Abstract
Introduction
Monoclonal antibodies (mAb) represent nowadays an add-on treatment option that was developed and initially introduced for patients with uncontrolled allergic eosinophilic asthma, 1 targeting specific molecules that participate in the activation of type-2 immune pathways responsible for airways inflammation. Over the past decade, the indications for mAb therapy were broadened; clinical trials initially aimed at asthmatic patients have shown surprisingly positive effects on comorbid chronic rhinosinusitis with nasal polyps (CRSwNP), significantly reducing disease burden.2-5 These findings paved the way for designing new trials to study the effect of mAbs, specifically in patients affected by CRSwNP (eg, LIBERTY SINUS-24 and -52, POLYP-1 and -2, SYNAPSE, OSTRO, etc.),6-9 eventually proving them to be effective. The evidence collected thus led to the approval of these drugs in patients who do not respond to standard medical and surgical treatments. In current clinical practice, the most frequently prescribed mAbs are Omalizumab (anti-IgE), Benralizumab (anti-interleukin (IL)-5 receptor α), Mepolizumab (anti-IL5), and Dupilumab (anti-IL4/IL13 receptor). Their effect in controlling type-2 inflammatory disease of the upper airways has also been reported in real-life scenarios,10-19 with satisfying results in improving nasal obstruction, hyposmia, and rhinorrhea. Although highly effective in most cases, some patients may still lack disease control in the nose and paranasal sinuses despite appropriate management of systemic comorbidities (eg, asthma). Consequently, the need for further surgery, albeit reduced, may still be advocated. 20 Regarding side effects, although none of the approved biologics have shown significant differences between each other or compared to placebo in clinical trials, a recent study from the University of Pennsylvania found that 24% of CRS patients who were on dupilumab for at least 6-month experienced adverse events requiring discontinuation. 21 Another analysis conducted in a real-life setting by the University of British Columbia recently found surprisingly high frequencies of adverse events among patients treated with dupilumab and mepolizumab, affecting 45% and 10% of the entire cohort, respectively. 22 Adverse events or nasal unresponsiveness to mAbs raises the question of what local treatment could be more beneficial, without switching to another mAb. In this scenario, could endoscopic surgery be effective in removing the inflammatory infiltrate that sustains the disease locally, thereby achieving the unmet effect of the mAb? In some tertiary centers, endoscopic sinus surgery (ESS) with non-mucosa sparing technique (eg, as reboot surgery)23-25 is performed with the aim of mechanically removing the inflammatory infiltrate in patients who have undergone multiple mucosa-sparing ESS procedures yet without satisfying results. So far, the results of reboot surgery have been promising; based on the rationale of sinus demucosalization, recent research has investigated the potential benefits of this approach in terms of symptom control and recurrence time in recalcitrant CRSwNP patients. 24 The aim of this study was to assess the outcomes of reboot surgery in a selected subset of patients locally unresponsive to mAb therapy.
Methods
This is a single-center retrospective observational study. Charts from all patients with recalcitrant CRSwNP who underwent reboot ESS in a tertiary rhinology center between January 2020 and January 2024 were reviewed. Inclusion criteria included: (1) having received multiple functional (eg, mucosa-sparing) nasal endoscopic surgeries in the previous years; (2) diagnosis of asthma under control with mAb, according to GINA recommendations (eg, Asthma Control Test ≥20, Asthma Control Questionnaire-5 <1.0, no risk for exacerbations) 26 ; (3) poor response at a nasal level [eg, nasal polyp score (NPS) ≥ 1 and SNOT-22 ≥ 40 or visual analog scale (VAS)-global ≥ 5, consistent with EUFOREA guidelines] 27 ; and (4) being addressed to partial reboot surgery as a rescue treatment for the nasal condition. Lung function was measured with a portable spirometer (CareFusion®, San Diego, CA, USA) 1 month prior to surgery and absence of bronchial obstruction was required for inclusion. Only patients with complete previous surgeries were sent for partial reboot surgery and enrolled in the study. The proper extent of previous ESS was defined as the absence of residual ethmoidal cells on CT-scan and nasal endoscopy. According to the European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS) guidelines, previous surgeries were considered complete if they included anterior and posterior ethmoidectomy, large middle meatal antrostomies, sphenoidotomy, and frontal opening (eg, a Draf IIa procedure), while preserving important landmarks such as the middle turbinate and mucosa in general. 