Abstract
Introduction
Chronic tympanic membrane (TM) perforations are the common entity in otologic clinic. The most common cause of TM perforations are chronic suppurative otitis media (CSOM).1 -5 Usually, the repair of chronic perforations depends on the reconstruction of the graft tissue; specifically, freshening or cauterization of the perforation edges is usually insufficient to promote healing. The course of the healing process of a perforation closed with a graft appears to be quite different from that associated with spontaneous healing. 6 Thus, questions remain with regard to the healing process involved in graft reconstruction following myringoplasty. Some scholars believe that the graft plays a scaffolding role, in which the proliferating epithelium of the perforation edges migrates along the lateral and medial sides of the graft, subsequently replacing the graft with new connective tissue.6,7 Others have suggested that the healing process of the TM starts from the fusion of the graft with the lamina propria layer of the remnant TM.8,9 Similar to the grafts used for a skin wound, the traditional view is that myringoplasty requires the removal of scar tissue at the perforation margin, or else the graft is unlikely to heal.10,11 However, even medium-sized perforations may become larger after trimming the margin or removing abnormal lesions of the remnant TM (eg, myringosclerotic plaques, atrophic lesions). 12 The healing process of a skin graft has classically been divided into 3 phases: imbibition, inosculation, and neovascularization. However, although healing via a graft is a unique physiological event following myringoplasty, little is known about this process. The conventional treatment is to place a dressing pack within the external ear canal (EAC) following the myringoplasty procedure, as this is believed to hold the graft in situ and improve the graft success rate. However, one drawback of EAC packing is the inability to assess graft healing in the early postoperative period. Although there are some accounts in the literature describing the TM healing process, changes in the graft were only assessed at 2 to 3 weeks postoperatively due to EAC packing, and trimming of the perforation margins was performed.12 -14 To date, no studies have evaluated the graft changes after myringoplasty without trimming of the perforation margin. Interestingly, failure to trim the perforation margin does not appear to affect the short- or long-term graft success rate.15,16 Similarly, the absence of EAC packing does not seem to have an effect on the graft success rate. 17 Without EAC packing, changes in the remnant TM and graft can be observed more readily. Thus, the objective of this study was to evaluate the graft healing process and graft success rate following myringoplasty, without trimming of the perforation margin and EAC packing.
Subjects and Methods
Ethical Considerations
Ethical approval for this study was obtained from the Medical Ethical Committee of Yiwu Central Hospital. Informed consent was obtained from all participants.
Patient Selection
The study subjects were recruited from consecutive patients diagnosed with chronic perforation who visited the Department of Otorhinolaryngology, Head and Neck Surgery. The inclusion criteria were as follows: adult chronic perforation with chronic otitis media, a dry ear for at least 3 months prior to surgery, air-bone gas <40 dB, and no synechia of epithelial or mucosal layers of the remnant TM by endoscopy examination. The exclusion criteria were revision cases, cholesteatoma, ossicular chain abnormity, middle ear inflammation, fungal otitis externa, and patients with other underlying diseases. Computed tomography scan of the temporal bone was performed prior to surgery to exclude the cholesteatoma of mastoid. The perforation size was categorized as small (<1/8 of the TM area), medium (1/8-1/4), large (1/4-1/2), or subtotal (1/2-3/4).
Surgical Technique
The surgical technique has been described in detail in a previous study. 17 Briefly, all patients underwent endoscopic perichondrium-cartilage underlay myringoplasty with no tympanomeatal flap elevation. A single-layer, perichondrial, composite cartilage graft was harvested from the ipsilateral tragus. The lateral perichondrium was peeled circumferentially, with pedicle attachment of the cartilage graft. The cartilage graft was shaped to be 1 mm larger than the perforation margins, with the perichondrium >1 to 2 mm larger than the cartilage graft. The perforation margins were not de-epithelialized. However, the epithelium was removed from the distal malleus handle if present. A channel for the malleus handle was made in the cartilage graft if any. The biodegradable NasoPore material was placed dry in the middle ear but not saturated with any medications. The perichondrium-cartilage composite graft was placed transperforation, and the cartilage graft was set medial to the remnant TM and the annulus. The channel in the cartilage graft accommodated the malleus if any. Also, the perichondrium was positioned medial to the remnant TM and the annulus but lateral to the cartilage. No EAC packing was applied.
