Abstract
Solitary myofibroma or infantile myofibroma is a rare spindle cell neoplasm that generally affects infants before the age of 2 years but cases in young children and adults have been described. Although the location of infantile myofibroma in the oral and maxillofacial region has been described, the intramasseteric location of the lesion is very uncommon. A thorough assessment of histopathological and immunohistochemical characteristics is necessary to have a correct diagnosis. Treatment relies on surgical resection. In this article, we present a rare clinical case of a 15-year-old patient with a myofibroma of the masseteric muscle and its management.
Introduction
Solitary myofibroma (SM) is a rare soft tissue spindle cell neoplasm that commonly presents in young children under the age of 2 years. 1 First described by Stout in 1954 as a congenital generalized fibromatosis and later termed as “infantile myofibromatosis,”2,3 these tumors are not only found in the pediatric population, besides rare SMs have been reported in adults.4-6 The most common location of the tumor is the head and neck region, and intramasseteric myofibroma is particularly rare. 7 Two forms are described: solitary and multicentric in which case it is named myofibromatosis. 8 Solitary type is more common and is localized mainly in the head and neck region. The clinical presentation can either be an aggressive exophytic cortical or periosteal and rarely a non-aggressive intraosseous type.9,10 This case report describes an uncommon presentation of a large SM in the masseter muscle in a 15-year-old patient and its management. It has allowed us to analyze this rarely encountered tumor. Diagnosis of myofibroma of the masseter muscle can be challenging because there is a broad differential for masses in the cheek region that can involve salivary glands, neural, lymphatic, bony, and vascular structures,11,12 Furthermore, it has morphological similarity with other spindle cell tumors of childhood. Careful assessment of histopathological features along with supportive immunohistochemistry is important for making the correct diagnosis.13,14
Case Report
A 15-year-old boy was referred to the emergency unit of the ENT and Head and Neck Surgery department, in Rabat, Morocco, for evaluation of a painful swelling of the right submandibular region. It started 4 months ago with a small swelling of the cheek that quickly grew in size and became painful with moderate trismus in the last 4 weeks according to his parents. The medical history was unremarkable.
Clinical examination showed a tense, firm, and fixed 6 cm mass in the right mandibular area overlaying the mandibular angle and inferior border of the mandible, and extended to the right submandibular region, painful during palpation. The overlying skin did not show a sign of local inflammation (Figure 1). There was no right facial nerve weakness and no palpable lymphadenopathy.
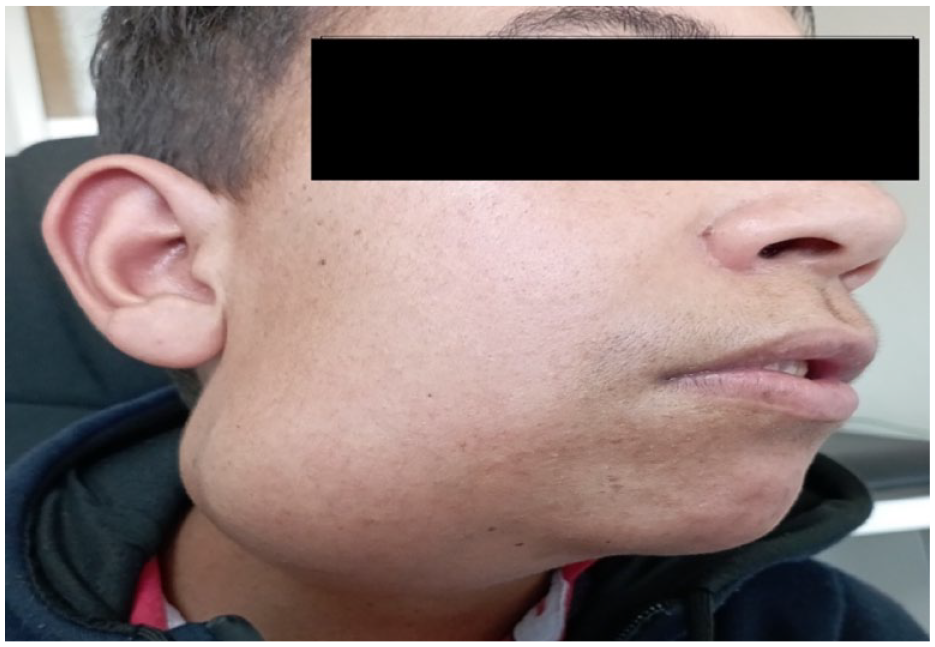
The clinical photograph shows diffuse swelling on the right lower third of the face and submandibular area, measuring about 6 cm × 5 cm.