28 No patient had previously undergone expanded endoscopic approaches (eg, Draf III, sphenoid nasalization, medial maxillectomy). Patients were referred for reboot surgery after a multidisciplinary consultation involving various specialists (eg, otorhinolaryngologists, pulmonologists, allergologists, immunologists). To avoid the risk of systemic condition control failure, patients were not recommended to be trialed with different mAbs other than the one they were already receiving. Preoperative examination was performed 2 months prior to surgery: data taken from patients’ charts were preoperative NPS, 22-items sinonasal outcome test (SNOT-22), and visual analog scale (VAS) for nasal symptoms, Lund–Kennedy scores for nasal crusts (LK-c) and synechiae (LK-s), and CT-scan Lund–Mackay score (LMS).29,30 The procedure, as per the original description by Alsharif et al, consisted of clearing all polyps and diseased mucosa down to the periosteum of all paranasal sinuses until the floor of the frontal sinus, to facilitate postoperative restitutio ad integrum of new uninflamed mucosa.23,31 All mucosa is cleared cranially from the skull base, laterally from the lamina papyracea and medially from the medial aspect of the middle turbinate (if preserved from the previous surgery and if preservable in the revision). The same is done in the frontal sinus after performing either a Draf IIb or Draf III, according to the extent of disease in this site; as in Alsharif et al, Draf IIb is done in partial reboot, whereas Draf III in complete reboot. 23 Our entire cohort underwent partial reboot; there is no clear indication for undertaking partial or complete reboot, but due to the limited involvement of the frontal sinuses in our subset of patients (eg, LM score for frontal sinus <2), we opted for the former. Postoperatively, at the 12-month follow-up, NPS, SNOT-22, LK-c, LK-s, and VAS assessment were repeated and compared to preoperative outcomes. As clinical symptoms showed marked improvement, the CT-scan was not repeated postoperatively. Histopathologic samples were collected under local anesthesia 2 months prior to surgery (from the polyp) and repeated 12 months postoperatively (from the middle meatus mucosa). Specimens were stained in May–Grünwald–Giemsa solution and analyzed with optic microscopy at 20× and 40× magnification to assess eosinophilic tissue infiltration. All surgical procedures, as well as follow-up examinations, were performed by the same surgical equip, made up of 2 expert rhinologists. The mAb therapy was not discontinued postoperatively to prevent the risk of systemic disease failure. Preoperative medical therapy consisted also of daily rinses with saline solution multiple times a day, as well as topical steroid (budesonide 0.5 mg/mL) administration via nasal douche (Rinowash, AirLiquide MedicalSystem, Bovezzo, Italy), which were continued in the postoperative period. As adequate control of the systemic condition was required for eligibility, no additional systemic medication other than mAb was assumed by the 2 patients who suffered from eosinophilic granulomatosis with polyangiitis (EGPA) (#1, #9; Table 1). To remove intranasal debris and to monitor surgical site healing, all patients were regularly followed up every 7, 14, 21, and 30 days, then every 3 months for the following year. All patients signed informed consent, and the study was conducted consistent with the declaration of Helsinki and its later amendments. Data analysis was conducted with IBM® SPSS Software for Macintosh, Version 26.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were calculated and expressed in terms of mean and standard deviation (SD) for parametric data. The Shapiro–Wilk test was used to ensure a normal distribution. Differences between continuous parametric variables were assessed with paired-samples t-test. Wilcoxon signed-rank test was used to measure differences between non-parametric variables.
Demographic and Clinical Characteristics of the Cohort.
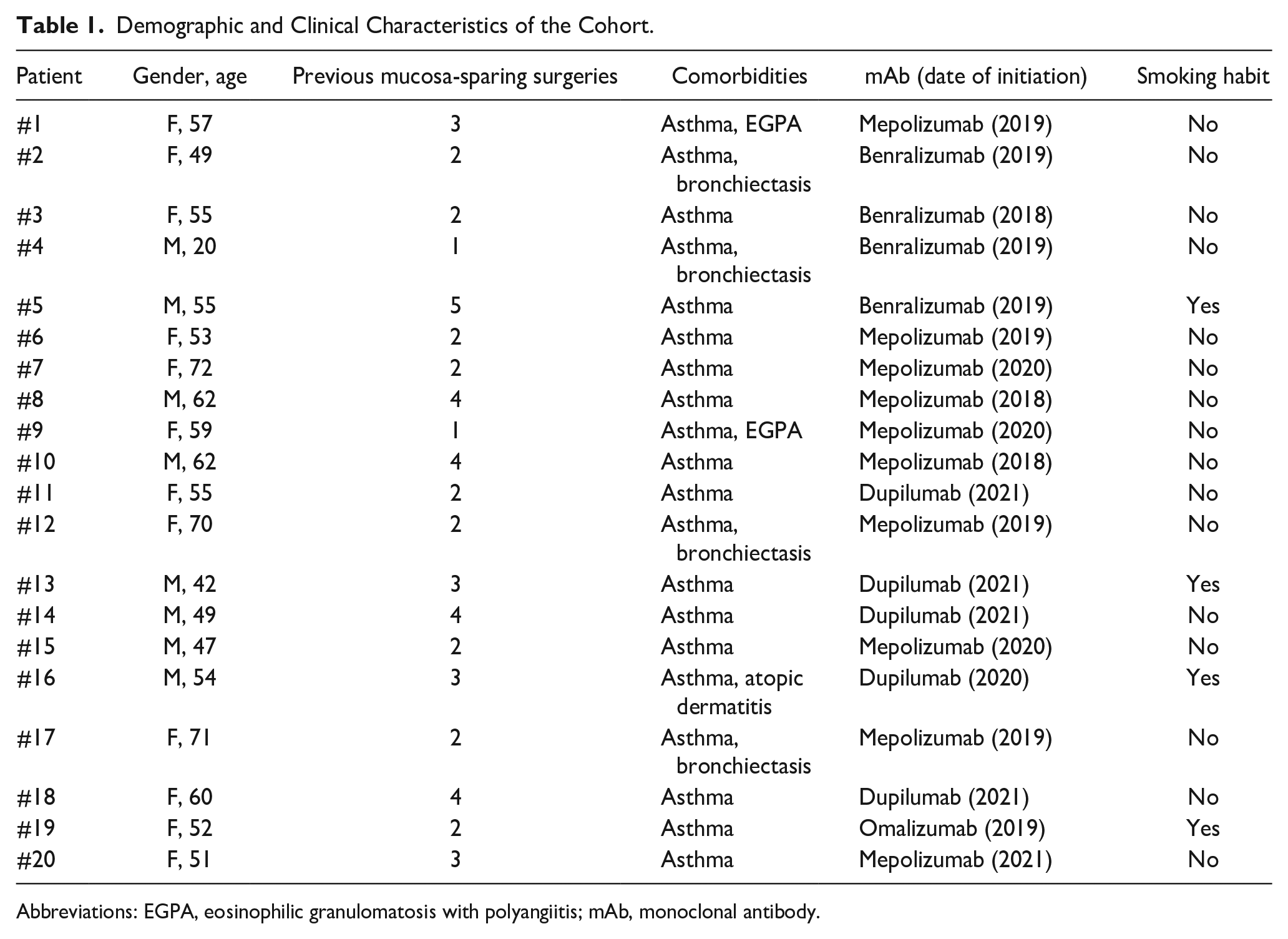
Abbreviations: EGPA, eosinophilic granulomatosis with polyangiitis; mAb, monoclonal antibody.
