Abstract
Introduction
Submandibular gland excision is a diagnostic and therapeutic procedure for a variety of salivary gland diseases. Gland excision is performed as a treatment for salivary gland stones, which cannot be recovered through sialendoscopy or transoral surgery because of their size or location. Furthermore, inflammatory causes can be an indication of gland excision. In the case of chronic submandibular sialadenitis, gland excision can be viewed as a therapeutic procedure. Compared to this, gland excision in cases of tuberculosis for example is a possible way to acquire a representative sample for further microbiological testing and diagnosis.
Most commonly, gland excision is performed in cases of solid salivary gland masses of unknown entities to obtain samples for histological classification.1-3 Apart from clinical examination, preoperative imaging is a key component for the initial assessment of masses as benign or malignant. High-resolution sonography is especially well-suited to depict submandibular gland masses. A typical ultrasonographic characteristic of malignant salivary gland masses is poorly defined borders. In the adjacent lymph nodes, a round shape and loss of the hilar structure can be a possible sign of malignancy.4-7 Salivary gland malignancies generally constitute 3% to 6% of all head and neck malignancies. In addition, the rate of distant metastasis is higher than in head and neck squamous cell carcinoma patients. 8 The most common benign tumors are pleomorphic adenomas with a rate of 60% to 70% of all masses (10% being located in the submandibular gland), followed by Warthin tumor and myoepitheliomas.7,9 Salivary gland masses are most commonly found in the parotid gland. But compared to a rate of 11% in the parotid gland, submandibular gland masses have a higher rate of malignancy with around 34% to 54%.10,11 The most common primary salivary gland malignancies are mucoepidermoid carcinomas followed by adenocarcinomas and adenoid cystic carcinomas. 8 Because of the high malignancy rate of submandibular gland masses, a timely sample acquisition is recommended. Fine-needle aspiration cytology and core needle biopsy are especially suited to acquire preoperative samples of the tumor. This allows the physician to adequately plan further diagnostic and surgical steps such as performing staging imaging or performing an additional neck dissection during the initial surgery. With the sensitivity and specificity of fine-needle aspiration cytology and core needle biopsy still being the subject of ongoing discussions in the literature, submandibular gland excision is an alternative to preserve the salivary gland mass for pathologic examination.10,12-14 While we have extensive knowledge about indications for parotid gland excision and the distribution of the histological findings of the surgical specimen, there are only a few systematical overviews for the surgical indications for submandibulectomies. The main goal of this study was to get an overview of all these indications with a special focus on solid submandibular gland masses and especially malignancies.
Methods
We analyzed all submandibular gland excisions performed at our hospital, a center for salivary gland disorders, during the last 20 years (January 2002-December 2021) with primary diseases and pathologies of the submandibular gland as the main cause. Cases in which gland excision was performed because of a neck dissection of Level Ib or metastatic disease of another primary malignancy not located in the submandibular gland, such as squamous cell carcinomas, were excluded. The underlying causes for gland excision were divided into 4 groups: Sialolithiasis, inflammatory causes, benign, and malignant masses. In the case of sialolithiasis, we investigated whether conservative procedures were performed beforehand. Inflammatory causes were divided into main 4 diseases. Benign and malignant lesions were furthermore divided into their different histological subtypes. Epidemiological information such as age and gender were investigated in addition to the T(Tumor)N(Lymph nodes)M(Metastasis) (TNM) classification, neck dissections performed, adjuvant radiotherapy, and recurrence rate.
The study protocol was in accordance with the Declaration of Helsinki. The Institutional Review Board of the Medical Faculty, Technical University of Munich, reviewed and approved the protocol (2023-108-S-SR).
Statistical analysis was done using version 28.0 of the Statistical Package for Social Sciences software (SPSS, Chicago, IL, USA). Descriptive data are reported as mean ± standard deviation, if not otherwise stated. The normal distribution of variables was tested using the Shapiro-Wilk-Test. P-values of <.05 were considered as statistically significant. Overall survival of patients with and without lymph node metastasis was compared using a Kaplan–Meier analysis.
