Abstract
Introduction
Pharyngeal packs are frequently employed in dental and otolaryngological procedures, including chin osteotomy, adenotonsillectomy, and nasal septoplasty.1,2 They may be applied by the surgeon or the anesthesiologist during general endotracheal anesthesia in the pharyngeal region to mitigate postoperative complications.3,4 In light of the presence of a physical barrier, the use of pharyngeal packs is considered essential to prevent the introduction of blood, which is known to possess potent emetic qualities in the gastrointestinal system, into the stomach via the esophagus. This measure is taken to reduce postoperative nausea and vomiting (PONV), thereby minimizing the risk of aspiration. In addition, the physical barrier also effectively impedes blood from entering the trachea, thus reducing the likelihood of respiratory complications.5,6 The incidence of PONV following general anesthesia is reported to be more than 30%, 7 and this rate increases significantly in dental and otolaryngological surgeries, exceeding 50%. 8 Therefore, PONV is one of the most distressing postoperative complaints while under general anesthesia,7,9 which causes significant discomfort and increases the possibility of dehydration, electrolyte imbalance, and aspiration of gastric contents.7-9 Given these implications, the extensive use of pharyngeal packs is considered instrumental in striving to completely avoid PONV in clinical practice.
On the other hand, the use of a pharyngeal pack was also linked to postoperative throat pain. The occurrence of postoperative sore throat following intubation is common, and it was notably exacerbated after the implementation of a pharyngeal pack. 10 Postoperative throat pain was attributed to the irritation of the packing material on the mucosa, 11 which may lead to difficulty in swallowing. Consequently, postoperative dysphagia hindered the intake of oral fluids and medication, leading to dehydration and electrolyte imbalance, ultimately counterbalancing the potential benefits of the pharyngeal pack in reducing PONV. Furthermore, the pharyngeal pack was considered a predisposing factor for postoperative aphthous stomatitis in patients who underwent otolaryngological procedures, 12 and the retention of the pharyngeal pack postoperatively may elevate the risk of severe complications. 13
Recent clinical investigations have noticed that pharyngeal packs do not yield any discernible benefit in reducing PONV and are significantly associated with an increased risk of throat pain.14-16 The clinical impact of pharyngeal packs on patients undergoing dental and otolaryngological procedures continues to be a matter of controversy. In this study, our objective was to rigorously assess the precise role of the pharyngeal pack in clinical practice by conducting a quantitative meta-analysis based on high-quality randomized controlled trials (RCTs). The findings of this meta-analysis are anticipated to offer the highest level of evidence to inform clinical decision-making in this area.
Materials and Methods
Obtaining Potential Eligible Studies
The current study was registered in the INPLASY database (INPLASY, Inc., Middletown, DE, USA) with the ID INPLASY202320002 and was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 statement. 17 To retrieve relevant literature, potentially eligible studies were sourced from globally recognized databases, such as PubMed, Embase, and Cochrane Central. MeSH terms were utilized both individually and in combination to identify RCTs that examined the clinical effectiveness of pharyngeal packing in dental and otolaryngological procedures (an example of the search strategy used in PubMed is presented in Supplemental Table S1). The retrieval timeframe extended up to December 2022, and complete texts in English were sought for thorough assessment. The title and abstract of each pertinent study were reviewed for initial screening, with full texts scrutinized in detail to determine final inclusion.
Inclusion and Exclusion Criteria
Trials were deemed eligible if they met the following criteria: (1) high-quality RCT design; (2) sole intervention involving the application of a pharyngeal pack; (3) focused on dental and otolaryngological surgeries; (4) availability of full English text; and (5) reporting of at least one pertinent parametric indicator.
The exclusion criteria were defined as follows: (1) trials deemed as low-quality RCTs or non-RCTs; (2) studies where the application of a pharyngeal pack was not the only intervention or studies comparing different types of packs; (3) unavailability of full English text; (4) the absence of pertinent raw data; and (5) studies deemed as irrelevant, including reviews, comments, or clinical case reports.
Parametric Data Extraction and Synthesis
In evaluating the clinical efficacy of pharyngeal packs in dental and otolaryngological procedures, we chose key parameters, notably PONV and postoperative throat pain, for inclusion in the final quantitative analysis. The main data, encompassing the incidence of nausea, vomiting, total PONV (excluding cases presenting both nausea and vomiting), the incidence of throat pain, and visual analog scale (VAS) scoring of throat pain, were extracted by two independent investigators for data synthesis. Any debates pertaining to data extraction were resolved through group discussion until a unanimous agreement was reached. The pooled estimation was carried out via quantitative meta-analysis using the extracted data, which was stratified according to postoperative hours (POH) 0.5, 2, 4, 6, 8, and 24, to assess the clinical effects at different time points. Subgroup analysis was also performed taking into account different surgical types, packing types, and packing placements.
Assessment of Quality of Methodological Process and the Grade of Evidence
The methodological quality of the studies was assessed using the Jadad scoring system, which employs a scale ranging from 0 to 5 points. 18 Trials with a Jadad score of 0, 1, or 2 were excluded due to their classification as low quality. In addition, the risk of bias for each eligible study was evaluated by referencing the Cochrane Risk of Bias assessment tool. 19 The level of quantitative evidence was graded using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system, 20 which reflects the reliability of the conclusion. Comprehensive measurement involved consideration of five degradation factors and three upgradation factors. 21 Initial assessments were completed by two principal researchers, with any conflicting judgments resolved through group discussion.
Statistical Analysis
Concerning the selected parameters, the risk ratio (RR) and its 95% confidence interval (CI) were employed for pooled estimation of dichotomous variables for assessing the risk of clinical incidence of nausea, vomiting, total PONV, and throat pain. In addition, pooled standard mean differences (SMDs) along with their associated 95% CIs were calculated to determine the final variation of the VAS of throat pain between the packing group and the non-packing group as a continuous variable. To calculate the VAS, data presented as medians and ranges were transformed into means and standard deviations using established formulas.22-24 The heterogeneity of the overall results was assessed using the Q statistic, with I2 > 50% or I2 < 50% denoting significant or insignificant heterogeneity, respectively. A random-effects or fixed-effects model was employed based on the significance of heterogeneity. In addition, the symmetry of the funnel plot and the Egger test were utilized to assess the publication bias in the included trials. Statistical analyses were conducted using the STATA software package (version 15.0, Statacorp LLC, College Station, TX, USA). For the assessment of bias risk and evidence recommendation, Review Manager Software (version 5.3, The Cochrane Collaboration, London, UK) and GRADE profiler (version 3.6, McMaster University and Evidence Prime Inc., Hamilton, ON, Canada) were applied, respectively.
Results
Characteristics of the Included Studies
We initially retrieved 1875 reports from online databases and identified 19 potential eligible RCTs for inclusion. Following the quality assessment using Jadad scoring, 9 low-quality RCTs were excluded, resulting in the inclusion of 10 high-quality RCTs comprising 13 comparisons and 1026 participants for the final pooled analysis (Supplemental Table S2) (Figure 1). Among the included trials, there were eight 2-arm trials and two 3- or 4-arm trials. Seven trials reported various nasal surgeries, while three investigated other dental and maxillofacial surgeries. The details of the included RCTs are presented in Table 1.
