Abstract
Benign tumors or malignant neoplasms must be evaluated in patients with unilateral nasal cavity mass lesions. The 3 most prevalent unilateral benign mass lesions in such individuals are nasal polyps (NPs) and inverted papillomas (IPs). Although rare, it should be kept in mind that sinonasal hamartomas can be occasionally diagnosed as well. Among sinonasal hamartomas, respiratory epithelial adenomatoid hamartoma is more prevalent, with seromucinous hamartoma (SMH) being the second most common. Unlike NPs, sinonasal hamartomas are benign tumors with growth potential, which means it should not be undertreated and warrants surgical removal for treatment. However, sinonasal hamartomas do not have local invasion or malignant transformation potential like IPs; hence, it is vital not to overtreat them. Therefore, understanding the histopathology of SMH and thereby establishing proper surgical planning prior to the surgery remains crucial in such cases. Here, we present a successfully treated case of SMH with a distinctive radiographic, gross, and pathological clinical image of SMH.
Keywords
Introduction
In otorhinolaryngology (ENT) practice, we often experience an unilateral nasal cavity mass lesion. In such cases, the ENT physician should be able to suspect benign or malignant lesions. In such situations, the most common benign lesions are inflammatory polyps, nasal polyps (NPs), or inverted papillomas (IPs). 1 However, despite their rarity, it is important to note that hamartomas in the sinonasal cavity can occasionally be diagnosed as well. 2
To date, 4 entities of hamartomas have been classified in the sinonasal tract based on their distinctive histological characteristics: respiratory epithelial adenomatoid hamartoma (REAH), seromucinous hamartoma (SMH), chondro-osseous and respiratory hamartoma (CORE), and nasal chondromesenchymal hamartoma (NCH). 3 REAH is the most frequently reported of these 4 histological subtypes, with SMH being the second most frequently reported. 4 In contrast to sinonasal REAH, which has been recorded often in earlier literature, fewer than 30 instances of sinonasal SMH have been published to date, causing a paucity of information describing the morphological and histological features of SMH.
Although sinonasal hamartomas are a genuine mass-forming lesion characterized by the proliferation of normal tissues, unlike NPs, they do not regress on their own and must be surgically removed. 5 However, they lack the capacity for local invasion or transformation into a malignant form, which is a typical feature of IPs. Therefore, sinonasal hamartomas should be surgically treated to alleviate the patient’s symptoms; however, the surgeon must bear in mind that these lesions are truly benign, and the planning for surgical excision should be less radical than when resecting malignant sinonasal tumors. 3
Herein in this case report, we provide a case of successfully treated SMH with the provision of a high-quality endoscopic, radiographic, gross, and histological images.
Case Presentation
A healthy 30-year-old male was referred to the ENT clinic for right-sided nasal obstruction and mucopurulent rhinorrhea over the last 6 months. The patient did not complain of any loss of smell, epistaxis, or facial pain. The patient’s past medical history did not reveal any trauma or sinonasal surgeries. On nasal endoscopy, a mass lesion was seen in the right nasal cavity. The mass was mobile, having a convoluted polypoid surface with a firm texture, and did not bleed upon palpation with bayonet forceps (Figure 1A).
