Abstract
Cogan syndrome (CS) is a rare systemic vasculitis characterized primarily by nonsyphilitic interstitial keratitis and vestibular and auditory dysfunction. In this article, we report the case of a 31-year-old male diagnosed with CS for 1 year. He was admitted to the hospital with fever, dizziness, headache, tinnitus, and hearing loss. After being treated with glucocorticoids, cellular immunosuppressants, and infliximab therapy, his symptoms were greatly relieved except for hearing loss. Then, he attempted to use tocilizumab (TCZ) which was ultimately effective in controlling the auditory dysfunction. In addition, we found 4 cases of TCZ for CS through a literature review and compared them with our patient. Although glucocorticoids are still the first-line treatment for CS, TCZ therapy provides fresh hope for patients who have refractory hearing impairment with hormone resistance, or whose hormone dosages cannot be lowered to maintenance levels.
Introduction
Cogan syndrome (CS) is a rare autoimmune disease of unknown mechanism that occurs mostly in young adults, with nonsyphilitic interstitial keratitis (IK) and vestibular and auditory dysfunction as the main clinical features. 1 Traditional CS therapies include glucocorticoids and immunosuppressive medications, but no standard treatment plan has been established. Some CS patients now struggle with the problem of hormone resistance following the use of conventional therapy or the difficulty of lowering hormones to maintenance dosages. Using biologics in CS therapy, including infliximab, rituximab, and tocilizumab (TCZ), develops novel remedies to these patients. 2 In this article, we report the experience of a CS patient with refractory hearing impairment. He was treated with TCZ and successfully stabilized after successive failures of glucocorticoid, cellular immunosuppressant, and infliximab therapy. We also provide 4 cases found in the literature search that used TCZ for CS and compare them with our case.
Case Presentation
A 31-year-old male with polyarthralgia, headache, bilateral ocular redness, decreased visual acuity, and bilateral hearing loss with tinnitus was diagnosed with CS for nearly a year and admitted with fever (maximum temperature 38.4°C), dizziness, headache, tinnitus, and hearing impairment. Physical examination showed hyperpigmentation of the chest and back, and both upper extremities and ophthalmologic examination showed bilateral uveitis and visual impairment. Laboratory examination revealed: erythrocyte sedimentation rate of 24 mm/hour (0-15 mm/hour), C-reactive protein of 8.64 mg/dl (0-5 mg/dl), interleukin-6 (IL-6) level of 33.5 pg/ml (<7 pg/ml). Immunological tests were weak positive for antinuclear antibodies (940 spots), and samples of rheumatoid factor, antineutrophil cytoplasmic antibody, human immunodeficiency virus, hepatitis B surface antigen and cerebrospinal fluid Gram stain, acid-fast staining, ink stain, bacterial and fungal culture, and cryptococcal capsular antigen were negative. Head-enhanced magnetic resonance imaging, computed tomography scan, and transthoracic ultrasonography were unremarkable.
On admission, he was treated with prednisolone (40 mg/day orally) and methotrexate (20 mg/week orally) but had difficulty lowering the hormone dosages to maintenance levels. His dizziness, headache, and tinnitus improved significantly after multiple intrathecal injections of methotrexate and dexamethasone, but his hearing impairment did not resolve. To further improve his hearing, 1 month after admission, the patient was started on infliximab (100 mg/month intravenously) for 2 doses but discontinued due to pyelonephritis. Then, we try to treat the patient with TCZ (460 mg/month, intravenous). Satisfyingly, the patient’s dizziness, headache, and tinnitus improved along with the relief of hearing impairment. We have not observed any side effects with the use of TCZ.
We also conducted a literature search using the terms “Cogan syndrome” and “tocilizumab” to identify reports of the use of TCZ for CS in the English-language medical literature on PubMed. A review of the literature in this area showed that 4 patients with CS had adopted the TCZ regimen (Tables 1 and 2).
Clinical Information of Cases Searched in PubMed.