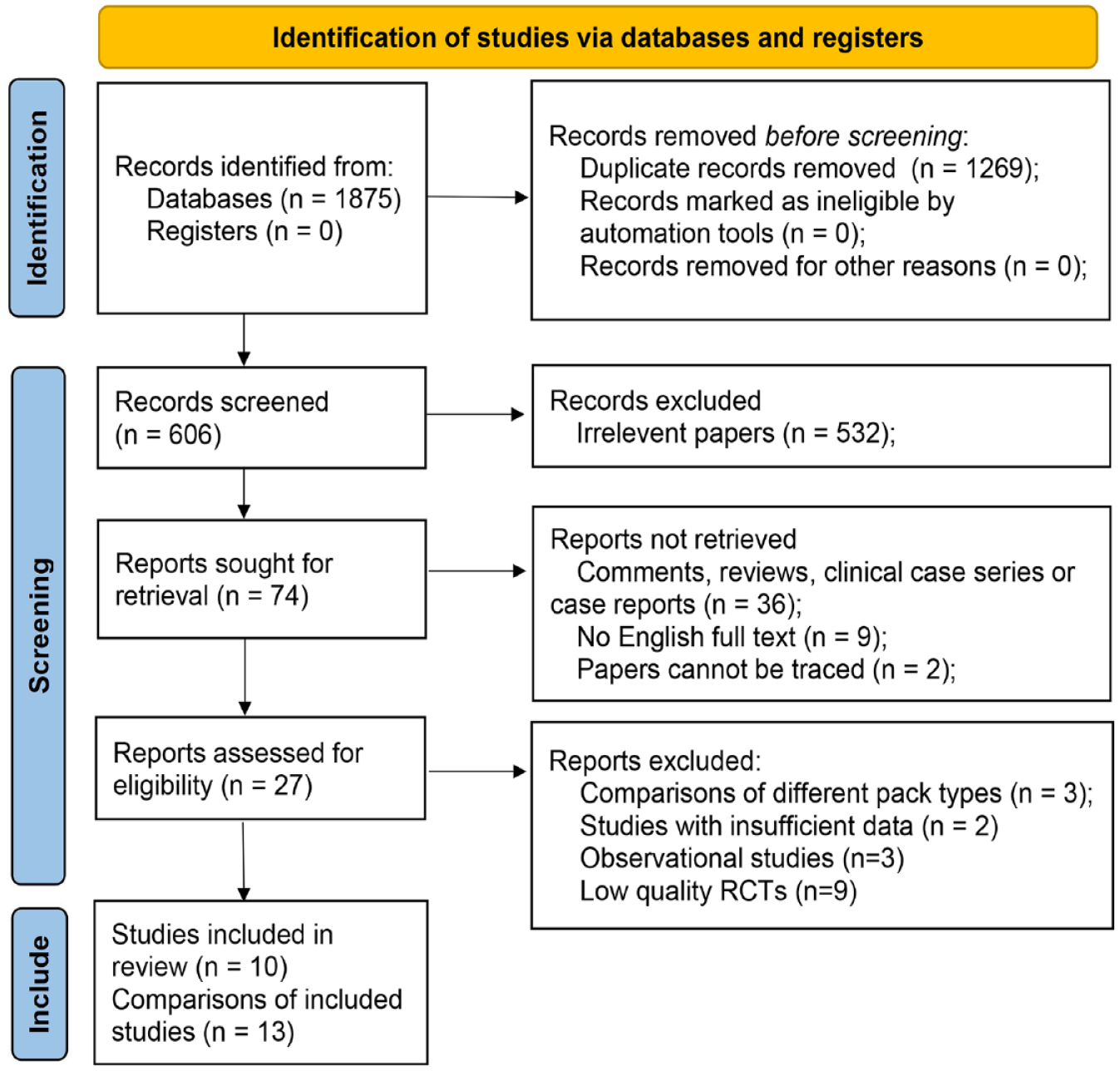
Flow diagram of the process of selecting studies for the current meta-analysis.
Characteristics of Included Studies.
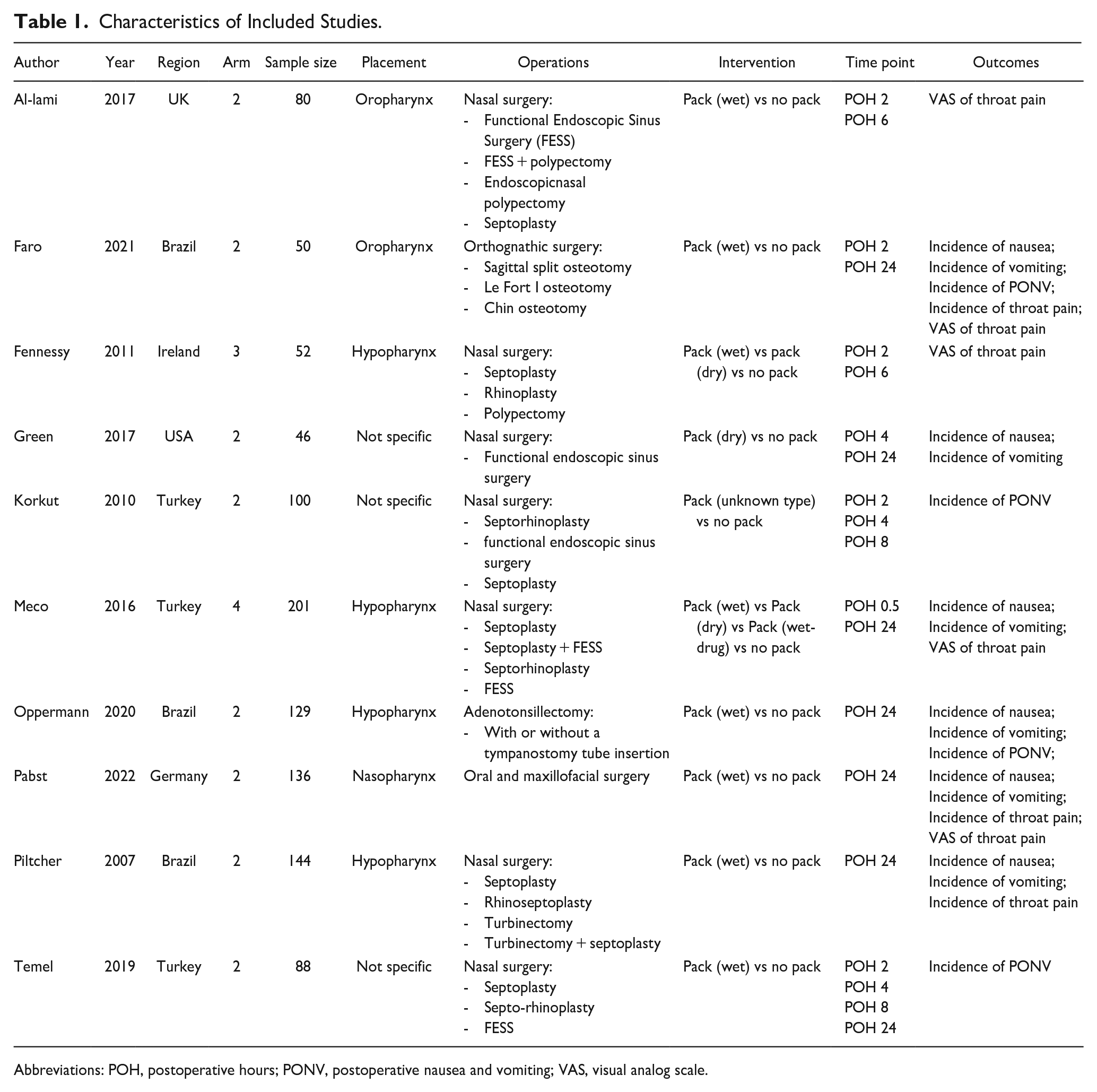
Abbreviations: POH, postoperative hours; PONV, postoperative nausea and vomiting; VAS, visual analog scale.
