Abstract
Introduction
Myringoplasty is the primary method to reconstruct chronic perforations of the tympanic membrane (TM). It involves the closure of perforations using various graft materials, such as temporalis fascia, perichondrium, and cartilage, applied through different techniques, including underlay, overlay, and over-underlay techniques.1-3 The graft healing process encompasses clinical inosculation, neovascularization, and epithelization.4,5 Successful grafting requires close contact between the graft and the remnant TM, which depends on ensuring sufficient remnant TM at the perforation margins, an adequately sized graft, precise graft placement, and preventing graft displacement.
Observing the anterior margins and accurately placing the graft in cases of anterior perforation with anterior wall protrusion is challenging and often results in high graft failure rates.1-3,6 Some researchers have advocated various techniques to repair anterior perforations and achieve high graft success rates. 6 However, these methods typically involve raising a tympanomeatal flap and drilling the anterior protrusion, which can extend the operation time and lead to complications such as operative pain, external auditory canal (EAC) stenosis, and chorda tympani nerve damage.7,8 Furthermore, anterior wall protrusion often leads to cracked tympanomeatal flaps, which fail to reinforce the graft adequately, and misplaced flaps can induce EAC cholesteatoma. Ungar et al. 9 reported 5 iatrogenic cholesteatomas originating from misplaced tympanomeatal flaps. Although some authors have suggested using a mucosal flap technique via the middle ear to repair anterior perforations,2,10 it remains difficult to identify the anterior annulus in patients with anterior wall protrusion. A systematic review and meta-analysis by Gülşen and Arıcı 2 confirmed that myringoplasty has a similar graft success rate to type I tympanoplasty, which involves raising a tympanomeatal flap. Lou et al. 4 were the first to promote a graft technique that preserves the perforation margins and found that it does not impede clinical inosculation or neovascularization, thereby benefiting the increase in the graft contact area at the anterior margins.11,12 Recently, some authors have recommended using a graft reinforcement technique in areas where graft coverage is insufficient to repair large, marginal, and subtotal perforations, achieving satisfactory results.13-17
To date, there have been no reports of using additional cartilage slice reinforcement for the repair of anterior perforations with anterior wall protrusion. This study evaluated the outcomes and complications associated with the endoscopic cartilage slice reinforcement technique of utilizing anterior margins to repair anterior perforations with anterior canal wall protrusion.
Materials and Methods
Ethical Considerations
Ethical approval for this study was obtained from the Medical Ethics Committee of the First People’s Hospital of Tongxiang, China. All participants provided informed consent.
Patients and Methods
This prospective case series study included consecutive patients who were diagnosed with chronic anterior perforations and treated at the Department of Otorhinolaryngology, Head and Neck Surgery, from February 1, 2017 to August 29, 2023. The inclusion criteria were as follows: aged > 16 years; chronic anterior perforation with anterior canal wall protrusion; dry ears for ≥3 months prior to surgery; positive Valsalva’s maneuver; and an air-bone gap ABG of <35 dB. The exclusion criteria included revision cases; presence of cholesteatoma or granulation tissue in the middle ear and mastoid; Eustachian tube dysfunction; pure-tone averages (PTAs) indicating a potential ossicular chain abnormality; acute inflammation; and fungal otitis externa. Cases in which the anterior margin of the perforation could not be visualized entirely with the endoscopic examination were considered as patients with anterior canal wall protrusion in this study.2,3
Perforations were classified into 4 sizes based on the affected area of the TM: small (<25%), medium (25%-50%), large (50%-75%), and subtotal (>75%). Data recorded for all participants included age, sex, cause and duration of perforation, side, myringosclerosis, operation time, and preoperative and postoperative hearing levels. Pure-tone hearing tests were conducted preoperatively and 6 months postoperatively. PTAs were calculated for both air conduction (AC) and bone conduction (BC) by averaging the thresholds at 500, 1000, 2000, and 4000 Hz. Preoperative and postoperative ABGs were calculated by subtracting the AC PTA from the BC PTA. ABG closure was determined by subtracting the postoperative ABG from the preoperative ABG.
Surgical Techniques
All operations were performed under general anesthesia by the same surgeon, using an endoscope. The perichondrium-cartilage graft was used to repair the perforation. The primary perichondrium-cartilage graft was placed trans-perforation without raising a tympanomeatal flap or trimming the perforation margins.
