Abstract
Empty nose syndrome (ENS) is a rare entity in patients who undergo sinonasal surgery due to over-resection of the turbinate. This syndrome leads to debilitating symptoms that include dry nose, painful nasal breathing, paradoxical nasal obstruction, crusting, and sleep disorder. The goal of surgical treatment is to reestablish the volume of the turbinates to rehabilitate the nasal resistance. Endonasal microplasty with cartilage implants on the lateral wall of the nasal cavity is useful for creating the neoturbinate. Here, we present 2 cases that describe the management of empty nose syndrome by endonasal microplasty using platelet-rich fibrin (PRF) scaffolds embedded with a diced cartilage graft. The integration of the PRF scaffolds with diced cartilage efficiently facilitated the reestablishment of the neoturbinate. This autologous biomaterial is suitable for the treatment of ENS.
Keywords
Introduction
Empty nose syndrome (ENS) is a rare debilitating disease characterized by dry nose, painful nasal breathing, paradoxical nasal obstruction, crusting, and sleep disorder. 1 ENS, first described by Eugene Kern in 1994, is an iatrogenic disorder resulting from over-resection of the turbinate during sinonasal surgery. Apart from nasal symptoms, patients with this crippling disorder usually experience anxiety and depression, which can even lead to suicidal ideas. 2 The diagnosis of ENS is difficult because not every patient experiences these debilitating symptoms after a radical turbinate procedure, and physicians can only make a diagnosis based on the subjective symptomatology. The Empty Nose Syndrome 6-item Questionnaire (ENS6Q) is a new and feasible measurement for identifying patients suspected of developing ENS. 3 A positive result of a cotton test is helpful for diagnosis and indicates its suitability for surgical treatment.
The aim of surgical intervention for ENS is to rebuild an increased nasal resistance by narrowing the airway with an implant secured within a submucosal pocket, resulting in the regeneration of the nasal mucosa, retention of more moisture in the nasal cavity, and an increase in the inspired air via laminar flow into the nasopharynx.4,5 Various materials, such as injectable fillers, synthetic implant materials (e.g., Plastipore, Gore-Tex, and hydroxyapatite), allograft materials (e.g., Alloderm and BoneSource), and autologous materials (e.g., bone, cartilage, muscle, and fat), have been used for surgical reconstruction of the missing turbinate.5,6