A facial computed tomography scan revealed a well-limited soft tissue mass, isodense to muscle with central necrosis which infiltrates the right submandibular salivary gland, coming in contact with the mandibular angle without any cortex erosion, the tumor is in contact with the anterior belly of the digastric muscle and creates a mass effect on the sternocleidomastoid muscle, the common carotid artery, and internal jugular vein and measures approximately 6 × 7 cm. There was no lymphadenopathy (Figure 2).
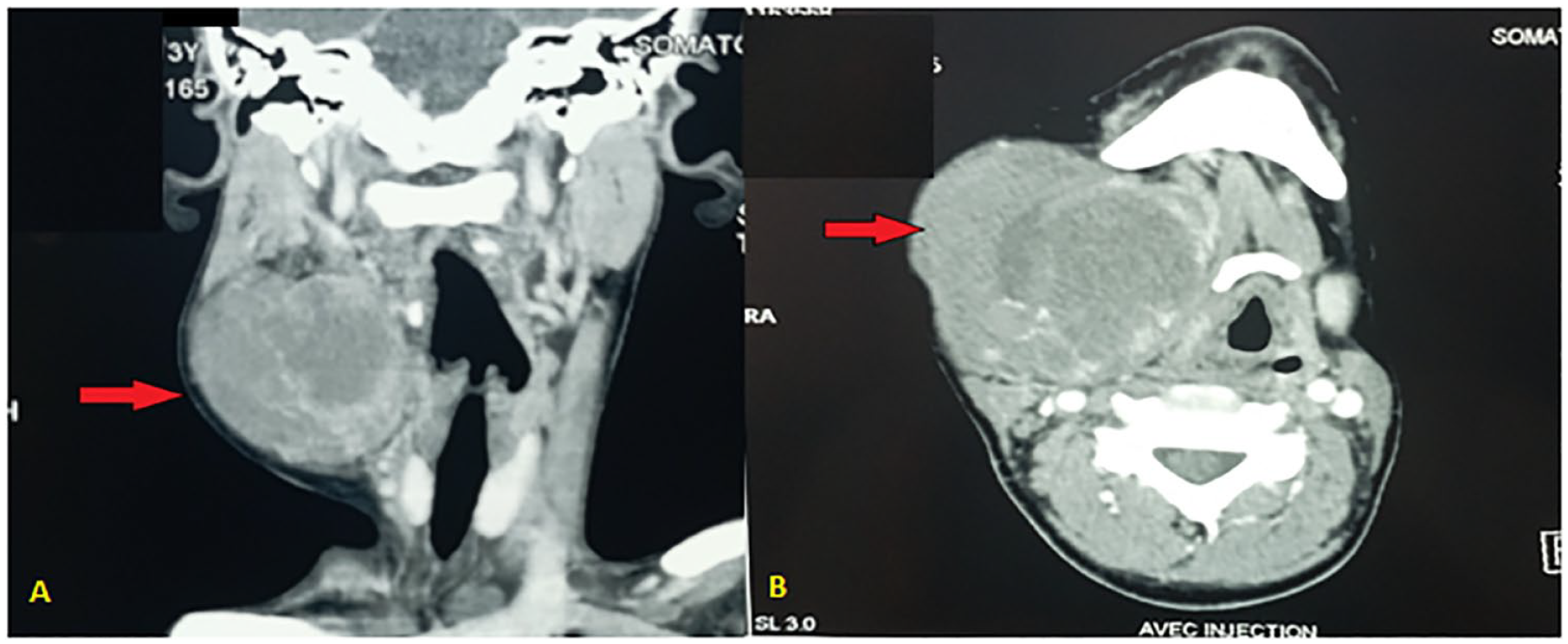
Axial (A) and coronal (B) CT scanner soft tissue window revealing a large mass in the right masseter muscle.