Postoperative Follow-Up
All patients were discharged on the day after surgery. Antibiotics were not given in all cases. Outpatient endoscopic inspections were scheduled at 1, 2, 3, and 4 weeks, and 3 and 6 months, postoperatively. Graft neovascularization was assessed by 2 otology specialists who were blinded to the treatment groups at 1, 2, 3, and 4 weeks postoperatively. Graft success was defined as the presence of an intact graft; graft failure was defined as the presence of residual perforation after surgery. Clinical neovascularization was defined as the presence of new capillaries and microvessels (of various diameters) on the graft surface by endoscope. A graft neovascularization score (ie, rate of neovascularization of the entire graft) was derived using a static endoscopic photograph, with the following grades: 0, no vascular proliferation; 1, neovascularization <50% of the graft area; 2, neovascularization >50%; and 3, extensive neovascularization. 17 The vascularization time of a graft was defined as the time required for the first confirmed appearance of new vessels on the graft following surgery.
Results
Demographic Data
Fifty-eight patients (58 ears) were included in this study. Of the 58 patients, 38 (65.5%) were female and 20 (34.5%) were male (mean age at surgery, 42.5 ± 2.1 years; range: 39-65 years). The left ear was evaluated in 37 (63.8%) patients and the right ear in 21 (36.2%). The perforation size was small in 9 ears (15.5%), medium in 17 ears (29.3%), and large in 24 ears (41.4%), with subtotal perforation seen in 8 (13.8%) ears. Of the 58 ears, varying degrees of myringosclerosis of the remnant TM was seen in 16 (27.6%) ears.
Endoscopic Observation
At 1 week postoperatively, the blood vessels of the remnant TM were dilated and congested, and the upper TM was most prominent. Clinical inosculation of the graft and remnant TM were observed in 9 small perforations and 17 medium-sized perforations (Figure 1). Neovascularization was initiated at the graft margins or most of the graft surface showed neovascularization. For 24 large perforations and 7 subtotal perforations, the blood vessels of the remnant TM were dilated and congested; graft neovascularization was not observed clinically, but a large amount of exudate was evident. The clinical inosculation of the graft and the remnant TM began in 11 large perforations, but was not seen in 13 large perforations or 7 subtotal perforations.
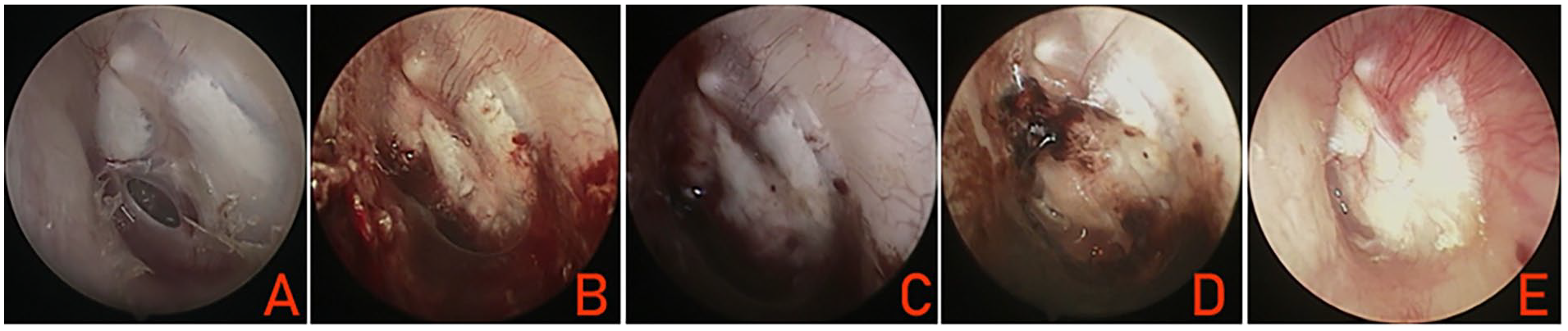
(A) Preoperative perforation. (B) At 1 week postoperatively. (C) At 2 weeks postoperatively. (D) At 4 weeks postoperatively. (E) At 2 months postoperatively.
