Abstract
Among the reconstructive options available for buccal mucosa defects with an intact mandible, free flap with microvascular anastomosis is the best option. However, in the developing world, with poor resources, limited in- frastructure, and high patient load, this cannot be offered to all patients. We report on the success of the masseter flap for reconstruction of such defects in carefully selected patients. Despite some known limitations, this flap is easy to learn and carries acceptable complications. The results of this flap may not be comparable to those of microvas- cular reconstructions, but they are better than those from other options such as skin graft, nasolabial flap, submental flap, etc. in terms of surgical time required, no donor site morbidity, and minimal aesthetic deformity.
Introduction
Adequate reconstruction of the defects after the exci- sion of carcinoma of the buccal mucosa is important for patients’ early recovery. Options available for the reconstruction of buccal mucosa defects with an intact mandible are split-thickness skin graft, nasolabial flap, tongue flap, submental flap, superficial temporal artery flap, pectoralis major myocutaneous flap, forehead flap, free radial forearm flap, anterolateral thigh flap, etc. 1 Free flap with microvascular anastomosis has revolutionized the reconstruction of such defects, with clear superiority over other forms of reconstruction. However, because of constraints of infrastructure and expertise, not all patients can be offered microvascular reconstruction, especially in resource-poor countries. Moreover, in less experienced hands, microvascular surgery has a high risk of flap necrosis. 2
We suggest the masseter flap as an alternative to the free radial artery forearm flap for such defects in selected patients. Despite some known limitations, this flap is easy to learn and carries acceptable complications.
Patients and Methods
Between 2004 and 2009, 17 patients with carcinoma of the buccal mucosa underwent masseter flap reconstruc- tion at our center. Of these 17 patients, 9 had the disease epicenter in the posterior part of the buccal mucosa/ retromolar trigone, 5 in the lower gingivobuccal sulcus, and 3 in the central part of the buccal mucosa. None of these patients had received neoadjuvant chemotherapy or radiotherapy. Most of them were evaluated for free flap but deemed unsuitable because of either significant comorbid conditions (12) or a donor-site–related prob- lem (3); in 2 patients, this approach was planned due to a long waiting period for a free radial forearm flap.
Of the 17 patients, 12 had T1-2 lesions, 3 had T3 lesions, and 2 patients had T4 lesions per the current American Joint Committee on Cancer classification. Neck dissection was performed according to the stan- dard guidelines (i.e., supraomohyoid neck dissection for necks without nodal metastasis, and modified neck dissection types I to III for node-positive necks, using a modified Crile incision.
All patients had the cheek flaps raised through the horizontal limb of the Crile incision to the lower border of the zygomatic arch, taking care to preserve branches of the facial nerve, including the marginal branch. For adequate exposure, 7 patients required a midline lip- splitting incision and 10 did not. Twelve patients had only a wide excision of the mucosal lesion, 5 required marginal mandibulectomy along with the wide excision of the mucosa, and 3 required additional removal of an island of overlying cheek skin for adequacy of margins. Masseter muscle was preserved only when it was considered oncologically safe. Once we were satisfied with the surgical clearance and integrity of the remnant masseter muscle, we opted to undertake reconstruction with the masseter muscle.
Technique
After removal of the tumor, the cheek flap is raised to the lower border of the zygomatic arch. The lateral surface of the masseter muscle is carefully separated from the cheek flap until the aponeurotic part of the muscle is seen near the lower border of the zygomatic arch. Starting from the lower border, the medial surface of the masseter muscle is detached from the lateral surface of the mandible with electrocautery. Once the muscle is raised to the sigmoid notch of the mandible, we can see the coronoid process and its muscular attachment. The fibers of the temporalis muscle and deep fibers of the masseter are detached from the coronoid process, and the process is removed with the help of a micromotor. The removal of the coronoid im- proves the reach of the muscle into the surgical defect and reduces the risk of postoperative trismus.
The mobilized masseter muscle is held with a Bab- cock forceps and pulled into the defect to estimate the adequacy of the muscle length and determine whether there is any need for further mobilization. To increase the muscle length, aponeurotic fibers can be sharply incised with a knife. Since the vessels enter the muscle from the undersurface, a few superficial fibers of the muscle also can be safely incised to increase the length. The muscle is placed in the surgical defect, and its lower border can be sutured to the floor of the mouth (to cover the exposed mandible) and superiorly to the remaining buccal mucosa using interrupted Vicryl 3-0 sutures (figure 1). In a few patients who had the skin excision en bloc with the primary tumor, their skin defects could be closed by a local rotation flap.
