Abstract
Introduction
An unintended consequence of improved cancer therapies is that a greater percentage of patients than ever before are suffering from long-term side effects of their respective treatments.1,2 These side effects have become more prevalent due to the success of cancer treatments, allowing patients to survive longer. Among the complications that can come with cancer therapies, medication-induced osteonecrosis stands out as one of the rare yet significant concern that arises in individuals who have undergone cancer therapy. 2 This report focuses on the case of a patient who developed osteonecrosis after receiving cancer treatments involving denosumab.
Currently, denosumab (Xgeva®) is a drug that is commonly used alongside chemotherapy to treat bone damage resulting from multiple myeloma (MM). It has been reported to induce medication-related osteonecrosis of the jaw (ONJ) in patients. 2 In this article, we present a case of osteonecrosis of the maxilla induced by adjunctive oncologic medication denosumab, and we review the relevant literature. We hope to illustrate a unique case of a common pathology presenting in an uncommon way as well as to discuss the possible mechanisms of medication-related maxillary osteonecrosis as well as the implications this may have on cancer survivors.
Case Presentation
We report the case of a 40-year-old male inmate with a history of MM who presented with a painful palatal lesion persisting for 6 months. The patient, with a concurrent history of schizoaffective disorder and a significant 30 pack-year smoking habit, reported bloody rhinorrhea and spontaneous loss of most dentition in the left maxilla (Figure 1A). His previous oncological treatment 3 years prior included chemotherapy, palliative radiation to the left hip and spine, and 17 cycles of monthly 120 mg subcutaneous denosumab therapy over 22 months. Denosumab was halted when a palatal lesion was reported. The palate lesion continued to grow in size after denosumab was halted beyond the normal half-life of denosumab.
