Abstract
Pseudomyogenic hemangioendothelioma (PMHE) is a rare vascular neoplasm with an intermediate to low-grade malignant potential. Only 5% of PMHEs occur in the head and neck. This tumor exhibits different histological patterns and mimics other vascular tumors, myoid tumors, or carcinomas. The distinction between these tumors can be a very challenging situation for pathologists. In this article, we report the first case, to our knowledge, of PMHE arising in the maxillary sinus, to highlight this uncommon entity and discuss differential diagnoses.
Introduction
Pseudomyogenic hemangioendothelioma (PMHE) was recognized as a distinct entity under the name epithelioid sarcoma (ES)-like hemangioendothelioma in 2003. 1 The term “pseudomyogenic” refers to its histopathologic pattern, which is similar to that of myoid and epithelioid tumors. 2 Histologically, PMHE is characterized by poorly defined nodules of plump spindle and epithelioid cells. It shows an abundant, densely eosinophilic cytoplasm that grows in sheets and fascicles. Its vascular differentiation is essentially inapparent, with rare intracytoplasmic lumens, making this type of tumor extremely difficult to diagnose. 3
Case Report
We report a case of extensive maxillary PMHE, in a 37-year-old woman without a clinical history. She was admitted for a swelling on the right cheek that progressively developed over 1 year (Figure 1A).
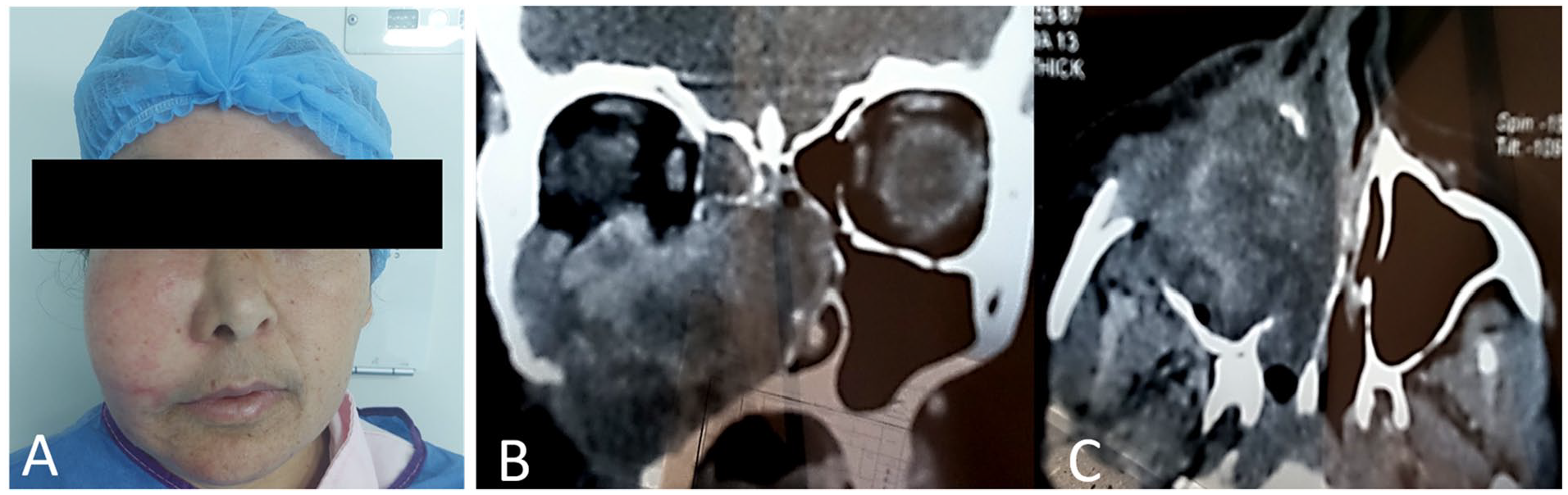
(A) Clinical photograph showing swelling in the right cheek. Axial (B) and coronal (C) CT scans show a large tissular mass that occupies the right maxillary sinus with extension to inferior wall of the orbit, the nasal septum, and the posterior wall of the ethmoidal sinus. CT, computed tomography.