Given the history of rapid growth, pain, trismus, and radiographic features, an infectious and inflammatory origin was first suspected and a fine-needle aspiration along with standard infection blood work was done but was non-contributory. The authors prompted surgical resection under general anesthesia with intraoperative histopathological examination. Given the location and the size of the mass, the submandibulectomy approach was considered adequate in accordance with oncology safety and risk to the facial nerve. Thus, a direct incision over the mass below the border of the mandible was made and an encapsulated mass was quickly identified. Samples for intraoperative histopathological examination were taken which came back in favor of a spindle cell tumor; thus, total resection including the ipsilateral submandibular gland was achieved with respect to the facial vein, hypoglossal nerve, and ligature of the Wharton duct. A drainage tube was put in place in the resection cavity before closure ( Figure 3).
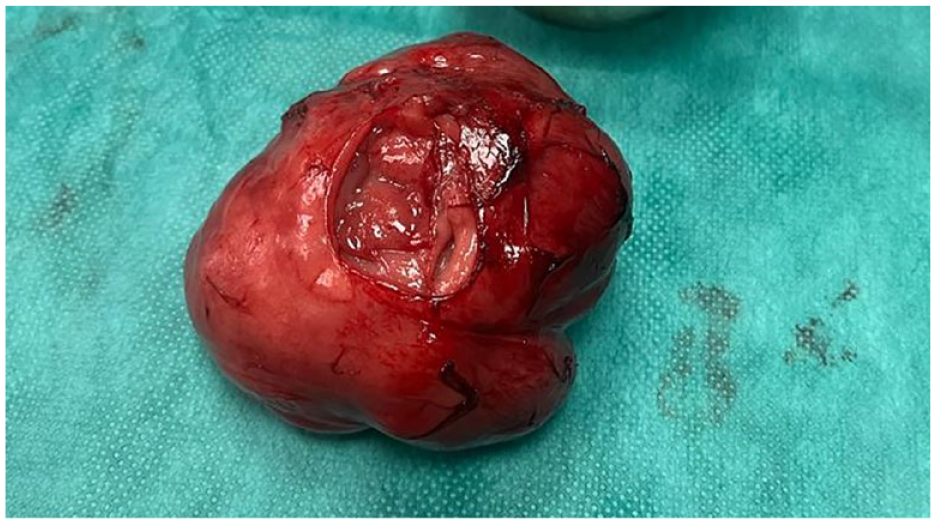
Gross image of intramasseteric tumor following removal (with removed extemporaneous examination.
The histological section showed a biphasic tumor with cells like spindle and round cells. Spindle cells are arranged in a diffuse sheet in streaming and fascicular pattern with long blunt-ended nuclei. There were small cells which are round with long nuclei and weakly eosinophilic cytoplasm. There were also slit-like spaces and dilated blood vessels with a hemangiopericytoma-like appearance. Few areas of hyalinized stroma were present. Immunohistochemical (IHC) staining showed positivity for alpha smooth muscle actin (α-SMA) and vimentin and negativity for S-100, CD34, and desmin. These findings supported the myofibroblastic nature of the cells (Figure 4).
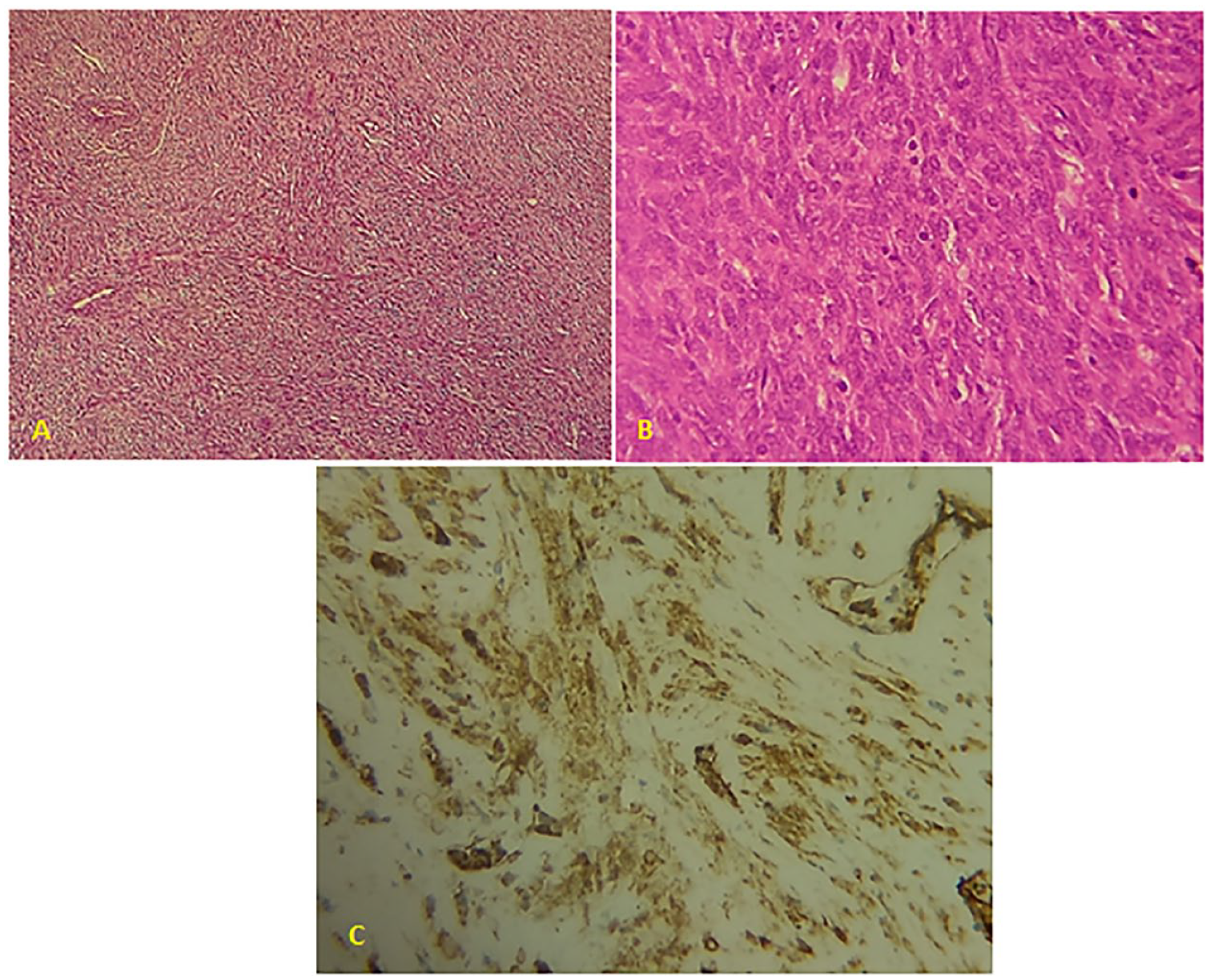
(A). The microphotography shows a spindle cell tumor with fascicular and hemangiopericytoma-like pattern (hematoxylin-eosin ×10). (B) At high magnification, tumor cells show long nuclei and cytoplasm (hematoxylin-eosin ×40). (C) Immunohistochemical staining shows the tumor cells expressing the alpha smooth muscle actin.
Following surgery, our patient recovered well and had improved trismus and swelling, and has been in follow-up for 6 months with no signs of recurrence.
Discussion
Myofibroma (unifocal) and myofibromatosis (multifocal) are rare spindle cell neoplasms composed of myofibroblasts15,16 These lesions show a predilection for the head and neck, with the oral lesions typically presenting in the mandible. 17 In our case, the lesion was located in the masseter muscle making it a unique presentation of SM. Generally, these lesions are slow growing in nature, particularly when confined to the mandible. 18 Symptoms of SM in the head and neck are nonspecific. 