Results
During the observation period, 359 submandibular gland excisions were performed (Figure 1), with 47% of the patients being men and 53% women. The mean patient age was 55 years (±16.8 years). The most common cause for gland excision was sialolithiasis with 36%. Intraparenchymal stone localization was the leading cause of gland excision in patients with sialolithiasis at 49.6% followed by unsuccessful gland-preserving procedures (15.5%). Recurrent sialolithiasis accounts for only 13.9% of these cases. Inflammatory causes (32% of all cases) could be divided as seen in Figure 2. Chronic submandibular sialadenitits was by far the most common inflammatory cause. Benign tumors account for 24.5% of gland excisions. The most common benign lesion by far were pleomorphic adenomas (78.4% of all benign tumors; Figure 3). Malignant tumors made up 7.5% of all cases. 33.3% of these malignant tumors were lymphomas. 66.6% were primary salivary gland malignancies. The most common primary submandibular gland malignancies were adenoid cystic carcinomas (44.4%), followed by adenocarcinomas (38.8%; Figure 4).
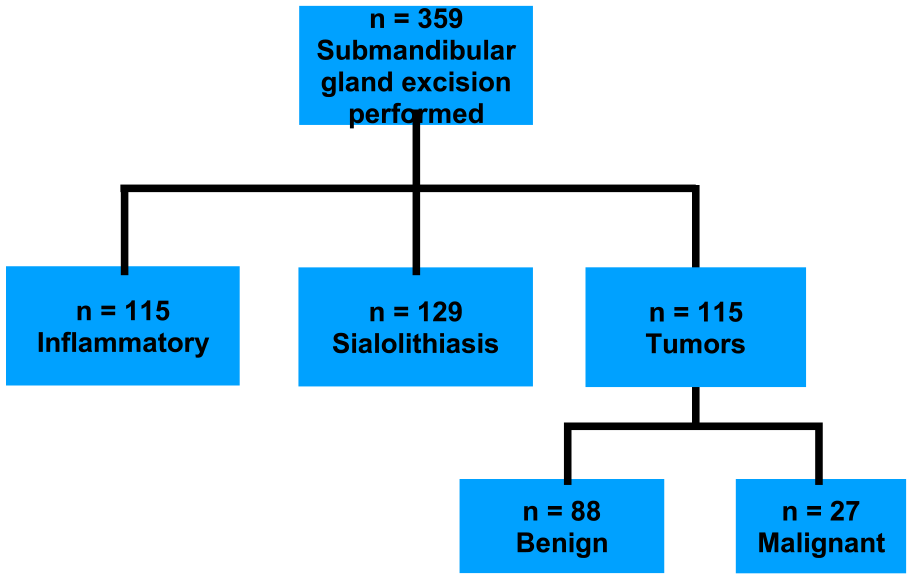
Overview of the underlying causes for gland excision.