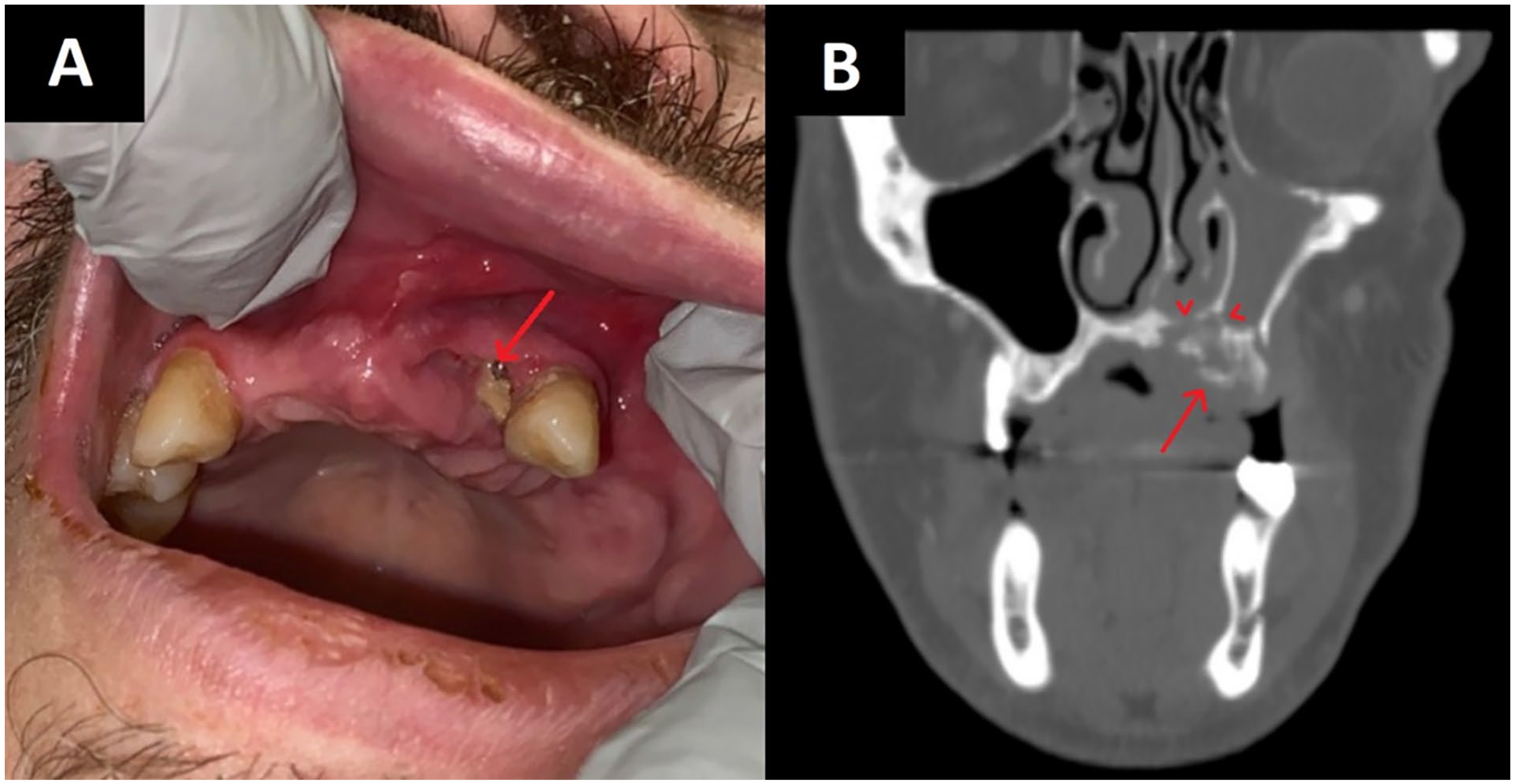
(A) Palatal lesion: Image of palatal lesion on presentation. Note the exposure of underlying maxillary alveolar ridge (red arrow). One adjacent tooth remained intact but loose. (B) CT scan: CT maxillofacial with contrast, coronal plane. Note destructive osseous changes to the left anterior maxilla with a 1.7 cm loose bony fragment (red arrow). In addition, erosion of the nasal floor can be seen with associated mucosal thickening of the left maxillary sinus and nasal floor (red arrowheads). CT, computed tomography.
Investigative computed tomography (CT) scan exhibited a nonspecific, destructive bony lesion of the hard palate (Figure 1B). An in-office biopsy of the lesion revealed benign squamous mucosa with dystrophic calcification and minimal inflammatory cells. Given the inconclusive nature of the initial investigation, surgical exploration was undertaken, during which several loose bony fragments were extracted and sent for pathological analysis. Initial pathologic evaluation raised suspicions of Actinomyces spp. colonization.
A definite diagnosis of maxillary osteonecrosis with Actinomyces colonization was subsequently confirmed. Intraoperatively, the remaining maxillary bone and surrounding soft tissue appeared healthy and well-vascularized. The cavity was packed with Gelfoam and Surgicel, and chromic gut sutures were used to close the overlying mucosa.
Following a 3 month treatment as per the PENTOCLO protocol, the patient demonstrated significant mucosalization of the previously exposed bone, along with a notable reduction in pain (Figure 2). A 1 year course of PENTOCLO protocol therapy was completed, and there was seen to be resolution of the palatal lesion.