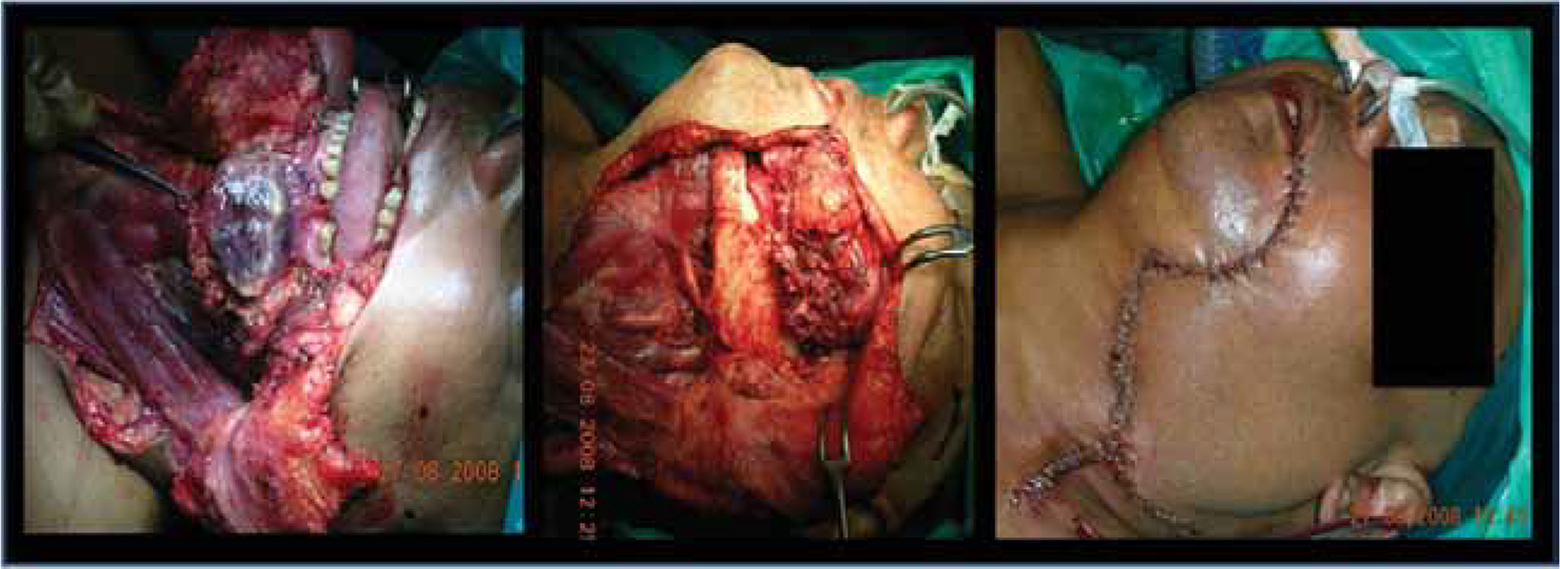
Intraoperative photos show a large buccal mucosa defect being reconstructed with a masseter flap.
Postoperative Management
All of our 17 patients had nasogastric tubes, and oral sips of water were started from the fifth postoperative day. Oral feeding was started as soon as patients were able to tolerate full liquid feeds, and the nasogastric tube was removed at that time. Jaw stretching exercises were started on the second postoperative day. Most patients were discharged as soon as the nasogastric tube was removed.
Data regarding the starting of oral feeds and mouth opening were collected at discharge, at the first follow- up visit at 3 months, and at 6 months. Nine patients received adjuvant radiotherapy according to the stan- dard guidelines.
Results
The mean duration for reconstructive surgery was 25 minutes (range: 12 to 46 minutes), and the mean time to starting oral feeding after surgery was 5.6 days (range: 3 to 14 days). Two patients had prolonged hospital stays due to delayed removal of the nasogastric tube (both had minor orocutaneous fistulas). The mean duration of hospitalization was 6.4 days (range: 5 to 7 days). In 11 patients, mucosalization (appearance of mucosal lin- ing) over the muscle of the flap occurred a mean of 22 days postoperatively (range: 16 to 42 days). Six patients showed weakness in the region of the marginal man- dibular nerve, which resolved completely in 3 patients after 25, 30, and 42 days, respectively.