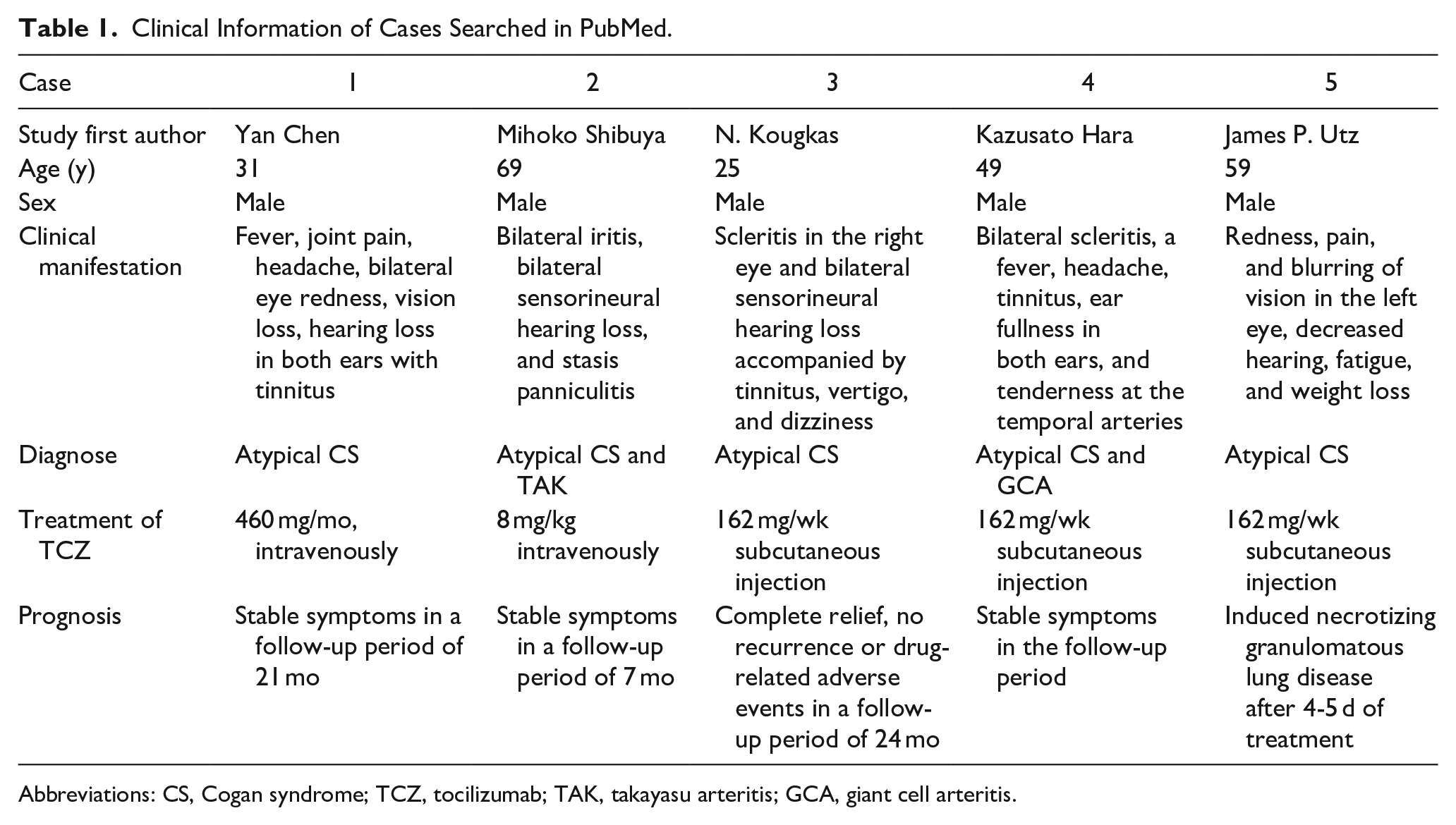
Abbreviations: CS, Cogan syndrome; TCZ, tocilizumab; TAK, takayasu arteritis; GCA, giant cell arteritis.
Laboratory Test Data on Admission.