Assessment Quality of Methodological Process and Bias
The Jadad scoring system evaluated 19 RCTs, of which 10 were categorized as high quality for final assessment: 2 of the 10 included studies received a rating of 5 points, while the other eight were rated as 3 or 4 points (Supplemental Table S2). In the risk of bias assessment, no high-risk factor was identified among all studies. Six trials reported randomization with both generation and concealment. In addition, eight reports indicated the use of double blinding for methodological processes and complete outcomes but only half of them explicitly addressed the issue of selective reporting bias (Figure 2).
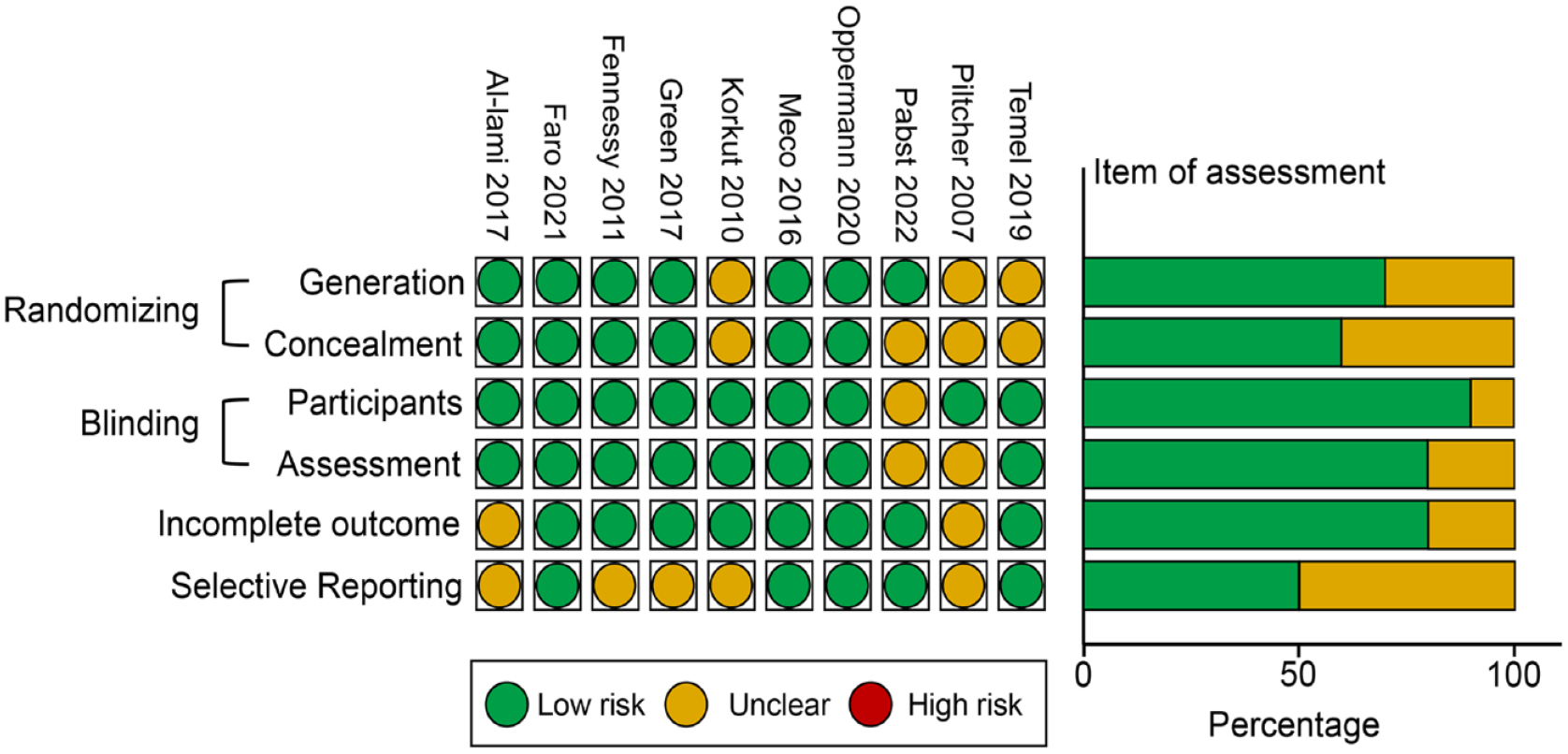
Bias assessment regarding each risk of bias item and summary of the included studies.
The Clinical Effects of Pharyngeal Pack on PONV and Throat Pain
We conducted a meta-analysis of 6 trials comprising 8 comparisons and 706 subjects to estimate the incidence of PONV. The results indicated that pharyngeal packing did not reduce the risk of nausea, with a P value of .272 (RR: 1.22, 95% CI: 0.86-1.73) (Figure 3), or the risk of vomiting, with a P value of .775 (RR: 0.93, 95% CI: 0.58-1.51), based on the fixed-effects model (Figure 4). Furthermore, the analysis of total PONV involved 4 trials with 367 participants. The results indicated that pharyngeal packing showed no effect in reducing the incidence of total postoperative PONV, with a P value of .118 based on the fixed-effects model (RR: 0.87, 95% CI: 0.74-1.03). In addition, no significant impact was observed at any of the time points (Figure 5).