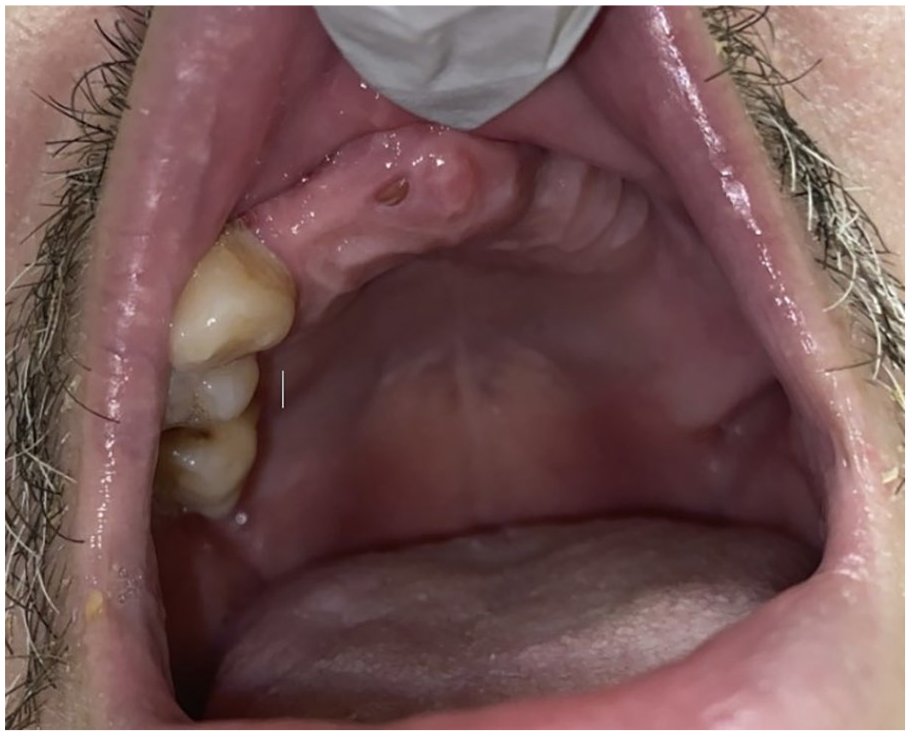
Post-PENTOCLO response: Image of palatal lesion 3 months after starting PENTOCLO protocol. Note significant healing with near-complete mucosal coverage of previously exposed bone.
Discussion
Denosumab is a total human immunoglobulin (Ig)G2 monoclonal antibody with high affinity to RANKL, competitively inhibiting its binding to receptor activator of nuclear factor κB (RANK), inhibiting osteoclast differentiation and subsequently inhibiting bone resorption, commonly used in treatment of osteoporosis and bone metastasis.3,4 Denosumab-induced ONJ is relatively rare, with the incidence to be about 1.7%. 5 While the exact mechanism of action of denosumab-induced ONJ is under debate, it is thought that by inhibiting osteoclast activity, it interferes with the normal bone remodeling process, leading to the possibility of accumulation of dead bone tissue and subsequently ONJ.6,7
There is no current standardized treatment protocol for management of medication-induced ONJ, and treatment options are largely dependent on the severity of necrosis. Rinsing with antiseptic chlorhexidine or chlorine dioxide, combination of local debridement and systemic antibiotic treatment,8,9 hyperbaric oxygen therapy, 10 vasodilators, antioxidants, 11 and other adjuvant therapy options have been reported as successful management options.
The PENTOCLO protocol, a combination of pentoxifylline, which is antioxidant, tocopherol (or vitamin E), antifibrotic, and clodronate, a bisphosphonate, has proven to be a safe and cost-effective treatment regimen for ONJ. 12
The complexity of this case initially prompted differential diagnoses such as squamous cell carcinoma, medullary plasmacytoma, osteomyelitis, and osteonecrosis. However, the destructive nature of the lesion, coupled with the absence of a significant soft tissue component, ruled out squamous cell carcinoma, despite the patient’s extensive smoking history. Similarly, the erosion of the bone, as opposed to thickening or sclerosis typical of osteomyelitis, eliminated the possibility of osteomyelitis. Though medullary plasmacytomas were considered, given their similarity to MM and appearance as lytic lesions on CT, they are not typically associated with Actinomyces spp. colonization, a key feature of osteonecrosis—hence, osteonecrosis remains the focal point.
Of interest is the role of denosumab, a monoclonal antibody used for MM among other conditions, in bone remodeling. Prolonged use of denosumab may trigger ONJ due to its antiresorptive effect in reducing bone turnover by inhibiting RANKL, a critical player in osteoclast differentiation and survival, and subsequently bone remodeling. Compared to other available antiresorptive or anti-remodeling therapeutic options, denosumab has been shown to have a greater capacity to suppress bone turnover than the commonly used bisphosphonates. 13
This case underscores the possibility for delayed ONJ and progression even after discontinuation of bone-modifying agent. Since 2010, several cases of ONJ associated with denosumab have been reported.14,15 Typical onset time of ONJ for denosumab-induced ONJ is about 1.3 years since starting of treatment. 16 Several other factors, including chronic masticatory load, oral bacterial colonization, dental trauma, and the anti-angiogenic effects of bisphosphonates, could contribute to this delay. Chronic masticatory load led to constant need for bone remodeling, and when continuously inhibited by medication, can lead to osteonecrosis. Similarly, following dental trauma, inhibited bone remodeling processes hinder healing. The oral cavity is constantly colonized by bacteria, and it was proposed that the presence of bacterial product promotes bone resorption and delays healing. 17 The dosage of denosumab also seem to make a difference, with 120 mg monthly having a significantly higher risk of developing ONJ comparing to bisphosphonate while denosumab 60 mg every 6 months has similar odds ratio of developing ONJ comparing to bisphosphonate.