At 2 weeks postoperatively, graft clinical inosculation and neovascularization were completed in 9 small perforations and 17 medium-sized perforations. For 24 large perforations and 7 subtotal perforations, in which the graft had been clinically incorporated to a considerable extent and neovascularization of the graft was initiated, there was a marked increase in exudate. Of these perforations, complete neovascularization was achieved in 9 large perforations.
At 3 weeks postoperatively, complete graft clinical inosculation was achieved in 57 ears. Complete neovascularization was achieved in 24 large perforations, and partial neovascularization was achieved in 7 subtotal perforations (Figure 2). Capillary diameter and functional capillary density within the grafts increased further.
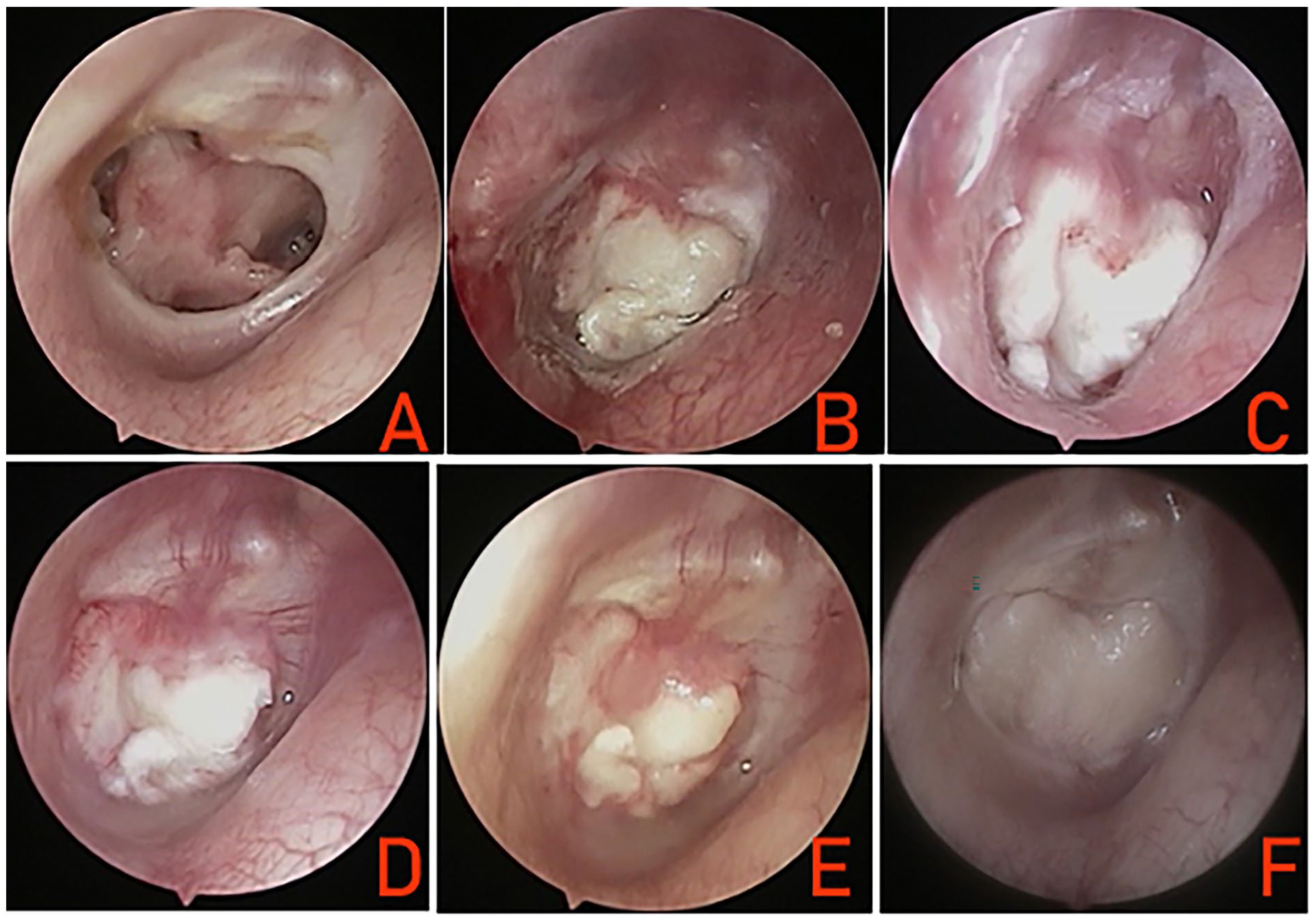
(A) Preoperative perforation. (B) At 1 week postoperatively. (C) At 2 weeks postoperatively. (D) At 3 weeks postoperatively. (E) At 5 weeks postoperatively. (F) At 12 months postoperatively.
