Abstract
Introduction
Parotid gland tumors are prevalent in the head and neck area and are the most frequent type of salivary gland tumors, making up around 80% of all tumors.1,2 The primary approach for treating these tumors is surgical removal, 3 although safeguarding the facial nerve during the procedure is a highly demanding task. The facial nerve traverses the parotid gland and is accountable for the contraction of facial expression muscles. Any surgical damage to this nerve might result in varying levels of facial nerve dysfunction in patients. Statistically, the occurrence of facial nerve issues following surgery for parotid tumors varies between 10% and 30%.4-6 Approximately 0% to 7% of patients experience permanent facial nerve palsy, whereas 65% experience temporary abnormalities in facial nerve conduction.7,8 The primary manifestation of facial nerve dysfunction is facial paralysis, muscle weakness, or facial muscle asymmetry.9,10 These symptoms not only affect the patient’s physical appearance but can also lead to various quality of life issues, including speech disorders, difficulty swallowing, and psychological problems such as reduced self-confidence, anxiety, depression, and social isolation.11-13
The advancement of machine learning (ML) technology has led to the emergence of medical prediction models that utilize big data for preoperative risk assessment. 14 The ML model can utilize the patient’s clinical characteristics, tumor attributes, surgical details, and other information to identify the crucial factors that influence the risk of postoperative facial nerve dysfunction. This enables doctors to receive personalized surgical strategies and postoperative management recommendations. Furthermore, this method can assist patients in gaining a more comprehensive understanding of the potential hazards they may encounter and enable them to establish realistic expectations for their recovery after surgery.
While ML has shown promise in predicting disease risk and development in other medical domains, there have been limited studies on using it to predict the likelihood of neurological impairment after parotid tumor surgery.15,16
Most current research focuses on individual factors and lacks comprehensive risk assessment models that incorporate multiple variables. This study aims to develop a ML model to predict postoperative facial nerve dysfunction in parotid tumor patients, enhancing clinical risk assessment for better treatment planning and reduced complications. By analyzing extensive preoperative, intraoperative, and postoperative data, the study intends to construct a highly accurate prediction model and identify key predictors using ML techniques.
Methods
Data Collection
Reevaluation of data collected within the case management system of the Guigang City People’s Hospital throughout the period from 2013 to 2023.
Inclusion criteria: Pathological examination confirming the diagnosis of a parotid gland tumor, age of 15 years or older, prior surgical treatment, and comprehensive case information including personal details, examination data, and presence of facial paralysis.
Exclusion criteria: Patients with facial paralysis resulting from intraoperative resection of the facial nerve, significant coexisting medical conditions such as severe heart disease, liver and kidney dysfunction, prior facial nerve damage or previous head and neck surgery, and history of radiotherapy in the parotid region.
This is an Institutional Review Board (IRB)-approved retrospective study (Approval No. ELW-2024-008-01), all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Variable Selection
This study employed case systems to collect personal and medical data, pathology reports to gather tumor features, surgical records to document procedural details, and testing systems to measure blood indicators. In total, there were 73 variables included in the study. These included demographic data, medical history (hypertension, diabetes, smoking, alcohol consumption), body mass index (BMI), hospitalization duration, hematological indicators [fasting glucose, blood counts, liver and renal function, neutrophil to lymphocyte ratio (NLR), platelet to lymphocyte ratio (PLR), monocyte to lymphocyte ratio (MLR)], and surgical specifics (tumor location, surgeon experience). Variables like tumor type, size, bilateral presence, mass effect, and pain were also analyzed, with BMI, NLR, PLR, and MLR derived from patient data.
Statistical Analysis
Statistical analysis was performed with SPSS Statistics 25.0 (IBM Corporation, Armonk, NY, USA) and R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria), setting the significance threshold at P ≤ 0.05. Discrete and continuous variables were, respectively, described using frequencies, percentages (%), and means with standard deviations (SD). R 4.3.2 was used for feature selection, as well as training and validation of models, with data split into training and test sets at a 7:3 ratio. Facial paralysis after parotid tumor surgery was analyzed as a binary outcome, applying binomial-LASSO regression for key predictor identification. Model development utilized a 10-fold cross-validation to optimize parameters across Logistic Regression (Logit), Random Forest (RF), XGBoost (XGB), Artificial Neural Network (ANN), and Support Vector Machine (SVM) algorithms. Performance was assessed via receiver operating characteristic (ROC) curves, area under the curve (AUC), sensitivity, specificity, F1 score, and accuracy, with Delong tests comparing AUC values among models.