17 The patients usually present with a slow-growing, painless mass but they may also present with symptoms related to a mass effect, 19 occasionally accompanied by an intraoral soft tissue mass lined by normal-appearing mucosa and rarely trismus can be associated. 20 Thus, the rapid growth of the mass in our patient and clinical features led to insufficient diagnosis prior to the excision. Radiologic features of myofibroma are typically an expansive soft tissue mass or a radiolucent lesion in the submandibular area associated with or without erosion of the mandible, frequently unilocular with well-defined borders, as was revealed in our case.4,16 With that said, it can be heterogeneous, poorly circumscribed, and show high vascularity. These features can lead to misdiagnosis as hemangioma or soft tissue sarcoma of childhood. 21 Magnetic resonance imaging can be useful in diagnosing the mass before surgery, clarifying relationships between the mass and adjacent structures, and determining the surgical approach to the mass. 12
The exact etiology of the condition is unknown in most cases reported as sporadic; although very low familial incidence has been reported, some reported cases suggest the possibility of a familial pattern of inheritance suggesting that myofibromatosis may be inherited as an autosomal dominant or recessive trait.18,22,23
The differential diagnoses for cheek masses include solitary vascular malformations (venous malformation of accessory parotid glands), venous malformation of the masseter muscle, (vascular leiomyoma), benign or malignant lymphadenopathy, masseter muscle hypertrophy, lipomas, neurofibromas, schwannomas, fibromas, malignant tumors arising from the muscles, buccal fat pad, or other structures, sialoceles, sialolithiasis, and other benign and malignant tumors arising from accessory parotid gland (APG). 12 Inflammatory or infectious etiologies were on the differential for this patient, albeit less likely. Klotz et al found in a retrospective study that the most common pediatric cheek masses were lymphatic malformations, followed by venous and arterial malformations. 24 Although the intramuscular location of the hemangioma is overall uncommon, when located in the head and neck region, the masseter muscle is the most common site as described in various studies and should be considered as a differential diagnosis.25,26
Histopathologically, the distinct features are a biphasic pattern of arrangement of spindle and round cells and the zoning phenomenon. However, since other tumors, such as leiomyoma, neurofibroma, nodular fasciitis, and solitary fibrous tumor, mimic the histopathologic findings of myofibroma, they had to be considered in the differential diagnosis. IHC becomes an invaluable tool in cases like this, to arrive at a final conclusive diagnosis.