The computed tomography scan showed a large tissular mass that appeared in the maxillary sinus with significant bone erosion and extension to the inferior wall of the orbit, the nasal septum, and to the posterior wall of the ethmoidal sinus. There was no muscle or skin involvement (Figure 1B).
Endoscopic biopsy revealed nonspecific ulcerative and inflammatory changes. Therefore, the patient benefitted from a partial surgical excision. Histological analysis of the sample showed spindle cell proliferation with an infiltrative growth pattern. Abundant myxohyaline stroma containing increased inflammatory changes and prominent neutrophil infiltration was noted. Focal areas of irregular vascular channels and clefts with hemangiopericytoma-like features were observed. Tumor cells showed an abundant eosinophilic cytoplasm, containing vesicular nuclei with small nucleoli, mimicking myoid cells (Figures 2 and 3). This pattern led us to discuss inflammatory myofibroblastic tumor (IMT), spindle cell carcinoma, sarcomas, or vascular tumor such as PMHE.
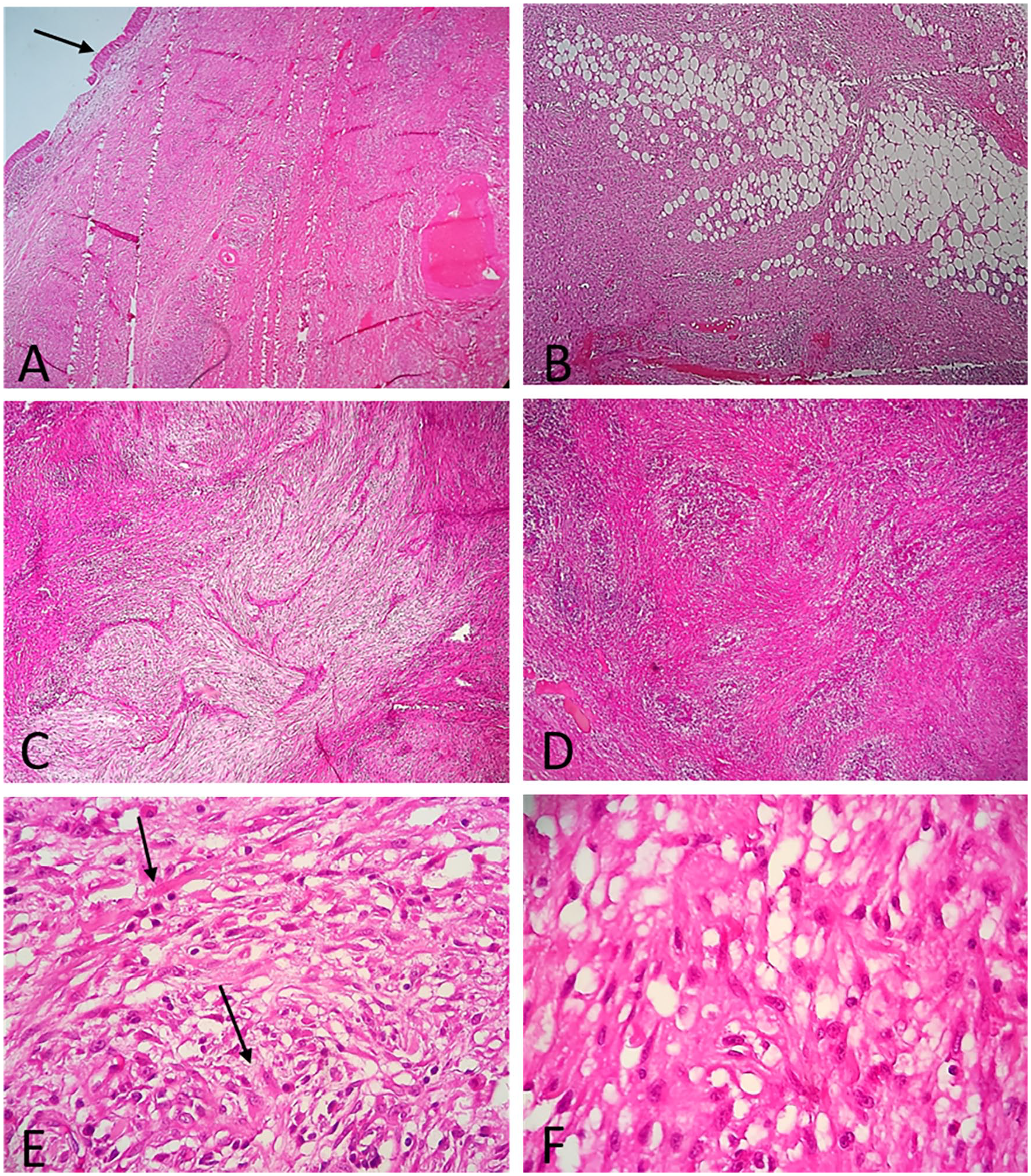
(A) Microphotography of PMHE showing spindle cell proliferation under the epithelium of sinus mucosa (arrow) with infiltrative growth pattern (B). Tumor is composed of sheets and fascicles, with some irregular vascular channels exhibiting a hemangiopericytoma-like appearance within a myxoid stroma (C) and increased inflammatory changes (D; hematoxylin-eosin ×10). The tumor cells show an abundant eosinophilic cytoplasm with small nucleoli, mimicking myoid cells (arrow) (E). Vascular clefts are observed (F; hematoxylin-eosin ×40). PMHE, pseudomyogenic hemangioendothelioma.