Result
Baseline Data of Patients Operated on for Parotid Tumors
This study comprised 403 individuals surgically treated for parotid tumors and 73 factors, with 56 (13.8%) developing postoperative facial paralysis. The data were divided into 2 sets: 283 cases for training and 120 cases for validation. The facial palsy group and nonfacial palsy group showed statistically significant differences in years of surgical experience, neutrophil percentage, basophils percentage, basophil count, red blood cell count, prothrombin time (PT), partial thromboplastin time, fibrinogen level, NLR, PLR, and tumor pain. However, the remaining variables did not show statistically significant differences (Table 1).
Baseline Clinical Characteristics of Parotid Tumors Patients.
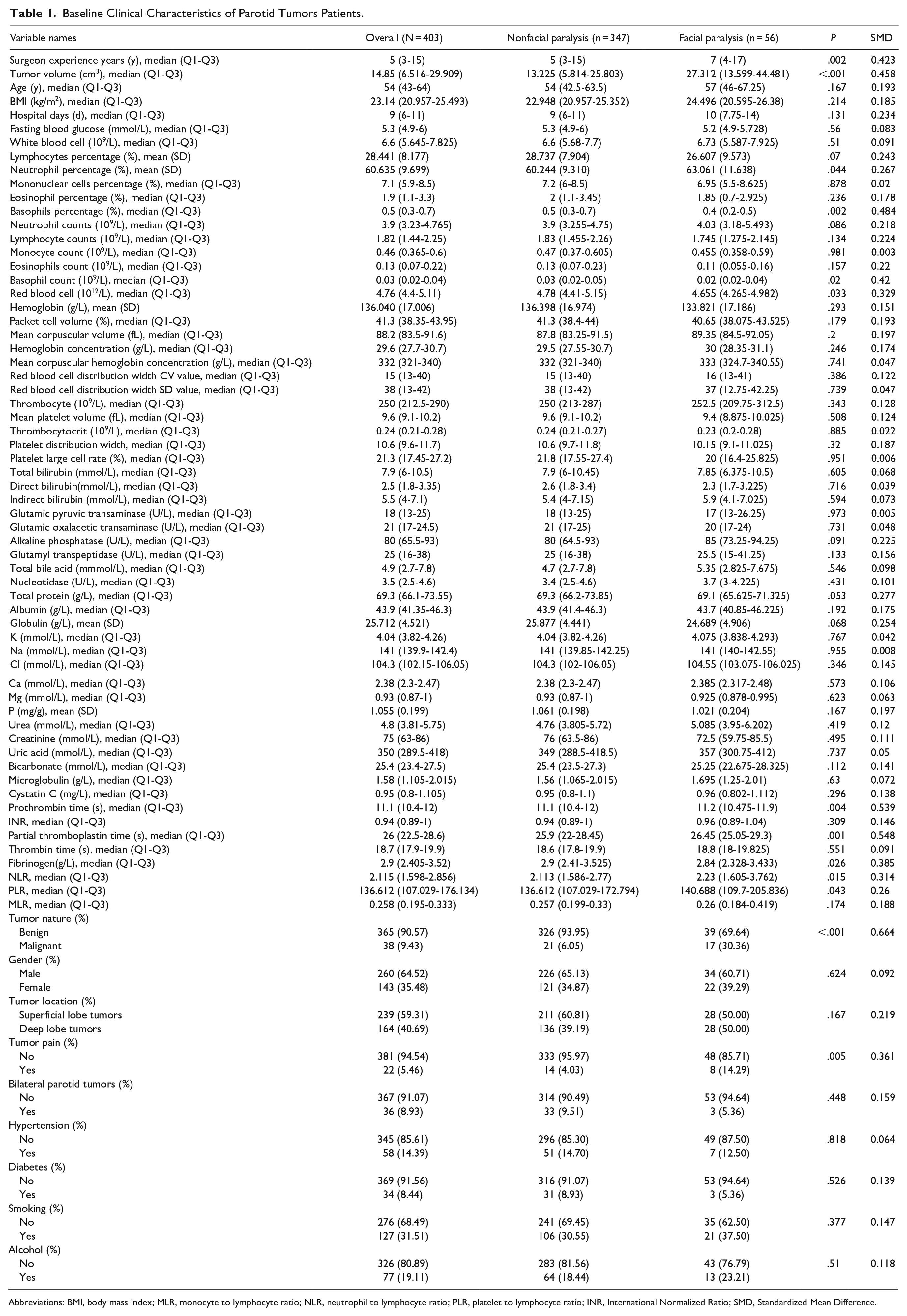
Abbreviations: BMI, body mass index; MLR, monocyte to lymphocyte ratio; NLR, neutrophil to lymphocyte ratio; PLR, platelet to lymphocyte ratio; INR, International Normalized Ratio; SMD, Standardized Mean Difference.