At 4 weeks postoperatively, complete neovascularization was achieved in all small-, medium-, and large-sized perforations. Of the 7 subtotal perforations, complete neovascularization was achieved in 5 ears, and no neovascularization was observed in the middle of the graft in 2 ears. Endoscopy results showed that clinical inosculation of the graft and the remnant TM involved the inosculation of all layers, but not of the graft or mucous layer. In addition, there was no effect of the absence of trimming of the perforation margin on the inosculation of the graft or remnant TM. Also, the myringosclerosis of the remnant TM did not affect the graft clinical inosculation and graft neovascularization time.
One subtotal perforation had postoperative infection within 1 week. Graft clinical inosculation did not occur, the upper portion of the graft was extruded by purulent otorrhea, and the remnant TM vessel congestion was not significant. The antibiotics and anti-inflammatory were given, the inflammation was controlled within 1 week after treatment. At 2 weeks postoperatively, graft clinical inosculation was not seen except for the posteroinferior graft and the remnant TM. Graft neovascularization was not seen clinically. At 3 weeks postoperatively, graft clinical inosculation was seen at the inferior and anterior margins of the graft. Graft neovascularization was not seen clinically. At 4 weeks postoperatively, graft neovascularization was seen clinically at the inferior and anterior margins of the graft. The upper part of the graft became pale and necrotic, resulting in residual perforation at 6 weeks postoperatively.
Graft Neovascularization Scores and Graft Success
Of the 58 ears, postoperative infection resulted in residual perforation in 1 ear, and an insufficient graft resulted in residual perforation in a large perforation without infection. The overall graft success rate was 96.6% (56/58).
We evaluated the graft neovascularization scores at 4 weeks postoperatively and the graft success rate at 6 months postoperatively. The myringosclerosis did not affect the graft neovascularization score. The graft neovascularization scores were as follows: score of 0 in subtotal perforation with postoperative infection, with a graft success rate of 0%; score of 1 in 13 ears, with a graft success rate of 100%; score of 2 in 38 ears with residual perforation of 1 large perforation, with a graft success rate of 97.4%; and score of 3 in 7 ears, with a graft success rate of 100%.
Discussion
Little is known about the healing process of grafts following myringoplasty. Although several studies have described the healing process following temporalis fascia myringoplasty, in these studies, only a few patients underwent trimming of the perforation margin and EAC packing.12 -14 With the development of endoscopic myringoplasty, endoscopic cartilage myringoplasty is becoming increasingly popular. To date, however, the healing process has not been clinically reported following cartilage myringoplasty. In addition, the absence of EAC packing did not affect the graft success rate.17 -19 The advantage of the absence of EAC packing is that changes in the graft and remnant TM can be observed directly. This study is the first to clinically evaluate the healing process of endoscopic cartilage myringoplasty.
We found that the cartilage grafts also underwent imbibition, inosculation, and neovascularization during the healing process. The blood vessels of the remnant TM were dilated significantly and congested at 1 week postoperatively, especially the upper TM and handle of malleus. A large number of capillary blood vessels were observed at the perforation margin. For small- and medium-sized perforations, the capillary vessel of the perforation margins tended to grow into the edges of the graft, accompanied by a large number of new vessels in the graft surface. This agrees with the study of Applebaum and Deutsche, 14 in which vascularization of the grafted fascia started as early as 7 days after surgery. Clinical inosculation of the graft and the remnant TM were achieved within 1 week for small- and medium-sized perforations. Osborn et al 20 found that angiogenesis of host blood vessels into the proximal region leads to inosculation between the host and donor vessels within the first week after graft placement. The introduction of the graft stimulates the perforation margins and creates a response similar to an infectious reaction, with the ingrowth of connective tissue with numerous capillaries. This vascular ingrowth is generated by the perforation edge, as well as by the tympanic edge through the rebased graft. 13 We speculated that the healing process following myringoplasty was similar to the healing of a skin graft, in which the dilatation and congestion of blood vessels in the remnant TM are firstly initiated, followed by the inosculation of the graft and the remnant TM and graft neovascularization.