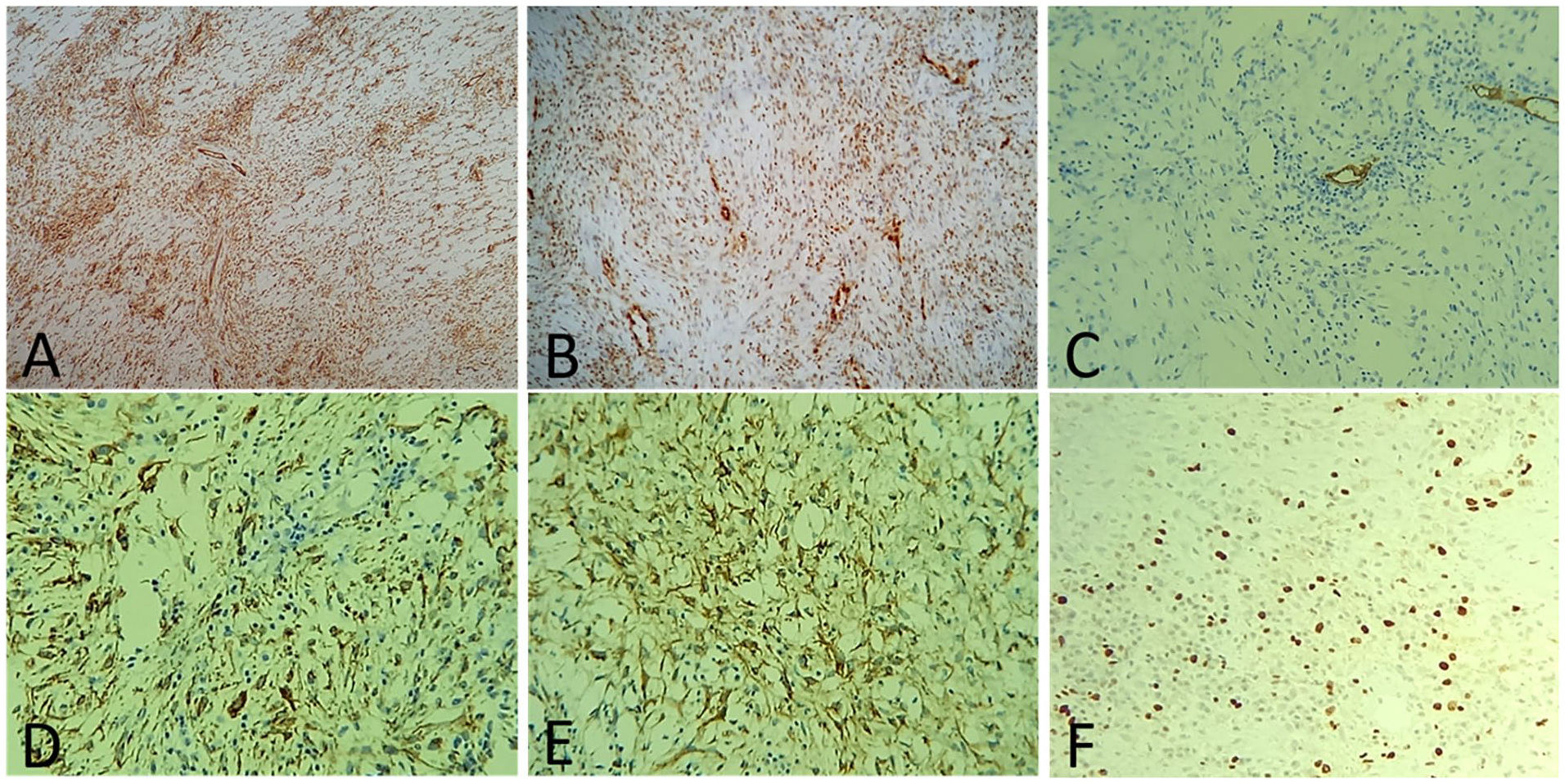
At immunohistochemistry, the tumor cells diffusely express CD31 (A) and ERG (B), while CD 34 (C) is negative. They express CK AE1/AE3 (D) and CK7 (E). Ki67 is low (F).
In immunohistochemistry, the tumor cells diffusely expressed epithelial markers (CK AE1/AE3, CK7) and vascular markers (CD31 and ERG).
CD34, P40, CK5/6, EMA, smooth and skeletal markers (SMA, desmin, myogenin, H-caldesmon), melanotic markers (melan A, HMB45), ALK, MUC4, beta-catenin, and IGG4 were negative. INI-1 expression was intact and Ki67 low. No ALK fusion was found in next-generation sequencing analysis.
Based on these histological and immunohistochemical features, the diagnosis of PMHE arising in the maxillary sinus was made. After surgery, the patient benefitted from radiotherapy with a significant reduction in tumor residue.