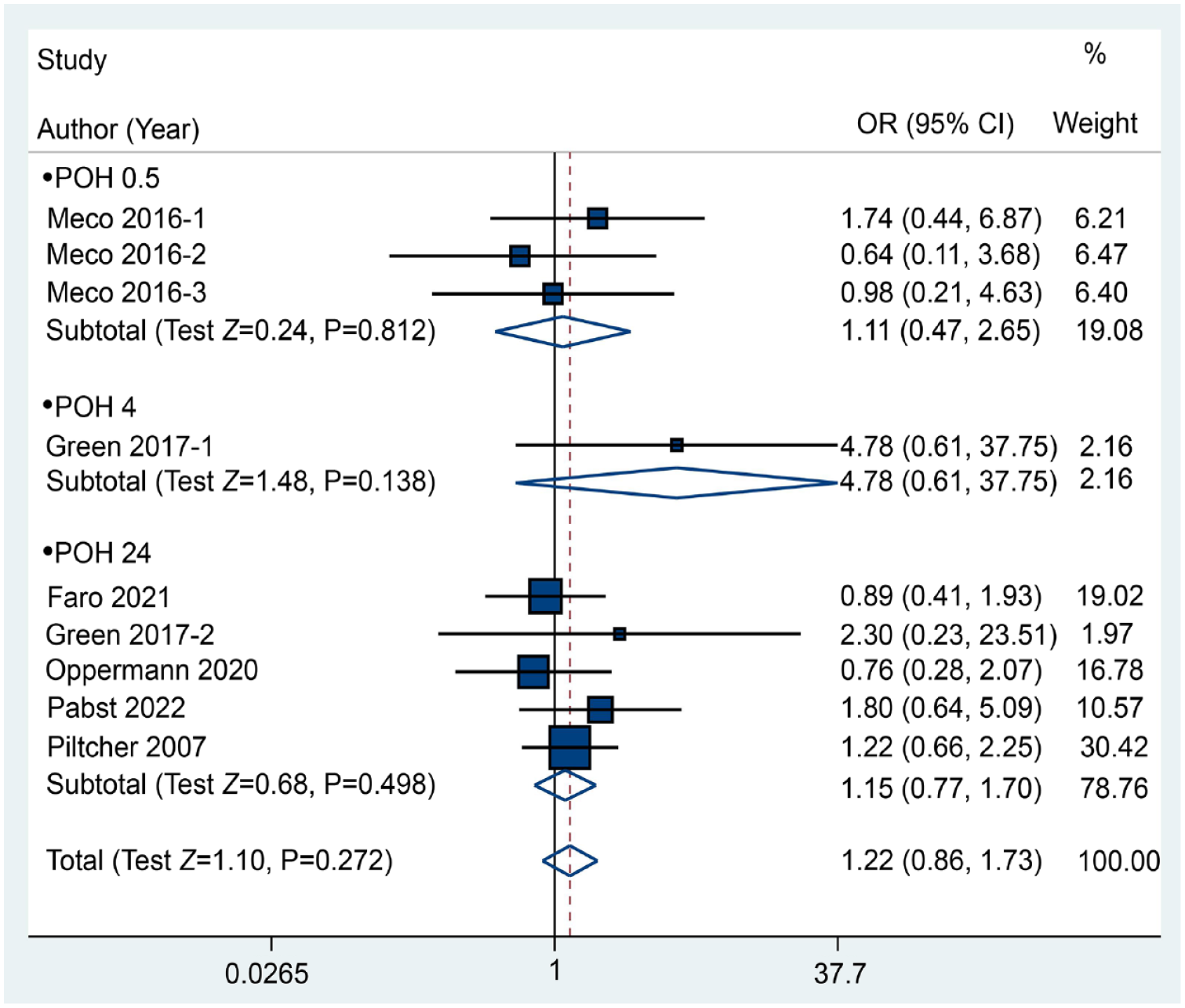
Forest plot between the packing and no packing groups with respect to the incidence of nausea.
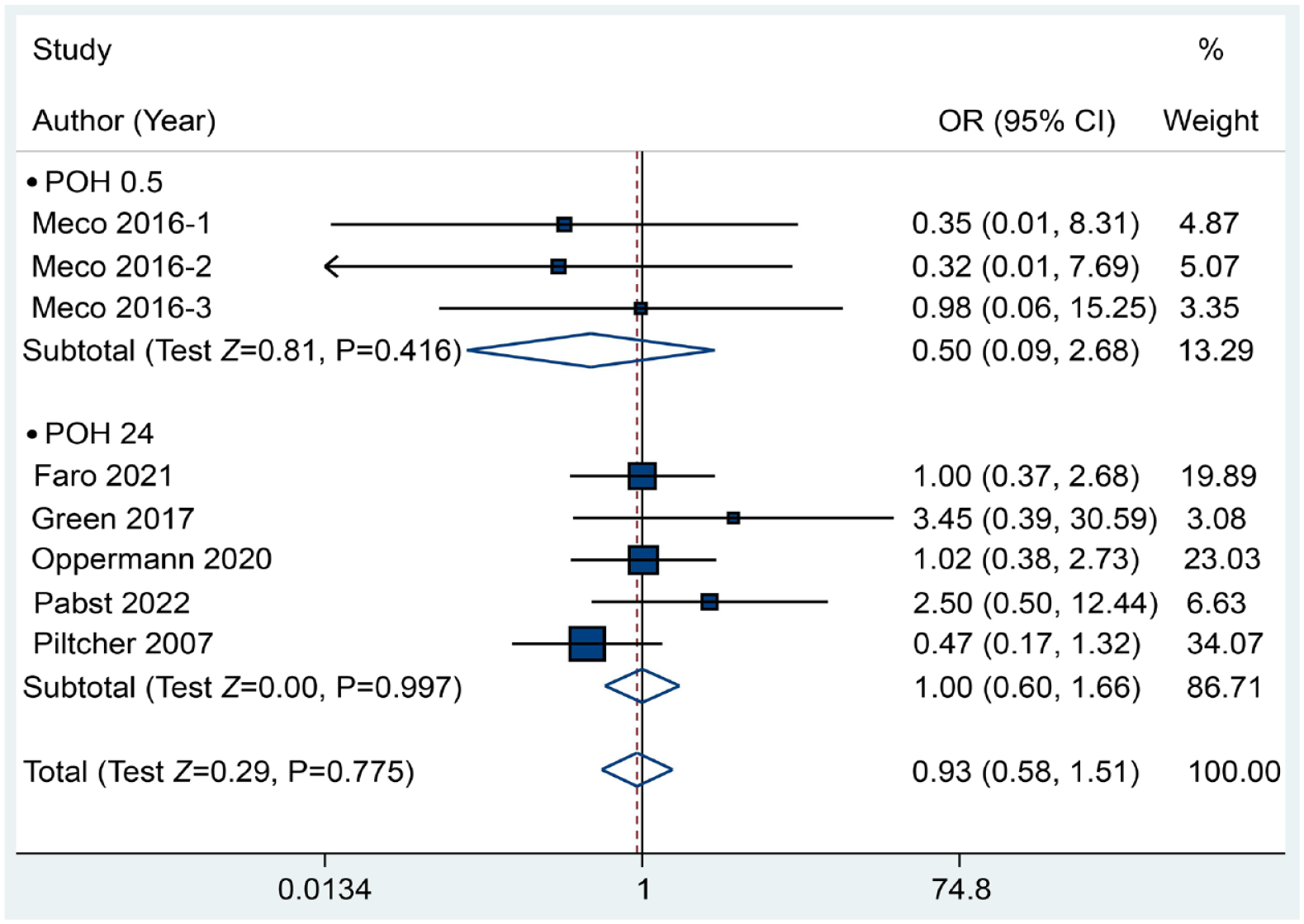
Forest plot between the packing and no packing groups with respect to the incidence of vomiting.