At present, research in tissue engineering and regenerative medicine is focusing on the development of cost-effective biomaterial scaffolds for the regulation of inflammation and the enhancement of wound healing processes. Platelet-rich fibrin (PRF) is an autologous biophysical and biochemical product that delivers growth factors, cytokines, and immune/stem-like cells for immunomodulation and tissue healing. The use of PRF has recently been recognized as a promising strategy for improving functional cartilage regeneration processes, including the enhancement of migration, proliferation, viability, and differentiation of chondrocytes.7,8 To our knowledge, this is the first study to address the clinical application of PRF and diced cartilage for reconstruction of the missing turbinate.
Surgical technique
The operations were performed under general anesthesia in an operating room. Conchal cartilage was harvested via the postauricular approach and then diced to make a sticky cartilage (Figure 1(A)). A venous blood sample was taken without anticoagulant into 10-mL sterile tubes and immediately centrifuged at 3600 rpm for 6 min. The supernatant was mixed with diced cartilage, and polymerization was allowed to proceed for 5–10 min for the fibrin network to stabilize the diced cartilage graft (Figure 1(B)). Illustration of the surgical technique: (A) The harvested conchal cartilage was diced to make a sticky cartilage. (B) Platelet-rich fibrin fluid was mixed with the diced cartilage, and the fibrin network stabilized the diced cartilage graft 5–10 minutes later. (C) The submucosal pocket was filled with the sticky cartilage graft. (D) The incision wound of the mucosa was covered with Hemopatch® to keep the cartilage implant in position.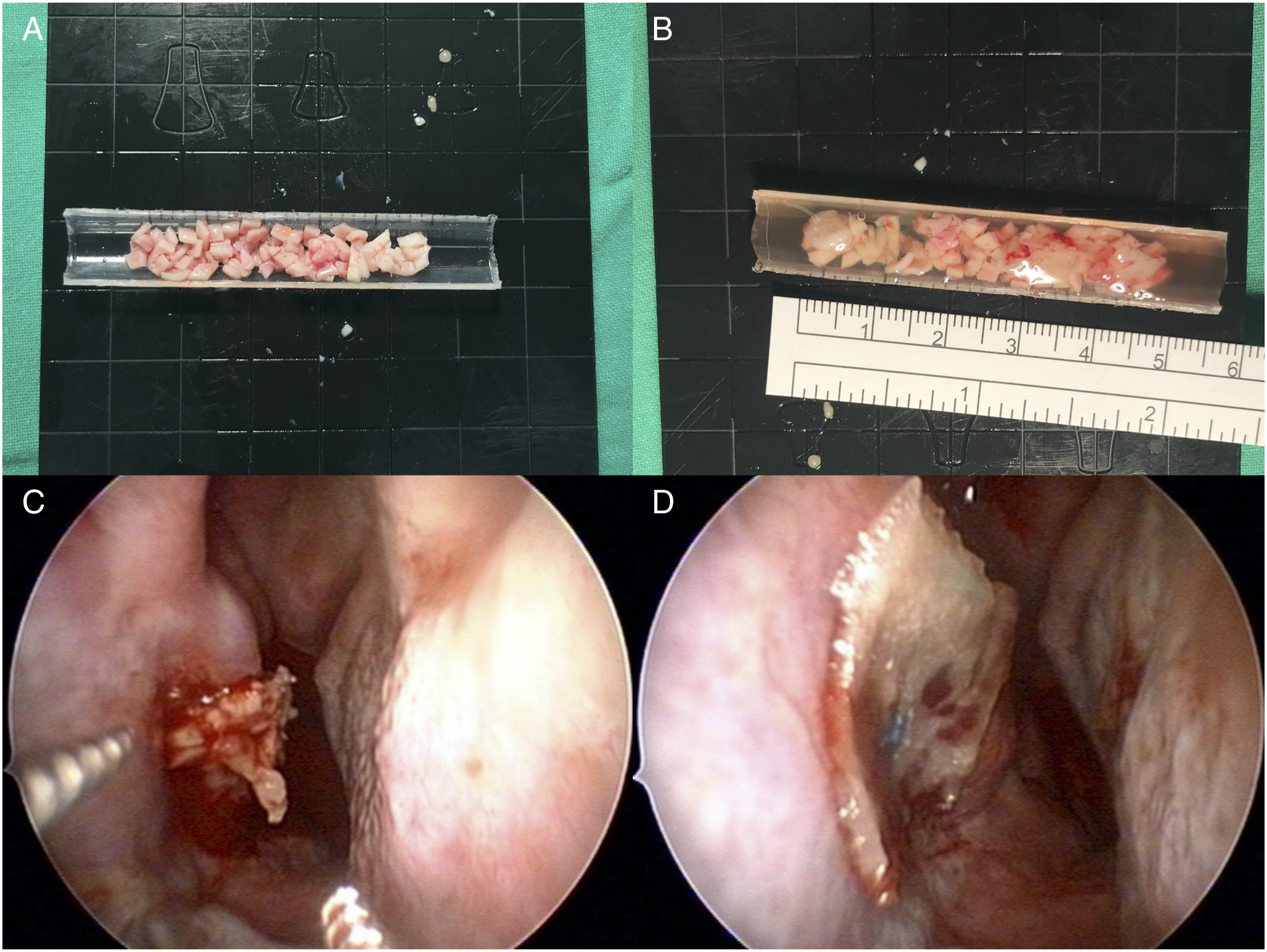
A submucosal pocket was made below the inferior turbinate remnant with a Freer septum knife. The mucoperiosteal flap was completely elevated as a tunnel into the lateral nasal wall, and the pocket was then filled with the sticky cartilage graft (Figure 1(C)). The mucosal incision wound was covered with Hemopatch® (polyethylene glycol–coated collagen patch, Baxter) to maintain the cartilage implant in position (Figure 1(D)).