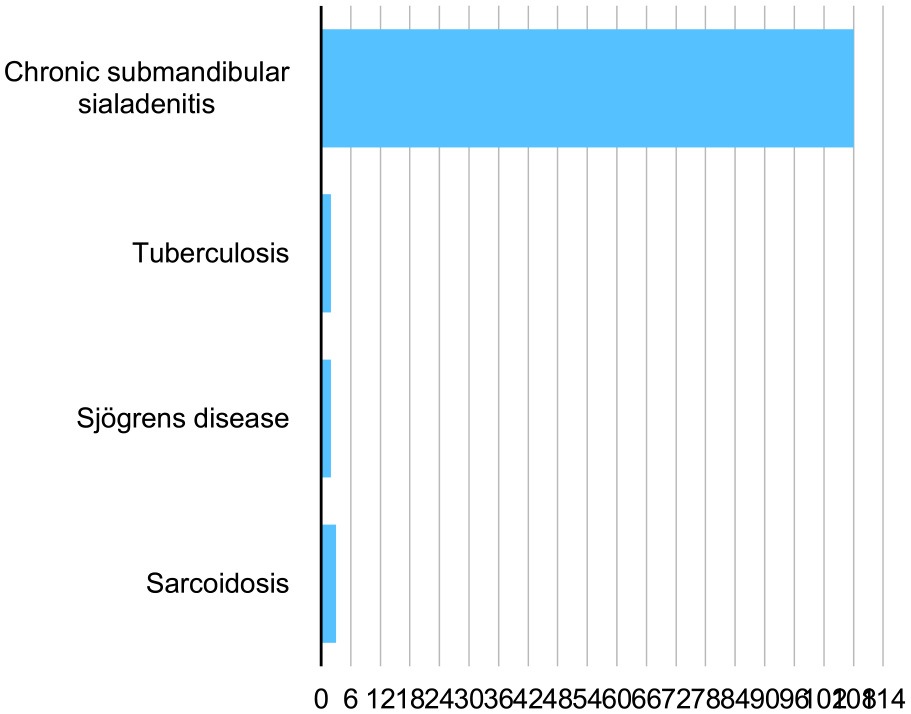
Inflammatory causes for gland excision.
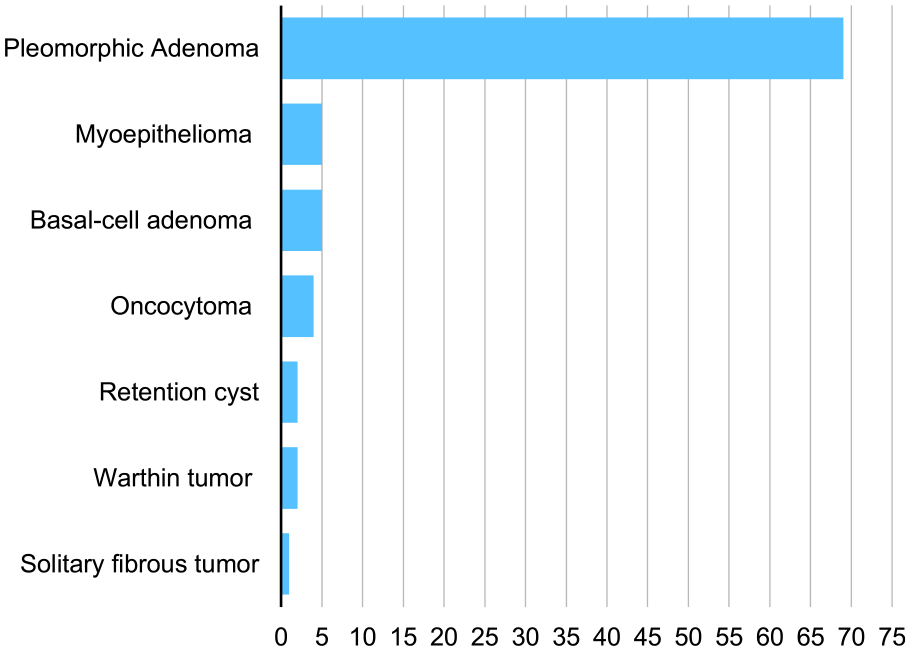
Benigne tumor case numbers.
