Abstract
Necrobiosis lipoidica is a chronic granulomatous disease historically associated with diabetes. Necrobiosis lipoidica commonly presents with erythematous papules or plaques on the anterior lower extremities, which can be ulcerated in up to 30% of patients. The pathophysiology of necrobiosis lipoidica is unknown but proposed to be predominantly linked to microangiopathy. No treatment option for necrobiosis lipoidica has shown consistent efficacy. Previous case reports have shown immune-modulating agents to be reasonable treatment options for ulcerative necrobiosis lipoidica. However, evidence for the tumour necrosis factor-alpha inhibitor, adalimumab, is limited and contradictory. We report a case of a 74-year-old type 2 diabetic female with a 2-year history of multiple ulcerated necrobiosis lipoidica plaques resistant to topical and systemic therapy. Treatment with adalimumab showed complete re-epithelization of all ulcers by week 28. Adalimumab may be an effective treatment option for ulcerated necrobiosis lipoidica that has failed traditional therapies. Further reports of adalimumab treatment of necrobiosis lipoidica and other chronic inflammatory wounds are needed.
Introduction
Necrobiosis lipoidica (NL) is a chronic granulomatous disease characterized by collagen degeneration within the dermis. It presents with well-circumscribed yellow-brown plaques commonly involving the lower extremities. 1 In approximately 30% of the cases, there is concomitant ulceration and subsequent treatment challenges.2,3 Multiple treatments have been used in the management of NL; however, the management of chronic, ulcerated lesions remains a challenge. Tumour necrosis factor-alpha (TNF-α) inhibitors have shown some promise in treating ulcerative NL in previous case reports; however, the literature in this area remains limited. Adalimumab is an anti-TNF monoclonal antibody approved for management of several cutaneous and systemic autoimmune diseases, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, plaque psoriasis and, more recently, hidradenitis suppurativa. Herein, we present a case of a woman with ulcerated NL refractory to topical and systemic treatments successfully treated with adalimumab. We also review the literature on anti-TNF therapy for NL and comment on the role of TNF in chronic inflammatory wounds.
Case
A 74-year-old Caucasian female presented with a 2-year history of multiple non-healing ulcerated plaques on her right pretibial area (Figure 1). The patient reported that these erythematous plaques on her lower extremities had started approximately 20 years ago as small scattered red papules which had enlarged over the years and subsequently ulcerated 2 years ago. Her medical history included an 8-year duration of poorly controlled type 2 diabetes, psoriasis of the scalp and ears, and remote hysterectomy for endometriosis. Her regular medications were metformin, rosuvastatin and enalapril. Her HbA1c was 10.1%, with rest of her blood work being unremarkable. She had previously been treated with topical and intralesional corticosteroids and multiple courses of oral antibiotics for superimposed infection.

Ulcerative necrobiosis lipoidica on right lower leg prior to initiating treatment with adalimumab.
On physical examination, she had seven ulcerated plaques of varying size on her right anterior and lateral lower leg, with the largest measuring 8.9 × 7.0 cm (Figure 1). There was associated serosanguineous drainage. Biopsy of the plaques showed histological changes consistent with NL, specifically necrobiosis of collagen throughout the superficial and deep dermis with surrounding palisaded histiocytes and multinucleated giant cells.
Initial management involved treatment with pentoxifylline 400 mg three times a day and hydroxychloroquine 200 mg twice a day with no response in 3 months; she eventually was unable to tolerate these oral medications, leading to discontinuation. She was also treated with a course of doxycycline with minimal effect. In addition, she received proper wound care and compression therapy for 2 years. Given the extensive burden of disease and the failure of all treatment options thus far, the patient was consented for adalimumab treatment. She was started on adalimumab with an initial dose of 80 mg subcutaneously, followed by 40 mg weekly thereafter. At week 4, her wounds were much improved, with no drainage and reduced pain. By week 11, only two open wounds remained. By week 28, she had complete re-epithelization of all wounds, with only atrophic scars remaining (Figure 2). She tolerated adalimumab well, with no adverse effects. Adalimumab was discontinued given the complete resolution of her chronic ulcers, and she remained healed over the last year and up to the publication of this report.
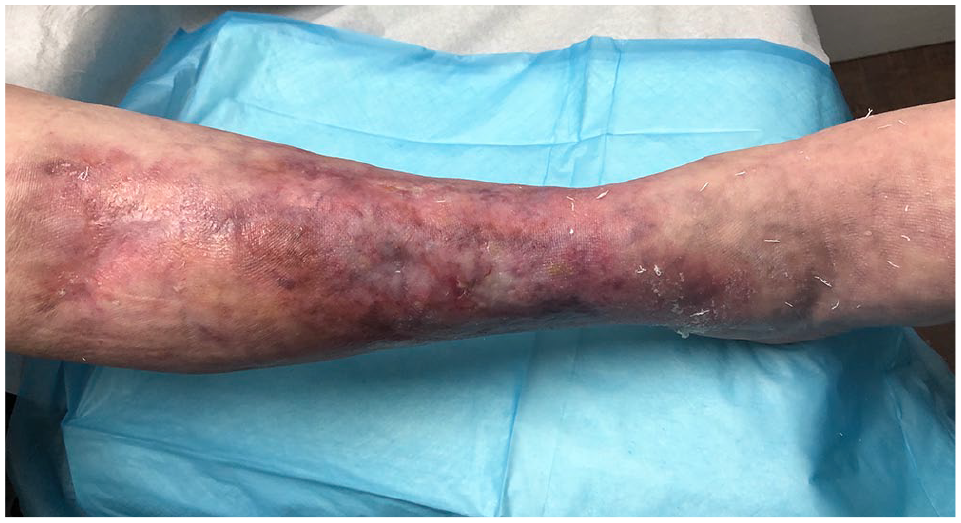
Right leg at week 28 (6 months) of adalimumab treatment, with all ulcers exhibiting complete re-epithelization.
