When we look at these studies, they mainly focus on the support and stability that the PDS plate provides to the cartilage structure in septoplasty/septorhinoplasty operations. A few studies examined cartilage/cartilage histopathology change using a PDS plate.
In the study by Boenisch and Nolst Trenite, 11 when a PDS plate was used together with a cartilage graft, it was stated that the PDS plate increases cartilage regeneration by acting as a temporary layer during the regeneration process. They also found that crushed cartilage became more mature due to renewal, and the transformation was almost similar to the original cartilage. 10 As a result of the study, they reported that they did not experience any foreign body reaction or infective process due to the complete resorption of the PDS plate.
In another study by Boenisch and Nolst Trenite, 11 they found that the rate of secondary deviation decreased and long-term stabilization increased with a PDS plate. Early inflammatory reaction, bleeding, septal abscess, or necrosis due to PDS plate use. They stated that they did not encounter complications such as implant rejection, septal perforation, or thickening of the septum mucosa in the long term. 11
It was revealed that a PDS plate is a practical material to support septal cartilage and L-strut. Using a PDS plate for rigid support instead of autologous rib cartilage, It has been reported that patient morbidity is reduced, surgical scars are reduced, and surgery time is shortened. They said they did not experience any foreign body reaction or septal abscess-like inflammatory process after using the PDS plate. Additionally, it has been reported that fewer cartilage resections were performed using PDS plates in the pediatric patient group.
In the study by Dayan and Ashourian, 6 significant results were obtained on the support and stability provided to the cartilage structure using the PDS plate in septoplasty/septorhinoplasty operations. 6 It has been stated that long-term stability increases when the 0.25 mm PDS plate is used together with the caudal septal extension graft in the endonasal septorhinoplasty operation. It has been reported that the technical difficulties that arise during the placement of septal extension grafts in endonasal surgery can be overcome more efficiently with a PDS plate. It has been stated that with the help of the PDS plate, the stability of the type projection increases in long-term follow-ups, and the variations that may occur in the long term are reduced. As a result of the study, it was reported that the PDS plate did not increase morbidity despite its success.
PDS plate was found to be effective and reliable. 1 It has been reported that cartilage loss occurred in the septum in the postoperative period in patients with a PDS plate, resulting in saddle nose deformity. As a result of our study, we did not detect a statistically significant effect of PDS plate use on cartilage regeneration. When the data were examined, we found that the histopathological cartilage viability score did not increase in the newly formed cartilage using a PDS plate; on the contrary, it decreased. As a result of histopathological cartilage viability score statistical analysis, a significant difference was obtained between the groups using cartilage+PRP and cartilage+PRP+PDS in the first month. When both groups were compared, it was determined that there was a statistically significant decrease in the histopathological cartilage viability score after using the PDS plate. When peripheral chondrocyte proliferation was examined using a PDS plate, we found that peripheral chondrocyte proliferation decreased statistically significantly at the end of the third month. When the relationship between PDS plate use and chondrocyte nucleus loss was examined, we could not reach a statistically significant result. These results are compatible with recent studies on the reliability of the PDS plate. 1 Our study will provide a new perspective to the literature regarding the effectiveness and reliability of PDS plates on cartilage regeneration.
As a result of our study, we obtained significant effects when we examined the groups in which the PDS plate was used. Significant macroscopic changes occurred in the groups in which the PDS plate was used. We found that in the groups where the PDS plate was used, the cartilage grafts changed shape, some cartilage grafts folded on themselves, and degenerative changes occurred in the cartilage. In the physical measurement title, a total of 3 specimens, 1 of 20 models in the first month, 1 of 20 specimens in the second month, and 1 of 20 specimens in the third month, could not be evaluated by pathology due to deformation and shape change of the cartilage. While removing the graft material containing a PDS plate, we observed that the graft integrated with the subcutaneous tissue and almost came out of the skin. Although the same surgical steps and techniques were used during the transplantation of all cartilage grafts, deformation and shape changes occurred in 3 cartilage grafts. These observations regarding the PDS plate were probably due to the foreign body reaction against the PDS plate and the secondary fibrosis. The results obtained after histopathological examination were compatible with this observation. In the groups where the PDS plate was used, foreign body reactions were significantly increased in all months compared to the other groups. However, there was an increase in the fibrosis rate in the groups where the PDS plate was used.
In comparison between groups, we found a statistically significant increase in the fibrosis rate in the PDS plate-used groups in the first and third months compared to the other groups. Although we detected an increase in foreign body reaction and fibrosis after PDS plate use, we did not see a statistically significant relationship between PDS plate and inflammation severity. These results have provided essential data to the literature regarding the development of foreign body reactions and fibrosis after using a PDS plate.