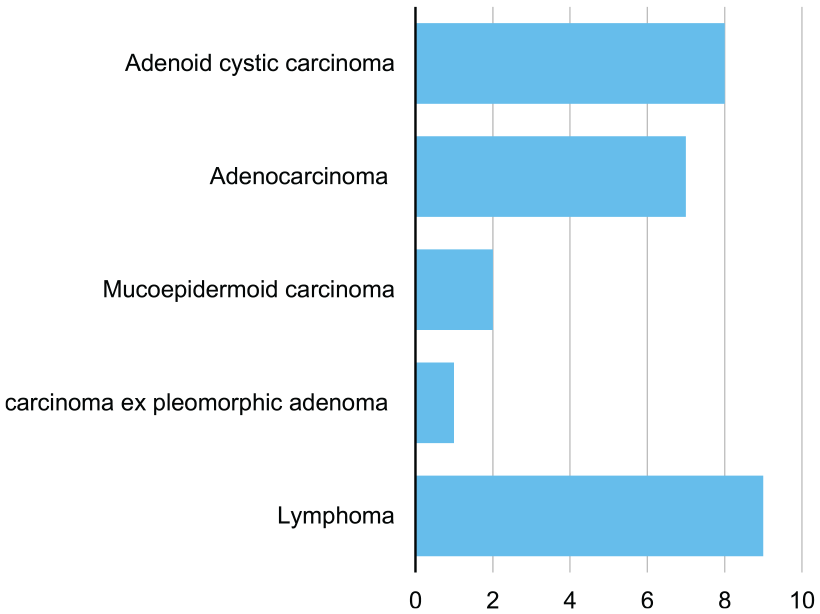
Malignant tumor case numbers.
Neck dissections were performed in 15 of 18 cases of primary salivary gland malignancies. In 2 cases, additional neck dissection was not performed because of distant metastasis being discovered by postoperative staging imaging. In another case, neck dissection was not performed because the tumor was a T1 low-grade adenocarcinoma. This is also the only of the 18 salivary gland malignancy cases where adjuvant radiotherapy was not administered. Four patients with primary salivary gland malignancies had lymph node metastasis. During the first 2 years, 5 patients showed recurrences (Table 1). Only one of these patients showed