Case reports
Case 1
A 32-year-old man had undergone septomeatoplasty at another hospital 5 years previously for assessment of his nasal obstruction. After surgery, the patient complained of persistent breathing symptoms, nasal congestion that was stuffier on the right side, oral mucosal dryness, poor sleep, and headache on inspiration. An examination revealed that only the caudal part of the patient’s right inferior turbinate remained (Figure 2(A)). The patient’s preoperative score on the ENS6Q was 18, and a positive cotton test was noted. Thus, an endoscopic inferior turbinate augmentation procedure was suggested. (A) Endoscopic view of the preoperative condition. Only the caudal part of the patient’s right inferior turbinate was retained. (B) Endoscopic view of the postoperative condition 1 year later, showing the submucosal implant secured on the lateral nasal wall.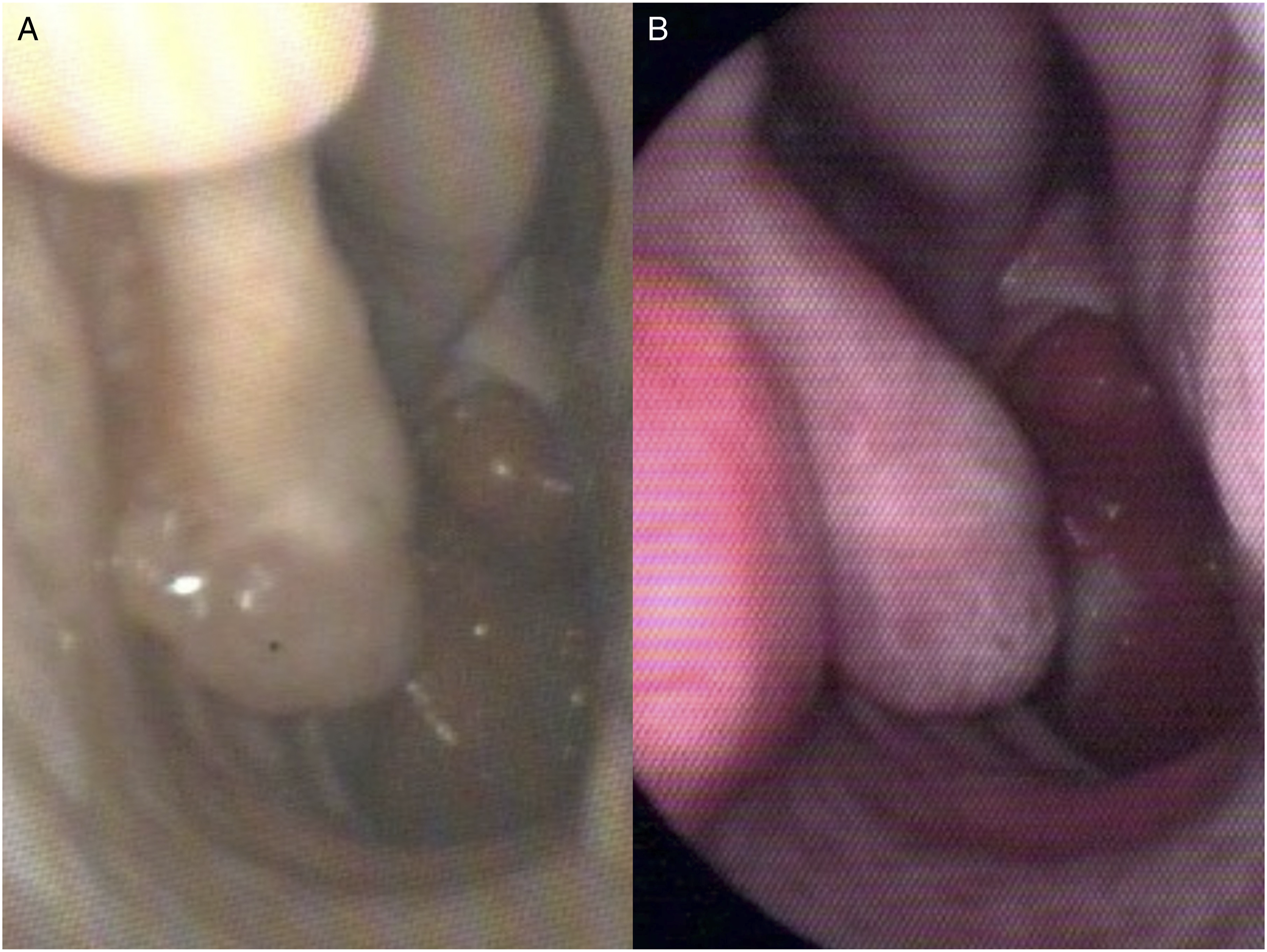
After the operation, the patient felt his breathing was improved, with a postoperative score of 4 on the ENS6Q, and the symptoms of headache and airway dryness were resolved. The conchal cartilage implant is still in place at an adequate amount at 1 year of follow-up (Figure 2(B)).