We performed an application that has yet to be tried in the literature to observe cartilage regeneration and the extent to which the gap on cartilage grafts is closed. To ensure standardization in evaluating renewal and physical change in cartilage grafts, we equalized the cartilages with 10 mm punch biopsy pens. Then, we drilled a hole in the middle of the equally sized cartilage with a 3 mm punch biopsy pen. Standardized cartilages of equal sizes with a perforation in the middle were created. In this way, it was determined how much of the space in the middle of the cartilage grafts was filled at the end of the working period. We made critical macroscopic observations while the cartilage graft materials were removed from the pockets at the end of the first, second, and third months. In the groups where PRP was used, the 3 mm space in the middle of the grafts was filled to a greater extent in all months compared to the other groups. In most of the preparations in the groups where PRP was used, the gap in the middle of the graft was closed entirely, and its back was not visible. We also examined this macroscopic subjective observation under the title of physical measurement, which is one of the histopathological study parameters. In our macroscopic observations, although a positive effect on cartilage regeneration was observed in the groups where PRP was used, no significant difference was obtained between the groups when the statistical results were examined. We attributed this result to the fact that although the graft materials prepared before the study were standardized, the sections obtained from the graft material in the physical measurement examination at the end of the study could not be standardized. Although the aim was to take sections from the middle point of the cartilage grafts during the histopathological examination, we thought the areas obtained might have been taken from the upper or lower part of the preparations because the cartilage regeneration rate of the units taken in the histopathological examinations of the preparations was not the same everywhere. The fact that cartilage regeneration is not homogeneous and at the same rate at all sections’ levels is an essential factor in obtaining these results.
Previous studies in the literature reported that the PDS plate remained intact for approximately 2 to 3 months and was utterly resorbed in 5 months.20-22 During this period, it has been accepted that the PDS plate acts as a temporary layer and supports cartilage regeneration. Boenisch and Nolst Trenite. 11 In their study, they reported that the PDS plate remained intact in the body for approximately 10 weeks, and it took about 5 months to absorb completely. In our study, different results regarding the resorption time of the PDS plate were obtained from the literature. The PDS plate was macroscopically almost completely resorbed in most specimens in the second month. In our study, no application different from the literature was used that could cause this period to be other.
All stages of the study were carried out under sterile conditions, and antibiotic prophylaxis was applied to all experimental animals in the postoperative period. When the literature is examined, the PDS plate was used in both human and experimental animal studies, and no different information was reported about the resorption time of the PDS plate in any of these studies.10,11,23 In these studies, PDS plates were used in many different parts of the body in human or experimental animal studies. We did not find any other information about the resorption time of the PDS plate in any of the studies placed in the skull, septum, or subperiosteally.24,25 We found the resorption time of the PDS plate to be shorter than the literature because we thought it might be due to the severe foreign body reaction against the PDS plate and the fibrosis that developed secondary to this. The results obtained after histopathological examination were compatible with this observation. In the groups where the PDS plate was used, foreign body reactions were significantly increased in all months compared to the other groups. However, there was an increase in the fibrosis rate in the groups where the PDS plate was used. In tissue-compatible implants, a foreign body reaction, albeit limited, may occur after implantation. However, Boenisch and Nolst Trenite 11 did not observe any significant foreign body reaction to the PDS plate in their study. Our study reached a different result than the literature regarding the absorption time of the PDS plate due to the severe foreign body reaction against the PDS plate. Our study provides additional information to the literature regarding PDS plate absorption time.
Conclusion
Our study hypothesized that using PRP and PDS plates with cartilage graft material may synergistically affect cartilage regeneration and histopathological properties. As a result of our study’s histopathological cartilage viability score statistical analysis, a significant difference was obtained between the groups using cartilage+PRP and cartilage+PRP+PDS only in the first month.
When both groups were compared, a statistically significant decrease was found in the histopathological cartilage viability score after PDS use. In examining peripheral chondrocyte proliferation, a statistically significant difference was found only in the third-month comparison. When the group using only cartilage was compared with the group using cartilage+PDS, it was determined that peripheral chondrocyte proliferation was significantly reduced at the end of the third month with PDS use. In evaluating the presence and severity of fibrosis and foreign body reaction, a statistically significant increase was detected using a PDS plate. No statistically significant difference was found between the groups using PRP and PDS plates regarding the presence, type, and severity of inflammation, chondrocyte nucleus loss, and physical measurement of the material.
In our study, in the groups where PRP was used, we saw that PRP had a macroscopic positive effect on cartilage regeneration. Still, we could not detect statistically significant findings when the data were examined. The result we obtained was different from the results of previous studies in the literature. The low number of rabbits in the groups was probably responsible for our failure to get statistically significant values. Different and statistically substantial values can be obtained in new studies by increasing the number of rabbits in the study groups.
Degeneration in the cartilage structure was observed macroscopically in the specimens where the PDS plate was used. There was shape change and cartilage deformation in the PDS plate specimens. When the results were examined, this observation coincided with the statistically significant increase in foreign body reaction and fibrosis in the PDS plate groups. However, these results contradicted our hypothesis before the study and the information in the literature.
These results provided different information to the literature. Our results will help provide preliminary information and guidance for future studies and offer a different perspective.
Footnotes
Acknowledgements
This article was produced from the ENT Specialty thesis of Ahmet Halit Aydın.
Author Contributions
The authors equally contributed to the planning, literature survey, experiment, and manuscript writing.
Availability of Data and Materials
All data for this study is presented in this paper.
Data Availability
All data obtained were presented in this article. Further inquiries may be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was carried out with the approval of Eskişehir Osmangazi University Animal Experiments Local Ethics Committee (HADYEK) dated June 20, 2017 and registration number 583-2/2017.
Ethics Committee Approval
Ethics committee approval was obtained from Eskişehir Osmangazi University Animal Experiments Local Ethics Committee (HADYEK) dated June 20, 2017 and registration number 583-2/2017.
Informed Consent
Not applicable.