lymph node metastasis at first diagnosis. Patients without lymph node metastasis showed a slightly better overall survival after 5 years compared to patients with lymph node metastasis (Figure 5) with a nearly significant P-value of .052. Gland excision due to sialolithiasis showed a normal distribution over our observational period (Shapiro–Wilk test, P = .95155).
Summarized Epidemiologic Data of the Entire Cohort 1.
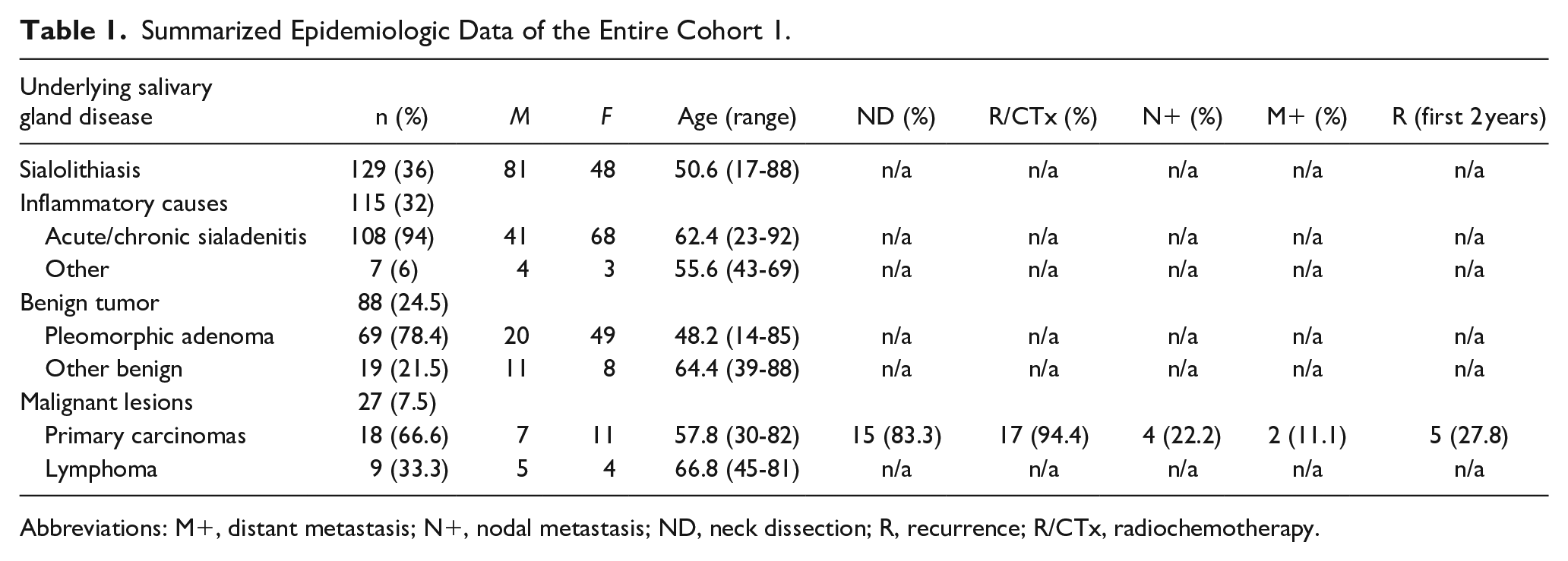
Abbreviations: M+, distant metastasis; N+, nodal metastasis; ND, neck dissection; R, recurrence; R/CTx, radiochemotherapy.
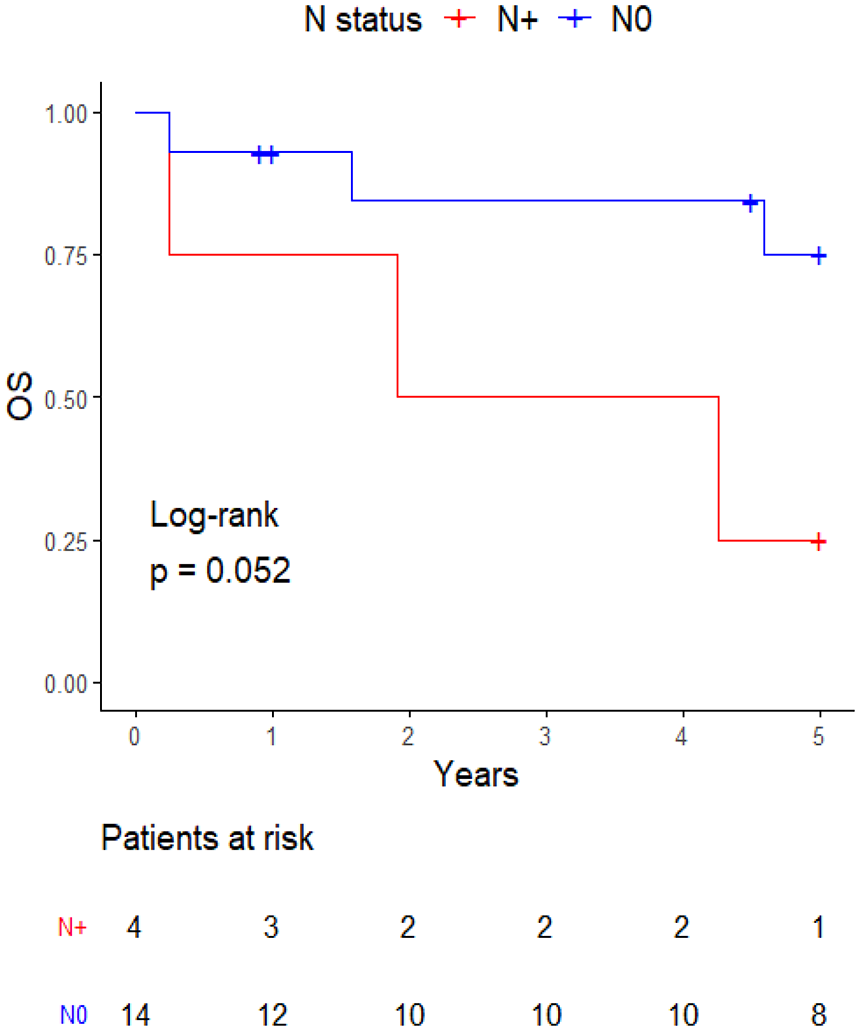
Comparison of the overall survival of N0 and N+ lymph node status.