Results
Out of 1284 patients who received ESS during the considered period, 59 of them (4.60%) underwent reboot surgery; among them, 20 (33.90%; mean age ± SD: 54.75 ± 12.9 years; 12 females) were under mAb treatment and agreed to be enrolled. All patients had undergone biological therapy for at least 1 year prior to referral (mean ± SD: 2.45 ± 1.05 years). Bronchial asthma was the most common comorbid systemic condition for which the molecular drug was indicated (Table 1). All patients were under topical nasal steroids (budesonide); however, most of them (85%, n = 17/20) had self-administered oral (prednisone) and/or intramuscular (triamcinolone) corticosteroids at least once in the 2-year preceding enrollment. Each patient had previously undergone multiple mucosa-sparing surgeries (mean ± SD: 2.65 ± 1.89). Only 2 patients (eg, case #4 and #9) had received comprehensive full-house functional endoscopic sinus surgery (FESS) only once before referral to our center (Table 1). Relapse of nasal symptoms occurred in all patients, on average 7.45 ± 5.74 months after mucosa-sparing ESS they had undergone over time.
Preoperative clinical scores were (mean ± SD): NPS = 4.95 ± 1.70; SNOT-22 = 63.90 ± 19.15; VAS-global (eg, “How bad is my nasal condition overall?”) = 7.06 ± 1.50; LMS = 19.0 ± 4.01. Preoperative CT-scans from patients #1 and #9 are shown in Figure 