Three patients developed major complications (one case each of major flap necrosis, parotid fistula, and hemorrhage). One patient, on the third postoperative day, developed hemorrhage from the raw muscle sur- face; it was controlled with cauterization. Two patients experienced partial necrosis of the muscle and devel- oped self-limiting orocutaneous fistulas. One of these patients required debridement under general anesthesia. One patient, as mentioned above, developed a major parotid fistula (>100 ml aspiration every day), and 5 had minor parotid fistulas (minor collection requiring aspiration only once a week). All fistulas were managed conservatively.
All 9 patients who were advised to undergo adjuvant radiotherapy completed it on time. With a median follow- up of 6.3 months (range: 3 to 8 months), 4 patients who had received adjuvant radiotherapy had mouth opening of <2 cm. Most patients achieved a mean inter-incisor distance of 3.5 cm (range 1.5 to 6 cm) at 6.3 months. All patients have normal swallowing and speech.
Discussion
Oral cancers have a high incidence in India, especially those located in the buccal mucosa due to the habit of tobacco chewing. 3 Reconstruction of buccal mucosal defects with an intact mandible is a challenging job. Although various nonmicrovascular options for re- construction have been reported, the free radial artery forearm flap appears to be the best choice in cases in which soft-tissue loss is minimal and the mandible is intact. However, because of infrastructure constraints and high complication rates in untrained hands, not all such patients can be offered this option. 2 Moreover, free radial artery forearm flap procedures may make the already-long waiting lists for surgery unwieldy in high-volume centers. We propose that the masseter flap can be attempted as an alternative in carefully selected patients, such as those with comorbid conditions, donor site problems, etc.
The masseter flap was described as long ago as 1867 by Lexer for the treatment of facial paralysis. 4 Conley and Gullane first described its use for oral cavity defects in malignancy in 1978. 5 It has been subsequently described by various authors for the reconstruction of oral and oropharyngeal defects. 6 -10 It can be used only in carefully se- lected patients in whom the masseter muscle can be saved without compromising their oncologic safety.
In our experience, masseter flap recon- struction is best suited for posterior buccal mucosal/retromolar trigone lesions that do not require removal of the masseter. Needless to say, it is unsafe to save the masseter muscle in the majority of such lesions. For that reason, despite our facility’s being a high-volume center, we could offer this procedure to only 17 patients. It is essential to know the anatomy of the masseter muscle before harvesting the flap. The masseter is a thick, quadrilateral muscle consisting of two portions— superficial and deep. The superficial portion, which is the larger of the two, arises via a thick, tendinous apo- neurosis from the zygomatic process of the maxilla, and from the anterior two-thirds of the lower border of the zygomatic arch. Its fibers pass downward and backward, to be inserted into the angle and lower half of the lateral surface of the ramus of the mandible.
The deep portion is much smaller and more muscular in texture. It arises from the posterior third of the lower border and from the whole of the medial surface of the zygomatic arch. Its fibers pass downward and forward, to be inserted into the upper half of the ramus and the lateral surface of the coronoid process of the mandible. The deep portion of the muscle is partly concealed in front by the superficial portion; behind, it is covered by the parotid gland.
Masseter muscle is innervated by the mandibular divi- sion of the trigeminal nerve. The masseteric branches of the transverse facial artery, facial artery, and internal maxillary artery supply the muscle. The first two arteries supply mostly the lateral muscle mass, and the masseteric branches of the internal maxillary artery supply the medial muscle mass. 7,8,11 The arteries enter the muscle through the sigmoid notch of the mandible. Therefore, extreme care should be taken not to raise the muscle above the sigmoid notch.
The masseter flap can be either superiorly based (as described above), inferiorly based, or as an island (based on masseteric branches of the internal maxillary artery). 9 The island masseter flap has more mobility as compared to the other two, but the blood supply is precarious. An intraoral approach has been described only for cases of facial palsy in which part of the muscle is transposed. 4
For reconstruction of mucosal defects, the masseter flap can he harvested only by an extraoral route after a cheek flap has been raised. This carries risks of damage to the mental nerve, branches of the facial nerve (espe- cially the mandibular branch), and the Stenson duct. 9 Most of the time during excision of oral cancers, the terminal end of the Stenson duct gets included in the specimen. We prefer to ligate the Stenson duct in those situations. Care should be taken to avoid injury to the duct while raising the muscle near the lower border of the zygoma. We find it extremely difficult to preserve the mandibular branch of the facial nerve while raising the cheek flap, and it is impossible when attempting a masseter flap. This flap becomes mucosalized (covered with mucosa) in 3 to 5 weeks (figure 2).