Discussion
In our study, the most common indication for submandibular gland excision was sialolithiasis with intraparenchymal stone localization accounting for 49.6% of these cases. With the advancement of sialendoscopy and combined endoscopic and surgical approaches, gland excision is gradually losing importance. Only 2% to 5% of patients with submandibular gland sialolithiasis require gland excision. While gland preservation should be prioritized because of surgical and postoperative complications, depending on the location, it may be the only viable treatment option. In 15.5% of cases, endoscopy and/or transoral surgery were unsuccessful leading to gland excision. The intraparenchymal location of stones makes it hard and nearly impossible to reach the concrement via endoscopy. In addition, the localization of stones through palpation becomes difficult limiting the possibilities of transoral surgical approaches.2,15-17 It needs to be mentioned that gland-preserving techniques were prioritized in every case of sialolithiasis. Even in some cases of intraparenchymal stone localization, transoral or combined approaches were initially performed but then expanded to gland excision during the same surgery when unsuccessful.
With the introduction of sialendoscopy, we would expect the numbers for gland excisions to decrease during the observational period because of continuous improvement of the endoscopes used and endoscopy becoming more available in general. But looking at the percentages throughout the years comparing gland excisions performed because of sialolithiasis to total gland excisions performed during 1 year, a normal distribution can be proven using the Shapiro–Wilk test (P = .95155). A possible explanation would be that sialendoscopy was first introduced in the 1990s. By the year 2002, this technique was already in use for more than 10 years having given the physicians enough time to adapt and learn the new procedures. 18 In addition, intraparenchymal localization seems to be an obstacle, which at this point of time cannot be overcome even with the constant advancement of the endoscopes and combined approaches. Looking at extracorporeal lithotripsy for submandibular stones, this method could not prevail as a valid alternative to other minimally invasive approaches such as intraoral surgery or endoscopic stone evacuation. Extra-corporeal lithotripsy has a lower success rate for submandibular sialolithiasis compared to parotid sialolithiasis. In addition, it regularly has to be performed a number of times before showing success. Compared to this, intraoral surgery shows much higher success rates for submandibular gland stones. Moreover, intraoral surgery can also be performed with local anesthesia while often needing only 1 session for it to show its effects. 19
Looking at submandibular gland masses, pleomorphic adenomas were by far the most common entities. In our study, they accounted for 60% of all tumors. These numbers are higher compared to the 36% described in the literature. 10 With a ratio of 1:2.5, women are affected more often in our study similar to the epidemiology mentioned in the literature. While Warthin tumors are the second most common entity for salivary gland masses in general (15%-35% of all masses), over 90% of them are found in the parotid gland. Extraparotid manifestations such as minor salivary glands are rare accounting for <10% of all cases. They are described in locations such as the lower lip or the hard palate for example. Even more rarely is a location in the submandibular gland having been described only in case reports and case series so far. In our cohort, 2 patients showed Warthin tumors of the submandibular gland. Analogous to the literature, these patients were both male and between 40 and 70 years of age. Even though manifestation of Warthin tumors inside the submandibular gland is possible and must be considered, it is still a rarity.10,20-22