Discussion
Ulcerative NL is notoriously challenging to treat, and response to current treatments is inconsistent. Traditional treatments for NL include topical and intralesional steroids, topical calcineurin inhibitors, anti-platelet drugs, immunosuppressants, phototherapy and hyperbaric oxygen therapy. 1 However, none of these therapies have shown consistent and effective results in the treatment of NL.
The difficulty in treating NL may stem from our limited understanding of the pathogenesis of the disease. Previous theories about the pathogenesis of NL had favoured microangiopathy, given the increased prevalence of the disease in insulin-dependent diabetes. 2 In a recent retrospective study of 236 American patients with NL, 58.5% had diabetes, with patients with diabetes developing NL earlier in life. 4 Despite this association, gaining control of blood sugars has not been consistently shown to effectively treat NL. 5 Doppler studies have also refuted the role of microvascular ischaemia in NL. 6 Other theories to explain the pathogenesis of NL include abnormal glucose transport by fibroblasts 7 and antibody-mediated vasculitis, with direct immunofluorescence microscopy demonstrating deposition of IgM, IgA, C3 and fibrinogen in blood vessels. 8
Histologically, NL is noted to have interstitial and palisaded granulomas within the subcutaneous and dermis tissues. TNF-α is a pro-inflammatory cytokine believed to have an important role in granulomatous inflammatory diseases. 9 TNF recruits inflammatory cells, such as macrophages and T cells, to the site of infection and also promotes increased macrophage phagocytic activity. Once the granuloma is formed, TNF acts as a survival factor promoting macrophage viability and thus maintenance of the granuloma.
In keeping with this, anti-TNF therapies are known to cause granuloma breakdown in infectious granulomatous disorder and subsequent dissemination of mycobacterium infections. 10 Using the anti-granuloma effect, anti-TNF has been used to successfully treat several cases of cutaneous granulomatous diseases, such as sarcoidosis 11 and granuloma annulare.12,13 We searched MEDLINE and Embase databases and identified several case reports supporting the use of TNF-α inhibitors for NL (Table 1). However, the majority of these reports use etanercept16,17,20,23 or infliximab,14,15,18,21,22,25 with the literature on use of adalimumab in NL being limited.
Summary of cases of tumour necrosis factor-alpha (TNF-α) inhibitor therapy for necrobiosis lipoidica (NL).

TB: tuberculosis; PUVA: psoralen and ultraviolet A.
We identified only two published case reports of adalimumab use in NL with contradictory findings. In Zhang et al., 19 a 29-year-old type 2 diabetic female with ulcerated NL of the lower extremities and trunk showed improvement with etanercept over a 2-month treatment period, but no improvement with adalimumab. In Leister et al., 24 a 71-year-old non-diabetic female with ulcerated NL of the lower extremity exhibited complete resolution of her NL with adalimumab, with no recurrence 5 months after completing the treatment. Evidently, in contrast to the many case reports that exist supporting the efficacy of infliximab and etanercept for NL, the literature on adalimumab is limited.
Wound healing is a complex process that progresses through four stages: haemostasis, inflammation, proliferation and remodelling. As reviewed in Weinstein and Kirsner, 26 a prolonged inflammatory phase involving TNF-α has been implicated in chronic wound development. Chronic wounds have higher systemic and local levels of TNF-α. 27 As wound healing progresses, levels of TNF-α decline. 28 Blockade of TNF-α has been noted to promote collagen deposition, 27 and case series have shown improved healing of chronic wounds with topical application of the anti-TNF agent, infliximab. 29 The success of TNF-α inhibitors in treating chronic ulcers has also been seen in other inflammatory ulcerative conditions such as pyoderma gangrenosum. 30 Traditionally, biologic agents were held before surgery due to a potential impact on wound healing or wound infection. However, recent data support that the risk of infection is not higher in patients going to surgery while they are on biologics, including anti-TNF agents. 31 Therefore, the role of TNF inhibitors in preventing and treating chronic wounds should be further explored. This study supports the role of anti-TNF in wound healing, particularly in inflammatory wounds.
In conclusion, NL is a chronic granulomatous disease of unknown aetiology, commonly seen in diabetic patients. Ulcerated NL is particularly challenging to treat and contributes greatly to the burden of disease. This case report demonstrates successful treatment of ulcerative NL with adalimumab. However, further reports of adalimumab use in NL with a larger sample size are needed. The response to TNF-α inhibitors in diabetic and non-diabetic patients should also be explored, given the possibility of different underlying disease processes in these two different patient populations. Finally, further research into the role of anti-TNF therapies in chronic wound healing is also needed.
Footnotes
Declaration of conflicting interests
Dr Alavi has consulted for AbbVie, Janssen, LEO, Galderma, Novartis and Valeant, and is also an investigator for AbbVie, Novartis, Regeneron, Pfizer, Boehringer-Ingelheim, Glenmark, Merck Serono, Roche, Xoma and Xenon. She received an unrestricted educational grant from AbbVie. Ms Sandhu has no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from the patient for the publication of this case report and the images contained within this report.