Discussion
Hemangioendothelioma is a vascular tumor of intermediate to low grade, characterized by proliferating neoplastic endothelial cells. Tumor cells may form small intracellular lumen, which can be seen as clear spaces, or vacuoles, that distort (or blister) the cell. 4
Enzinger and Weiss have classified hemangioendothelioma into epithelioid, kaposiform, hobnail, composite, and ES-like hemangioendothelioma.4,5 The latter is currently recognized as PMHE in the fifth edition of WHO Classification of Tumours of Soft Tissue and Bone and is defined as a rarely metastazing endothelial neoplasm that occurs more frequently in young adult men. It often presents as multiple discontiguous nodules in different tissue planes and usually arises in the lower extremities (55% of cases). It rarely occurs on the head and neck (5% of cases). 6
In the study of Mittal et al, 7 to date 2021, only 12 cases of PMHE have been reported in the head and neck region, including scalp, nose, and oral cavity involvement. 7 To our knowledge, this is the first case of PMHE that arises in the maxillary sinus. In this location, the pathological diagnosis can be a challenge. This tumor, unlike other vascular tumors, lacks vascular differentiation with multicellular vascular channels or intralesional hemorrhage 3 and mimics myoid tumor, carcinoma, or ES. 6
Histologically, it is composed of sheets and loose fascicles of plump spindle cells with abundant, brightly eosinophilic cytoplasm mimicking rhabdomyoblasts. A minor component of cells with epithelioid cytomorphology is often present, but sometimes epithelioid cells predominate. Tumor cells contain vesicular nuclei with generally small nucleoli. The degree of nuclear atypia is usually mild, and mitotic activity is scarce. 6
This infiltrative tumor often has a stromal neutrophilic infiltrate, and sometimes it has a myxoid change in the matrix. 3 In our case, the tumor showed significant inflammatory, fibrohyaline, and myxoid changes, leading us initially to think it as IMT. However, both tumors have different immunohistochemical phenotypes: IMT expresses SMA diffusely, while focal expression of SMA is observed in one-third of PMHE. 6 In addition, PMHE expresses FLI-1 and ERG (in all cases) and CD31 (in 50% of cases), confirming its endothelial differentiation. 2
This immunoprofile leads us to discuss other vascular tumors, such as epithelioid hemangioendothelioma or epithelioid angiosarcoma; however, the inconsistency of CD31 reactivity and complete lack of CD34 expression, as well as lack of apparent histologic evidence for endothelial differentiation, are distinct features of PMHE. 2
In some cases, the pattern of spindle cells can be confused with a myoid tumor such as leiomyosarcoma or rhabdomyosarcoma. In such cases, PMHE negative staining for specific markers of the skeletal muscle, namely desmin, myogenin, and MyoD1, rule out these differential diagnoses. 7
PMHEs are characterized by the fact that they show a broad expression of cytokeratin AE1/AE3. 2 This expression leads to misdiagnosis of carcinoma, especially at mucosal sites, where carcinomas are more common. AE1/AE3 is particularly common in epithelioid vascular neoplasms. 8 Interestingly, high molecular weight cytokeratin (CK14 and 34βE12) is not detected in vascular neoplasms. 8 Furthermore, EMA staining is usually weak and pancytokeratin MNF-116 is negative in PMHE. 2
Differential diagnoses also include ES. Histologically, ES tends to be composed of smaller, well-defined nodules rather than the sheetlike, vaguely nodular growth seen in PMHE; it shows greater nuclear atypia in addition to geographic necrosis. Although there is an immunophenotypic overlap with PMHE, ES typically lacks reactivity to CD31, FLI1, and SMARCB1 (INI-1), unlike PMHE. 2
Recently, SERPINE1-FOSB has been identified as a specific fusion gene in PMHE. 9 Therefore nuclear staining for FOSB is a consistent finding 6 to confirm the diagnosis, but it is also expressed in epithelioid hemangiomas and osteoblastomas. 7
Unlike most differential diagnoses, which are relatively aggressive with high-grade malignancy, PMHE has an indolent clinical course with potential for local recurrence and minimal risk of distant metastasis. 7 Thus the distinction between PMHE and the other tumors previously described is very important and significantly affects the management and prognosis.
Conclusion
In this article, we report the first case, to our knowledge, of PMHE arising in the maxillary sinus. Heterogeneous morphological features and significant inflammatory changes within the tumor make the diagnosis of PMHE a challenge for the pathologist, especially in such unusual location. Immunohistochemical staining should be performed to rule out others differential diagnosis including IMT, spindle cell carcinoma, or sarcomas.
Footnotes
Acknowledgements
Authors thank Dr B. Dani for his contribution in the management of the patient.
Author Contributions
All authors have read and approved final manuscript. HEO wrote the manuscript; OH and AH provided clinical and surgical information; FZ approved the diagnosis; MB and NC supervised the work.
Data Availability
No new data were generated or analyzed to support of this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This report was prepared in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report.