Case 2
A 55-year-old man had undergone endoscopic sinus surgery, septomeatoplasty, and uvupalatopharyngoplasty at another hospital 7 years previously. In the most recent 2 years, he had developed facial pressure and headache on inspiration, suffocation, and waking up at night with a strange loud snoring. At the same time, the patient felt anxiety and depression and had difficulty concentrating on his daily work. An examination revealed a total resection of the patient’s bilateral inferior turbinates totally (Figure 3(A)). The patient’s preoperative ENS6Q score was 23, and a positive cotton test was noted. Thus, an endoscopic microplasty of the inferior turbinate was arranged. After the operation, the patient felt more energetic and slept better. The strange loud snoring disappeared, and he had a postoperative ENS6Q score of 8. The headache, facial pressure, and sadness were resolved. The conchal cartilage implant remains secured in place at an adequate amount after 1 year of follow-up (Figure 3(B)). (A) Preoperative right nasal cavity with the remnant inferior turbinate. (B) The submucosal implant persists reliably on the right lateral nasal wall.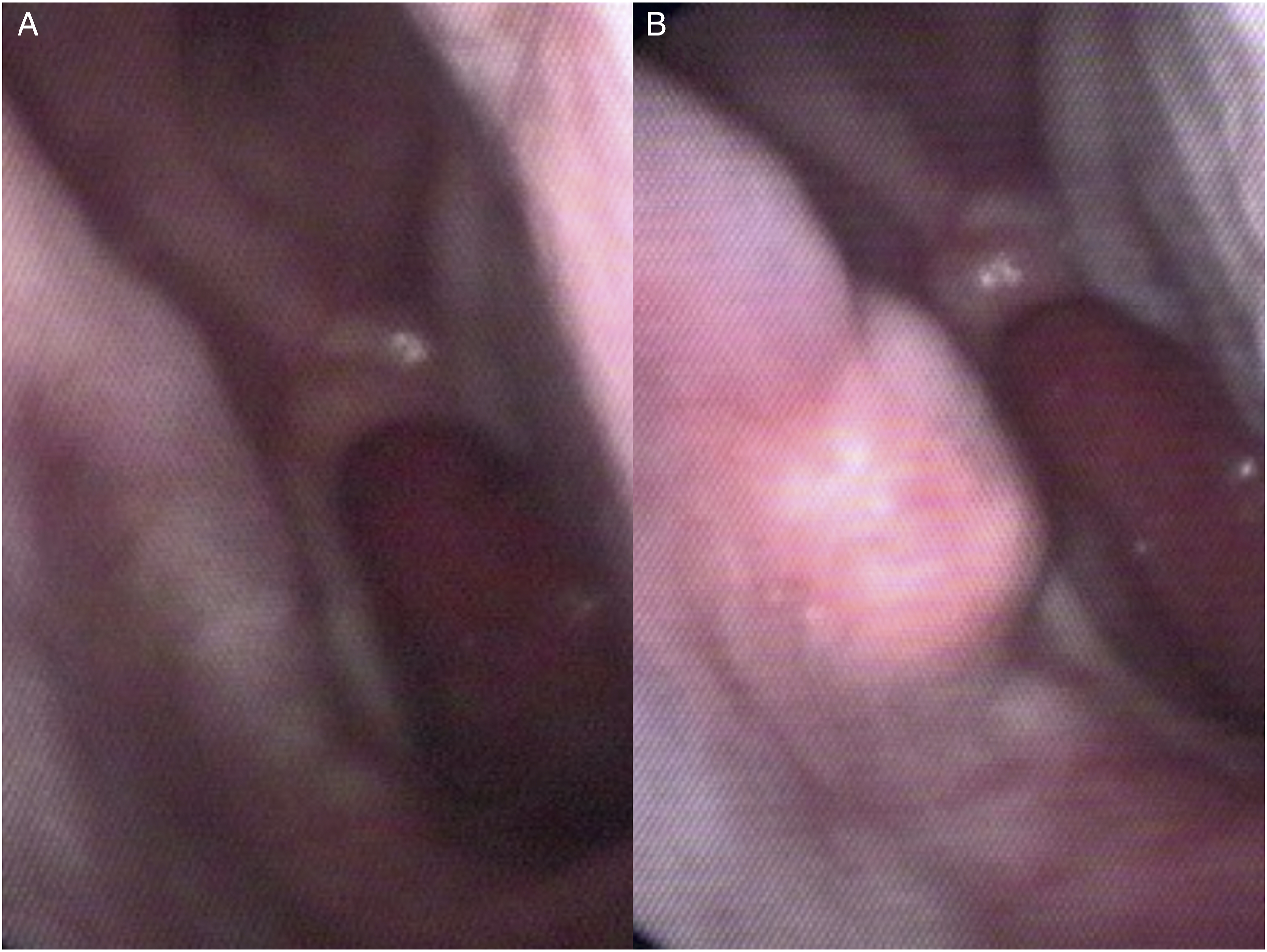
Discussion
Many treatment modalities are available for ENS, but resolving all the patient’s debilitating symptoms is difficult. The goal of medical therapy focuses on the moisturization of the nasal cavity by nasal irrigation and the application of nasal moisturizing ointments to relieve the mucosal dryness, prevent crusting, and decrease facial pain. However, the results of conservative treatment are poor in most ENS patients. 5 Surgical treatments that reestablish the volume of the turbinates in order to rehabilitate the nasal resistance can include filler injections or submucosal implantation of synthetic biomaterials, allograft materials, and autologous materials. The acceptance of surgical management for ENS has increased recently with advances in endonasal microplasty techniques and with growing evidence of favorably improved outcomes in nasal symptom scores and in comorbid mental health domains.5,6
Jang 4 and Jung 5 reported significantly improved results in their studies on endonasal microplasty with cartilage implants, and they indicated that cartilage is an ideal material, with long-term positive outcomes, for nasal implantation. They also concluded that the lateral wall of the nasal cavity was most suitable for placing the cartilage implant, and placement on the caudal one-third to one-half of the nasal cavity was sufficient for creating the neoturbinate.
Several recent studies in humans and animals have identified PRF as a promising biomaterial for improving functional cartilage regeneration. Bioactive growth factors and cytokines, including transforming growth factor ß (TGF-ß), basic fibroblast growth factor, insulin-like growth factor 1 (IGF-1), vascular endothelial growth factor, and platelet-derived growth factor, from the PRF in the scaffolds can stimulate the proliferation of chondrogenic cells, the deposition of cartilaginous extracellular matrix, and the upregulation of chondrogenic gene expression.7,8 The natural three-dimensional fibrin structure is also favorable for the maintenance and continued release of growth factors and cytokines over time.9,10