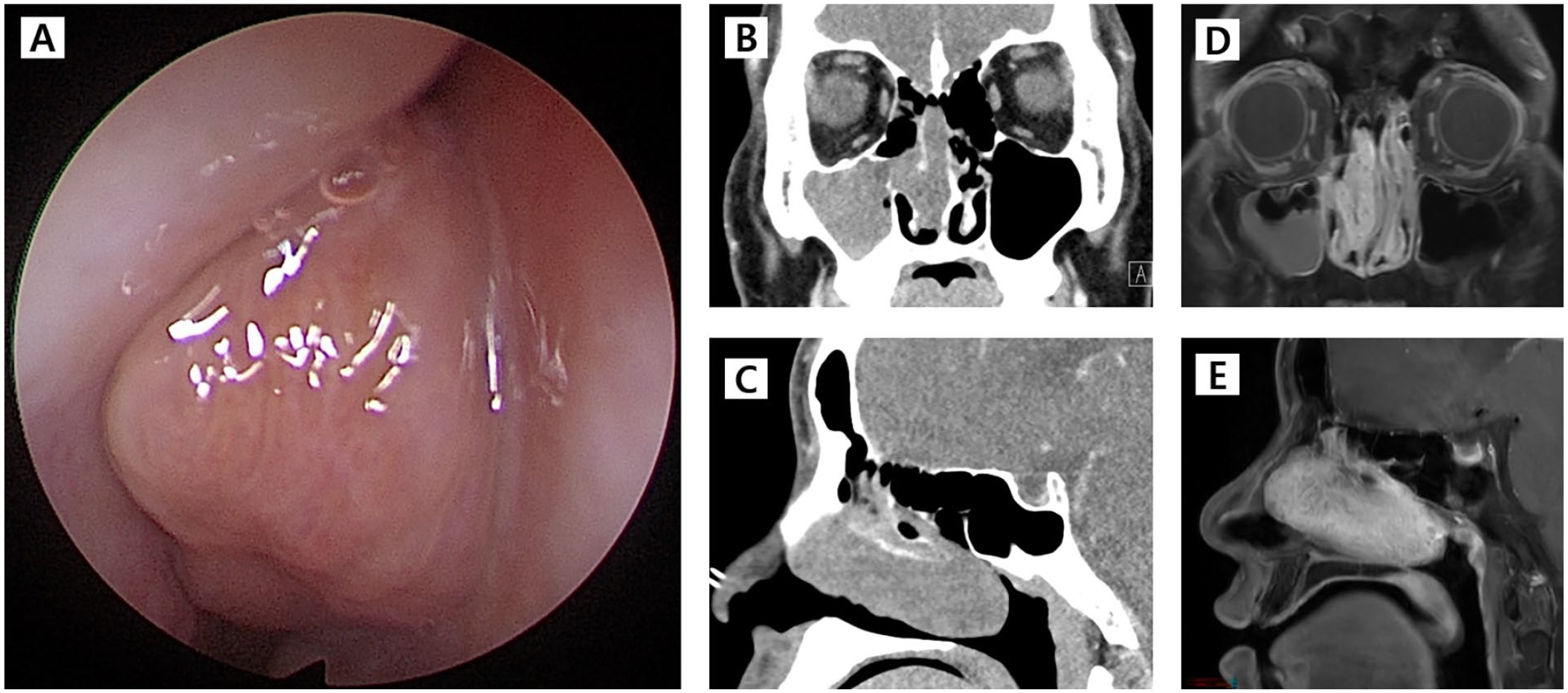
Nasal Endoscopy and radiological images of seromucinous hamartoma. (A) A firm, polypoid, convoluted mass in the right nasal cavity. (B) and (C) Contrast-enhanced computed tomography images showing a mass lesion in the right nasal cavity, surrounding the right middle turbinate, elongating from the anterior choana to the posterior choana. (D) and (E) Gadolinium-enhanced magnetic resonance image showing a rapid wash-in and progressive enhancement with the convoluted cerebriform enhancement pattern of the mass lesion.
On the contrast-enhanced computed tomography, a mass lesion in the right nasal cavity surrounding the right middle turbinate, elongating from the anterior choana to the posterior choana, was noticed (Figure 1B and C). On a gadolinium-enhanced magnetic resonance image, a mass showing a rapid wash-in and progressive enhancement with the convoluted cerebriform enhancement pattern was identified (Figure 1D and E). The in-office punch biopsy of the mass revealed nasal mucosal tissue with Schneiderian epithelial hyperplasia and glandular hyperplasia without any malignant histologic features.
Under general anesthesia, an elective endoscopic endonasal resection of the mass with a middle meatal antrostomy was performed, and the surgery underwent without complications. The mass attached to the lateral nasal wall and was taken out in an en bloc manner (Figure 2A, medial surface and B, lateral surface); the mass was colored yellow–orange and had a rubbery, firm texture with a polypoid surface. The obtained surgical specimen was sent to the pathology lab for histological confirmation (Figure 2C, ×4 and D, ×100). The final pathology report confirmed a diagnosis of SMH. The patient was free of nasal symptoms following surgery, with no recurrence at an 8-month follow-up.