Denosumab has a half-life of less than 30 days. 18 While denosumab does not have the potential effective of being stored in bones and slowly released, 17 a study has shown through long-term follow-up, that after denosumab use was discontinued in osteoporosis patient, starting from 2 years posttreatment until even 10 years after, there is persistent decrease in bone remodeling, indicating prolonged remodeling inhibition beyond the discontinuation of denosumab. 19 This prolonged inhibition of bone remodeling might contribute to the delayed onset of ONJ.
It is generally thought that development ONJ in MM, when treated with antiresorptive agents, favors the mandible over maxilla with a ratio of approximately 2:1.20,21 In a 5 year retrospective cohort study, the ratio of mandibular over maxillary ONJ development in patients treated with denosumab was 3:1. 22 The mandible overall endures more mechanical stress and bone turnover than the maxilla, which makes it more susceptible to ONJ. It has been proposed that, compared to the maxilla, the mandible is relatively denser and more poorly vascularized, especially in the posterior region of the mandible, where the bone is denser and more compact, and thus even more susceptible to ONJ. 23
Understanding Denosumab and Preventive Strategies for Osteonecrosis
Recent studies have shed light on the nuanced mechanisms of denosumab, particularly its long-term impact on bone metabolism.24,25 Unlike bisphosphonates, denosumab does not incorporate into the bone matrix, but its inhibitory effects on RANKL can lead to a significant, albeit reversible, reduction in bone turnover. This is crucial in understanding the delayed onset of osteonecrosis, as the suppressed bone remodeling may persist even after the cessation of therapy, potentially contributing to the risk of osteonecrosis.
Considering these findings, it is imperative to adopt preventive strategies in the management of patients on denosumab therapy. Regular dental evaluations and maintaining rigorous oral hygiene are paramount, given the susceptibility of the jaw to infections and necrosis. 26 In addition, minimizing invasive dental procedures during denosumab treatment and closely monitoring bone health through periodic assessments can be vital in early identification and prevention of osteonecrosis.
Emerging research indicates that genetic predisposition plays a role in the susceptibility to osteonecrosis, particularly in patients receiving antiresorptive therapies like denosumab.27,28 Specific genetic markers, such as polymorphisms in the RANK, RANKL, and OPG genes, have been associated with an increased risk of osteonecrosis. This highlights the potential for personalized medicine approaches, where genetic screening could help identify patients at higher risk and guide the choice and duration of antiresorptive therapies.
Conclusion
With advancements in cancer care resulting in increased survivorship, there is a surge in once-rare treatment complications. This article highlights delayed onset medication-related osteonecrosis of the maxilla, a complication which had previously been infrequent. As survival rates and oncologic outcomes improve, there is an evolving landscape of complications in head and neck surgery. Therefore, a heightened clinical vigilance remains crucial, especially in evaluating patients with bony head and neck lesions who have undergone treatments such as denosumab, known for its antiresorptive properties affecting bone health. Adapting to these emerging challenges and understanding the evolving complications is critical for providing patient-centered care. Emphasizing the importance of continued research, this case calls for a deeper understanding of delayed manifestations, refining diagnostic protocols, and enhancing therapeutic strategies. These efforts are pivotal for providing comprehensive care to the ever-increasing population of cancer survivors, aligning with the ultimate goal of improving patient outcomes and their quality of life.
Footnotes
Authors’ Note
This study was presented at American Academy of Otolaryngology–Head and Neck Surgery annual meeting 2021. This article has not been published and is not under consideration elsewhere.
Data Availability
All data underlying the results are available as part of the article and no additional source data are required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Permission was obtained from the patient to use his images.