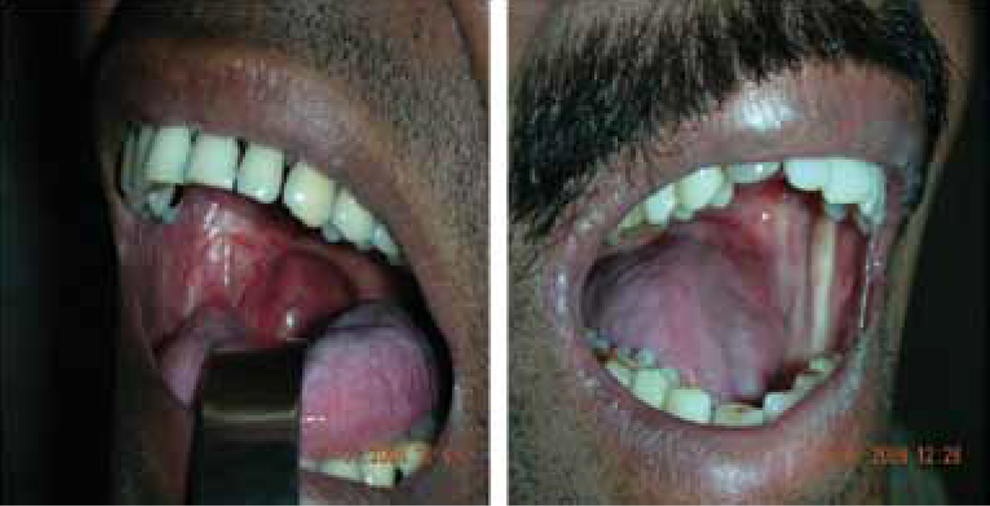
Intraoral photos show patients who have undergone masseter flap re- construction 6 to 12 months previously. Note the good mouth opening.
We have found that there is acceptable aesthetic deformity in the postoperative period (figure 3). A major drawback is trismus, which occurs as a result of postoperative fibrosis of muscle that seems to be aggra- vated by adjuvant radiotherapy. 9 Early and aggressive jaw stretching is the only solution we have found to be useful. At 6.3 months postsurgically, patients could achieve a mean mouth opening of 3.5 cm (range: 1.5 to 6 cm). Other significant complications were partial muscle necrosis and parotid fistulas, which were man- aged conservatively. Complete flap necrosis requiring revision has not been encountered to date.
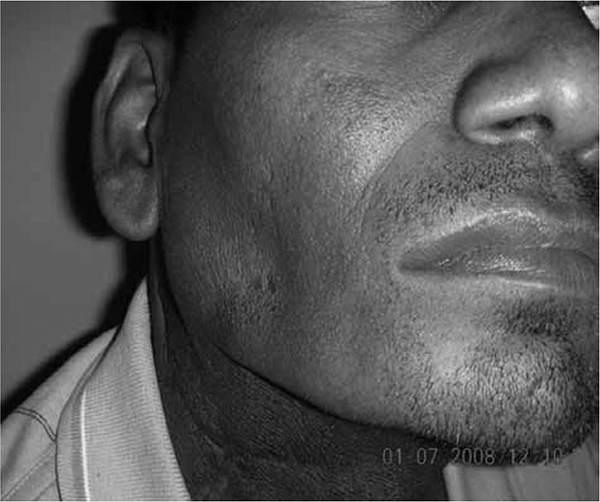
This patient underwent masseter flap reconstruction 6 months previously, followed by adjuvant radiotherapy for a right- sided retromolar trigone lesion.
The results of reconstruction with this flap may not appear exciting compared to those obtained after mi- covascular reconstructions. However, this flap is better than other reconstruction methods (such as skin grafts, nasolabial flaps, submental flaps, etc.) in terms of sur- gical time required, lack of donor site morbidity, and minimal aesthetic deformity. No single reconstruction technique can be universally applicable in all cases. We report that the masseter flap is a good tool in the surgeon’s armamentarium.