The perforation margins were not trimmed, and sclerotic plaques on the remnant TM were preserved. The epithelium was removed from the distal malleus handle if present. An ipsilateral tragal cartilage with a one-sided perichondrium composite graft was harvested without thinning the cartilage. The lateral perichondrium was raised circumferentially, and the cartilage graft was trimmed to match the size of the perforation. A notch was created in the cartilage to accommodate the malleus handle if necessary. All cartilage slices were preserved.
The middle ear was packed with bioresorbable synthetic polyurethane foam (NasoPore; Polyganics, Groningen, The Netherlands). The cartilage composite graft was inserted through the perforation. The cartilage graft was positioned medial to the anterior margins, anterior annulus, and posterior remnant TM, with a notch accommodating the malleus if present. All of the free perichondrium was also placed medial to the anterior margins, anterior annulus, and posterior remnant TM, but lateral to the cartilage. Excess-free perichondrium was not removed but accumulated at the perforation margins. Next, a piece of cartilage slice was selected and trimmed to reinforce the anterior graft and placed lateral to the anterior margins, anterior annulus, and anterior perichondrium (Figure 1). The EAC was also packed with bioresorbable synthetic polyurethane foam. The tragus incision was not sutured but instead covered with gauze soaked in erythromycin ointment.
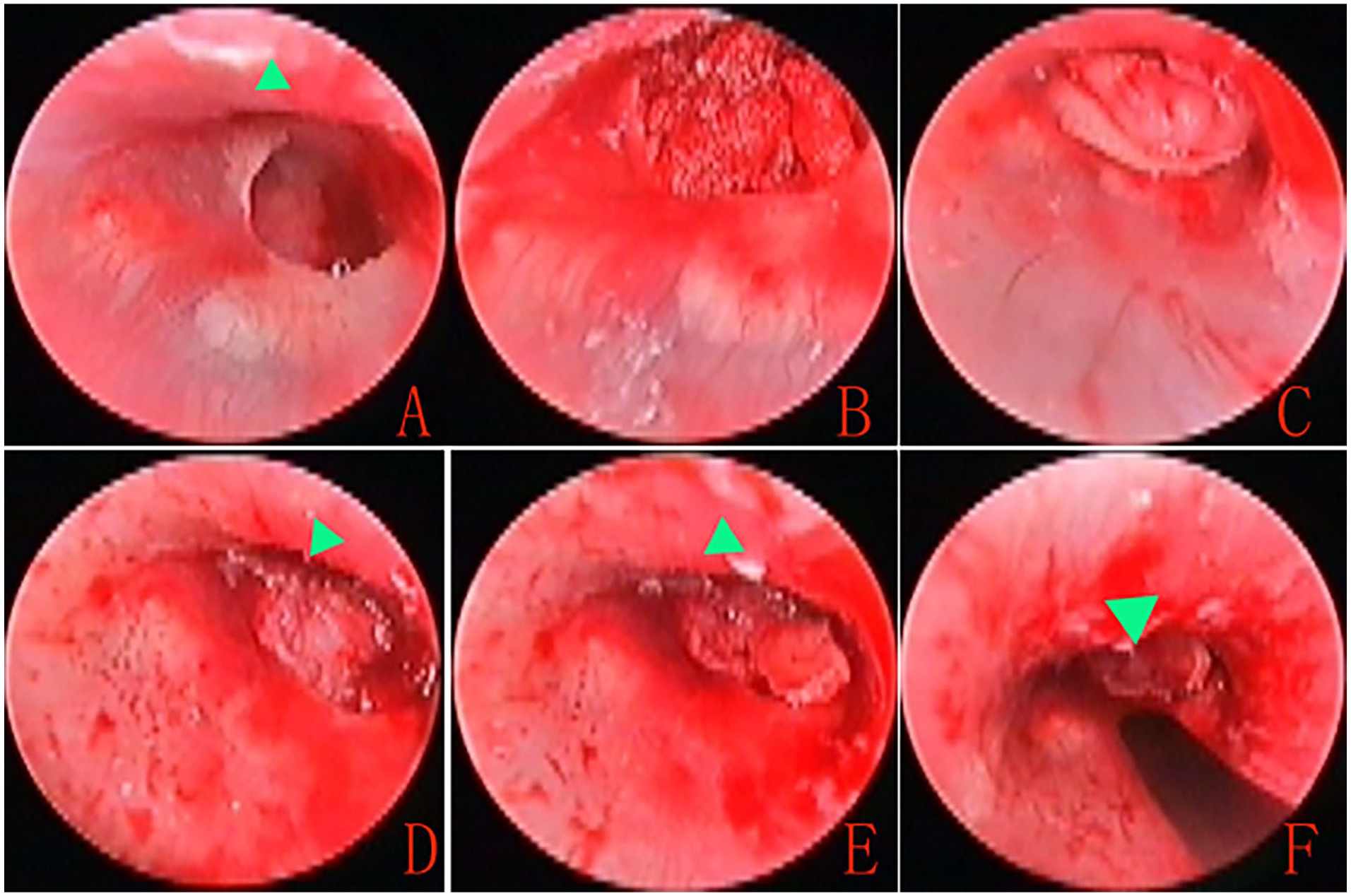
Preoperative perforation (A), middle ear packing (B), graft placement (C and D), and additional cartilage slice placement (E and F).