Variable Screening and Modeling
The Binomial-LASSO regression method was chosen to analyze the variables in this study. A total of 73 variables were included in the LASSO regression analysis. Through cross-validation, the optimal log lambda value of −3.61 was determined (Figure 1). At the log optimal lambda value, a comprehensive screening of 8 variables was conducted to identify nonzero variables. These variables include tumor nature, tumor pain, surgeon experience years, tumor volume, basophils percentage, red blood cell count, partial thromboplastin time, and PT. All 8 variables were included in the modeling process.
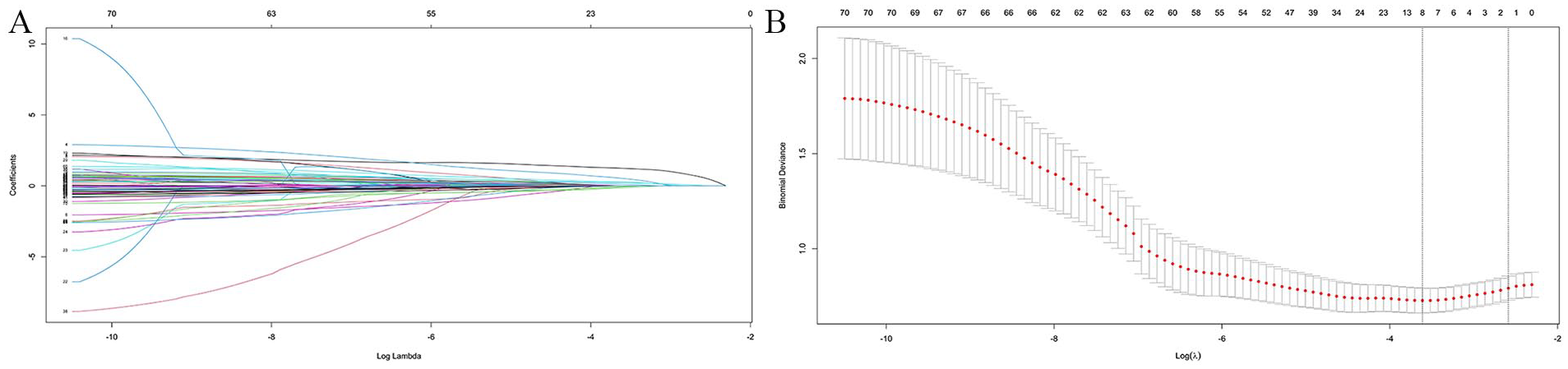
Variable selection based on LASSO regression. (A) LASSO coefficient curves for 73 variance variables. Eight variables with nonzero coefficients were selected based on the optimal log lambda value. (B) Determination of the optimal lambda value of the LASSO model by tenfold cross-validation.
Model Evaluation
Our study applied Logit, RF, XGB, ANN, and SVM algorithms to predict facial nerve dysfunction post-parotid tumor surgery, assessing model performance via ROC curves and AUC values (Figure 2): Logit (AUC = 0.829), RF (AUC = 0.752), XGB (AUC = 0.799), ANN (AUC = 0.829), and SVM (AUC = 0.724). Delong’s test indicated that the AUC values of the ANN and logit models were significantly better than those of the SVM model (P ≤ .05), but there were no significant differences in AUC values compared to the other models (P > .05). The models showed variable performance across training and validation sets (Table 2), with both the ANN and Logit models demonstrating the smallest discrepancies.