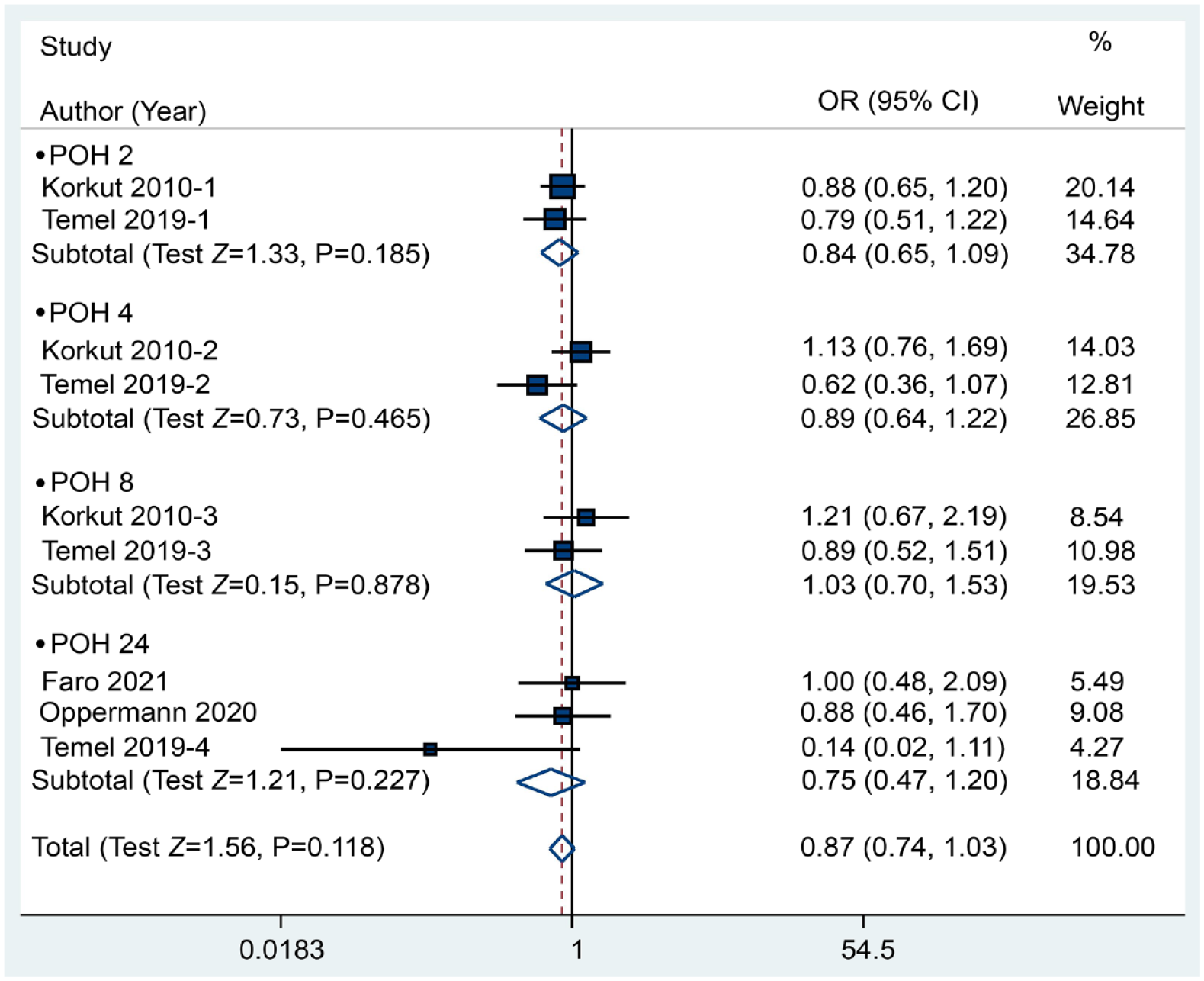
Forest plot between the packing and no packing groups with respect to the incidence of total postoperative nausea and vomiting.
To identify the influence of pharyngeal packing on postoperative throat pain compared with no packing, we evaluated the incidence of throat pain from 3 RCTs containing 4 comparisons. The results indicated no clinical benefit of packing in reducing postoperative throat pain, yielding a P value of .149 (RR: 1.22, 95% CI: 0.93-1.61) based on a random-effects model (I2 = 74.3%). Conversely, pharyngeal packing may increase the incidence of throat pain at POH 2, with an RR of 1.89 and a P value of .034 (Figure 6). Moreover, 5 included RCTs containing 519 patients compared the VAS of postoperative throat pain between the pharyngeal packing and no packing groups, and we discovered that the application of pharyngeal packs seemed to significantly increase the VAS of throat pain [SMD (95% CI) = 0.21 (0.07, 0.34), P = .003], although an insignificant difference was observed at each time point except POH 24 (Figure 7).
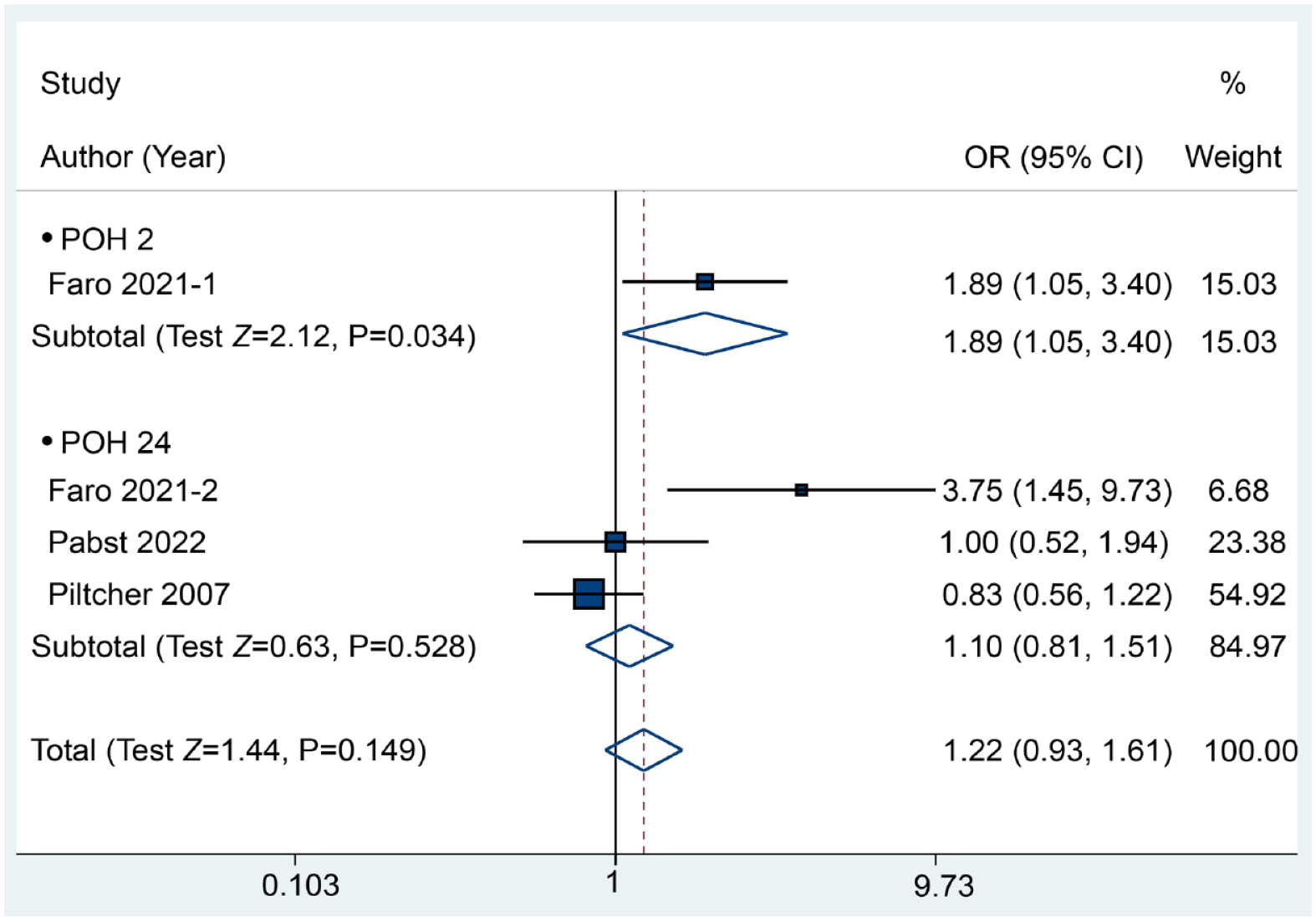
Forest plot between the packing and no packing groups with respect to the incidence of throat pain.