Postoperative Follow-Up and Outcomes Assessment
Patients were discharged the day after surgery. The biodegradable synthetic polyurethane foam was removed 2 weeks later. Endoscopic examinations were performed at the outpatient clinic at 2 and 4 weeks, and at 3 and 6 months after surgery. Any intraoperative or postoperative complications were documented. Audiometric evaluations were conducted 6 months after surgery to assess operation time, graft success rate, audiometric outcomes, and any complications. Graft success was defined as the presence of an intact graft without residual or re-perforation. The operative time was defined as the duration from the start of surgery following anesthesia induction to graft placement. Postoperative success, based on audiometric criteria, included an ABG ≤ 20 dB.
Statistical Analyses
Statistical analyses were performed using SPSS Statistics (version 20; IBM Corp, Armonk, NY, USA). Data are expressed as mean ± standard deviation and number (%). Differences between the preoperative and postoperative ABG were analyzed using a paired-sample t-test. Differences in which P < .05 were considered statistically significant.
Results
Demographic Characteristics and Operation Time
The study population included 38 patients with chronic anterior perforations and anterior canal wall protrusion (21 males and 17 females; average age 42.1 ± 9.7 years), with 22 patients having left-side involvement and 16 on the right. The mean duration of perforation was 9.2 ± 3.7 years. Perforations were medium in 11 (28.9%) patients, large in 25 (65.8%) patients, and subtotal in 2 (5.3%) patients. The cause of perforation was mucosal chronic otitis media (COM) in 35 (92.1%) patients, traumatic perforation with failure to close for 1 year in 2 (5.3%) patients, and ventilation tube removal after 3 years in 1 (2.6%) patient. The mean operation time was 27.2 ± 4.6 min.
Endoscopic Observation and Graft Take Rate
At 2 weeks postoperatively, the endoscope revealed clinical inosculation between the perichondrium graft and the remnant TM, as well as neovascularization of the graft. At 3 months postoperatively, the reinforcement cartilage slice and perichondrium were on the same plane in 34 (89.5%) patients, while the reinforcement cartilage slice was higher than the perichondrium plane in 4 (10.5%) patients. Two patients had fissure-like perforations at the anterior-inferior and posterior-inferior margins, but no re-perforations were observed. The graft success rate was 94.7% (36/38) at 6 months postoperative. Figure 2 shows the healing process.