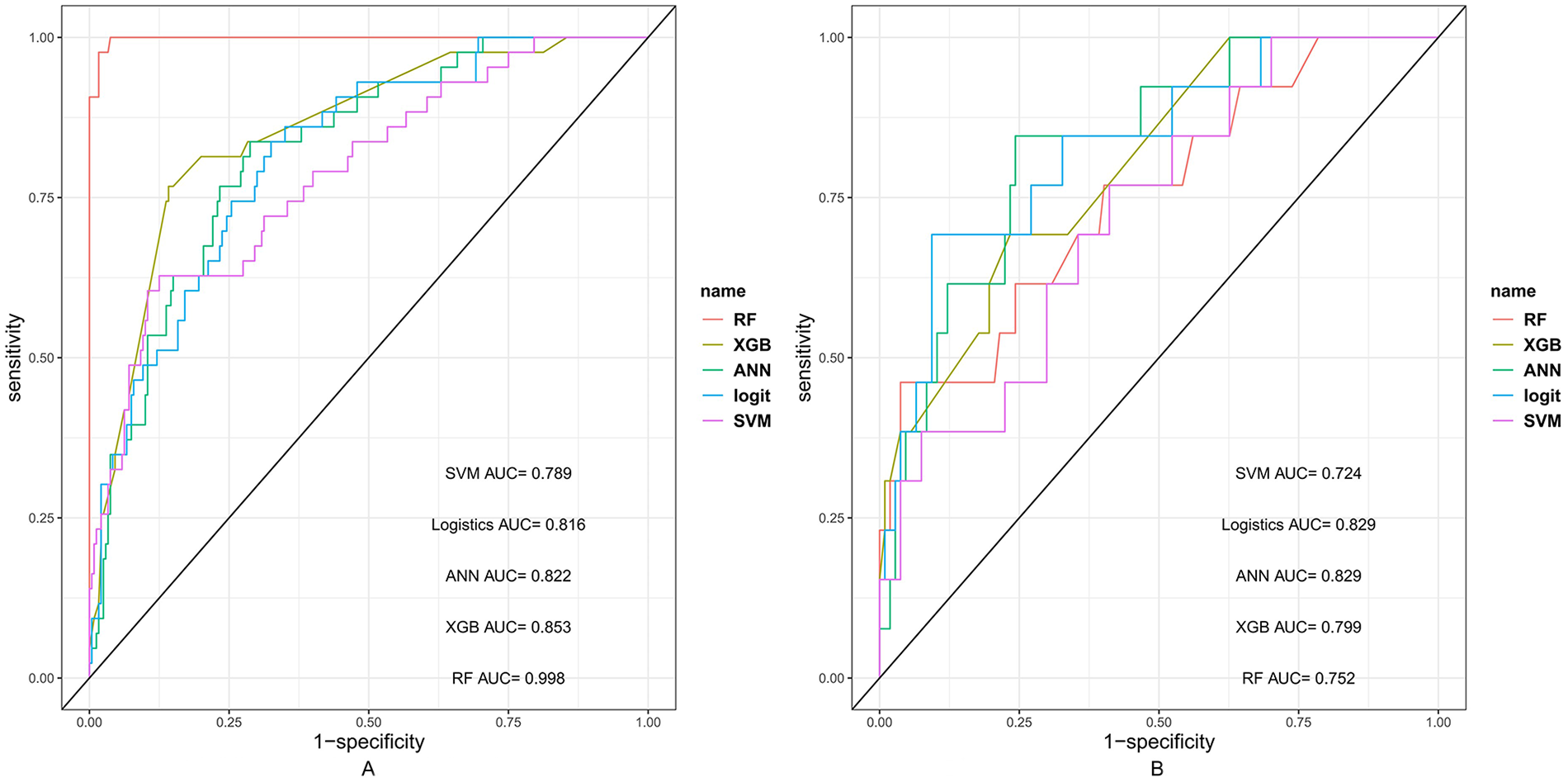
The ROC curves of 5 models in (A) train set and (B) validation set.
Performance of 5 Algorithmic Models on the Training and Validation Sets.