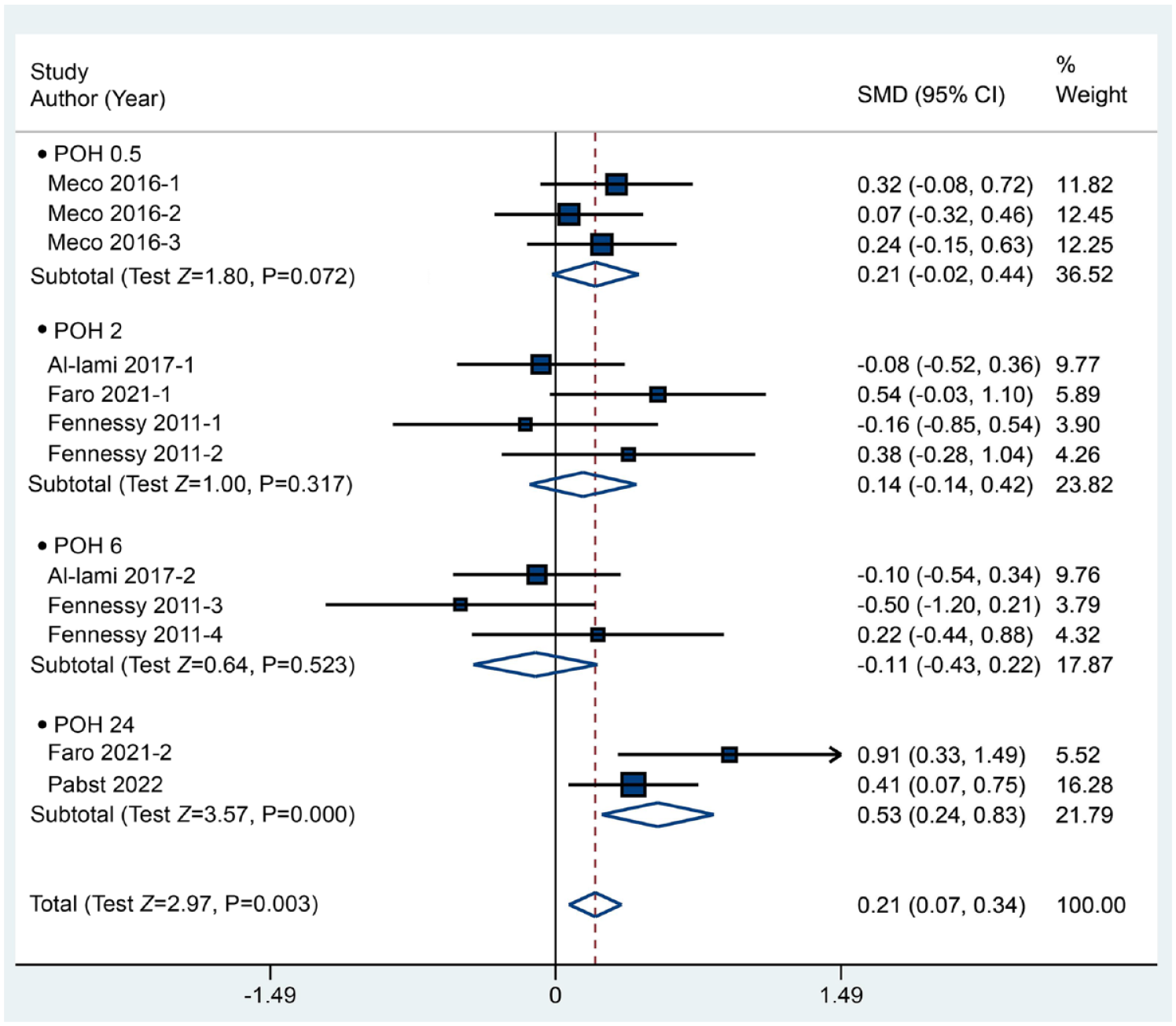
Forest plot between the packing and no packing groups with respect to the visual analog scale of throat pain.
Publication Bias and Recommendation of Evidence
To evaluate publication bias for each outcome, we conducted funnel plots and the Egger test. Our analysis did not show significant asymmetry in the funnel plots (Supplemental Figure S1), and the Egger test did not produce statistically significant results (Supplemental Figure S2). Following the bias assessments, we proceeded to analyze the overall evidence regarding five primary outcomes, namely the incidence of nausea, vomiting, total PONV, throat pain, and VAS scores for throat pain. The evidence concerning nausea, vomiting, and total PONV was rated as moderate due to the range of CIs, which could potentially introduce imprecision. However, the evidence for postoperative throat pain was rated as low grade and was not recommended due to the potential imprecision and high heterogeneity (Table 2).
The Quality and Recommendation of the Evidence According to the GRADE System.
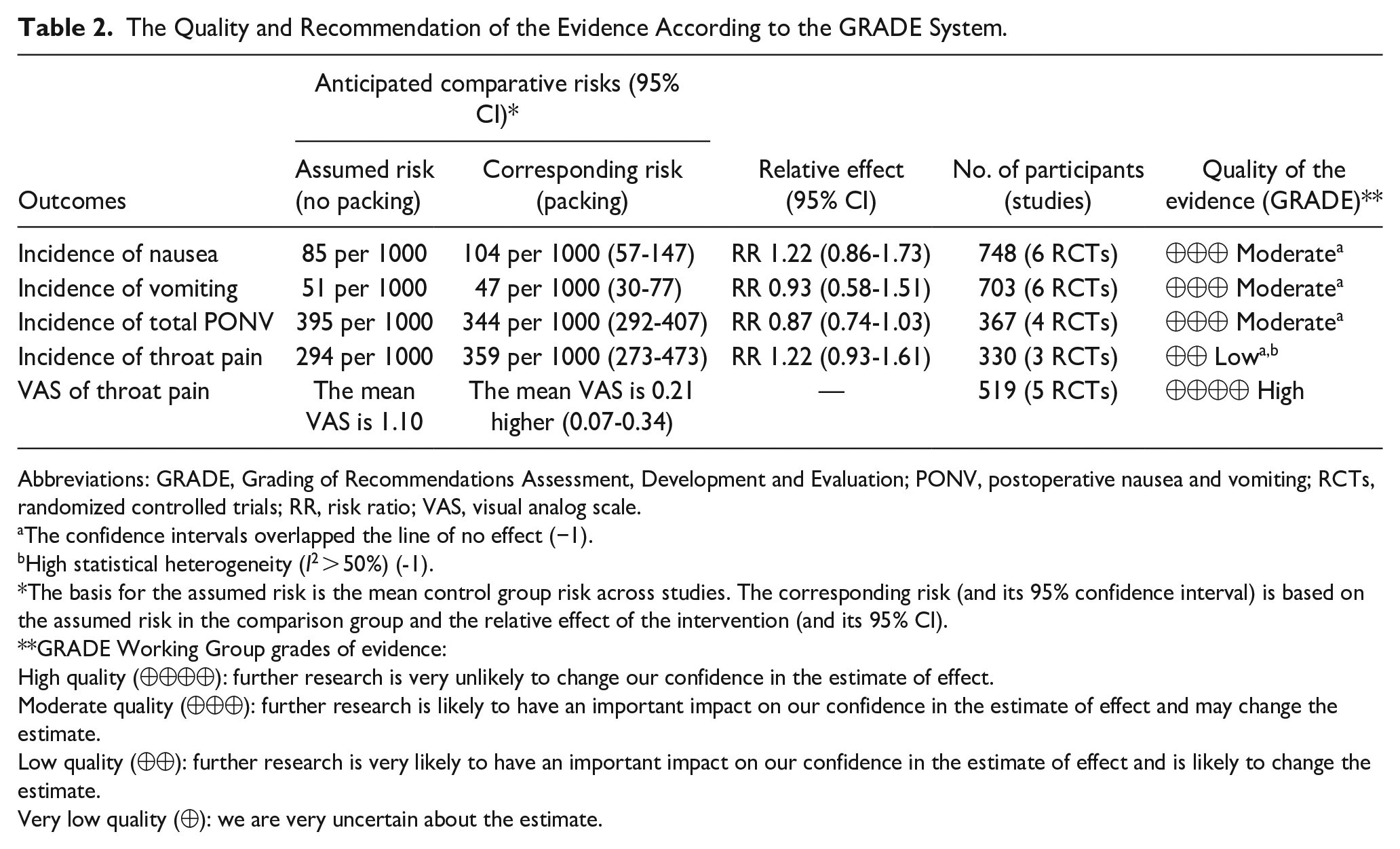
Abbreviations: GRADE, Grading of Recommendations Assessment, Development and Evaluation; PONV, postoperative nausea and vomiting; RCTs, randomized controlled trials; RR, risk ratio; VAS, visual analog scale.