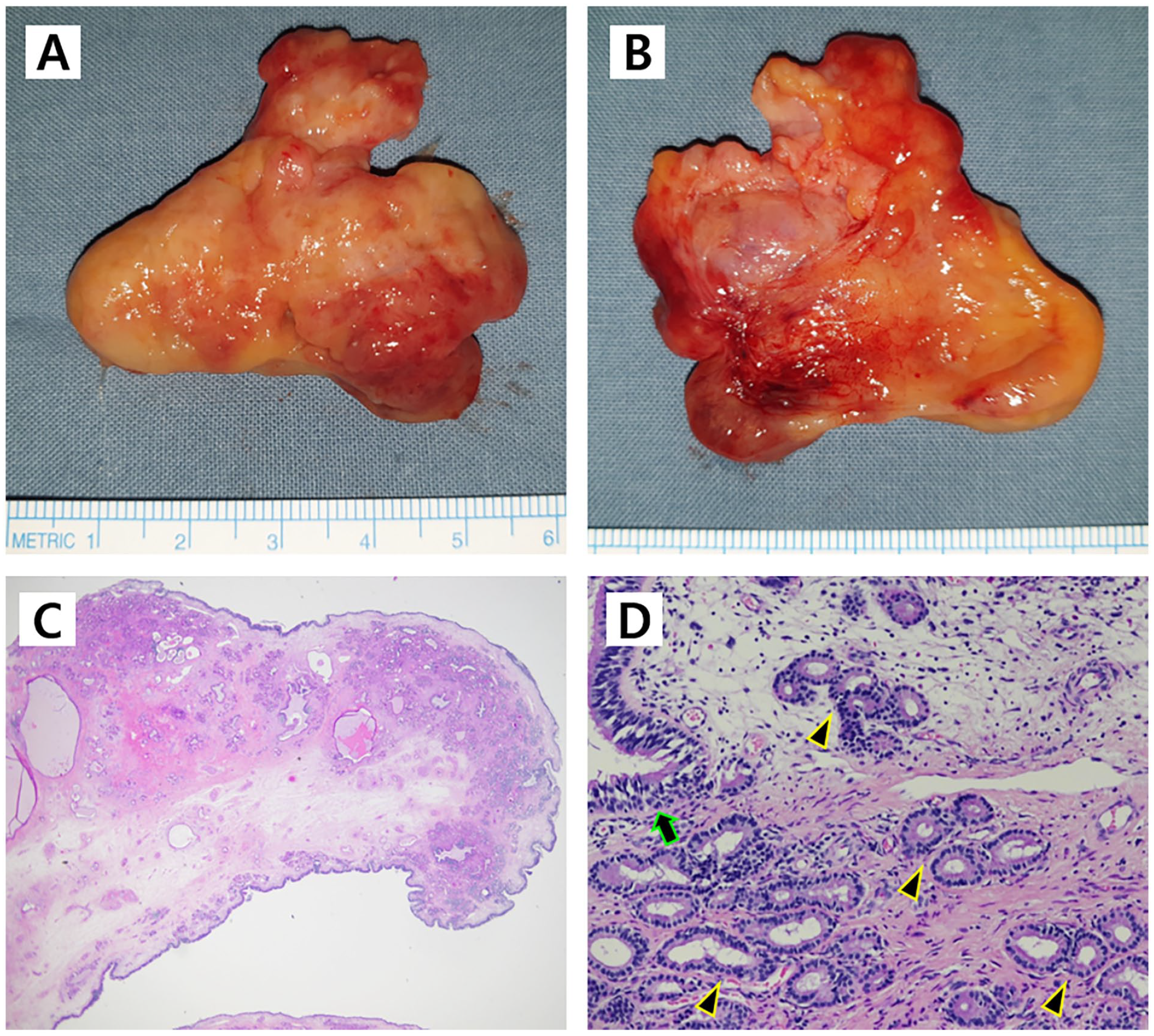
Gross and histological images of SMH. (A) and (B) Gross photographs of an excised surgical specimen of sinonasal SMH. (C) and (D) Hematoxylin–eosin stain pathology image showing a mass lined up with respiratory epithelium (arrowhead) in a fibrous background, concurrently showing lobular proliferation of seromucinous glands (arrows).