Analysis of a combination of markers (α-SMA, vimentin, desmin, S100, and CD34+) for positive and negative findings will aid in differentiating between these neoplasms. Very strong positivity to α SMA in most areas and to vimentin in a few areas is noted in myofibromas. Leiomyoma shows positive staining for desmin, neurofibroma to S100, and solitary fibrous tumor to CD34+. Faint positivity to α SMA is also noted in nodular fasciitis. 27 Based on the IHC findings in the present case, leiomyoma, neurofibroma, nodular fasciitis, and solitary fibrous tumor were ruled out and a definitive diagnosis of myofibroma was made. It is evident that IHC methods also have a limitation due to overlapping expression and no single marker alone can reliably be used to validate the presumptive diagnosis.28-30 IHC features of IM are not specific. The tumor cells are vimentin and SMA positive and negative for CD34, S100, desmin, Bcl 2, and CD99.31,32 In the recent WHO classification of soft-tissue tumors, 2013 and 2020, IM has been grouped under tumors of pericytic origin.33,34
Treatment is based on surgical excision, surgical approach varies from direct incision over the mass, standard parotidectomy approach, and transoral approaches. A parotidectomy approach which is the most common for mid-cheek masses is effective oncologically and safe for facial nerve preservation. 11 Transoral approaches are prone to have less cosmetic morbidity and also avoid the risk of Frey syndrome, and it is successful with the potential for less disruption to facial nerve branches. 35 In the present case, given the volume, the location of the mass in the submandibular region, and the submandibular gland being taken in the mass a submandibulectomy approach was judged appropriate. Systemic therapy is recommended for multicentric visceral disease due to the significant mortality associated (73%-93%) with cardiopulmonary or gastrointestinal involvement.36,37 Six months of follow-up in our patient has been uneventful.
Conclusion
In conclusion, we report a unique case of intramasseteric myofibroma of a 15-year-old patient. Although rare, an intramasseteric myofibroma should be considered as a differential diagnosis when faced with a slow-growing mass in the submandibular region. CT scan and Magnetic resonnance imagin (MRI) are useful for diagnosis, but only IHC features can determine the exact diagnosis. Surgical excision with direct incision, parotidectomy approach, transoral approach, or as in our case submandibulectomy approach is the adequate treatment.
Footnotes
Conception and Design
Zakaria El Hafi, Eden Ayele Habte, Khalil Hjaouj.
Administrative Support
Razika Bencheikh, Mohamed Anas Benbouzid, Abdelilah Oujilal, Nadia Cherradi, Hafssa EL Ouazzani, and Leila Essakalli.
Provision of Study Materials or Patient
Collection and assembly of data: all authors.
Data analysis and interpretation: all authors.
Manuscript writing: all authors.
Final approval of manuscript: all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval Obtained
This study was approved by Mohammed V University in Rabat; Morocco
Informed Consent
Written information was obtained from the patient for publication of this case report and accompanying images.
A copy of the written consent is available for review by the editor-in-chief of this journal on request.
All data are available in the patient’s medical file Provenance and peer review. Not commissioned, externally peer-reviewed.
All authors approved the final version of the manuscript.