1. Post-reboot scores, at the 12-month follow-up, were: NPS = 0; SNOT-22 = 17.90 ± 6.80; VAS-global = 1.91 ± 1.17. Pre- and postoperative outcomes are presented in Table 2. Differences between pre- and postoperative outcomes were statistically significant for parametrical variables at paired-samples t-test (Table 3). The mucosal healing process over different timepoints is illustrated in Figure 2. Endoscopic nasal examination at the 12-month follow-up showed healed nasal epithelium, with no sign of polyp recurrence (NPS = 0) in the overall sample (Figure 2). None of the patients required the adjuvant administration of oral corticosteroids or systemic antibiotics to control nasal symptoms.
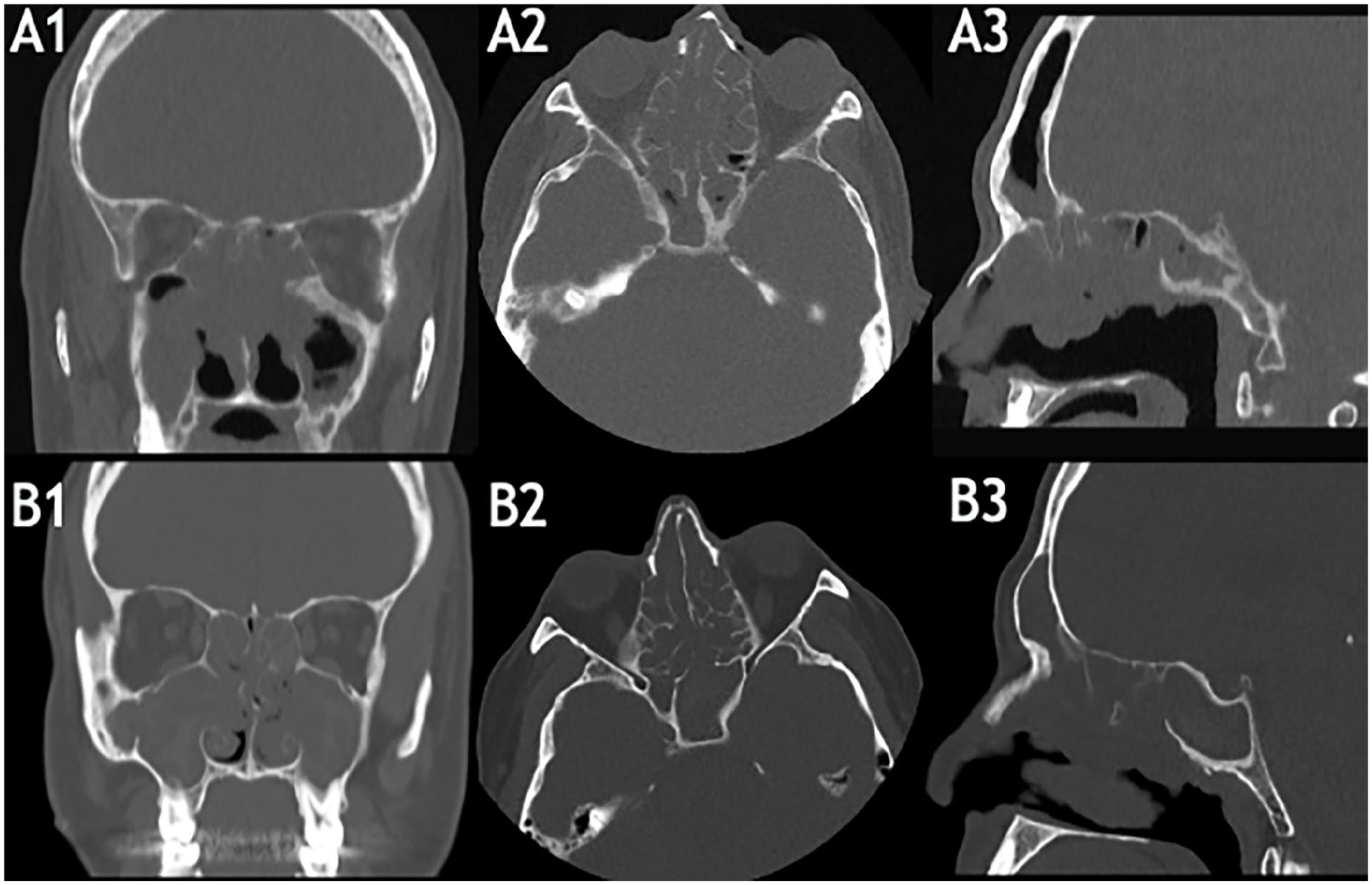
Preoperative CT findings (A, Patient #1; B, Patient #9) confirming diffuse opacification of the paranasal sinuses (LMS: Patient #1 = 18, Patient #9 = 24) despite the adequate completeness of previous surgeries; A1, B1: coronal planes, A2, B2: axial planes, A3, B3: sagittal planes. LMS, Lund–Mackay score.
Pre- vs Post-Reboot (after 12 months) Characteristics of the Cohort.
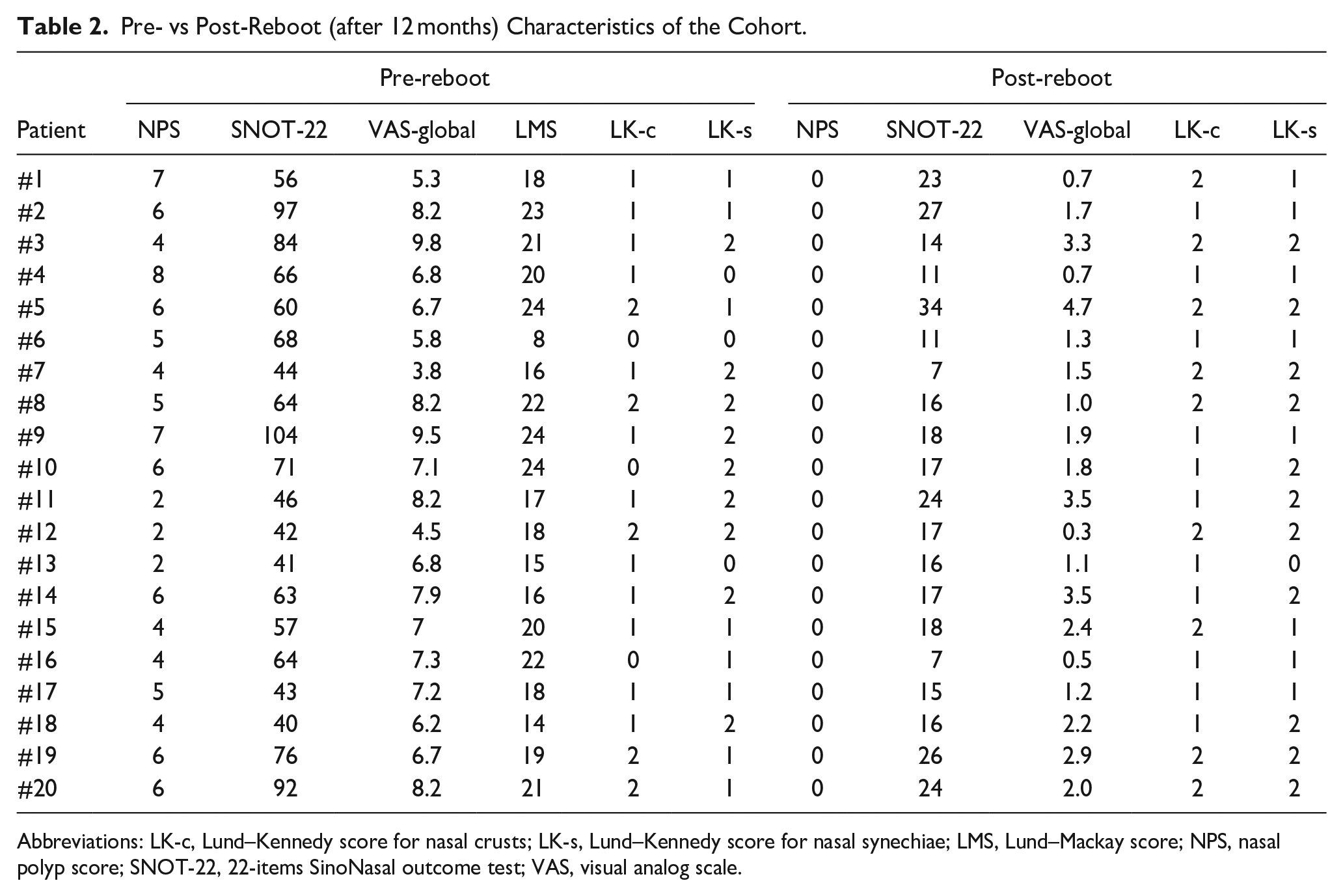
Abbreviations: LK-c, Lund–Kennedy score for nasal crusts; LK-s, Lund–Kennedy score for nasal synechiae; LMS, Lund–Mackay score; NPS, nasal polyp score; SNOT-22, 22-items SinoNasal outcome test; VAS, visual analog scale.