Platelet-rich fibrin has been widely investigated for cartilage repair, but the potential variation of PRF products between different preparations and patients may be a challenge. The effects of PRF on biological activities depend on the amounts of platelets and the growth factor concentrations, and as these vary from person to person, they may influence the therapeutic effects.7,9 The variation in platelet derivatives from different human populations is therefore an inevitable limitation of PRF. However, with a standard preparation protocol, we can still obtain essential growth factors within acceptable concentration ranges among different patients.
The fibrin glue of PRF provides growth factors and cytokines to create a scaffold that allows stabilization and diffusion of nutrients to the cartilage implants. Diced cartilage with PRF has been confirmed as a safe, reliable, and easily reproducible technique for dorsal nasal augmentation in aesthetic and reconstructive rhinoplasty.11,12 To our knowledge, ours is the first study to address the clinical application of PRF and diced cartilage for the creation of a neoturbinate. In our literature review, cartilage implants were usually rolled into a spherical kidney-shaped structure (conchal cartilage) or carved into round shapes to simulate the neoturbinate.4-6 In our study, we diced the cartilage and mixed it with PRF as a gel, and we needed to make just a small opening for implantation. The use of a small opening then allowed us to use Hemopatch® instead of suturing to close the wound. Hemopatch® is a polyethylene glycol–coated pad derived from bovine collagen that has a coagulation and sealing effect. Its properties allow the pad to seal a wound for hemostasis, with hemostatic success achieved in 3 min. 13 Its good adherence to wound surfaces without further bleeding and its ease of use without associated complications provides a rapid, effective, and noninvasive method of hemostasis in our patients.
One drawback of cartilage grafting is the possibility of graft resorption and necrosis after implantation. Optimizing the survival of transplanted chondrocytes is most important for successful cartilage implantation, and the use of PRF in cartilage regenerative medicine is paving the way for new therapeutic strategies that may overcome this limitation. The present cases show that the integration of PRF scaffolds and diced cartilage efficiently facilitates the reestablishment of a neoturbinate. PRF serves as a potent bioactive scaffold that enhances the migration, proliferation, viability, and differentiation of chondrocytes.
Conclusions
The findings of the present study suggest that the combination of PRF scaffolds with diced conchal cartilage grafts is suitable for endonasal microplasty in the treatment of ENS. Further studies are necessary to identify more practical and effective protocols for the PRF manufacturing process and to compare the effectiveness of using PRF scaffolds with diced conchal cartilage grafts and other cartilage implants for the management of ENS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