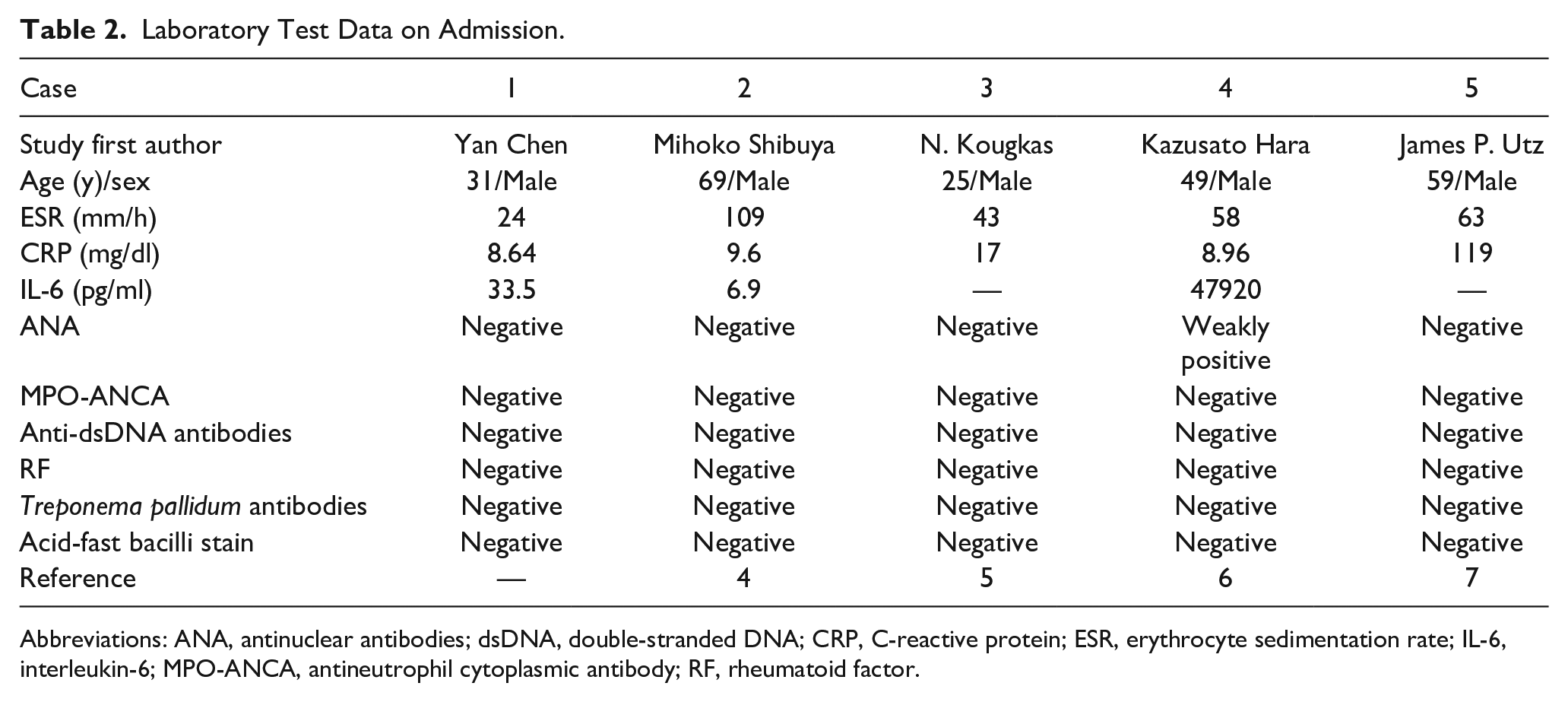
Abbreviations: ANA, antinuclear antibodies; dsDNA, double-stranded DNA; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IL-6, interleukin-6; MPO-ANCA, antineutrophil cytoplasmic antibody; RF, rheumatoid factor.