Differences Between Pre- vs Post-Reboot Outcomes for Parametrical Variables.
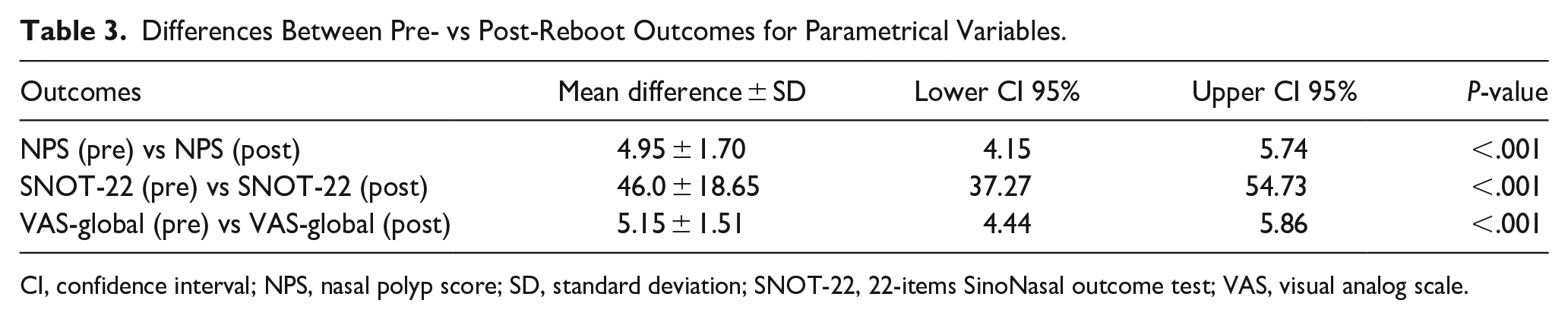
CI, confidence interval; NPS, nasal polyp score; SD, standard deviation; SNOT-22, 22-items SinoNasal outcome test; VAS, visual analog scale.
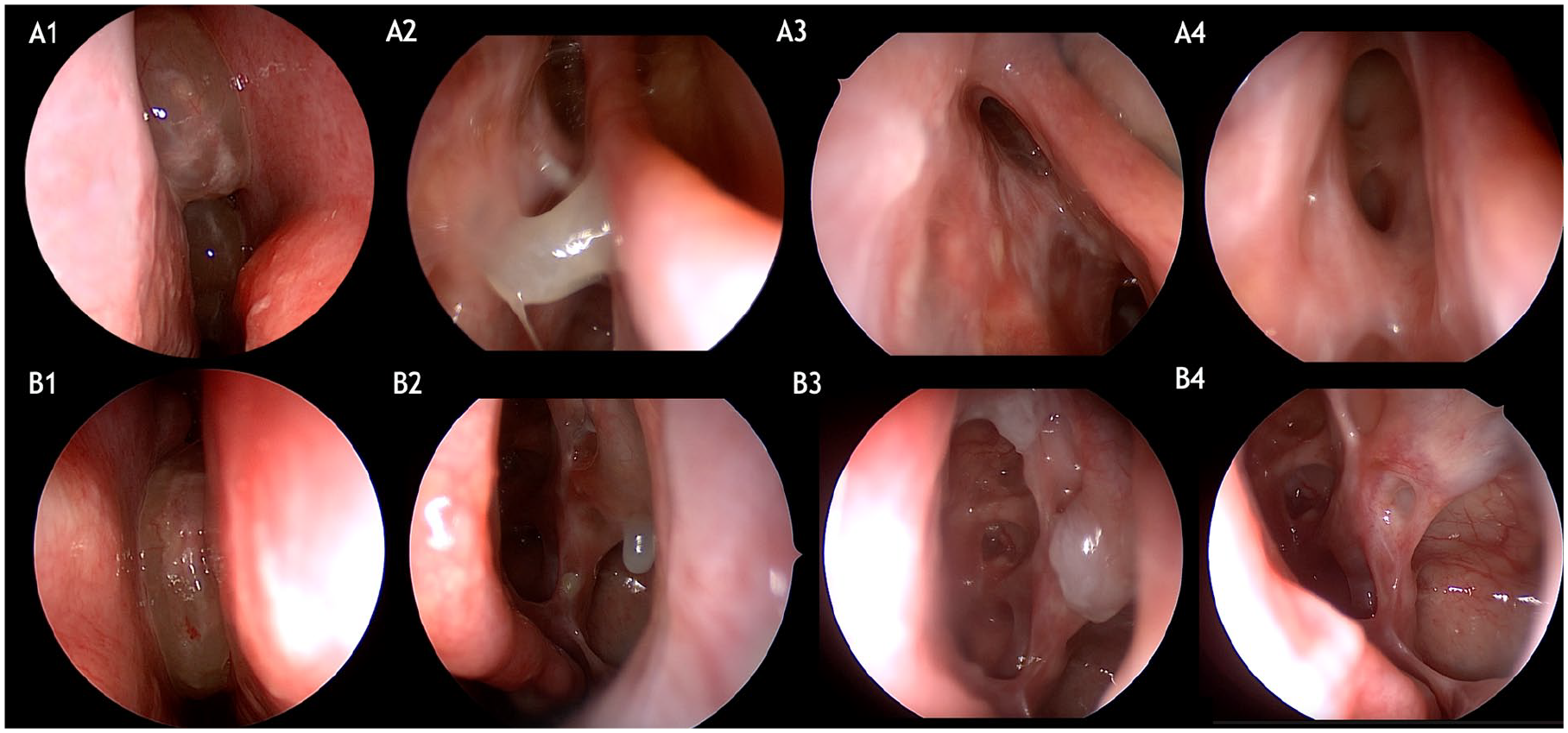
Preoperative endoscopic nasal examination and at different timepoints. (A, B) Pre-surgical examination showing nasal mucosal edema with polypoid degeneration; NPS: 3 (A, Patient #1), 4 (B, Patient #9). A2-4: Postoperative healing process of the frontal sinus mucosa (A2: 21 days, A3: 3 months, A4: 12 months). B2-4: Postoperative healing process of the maxillary and ethmoidal sinus mucosa (B2: 21 days, B3: 3 months, B4: 12 months). Mucosal regeneration occurred gradually. Early postoperative findings included diffuse edema and crusts (A2, B2). By 3 months, edema was significantly reduced, and there was progressive improvement in nasal epithelium regeneration (A3, B3). By 12 months, the healing process was complete (A4, B4). NPS, nasal polyp score.
No major surgical complications (eg, injuries of the lamina papyracea, anterior skull base, ethmoidal arteries, infraorbital nerve, lateral intra-sphenoid structures, etc.) nor long-term morbidity (eg, chronic osteitis, exacerbations of lower airways diseases, etc.) were observed during follow-up. Lund–Kennedy score for nasal crusting was 1.10 ± 0.64 preoperatively and 1.45 ± 0.51 at 12-month follow-up, with no statistically significant difference (P = .071). Similarly, no differences were observed for LK-s (pre-op: 1.30 ± 0.73, post-op: 1.50 ± 0.61; P = .372). Preoperative tissue biopsies revealed a markedly high density of eosinophils in all patients, ≥10 per high power field (HPF; range 10-60), despite the ongoing mAb treatment. Nasal specimens collected postoperatively showed remarkably reduced eosinophilic infiltration in that same tissue (≤1/HPF, 100% of cases; Figure 3).
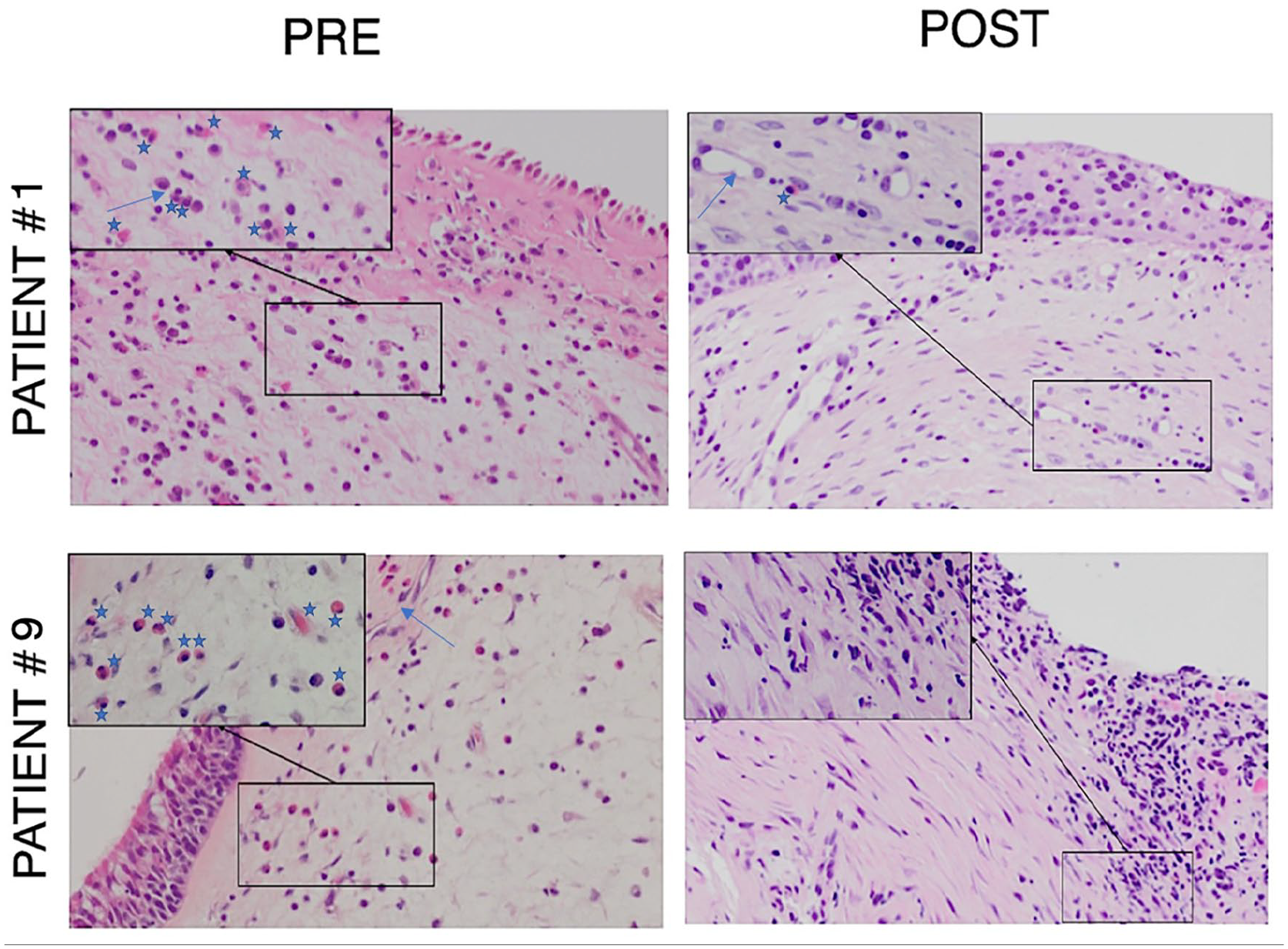
PRE: Preoperative histopathological samples showing typical EGPA perivascular eosinophilic pattern (patient #1: 10/HPF; patient #9: 12/HPF). POST: Postoperative histopathological samples depict markedly reduced eosinophilic infiltration (≤1/HPF); objective magnification at 20× (at 40× in the boxes); star: eosinophil, arrow: vessel. EGPA, eosinophilic granulomatosis with polyangiitis; HPF, high power field.
Discussion
Long-term management of CRSwNP has always been significantly challenging for clinicians. 32 In this study, we aimed to analyze a specific subset of patients, namely those who received mAb therapy for a comorbid condition, such as asthma or systemic vasculitis (EGPA), yielding good outcomes on the comorbidity, but still with a limited response at the nasal level. 