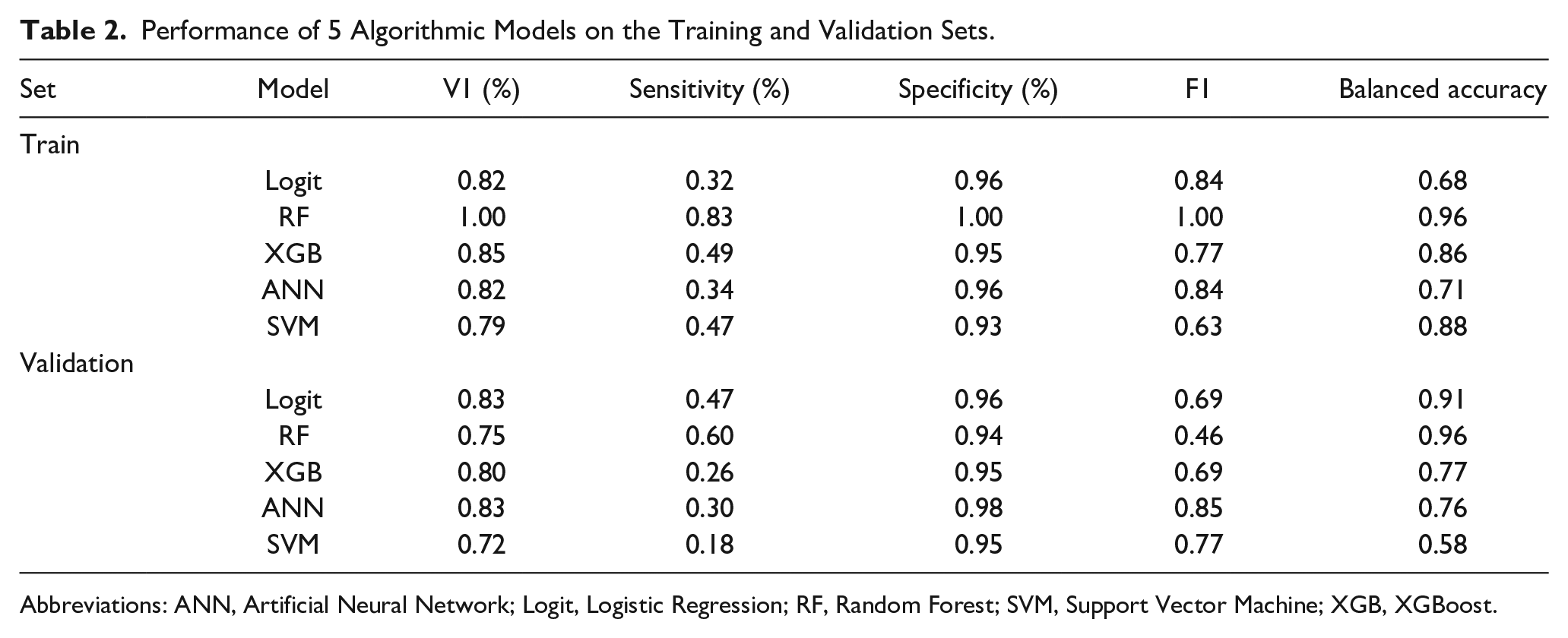
Abbreviations: ANN, Artificial Neural Network; Logit, Logistic Regression; RF, Random Forest; SVM, Support Vector Machine; XGB, XGBoost.
Discussion
In the field of head and neck pathology, particularly in the diagnosis and treatment of parotid gland tumors, ML technology plays a crucial role in developing high-precision predictive models. For instance, researchers have utilized convolutional neural networks to develop models that distinguish between pleomorphic adenomas and malignant parotid tumors, achieving classification accuracies as high as 90%. 17 In addition, ML models that utilize radiomics characteristics extracted from computed tomography scans are capable of distinguishing between low-grade and high-grade mucoepidermoid carcinomas. 18 ML models aid clinicians in forecasting the advancement of diseases and refining treatment approaches. For instance, the models created by Zhang et al have the ability to quickly and precisely differentiate between benign and malignant parotid tumors, therefore aiding in clinical diagnosis. 18 This showcases their considerable capacity to accomplish precision medicine and individualized treatment approaches in the realm of head and neck pathology.