This study found that the time required for inosculation and vascularization of the graft usually varies depending on the individual. Also, similar to the findings of a previous study, 12 the inosculation and vascularization times differed significantly according to the perforation size in this study. The inosculation in large and subtotal perforations was delayed significantly compared with that of small perforations, with complete inosculation taking 2 weeks and complete neovascularization taking 3 to 4 weeks. This may be related to the graft size, as the graft requires a remodeling process prior to inosculation such that the remodeling time for a large graft is longer than that of a small graft. Tuba and Kálmán 21 suggested that the graft must reach a stage of maturation to receive the ingrowing vessels. Krum and Rosenstein 22 found that mature autonomic tissue stimulates the growth of adjacent host vessels when transplanted to undamaged brain surfaces. In addition, we speculated that a larger graft would require remodeling by massive plasma exudation. Some scholars believe that the graft should include “plasmatic circulation,” revascularization, and organization; specifically, they found in animal experiments that the nutrition provided by plasmatic circulation is necessary for graft vitality. 13 An improvement in the plasmatic circulation can be obtained by filling up the tympanic cavity with nutrient solution. 13
Different from previous studies, endoscopic observation showed clinical inosculation of the graft and the remnant TM of all layers, with the exception of the graft and mucous layer. Some scholars believe that the graft is replaced by new connective tissue following inosculation.6,7 We found that the cartilage graft was not clinically replaced by new connective tissue. Trakimas et al 23 also found that temporalis fascia does not remodel significantly, nor change its thickness or fibrous structure following a successful tympanoplasty. In addition, this study suggested that the absence of trimming of the perforation margin did not prevent the inosculation of the graft or the remnant TM, nor graft neovascularization. Sagiv et al 24 demonstrated that the scar tissue of perforation margins has vascular and proliferative activity. Graft survival depends on the congestion of the remnant TM and the inosculation of the graft and the remnant TM, followed by graft neovascularization. Therefore, the results of this study further confirmed our previous conclusion that the graft success rate is not dependent on trimming of the perforation margin.
Also, no significant relationship was observed bet-ween the vascularization time and myringosclerosis in the remnant TM. This finding agrees with the results of a previous study. 12 Thus, myringoplasty should preserve the myringosclerosis in the remnant TM as much as possible. Similar to the findings of Noh and Lee, 12 there was no correlation between the graft success rate and graft neovascularization. This study found no significant difference in the graft success rate among 3 types of neovascularization. Graft survival depended on graft neovascularization and inosculation. Moreover, graft success depended not only on graft survival but also graft size. Although the graft was alive, a posteroinferior residual perforation was found in a large perforation because of the insufficient size of the graft.
Noticeably, postoperative infection was observed in 1 subtotal perforation within 1 week after surgery. Although the inflammation was controlled in the case within 1 week of antibiotics and anti-inflammatory treatment, it resulted in incomplete inosculation and failed neovascularization, in turn resulting in residual perforation. This result suggests that postoperative infection prior to graft inosculation may result in graft collapse. The infection may result in chronic hypoxia, which impairs neovascularization. 25 For this reason, decisive therapeutic steps should be taken when the first indication of inflammation occurs, 13 antibiotics and anti-inflammatory are given in all for at least 1 week, as it could result in failed inosculation and neovascularization if the graft remodeling fails.
One advantage of the current study was that no patient was lost to follow-up. In addition, the graft change could be observed endoscopically at any time points due to the absence of EAC packing. The limitations of this study include a small sample size, a descriptive study of clinical morphological observation only; there were no attempts to objectively quantify the inosculation or neovascularization of the graft.
Conclusions
The graft healing process experienced the dilation of the blood vessels of the remnant TM, graft clinical inosculation, and neovascularization following cartilage myringoplasty without trimming of the perforation margin and EAC packing; however, the graft success rate was not related to the endoscopic graft neovascularization scores.
Footnotes
Author Contributions
MW: interpretation of data for the work, design of the work, analysis of data for the work, drafting the work, agreement to be accountable for all aspects of the work, and final approval of the version to be published; ZL: interpretation of data for the work, design of the work, and final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by Health Commission of Zhejiang Province, China (Grants#2021KY1186), Technology Agency of Jinhua City, China (Grants#2022-3-042).