33 This group of patients represents a tough category for physicians to treat; despite satisfactory outcomes in the lower respiratory tract, the disease burden remains high when unresponsiveness occurs on the upper airways, raising the question of whether treatment adjustments are necessary. The reason why type-2-driven inflammation might be under control with mAb therapy in the bronchi and lungs, while resistant within the nose and paranasal sinuses, is still debated. Fortunately, non-responders to mAbs are rare, but still not less relevant than others, as their need for care and symptom control are even more challenging. It is pivotal to develop therapeutic strategies to reduce the disease burden in such patients, especially those who had already undergone multiple mucosa-sparing procedures. In this scenario, reboot surgery might represent a “rescue approach,” or an “adjuvant” option, in patients whose peripheral and systemic conditions are well controlled, but who still suffer from excessive nasal inflammation and accompanying symptoms. Reboot surgery was described by Alsharif et al for patients with uncontrolled CRSwNP, aiming to offer longer-term polyp-free status in patients with a high inflammatory load after failure of standard-ESS and topical medical therapy.23,34 It consists in removing of the whole mucosa down to the periosteum of the sinuses to facilitate restoration to a non-inflamed state. Its added value is thought to consist specifically in the mechanical removal of chronically accumulated eosinophilic infiltrates, as well as dysfunctional microbiota and intramucosal germs. This approach may allow better and long-lasting control of nasal manifestations in patients for whom mAb therapy shows unsatisfactory results. To guarantee the homogeneity of our sample, our entire cohort underwent partial reboot surgery, which includes a Draf IIb procedure rather than a Draf III, in accordance with Alsharif et al. Due to the limited involvement of the frontal sinuses in our sample (LMS < 2), a Draf III was not warranted to effectively remove the entire diseased frontal mucosa. Importantly, no cases of disease relapse were observed locally in the frontal sinus during the follow-up. According to larger cohort studies following the first publication, reboot surgery has shown significantly lower recurrence rates, longer recurrence-free survival, improved quality of life (QoL) as well as zeroing of oral corticosteroid uptake over 2 years postoperatively in patients with recalcitrant CRSwNP, who were not eligible for or unwilling to undergo mAb therapy.25,35 Likewise, our previous experience with reboot surgery in patients who were not under biologics showed promising results in terms of disease control. 24 However, this article presents the first reported series focusing specifically on patients already under mAb therapy for a comorbid condition. In this selected group of patients, at 12-month postoperatively, reboot surgery determined a statistically significant reduction in endoscopic findings (NPS—Figure 2) as well as in QoL outcomes (SNOT-22; VAS—Tables 2 and 3). Similarly, tissue eosinophil count on postoperative specimens was markedly reduced (Figure 3). Moreover, although it was not a specific outcome of the study, during postoperative follow-up, patients did not require oral corticosteroids to control nasal symptoms. We observed a slightly prolonged healing process compared to mucosa-sparing functional procedures, as patients required serial medications for up to 90 days postoperatively. Nasal medications consisted of daily rinses with saline solution multiple times a day, as well as topical steroid (budesonide 0.5 mg/mL) administered via nasal douche twice