Facial paralysis after surgery to remove a tumor in the parotid gland is a significant consequence, which can be influenced by various variables. Prior studies have revealed various crucial criteria, such as the size and location of the tumor, the scope of the surgical procedure, the expertise of the surgeon, and the specific surgical procedures employed.5,19-21 For example, tumors that have a significant dimension or are situated deep within the parotid gland elevate the likelihood of facial nerve damage since they require more extensive surgical removal. 22 The utilization of skilled surgeons and the implementation of facial nerve monitoring tools can significantly decrease the occurrence of facial paralysis.23-25
Through this study, we have successfully identified 8 characteristics that are strongly linked to the occurrence of facial paralysis after surgery. These findings are consistent with earlier research, especially in relation to the effects of tumor size and surgical techniques. 25 Contrary to earlier discoveries, the position of the tumor did not appear to be a significant factor in predicting outcomes. This could be due to the fact that other factors in the model, such as tumor size and the surgeon’s skill, may have already taken into consideration the influence of the tumor’s location. In addition, our study discovered other possible factors that may have an impact, such as the proportion of basophilic granulocytes and certain coagulation indicators. This suggests that the likelihood of postoperative facial paralysis could be linked to the patient’s inflammatory response and coagulation condition. Our work not only validates established risk variables but also offers novel perspectives on the intricate multifactorial characteristics of postoperative facial paralysis. This is vital for enhancing surgical preparation and minimizing postoperative sequelae.
This study utilized ML techniques to develop a model predicting postoperative neurological impairment in parotid tumor patients, incorporating 8 predictors. Although the performance varied among Logit, RF, XGB, ANN, and SVM models, it is noteworthy that the ANN and Logit models outperformed the SVM in terms of AUC values. However, the ANN and Logit models showed comparable predictive abilities to each other and to the RF and XGB algorithms, indicating similar efficacy across these models. This outcome underscores the importance of considering factors beyond performance, such as interpretability, computational efficiency.
The biological behavior of a tumor and its impact on surrounding structures are directly influenced by the nature, induration, and volume of the tumor. A larger tumor volume increases the likelihood of invasion and raises the risk of intraoperative injury to the facial nerve. Furthermore, the significance of operator expertise underscores the crucial function of a proficient level of skill in mitigating surgical problems and safeguarding the facial nerve. Because of their invasive characteristics, malignant tumors have a greater tendency to spread via nerve tissue, specifically affecting the facial nerve. As a result, individuals with malignant tumors are more prone to experience facial palsy. The current study’s results correspond with the findings of Kim et al, 9 Ruaux et al, 26 and Wiertel-Krawczuk et al, 27 which indicate that malignant tumors are a prevalent and separate risk factor for facial paralysis following parotid surgery. The current study demonstrated a substantial correlation between the volume of parotid tumors and the likelihood of experiencing facial paralysis. Furthermore, the size of the tumor plays a crucial role in evaluating the surgical risk and predicting the prognosis. Tumors of greater size pose a higher risk of surgical nerve damage due to their proximity to the facial nerve. In addition, larger tumor volumes can directly exert pressure on the facial nerve, hence increasing the likelihood of facial paralysis. In addition, the presence of discomfort in the tumor may suggest the occurrence of local inflammation or accelerated expansion, resulting in heightened pressure on adjacent tissues and consequent facial paralysis. Pressure pain can also suggest a loss of distinction between the tumor and nearby structures, which raises the likelihood of surgical damage to the facial nerve. These findings offer surgeons crucial factors to contemplate when preparing and performing parotid tumor surgery. They underscore the significance of preoperative risk evaluation and intraoperative neuroprotection, as well as the necessity for personalized treatment strategies to reduce the likelihood of facial paralysis. Furthermore, it is emphasized that including these risk variables into surgical planning and patient counseling is crucial for a more precise assessment and control of the risk of facial paralysis. 9
Regarding hematologic parameters, alterations in partial thromboplastin time, PT, and red blood cell count can indicate the patient’s overall condition and blood clotting ability, which are crucial for controlling bleeding during surgery and after surgery, promoting tissue healing, and managing inflammatory reactions. The presence of basophil percentage suggests that inflammation may have an influence on neurological rehabilitation. Modifications in red blood cells indicate changes in the small-scale environment of the blood, which can affect the blood supply around tumors and the circulation in tiny blood vessels. These factors are crucial in the process of nerve injury and healing. Significantly prolonged or decreased PT and activated partial thromboplastin time (APTT) can both have a substantial effect on the safety of the surgical procedure and the subsequent healing of brain tissues. Increased risk of bleeding and hematoma is associated with the former, while increased risk of thrombosis is associated with the latter. Both of these factors might indirectly affect the health of the facial nerve and its repair process. The findings of the current investigation are consistent with the results reported by Dulguerov et al, Yuan et al, Ruohoalho et al, and others.4,11,12,26 Surgical procedures for parotid tumors can cause nerve stretching and disrupt the blood supply to the nerve, increasing the likelihood of nerve damage due to compression and lack of blood flow. Factors such as coagulation, the presence of dead tissue near the nerve, and fibrosis play a significant role in the development of facial nerve dysfunction after surgery. Abnormalities in the PT and APTT are also important indicators of this dysfunction. Abnormalities in the blood clotting system, which can affect the blood vessels and overall health of the facial nerve, may cause facial paralysis in individuals with parotid tumors. These abnormalities serve as markers for the underlying pathways of the blood clotting system.