The confidence intervals overlapped the line of no effect (−1).
High statistical heterogeneity (I2 > 50%) (-1).
The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
GRADE Working Group grades of evidence:
High quality (⊕⊕⊕⊕): further research is very unlikely to change our confidence in the estimate of effect.
Moderate quality (⊕⊕⊕): further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate.
Low quality (⊕⊕): further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate.
Very low quality (⊕): we are very uncertain about the estimate.
The Results of Subgroup Analysis
To further explore potential factors that may influence the main results of the current meta-analysis, we conducted subgroup analyses based on surgical type, packing type, and packing placement. While comparing the incidence of nausea and vomiting, no significant differences were observed between the packing and no packing groups across various subgroup categories. Notably, we observed that wet packs may reduce the risk of PONV (P = .027). However, in terms of the impact of pharyngeal packing on throat pain, we discovered that the packing route may increase the incidence and VAS of pain during oral and orthognathic procedures (P = .008 and .000), as well as during nasopharynx or oropharynx placement (P = .008 and .006). In addition, the use of wet packs may also increase the VAS of throat pain (P = .009) (Table 3).
The Results of Subgroup Analysis Regarding Surgery, Packing Type, and Packing Placement.
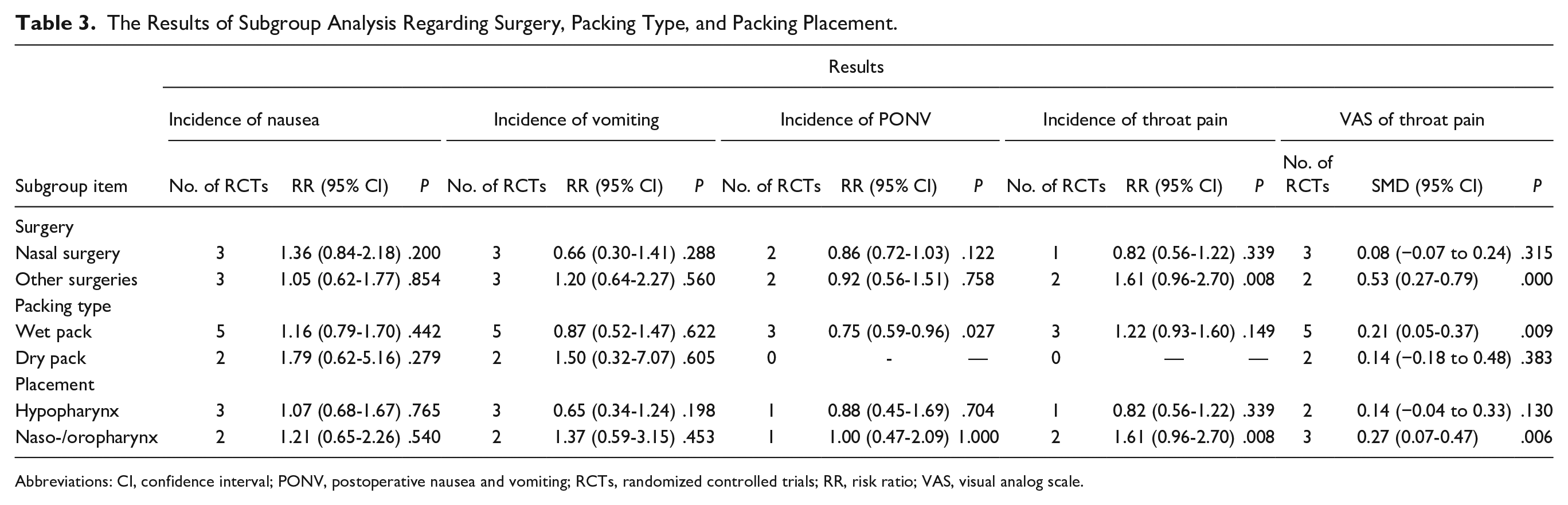
Abbreviations: CI, confidence interval; PONV, postoperative nausea and vomiting; RCTs, randomized controlled trials; RR, risk ratio; VAS, visual analog scale.
Discussion
Dental and otolaryngological surgeries involve a narrow range of procedures; yet, they are associated with the potential for diverse postoperative complications, owing to their proximity to the junction of the esophagus and trachea and their heightened sensitivity. As previously indicated, pharyngeal packing is theoretically posited to lower the occurrence of PONV by serving as a barrier against surgical debris, blood, and secretions from entering the esophagus or being absorbed in the stomach.5,6,10
The clinical rationale behind the use of pharyngeal packing was based on the theory that PONV could be attributed to the ingestion of surgical blood and secretions.3,25 However, to date, quantitative confirmation of whether the volume of surgical fluid during oral or maxillofacial operations is sufficient to induce PONV has not been established. 26 While we acknowledge that oral or maxillofacial surgeries may result in considerable blood loss, the impact of this blood loss on hemodynamic stability, possibly leading to PONV, dehydration, or electrolyte imbalance, is contingent on the volume lost and the patient’s weight. 27 In other words, varying volumes of bleeding may give rise to different degrees of PONV and consequently influence the efficacy of pharyngeal packing. Hence, in the context of oral or maxillofacial surgeries, the utility of pharyngeal packing may be limited in preventing PONV.
On the other hand, the effectiveness of the pharyngeal pack as a barrier against blood aspiration remains uncertain. Powell et al 16 reported that the use of pharyngeal packing did not diminish the ingestion of blood during orthognathic surgery, thus indicating no significant impact on reducing the incidence of PONV. By contrast, Temel et al 28 stated that pharyngeal packing may substantially reduce the gastric volume in nasal surgery, thereby serving as a preventive measure against PONV. This contradictory conclusion seemed to be associated with different surgical types, yet the results of our subgroup analyses did not support this association. Similarly, based on our subgroup analysis results, we have discovered that the placement of pharyngeal packing, whether in the hypopharynx or the naso-/oropharynx, seems to have no substantial influence on the amelioration of PONV. In addition, a previous study on sinonasal surgeries also observed that no significant differences were yielded between the placement of the nasopharynx and placement of the hypopharynx, 29 thus indicating that the role of placement site may not be particularly impactful in the context of PONV. In addition, a pharyngeal pack was helpful to seal the tracheal tube for pediatric patients to prevent endotracheal tube leakage.1,30 The pharyngeal pack may be functional in preventing contamination of the upper aerodigestive tract to reduce the incidence of PONV.14,31 However, this hypothesis still needs further verification.