a day (Rinowash, AirLiquide Medical System, Bovezzo, Italy). During the immediate postoperative period (up to the first month), topical antibiotics (ceftazidime 0.5 mg/mL) were added to enhance the local healing process. Nonetheless, no long-term morbidity was observed in our sample. Although Lund–Kennedy scores were higher after revision surgery than after FESS, the differences were not significant (P > .05). In a previous study by Vlaminck et al, local tissue eosinophilia (LTE) and the presence of eosinophilic mucin (EM) were closely correlated with the recurrence of CRSwNP: LTE alone was associated with a 48% risk of recurrence, whereas LTE and EM together were associated with a recurrence rate of 56%. No such association was found in patients with CRSsNP, suggesting that eosinophils and/or EM do not contribute to recurrence in CRSsNP. 36 Matsuwaki et al also concluded that the presence of eosinophils in the nasal mucosa has a positive predictive value for recurrence of 85.7% within 5 years after surgery. 37 In their research, mucosal eosinophilia was defined as the evidence of more than 5 cells per HPF; interestingly, in our series, all patients had no less than less than 10 eos/HPF, reaching up to 60 eos/HPF. Mucosa-sparing ESS involves opening paranasal sinus “prechambers” to preserve physiological mucociliary clearance of the sinus cavities, restore drainage and ventilation, and allow uniform distribution of topical medication across the nasal mucosa. This approach has proven effective in CRSsNP and in most CRSwNP cases when performed in a comprehensive fashion (or “full-house FESS”), as stated by the EPOS 2020 steering group. 28 Considering its lower risk of surgical complications, the functionality criterion should be respected in CRSwNP patients who have undergone no or incomplete previous surgeries. However, in this selected subset of patients, the completeness of previous ESS was confirmed, necessitating a more radical approach (Figure 1). Despite being macroscopically complete, a comprehensive mucosa-sparing technique still harbors a significant risk of relapse, especially in severe type-2-related CRSwNP.38,39 Specifically, in this sample, all patients experienced early recurrence of symptoms (on average 7.45 ± 5.74 months after previous functional surgeries). It is the opinion of the authors, supported by current literature, that for this specific group of patients, more extensive approaches including larger sinusotomies (eg, a frontal Draf IIb) and non-mucosa sparing techniques aimed at regenerating the diseased nasal epithelium may be warranted.40-42 Despite the outstanding results of mAbs, it might be speculated that not all patients who are already under biologic therapy for comorbid condition should switch to a different mAb in case of ineffectiveness over the concurrent CRSwNP. Albeit rarely, it is described that anti-IL-4/13 might cause complications such as hypereosinophilia, with worsening of asthma symptoms, tissue eosinophilic infiltration, and EGPA-like symptoms. This risk is highest especially in steroid-dependent asthmatic patients who switch from anti-IL-5 to anti-IL-4/13 treatment. 43 For this reason, in patients whose lower airways inflammation is well controlled, the choice to switch to a different mAb should be well pondered and, in absence of contraindications, a surgical strategy should be also considered.