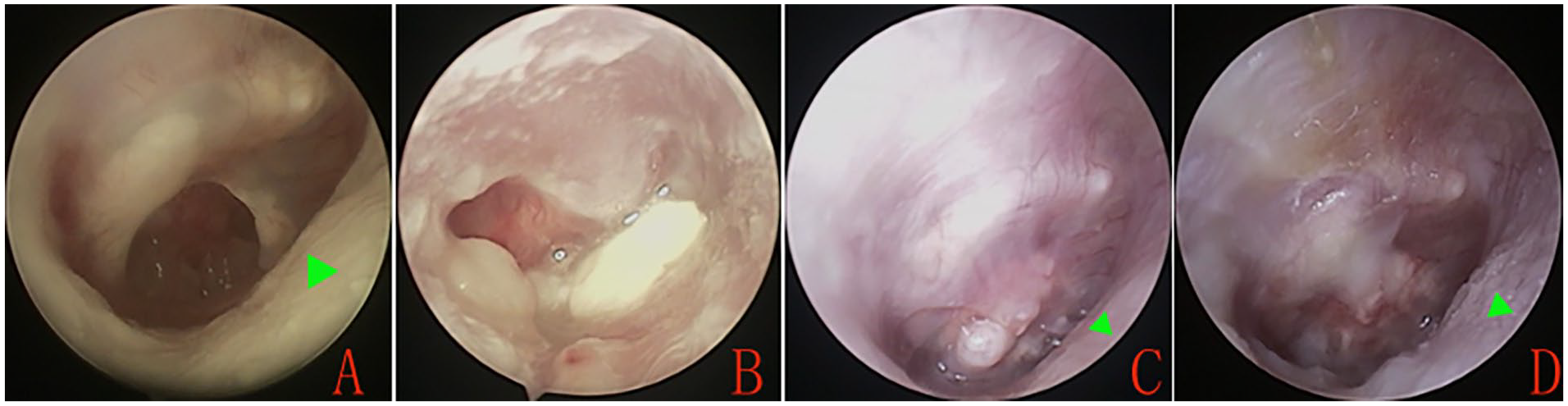
Preoperative perforation (A), postoperative at 2 weeks (B), 4 weeks (C), and 3 months (D). Blue triangles indicate the anterior wall protrusion.
Hearing Gain and Complications
The mean preoperative AC was 31.2 ± 1.3 dB, and the mean postoperative AC was 19.4 ± 3.5 dB; this difference was significant (P < .001; paired-sample t-test). The mean preoperative BC was 11.6 ± 2.7 dB, and the mean postoperative BC was 12.1 ± 1.9 dB; this difference was not significant (P = .417; paired-sample t-test). The mean preoperative ABG was 19.8 ± 4.2 dB, and the mean postoperative ABG was 8.6 ± 2.9 dB; this difference was significant (P < .001; paired-samples t-test). The ABG gain was 11.8 ± 5.1 dB.
A preoperative individual audiometric assessment revealed an ABG ≤ 10 dB in 7.9% (3/38) of patients, 10-20 dB in 57.9% (22/38), and 20-30 dB in 34.2% (13/38). Postoperatively, the individual ABG was ≤10 dB in 60.5% (23/38), 10-20 dB in 36.8% (14/38), and 20-30 dB in 2.6% (1/38). The successful surgery rate (postoperative ABG ≤ 20 dB) was 97.4% (37/38).
No complications such as altered taste, vertigo, or tinnitus were reported during the follow-up period. In addition, no cases involved graft lateralization, significant blunting, graft atelectasis, graft adhesions, or effusion. However, myringitis was observed in 4 (10.5%) patients, which was controlled by topical ofloxacin ear drops plus dexamethasone.
Discussion
The graft success rate in this study was similar to that for other techniques for repairing anterior perforation. Visvanathan et al. 6 conducted an evidence-based systematic review of closure of an anterior perforation and summarized 3 graft techniques with their graft success rates: 84.6%-94% using anterior anchoring techniques, 97%-98% using lateral graft tympanoplasty, and 88%-96% using techniques for large and subtotal perforation. Compared with these conventional techniques, the endoscopic cartilage slice reinforcement technique has the advantage of not raising a tympanomeatal flap via an EAC incision, thereby reducing the complications associated with the EAC. Although some authors applied a mucosal flap without an EAC incision to repair anterior perforation and reported graft success rates of 93.7% and 92.2%,2,10 raising a mucosal flap increases the risk of intraoperative bleeding and chorda tympani nerve injury.
It is difficult to completely expose the anterior margin, even with an endoscope, if the prominent bone wall is not drilled for anterior perforations with anterior prominence. Thus, it can be difficult to trim the anterior margin completely, especially for beginners. Lou et al. 11 were the first to advocate myringoplasty while preserving perforation margins, finding that it did not affect graft neovascularization or short- and long-term graft success rates.4,11,12 The results of the present study support their findings, demonstrating that all grafts achieved inosculation with the remnant TM and neovascularization. Avoiding the need to raise a tympanomeatal flap and trim the perforation margin can reduce the operation time compared with techniques that involve raising the tympanic flap and trimming the margin.4,11 The mean operation time in this study was 27.2 ± 4.6 min.