Among the clinical RCTs, several studies have demonstrated that the use of pharyngeal packs does not confer any advantage in decreasing the likelihood of PONV.10,32 Moreover, an investigation reported results suggesting that pharyngeal packs may even increase the incidence of PONV in oral surgery. 33 Among the high-quality RCTs included in our analysis, only one publication indicated a potential benefit in reducing PONV, 28 while the remaining studies did not6,14,15,26,34-36 In the present meta-analysis, we aggregated the estimated incidence of nausea, vomiting, and total PONV, and none of these outcomes showed statistical significance. Current evidence supports the limited effectiveness of pharyngeal packs in reducing the overall risk of PONV, aligning with the findings of the majority of published investigations.
Similar to the conflicting evidence regarding PONV, certain studies have suggested that the prevalence and severity of throat pain are closely linked to the use of pharyngeal packs,34,37 while others have presented opposing viewpoints.38,39 As previously mentioned, throat pain may be elicited by irritation from the packing material on the mucosa and is also associated with the duration of surgical procedures. 11 However, some argue that the application of a pharyngeal pack is not the sole factor contributing to throat pain and that the type of pack may also play an important role. Some trials deduced that wet pharyngeal packs may result in significant throat pain, but dry packs may not,6,34,35 which was partially consistent with our results of the subgroup analysis. Nevertheless, this hypothesis has not been consistently replicated in other investigations.26,36,40
According to the results of the subgroup analysis in our study, surgical type and placement may be other factors influencing throat pain. The consistent outcomes observed for surgical type and placement in our subgroup analysis may be attributed to the utilization of the same data source literature for data synthesis. Our findings suggest that the placement of hypopharyngeal packing does not appear to have a significant impact on postoperative throat pain, while the placement of naso-/oropharyngeal packs appears to have notable effects (Table 3). Among the included studies, both oropharyngeal and nasopharyngeal packing were associated with increased postoperative throat pain.11,26,34 In a comparative study of placement sites, Rizvi et al 41 reported a higher likelihood of postoperative throat pain among patients with oropharyngeal packs compared to those with nasopharyngeal packs at all-time intervals after extubation. In addition, Alfiky et al 29 suggested that there was no significant difference in the impact on postoperative throat pain between nasopharyngeal and hypopharyngeal packing. However, due to the limited sample size in these two studies, their respective conclusions remain inconclusive. Therefore, in light of the existing evidence, we posit that different placement sites, particularly naso-/oropharynx packing, may potentially influence postoperative throat pain, requiring further validation to reach definitive conclusions.
The investigation on the association between specific surgical procedures and postoperative throat pain following the use of pharyngeal packs has been scarce, posing challenges for comprehensive discussion. An alternative viewpoint posited that postoperative throat pain could be effectively managed and that drug-soaked packs might mitigate the frequency or severity of throat pain.42-45 Nevertheless, the impact of drug-soaked pharyngeal packs on throat pain has been inadequately studied, with only a single 4-arm RCT currently addressing these effects, and finding no significant impact on the severity of throat pain. 6 Moreover, other adverse events, such as dysphagia and foreign body sensations along with throat pain, were not given enough attention in the analysis. Pabst et al asserted that dysphagia and foreign body sensations, in addition to throat pain, were the most prevalent side effects following the use of pharyngeal packs. These side effects may lead to further consequential complications, particularly in oral and maxillofacial procedures. 26 Therefore, these adverse events may be closely related to the incidence and level of postoperative throat pain and may help accurately reflect the throat pain affected by pharyngeal packs in other aspects. Together, the application of pharyngeal packs seemed to be one of the important factors influencing postoperative throat pain, although its predominant role could not be determined. However, it can be confirmed that pharyngeal packs presented no benefit in reducing the risk and level of throat pain based on current evidence.
A previous meta-analysis assessed the clinical effectiveness of pharyngeal packs in nasal surgery. 5 However, this review was significantly limited by the inclusion of numerous low-quality trials and its exclusion of several relevant RCTs based on predetermined criteria. Furthermore, it provided inaccurate information regarding quality assessment and clinical features. Another recent meta-analysis examined the use of pharyngeal packs in dental and otolaryngological surgeries. Nevertheless, its focus was solely on estimating the occurrence of throat pain at different points in time, failing to deliver a comprehensive analysis. 46 These limitations collectively raise concerns about the robustness of the respective conclusions. In our study, we conducted a comprehensive analysis of PONV and throat pain based on high-quality RCTs for the first time. No evident bias was detected, and the overall evidence holds substantial clinical relevance, notwithstanding some inevitable limitations. First, we focused solely on analyzing PONV and postoperative throat pain as the most prevalent parametric data, while other indicators, such as dysphagia, antiemetic usage, or intraoperative bleeding, were not evaluated due to limited data sources. Notably, the results of our subgroup analysis indicated that wet packs may reduce the incidence of total PONV, although they could not simultaneously reduce the incidence of nausea and vomiting. This may be due to the limitations of small coverage reports but it may be an interesting direction for future clinical research. In addition, the results of pooled estimations for certain parameters may be susceptible to undetectable bias owing to the limited number of studies available.
Conclusions
Our findings substantiate that the use of pharyngeal packs does not mitigate the likelihood of postoperative nausea, vomiting, or throat pain. Furthermore, their utilization may actually escalate the severity of postoperative throat pain. Our concluding remarks necessitate the inclusion of additional high-quality RCTs to facilitate a more comprehensive analysis of clinical efficacy based on other pertinent parameters. At present, our study warrants the conclusion that the application of pharyngeal packs is not recommended in dental and otolaryngological surgeries.
Supplemental Material
sj-doc-1-ear-10.1177_01455613231223352 – Supplemental material for The Pharyngeal Packs for Dental and Otolaryngological Surgery: A Meta-Analysis of High-Quality Randomized Controlled Trials
Supplemental material, sj-doc-1-ear-10.1177_01455613231223352 for The Pharyngeal Packs for Dental and Otolaryngological Surgery: A Meta-Analysis of High-Quality Randomized Controlled Trials by Xiaojuan Xie, Yao Yao, Min Shi, Haifeng Ding, Daijuan Zhang, Yingying Jiang and Tao Guo in Ear, Nose & Throat Journal
Supplemental Material
sj-docx-2-ear-10.1177_01455613231223352 – Supplemental material for The Pharyngeal Packs for Dental and Otolaryngological Surgery: A Meta-Analysis of High-Quality Randomized Controlled Trials
Supplemental material, sj-docx-2-ear-10.1177_01455613231223352 for The Pharyngeal Packs for Dental and Otolaryngological Surgery: A Meta-Analysis of High-Quality Randomized Controlled Trials by Xiaojuan Xie, Yao Yao, Min Shi, Haifeng Ding, Daijuan Zhang, Yingying Jiang and Tao Guo in Ear, Nose & Throat Journal
Footnotes
Authorship Contribution
TG and YYJ designed the research. XX, YY, MS, HD, DZ, and JY performed the research and data collection. XX, YY, and HFD contributed analytic tools and data analysis. XX, TG, and YYJ wrote the paper. All the authors have read and approved the final manuscript.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author. Data sharing does not applicable to this article as no datasets were generated or analyzed during current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural Science Foundation of Shandong Province (ZR2020MH415).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