This study has some limitations. Due to strict inclusion criteria, we were able to enroll a limited sample of 20 patients; thus, our results should be interpreted in the context of a clinical experience and are not suitable to draw definitive conclusions. From this perspective, we did not include measures of disease control, such as those proposed in the 2020 EPOS document, since recent research suggests that these guidelines may not align with patients’ self-assessment of their CRS control. 44 Accordingly, we prioritized reporting patient-reported outcome measures (eg, SNOT-22, VAS), which are regularly administered in our clinical practice. Due to the scarcity of sinonasal symptoms at the 12-month follow-up examination, and to avoid unnecessary exposure to further ionizing radiation, patients were not subjected to postoperative CT examinations. Instead, objective findings were assessed by nasal endoscopy and histopathologic sampling. Additionally, we included subjects assuming different types of mAbs, potentially introducing some selection bias. Nevertheless, in this article, we aimed to present a possible surgical solution to selected cases of nasal unresponsiveness to biologic treatment, regardless of the specific mAb used. Finally, as we presented a real-life mono-institutional experience, we included patients who were treated with benralizumab for their concurrent CRSwNP, in accordance with the indications from the Italian regulatory drug agency (Agenzia Italiana del Farmaco, AIFA). It is important to note that this specific monoclonal antibody is currently not approved by the Food and Drug Administration in the United States for this population. Further investigations with larger multicentric cohorts, including patients locally unresponsive to a specific molecular drug as well as direct control groups undergoing further functional surgery are needed to ascertain the potential role of non-mucosasparing techniques (eg, reboot) in aiding disease control in patients under mAb treatment locally unresponsive at a nasal level.
Conclusion
Our analysis showed that reboot sinus surgery may be an effective solution in selected cases of recalcitrant CRSwNP. Given the rationale behind sinus demucosalization and the optimal results our patients experienced, the query of whether reboot surgery has a rescue role in the context of mAb nasal unresponsiveness should be raised, especially in patients who have already failed multiple ESS procedures. In such cases, ENT surgeons might reconsider their approach to sinus surgery, deviating from standard mucosa-sparing techniques when these prove ineffective. Further investigations with larger cohorts and longer-term follow-ups are needed to confirm our preliminary results.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the ethical standards of the Declaration of Helsinki and its later amendments, and the institutional ethics committee approval was obtained (protocol number: ICH/320/21).
Informed Consent
Informed consent was signed by every patient enrolled.
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