The graft success rate in this study was comparable to those of other graft reinforcement techniques that involve raising a tympanomeatal flap. Some scholars recommend a perichondrium reinforcement technique for repairing large perforations, in which a secondary perichondrium patch is placed over areas where the graft does not tightly fit the residual TM or if the marginal perforation has no TM remnant to support the graft. Shakya et al. 13 and Wang et al. 14 reported graft success rates of 95% and 97.5%, respectively. Others have reported on the cartilage reinforcement technique, with the cartilage graft placed on the medial side of the fascia temporalis. Tek et al. 18 reported a success rate of 86.5%, Kouhi et al. 19 reported a success rate of 93.4%, and other authors reported success rates of 94.7%-98.4%.15,20,21
Lou et al.16,17 first reported a cartilage reinforcement technique without raising a tympanomeatal flap for repairing subtotal and large perforations, achieving graft success rates of 94.7% and 100.0%, respectively. The endoscopic cartilage slice reinforcement technique is similar, but Lou et al. 16 did not apply their approach to repair anterior perforations with anterior prominence; they primarily used it for reinforcing first insufficient grafts and marginal perforations. 17 We were the first to apply this technique to repair anterior perforations with anterior prominence and obtained a graft success rate of 94.7%. In addition, unlike in previous methods, we did not remove the excess perichondrium; instead, we placed the excess at the perforation margins, a technique similar to “tuck grafts,” which are known to improve the graft success rate. 22
The current endoscopic cartilage slice reinforcement technique yielded outcomes comparable to those of other reinforcement techniques. It avoided raising a tympanomeatal flap via the EAC, preserved the perforation margins, and increased the contact area between the graft and remnant TM, thereby improving the graft success rate. Although the anterior prominence affected the surgical field, an additional cartilage slice was carefully placed through the space between the anterior wall and remnant TM, compressing the anterior annulus, remnant TM, and anterior perichondrium. This acted as reinforcement for the previously placed perichondrium and cartilage graft. Two patients in this study had residual fissure-like perforations. We speculate that the placement of the additional cartilage slice resulted in displacement of the cartilage. Therefore, we emphasize that the additional cartilage slice should be gently placed until it meets bone wall resistance, without excessive pressure to avoid displacement of the underlying graft.
The hearing improvements noted in this study were similar to those of previous techniques for repairing anterior perforation6,13-21; postoperative hearing significantly improved compared to preoperative levels and the successful surgery rate was 97.4% (37/38). An additional cartilage slice was placed only over the anterior annulus and anterior remnant TM, which did not affect the vibration and conduction of the ossicular chain. Furthermore, the reinforcement cartilage slice in 89.5% of patients in this study showed inosculation with the remnant TM without projecting beyond the TM plane; it protruded from the perichondrium in only 10.5% of patients, which did not affect hearing improvement. The cartilage is significantly higher than the TM plane in the butterfly cartilage technique, but to date, no English-language studies have reported that the butterfly technique is detrimental to hearing improvement. 23 However, this cartilaginous reinforcement technique did not improve the audiometric and successful surgical results compared with other techniques. No cases of graft lateralization, significant blunting, graft atelectasis, or graft adhesions were observed during the follow-up period. However, 4 cases of postoperative myringitis were found, we speculated that the uncovered cartilage was a risk factor for myringitis, a piece of cartilage was exposed but not covered by perichondrium, which could delay the epithelization, thereby resulting in myringitis.
One limitation of this study is the small sample size of anterior perforations with anterior wall protrusion, which may be why it is rare as a single-series study.
Conclusion
Endoscopic cartilage slice reinforcement for the anterior margin is a simple, effective technique for repairing anterior perforations with anterior protrusion. It achieves a high graft success rate, better hearing improvement, and minimal complications.
Footnotes
Authors’ Note
Authors’ contributions
Xionghui Hu, MD and Fang-Luo, MD: Interpretation of data for the work, design of the work, analysis of data for the work, drafting the work, and final approval of the version to be published; Di-He, MD: Analysis of data for the work, drafting the work and technical the diagram, and final approval of the version to be published; agreement to be accountable for all aspects of the work, design of the work; Wenlong Jiang, MD: Interpretation of data for the work, final approval of the version to be published.
Data Availability Statement
All data generated or analyzed during this study are included in the published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical/Consent Statement
The study protocol was reviewed and approved by the Institutional Ethical Review Board of the First People’s Hospital of Tongxiang. All participants provided written informed consent.