Our investigation revealed that the percentage of basophils were significant indicators for predicting facial nerve impairment following surgery for parotid tumors. Currently, a number of hematologic markers, such as PLR and NLR, have been shown to be helpful as prognostic predictors of many diseases, including inflammatory disorders associated with cancer and infections.28,29 The rise in basophil percentage may indicate the presence of inflammation in the body, which is directly linked to the healing process of nerve damage. This factor ultimately affect the functional recovery of the facial nerve. Monitoring this indicator offers a fresh viewpoint on postoperative care, highlighting the importance of preserving the patient’s metabolic balance to enhance the recovery of nerve function.
In summary, our research highlights the significance of various parameters in predicting the occurrence of facial nerve dysfunction after surgery for parotid tumors. This underscores the necessity for a thorough evaluation strategy in clinical settings. By incorporating these variables into a prognostic model, we anticipate enhancing the precision of preoperative guidance, optimizing surgical preparation, and reducing the likelihood of facial nerve dysfunction through meticulous postoperative care, ultimately enhancing the well-being of our patients. The successful execution of this strategy necessitates a tight-knit partnership among a multidisciplinary team, comprising surgeons, anesthesiologists, nursing personnel, and rehabilitation experts, who collaborate harmoniously to deliver holistic care and assistance to patients.
Limitation
Our study encountered limitations in forecasting postoperative facial paralysis among parotid malignancy patients. The cross-sectional survey design precluded causal inferences. Moreover, as a retrospective analysis, the scope was restricted by the availability and quality of historical data, influencing variable selection. The binary consideration of postoperative facial palsy, necessitated by the small sample size, enabled sensitive predictions but limited detailed exploration. Future studies should leverage larger, multicentric prospective cohorts and incorporate comprehensive evaluations of facial palsy severity to enhance predictions of its postoperative occurrence.
Conclusion
Our study included a cohort of 403 individuals who had undergone surgery for parotid tumors. Out of these patients, 56 experienced facial nerve impairment. An ML approach was utilized to develop a risk prediction model for postoperative facial nerve dysfunction using real-world clinical data. The efficacy of the model in predicting the likelihood of postoperative facial nerve dysfunction in patients with parotid tumors has been confirmed. The model demonstrates superior accuracy and reliability when compared to conventional clinical prediction approaches. Hence, the model can serve as a potent instrument to aid physicians in formulating precise treatment plans and prognosis assessments.
Footnotes
Acknowledgements
We express our heartfelt gratitude to all the clinical workers involved in the data collection process, whose diligent work made this research possible. Special thanks to Dr Shuai Jin for his expert guidance in the methodology of machine learning techniques, whose insightful and valuable advice greatly enriched the content and methods of our study. In addition, we would like to thank Professor Jiping Su for his precious revision suggestions during the manuscript writing stage, whose meticulous review and professional advice significantly improved the quality of our article. We deeply appreciate the support and assistance from all of them.
Author Contributions
Lianzhao Yang and Ruilin Li initiated this project, while Shuimei Li, Sujuan Xu, Chenxingzi Wu, and Wenjuan Wang were in charge of data collection and organization. Zhanhang Zheng and Shuhong Qin were involved in the design of the model and the analysis of the data. The research report was co-authored by Ruilin Li and Zhanhang Zheng. Ruilin Li contributed to the review and revision of the manuscript, while Lianzhao Yang oversaw the entire effort. Ruilin Li and Zhanhang Zheng have contributed equally to this article. The final article was reviewed by all authors and unanimously approved for publication.
Data Availability Statement
All data generated during this study are included in this article. Further enquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our project has received support from the High-Level Talent Cultivation Innovation Team Fund of Guangxi University of Chinese Medicine, under grant number 2022A010.
Ethical Statement and Inform Consent
This study received ethical approval from the Guigang City People’s Hospital IRB (Approval No. ELW-2024-008-01) on April 2, 2024. This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