In our cohort, adenoid cystic carcinoma and adenocarcinoma were among the most common primary salivary gland malignancies similar to the published literature.1,23-25 With 23.5% of all submandibular gland masses being malignant, our ratio was lower than described in the literature. 11 A possible explanation would be the rise of ultrasound-guided core needle biopsy. It can easily be used to acquire a sample preoperatively allowing the physician to plan further diagnostics and treatment. This way malignancies can be diagnosed before gland excision. If the following staging imaging shows distant metastasis, definitive radiochemotherapy is administered without gland excision ever being performed. In our study, we only looked at cases in which submandibulectomy was performed. Furthermore, patients unsuitable for surgery due to their comorbidities, functionally unresectable disease, and patients denying an operative concept in general, were not included, as we searched for our cohort using the German procedure classification code for submandibulectomy and not the ICD-10 codes of the different salivary gland carcinomas.14,15,26,27 Nearly all of our tumors were still operable at the time of diagnosis explaining the high ratio of neck dissections performed. The only exceptions for neck dissections performed were 1 patient with a low-grade T1 N0 M0 adenocarcinoma and 2 patients with an M+ status discovered after completion of the staging imaging following the initial submandibular gland excision. In addition, adjuvant radiotherapy was administered in all cases except the T1 N0 M0 low-grade adenocarcinoma, where only follow-up examinations were performed. 14 27.8% (n = 5) of patients showed recurrences within the first 2 years. In only 1 of these 5 cases, a surgical R1 resection was performed. This aspect underlines the aggressiveness of salivary gland malignancies irrespective of the R status and showcases the unpredictability of malignant tumor disease but also the importance of timely therapy. In addition, 4 of the patients had N0 status. Patients without lymph node metastasis showed a better 5-year survival with a nearly proven significance (P = .052; Figure 5). While it would be logical to assume a worse prognosis for malignancies showing lymph node metastasis compared to N0 tumors, in the end, there was no clear significant correlation. This aspect correlates with the European guideline for major salivary gland malignancies, as neck dissections are recommended for all high-grade carcinomas of the submandibular gland regardless of the N status once again showcasing the aggressiveness of these malignancies. 14
Although our main approach toward salivary gland malignancies is modeled toward the ESMO (European Society for Medical Oncology) guideline for salivary gland carcinomas, there are some differences to be mentioned. Fine-needle aspiration is not our preferred form of biopsy for salivary gland masses if malignancy is suspected. In our experience, core needle biopsy leads to less non-diagnostic results avoiding a delay of the upcoming therapy. We take up to 3 samples generating an adequate amount for the pathologist to work with. Considering the higher diagnostic yield and only slightly higher risk for complication, we deem core needle biopsy superior to fine-needle aspiration.14,15,26,27 In cases of skin ulceration, incisional biopsy is the preferred approach. If clinical findings highly suggest malignancy (eg, paralysis of the facial nerve), staging imaging is launched even before the pathologic report of the core needle biopsy is finished to avoid a therapeutic delay. In some cases, the recommended additional FDG-PET-CT for N+ carcinomas is delayed until after surgery due to the sometimes limited availability, allowing a timely treatment in particular for large T3/T4 carcinomas already infiltrating neural structures. This approach is only viable if distant metastasis is ruled out beforehand by conventional CT scans. As for the primary salivary gland carcinoma, PET CT has been shown to not improve discrimination between benign and malignant lesions compared to regular CT scans.14,28,29
Conclusion
Submandibulectomy is a viable treatment option for sialolithiasis if gland-preserving approaches have already failed. Although most of the benign masses are pleomorphic adenomas, Warthin tumor, though rather rarely, can manifest in the submandibular gland. Core needle biopsy is a valid option to acquire preoperative samples of salivary gland masses. In our study, we investigated one of the largest cohorts of patients, who underwent submandibular gland excision.
Footnotes
Acknowledgements
None.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Technical University Munich paid the publication costs.
Ethical Statement
The Institutional Review Board of the Medical Faculty, Technical University of Munich, reviewed and approved the protocol (2023-108-S-SR).
Patient Consent
Since all patient information was anonymized and the research involved no risk to the patients, patient consent was not gathered.