Discussion
CS was first described by Dr David Cogan in 1945 as “a syndrome of non-syphilitic IK and Meniere’s vestibular auditory symptoms.” 3 In 1980, Haynes et al 4 expanded the definition of CS, dividing it into typical CS and atypical CS. The pathogenesis of CS is unclear. According to some research, it may be mediated by hypersensitive responses to 1 or more infectious pathogens linked to vasculitis. Epidemiology shows that CS often occurs after upper respiratory tract infections. Thus, viral infection is likely to trigger an antibody response that generates specific antibodies that cross-immunize with the presence of autoantigens on endothelial and supporting cells of the sensory nerve epithelium of the inner ear and occasionally with similar proteins in other organs, 5 which associated hearing and vision symptoms. It has been demonstrated that the specific antibody associated with the pathogenesis of CS is an immunoglobulin G antibody against DEP-1/CD148 autoantigen on endothelial and supporting cells of the sensory nerve epithelium of the inner ear, which induces an inflammatory response in the inner ear, leading to hearing loss. 1
Prednisolone is a conventional treatment of CS, and there is no proof that alternative treatment approaches are useful due to resistance to hormone therapy. Immunosuppressants are added if the expected outcome of treatment is not reached. 6 Before admission, our patients were given prednisolone (40 mg/day orally) and methotrexate (20 mg/week orally) but still endured the torment of arthritis, headache, syncope, and other symptoms of systemic vasculitis. Four other cases of patients retrieved from the PubMed search also faced problems with uncontrolled symptoms or failure to reduce hormones to maintenance doses after treatment with glucocorticoids and immunosuppressants.2,7 -9 Therefore, we tried to find a new solution to solve these problems.
CS is a rare form of variant vasculitis that falls under the category of systemic vasculitis according to the classification of vasculitis developed by the Chapel Hill Consensus in 2012. Although patients should initially consider infliximab therapy as a biological therapy for hearing loss, our patient developed severe pyelonephritis after 2 doses of infliximab and had to abandon this regimen. We have learned from the treatment options for other vascular diseases. TCZ has been proven effective for the treatment of systemic vasculitis such as takayasu arteritis (TAK) and giant cell arteritis (GCA). 10 TCZ is a recombinant humanized monoclonal antibody against the IL-6 receptor that inhibits membrane-bound and soluble IL-6 receptors and suppresses T-cell activation and antibody secretion, as well as reducing myeloid circulating cells, 11 thereby effectively suppressing the autoimmune response. The IL-6 level of our patient and 2 patients retrieved from PubMed (cases 2 and 3)7,8 was above the normal range on admission (Table 2), and 2 other patients were admitted with unknown IL-6 levels. Our patient includes 3 patients identified in the literature review (cases 2, 3, 4),7,8,12 who had TCZ therapy and experienced symptom alleviation, especially in whom hearing levels were greatly improved after treatment. This phenomenon suggests that elevated IL-6 in those patients may be related to the pathogenesis of CS. Notably, IL-6 can also be referred to as B-cell stimulating factor 2, suggesting its ability to induce the differentiation of activated B cells into antibody-producing cells, 13 which may be involved in the production of specific antibodies related to CS pathogenesis.
However, our report does have certain limitations, as our patient experienced an improvement in hearing impairment after TCZ treatment but still did not return to normal levels. Among the patients we retrieved, 1 patient had complete resolution of visual and hearing impairment (case 4), 12 2 patients had sustained improvement in visual and hearing impairment (cases 2, 3),7,8 and 1 developed drug toxic lung injury after drug administration (case 5). 9 Further scientific studies are required to ascertain the precise mechanism of TCZ treatment for CS and whether IL-6 plays a role in the onset of CS.
Although randomized controlled trials are lacking, treatment of CS has shown some effectiveness. The data reported so far on the successful treatment of CS with TCZ are encouraging, and treatment duration and monitoring of disease activity are issued for future research. However, randomized controlled trials are needed to confirm the efficacy of TCZ and to avoid publication bias. In addition, patients treated with TCZ should be informed of the possible risk of side effects of the respective drug, and clinicians need to be highly vigilant and diagnose early to prevent irreversible tissue damage.
Conclusion
The treatment of CS is a challenging task. Due to its rarity, no standard treatment strategy has been established, although corticosteroids are commonly used as first-line therapy. The use of TCZ as second-line therapy offers fresh hope for patients with refractory hearing impairment who have glucocorticoid resistance or difficulty in dose reduction.
Footnotes
Data Availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Research project plan of the Sichuan Medical Association (S20001); Second Edition of Key R&D Projects of Sichuan Provincial Department of Science and Technology (2021YFS0165).
Ethical Approval
Ethical approval is not applicable for this article.
Informed Consent
Informed consent was obtained from the patient and her daughter in the study.