Discussion
Hamartomas, which are defined as tumorous lesions comprised of a collection of normal tissue components of the involved organs, can occur throughout the whole body. 6 Although the cellular components of hamartomas are composed of normal cells, there are aberrations in the proliferation, distribution, and ratio of cellular components within this benign tumor lesion. 6 When it comes to the sinonasal tract, 4 subtypes of sinonasal hamartomas have been identified depending on the features shown on the pathology. With REAH being the most prevalent, SMH is the second most frequently reported entity, followed by CORE and NCH, which are relatively uncommon. 2
SMH, a subtype of sinonasal hamartoma, is given its name due to its histological appearance. First described in 1974, SMH features a convoluted, polypoid surface in gross, whereas inflammatory polyps tend to have a smooth surface. The histology of SMH reveals a mass lined with respiratory epithelium with a fibrous background, with the hyperplasia of seromucinous glandular structures arranged in a lobular pattern, which is a distinctive feature to diagnose SMH from other sinonasal hamartomas. 2
One distinctive clinical importance of SMH from other sinonasal hamartomas is that SMH may be often confused and misdiagnosed with low-grade sinonasal adenocarcinoma (LGSAC), especially the non-intentional type. 7 LGSAC is a truly malignant neoplastic condition. Thereby it is crucial to distinguish SMH and LGSAC in the obtained surgical specimen to establish a proper surgical resection margin and further treatment planning. Nevertheless, both SMH and LGSAC exhibit similar features on histology, making it challenging to distinguish these 2 tumors.
Both tumors reveal respiratory epithelial hyperplasia accompanied by glandular hyperplasia, which the cells are cytologically bland and monomorphic cuboidal to columnar cellular lining are present. 7 One crucial tip to separate those 2 lesions is that LGSAC distinctively features a dense, back-to-back glandular proliferation with papillary projections and a tufted epithelial lining, which are never featured in SMH. However, since LGSAC does not exhibit some typical features shown in malignant tumors, such as mitoses, necrosis, perineural invasion, or lymphovascular invasion, 8 acts as a hurdle to clearly distinguishing this tumor from SMH in a real world. Moreover, immunohistochemistry is not beneficial in distinguishing those 2 tumors, as both SMH and LGSAC show negative p63 9 and CK 20 staining, and positive stain results with S-100, CK7, and CK19.10,11 Therefore, ENT surgeons and surgical pathologists should be fully aware of existence of both tumors and should be able to suspect and communicate in cases showing such pathology.
Although sinonasal hamartomas are much less frequently diagnosed compared to inflammatory polyps or IPs, precisely identifying this tumor preoperatively and understanding the pathology of this tumor is essential for otorhinolaryngologists so the patients are not “overtreated” or “undertreated”. 3 Since sinonasal hamartomas are a truly benign but neoplastic lesion, they may cause and worsen symptoms such as nasal obstruction or secondary sinusitis, as in our patient, unless completely removed. Therefore, it is crucial to completely remove the lesion with surgery in order to manage the patient’s discomfort. Nevertheless, the reported recurrence rate of sinonasal hamartoma is extremely low, as is its non-malignant transformation potential, which is contrary to that of IPs. In contrast to the removal of IPs, which frequently necessitates extensive surgical approaches to eradicate the tumor and its attachment site, 12 radical approaches such as external approaches or skull base manipulations may not be required for the management of sinonasal hamartoma. 13
Recently, SMH and REAH have received more attention worldwide because they should be managed properly, apart from polyps or IPs. In 2017, the fourth edition of the World Health Organization Classification of Head and Neck Tumors announced SMH as one of the subtypes of REAH, highlighting the distinctive seromucinous glandular proliferation and separating it from other subtypes. 14 Accordingly, a review article has been published addressing the distinctive radiological features of SMH and REAH was published in the American Journal of Neuroradiology. 15 Therefore, understanding the benign pathology and histological features of SMH is of critical importance. As our case very well demonstrates successfully managed SMH, we believe that our high-resolution clinical and pathological images would provide guidance in properly recognizing and managing SMH.
Footnotes
Data Availability
All relevant data are included in the manuscript. The raw clinical and image data used and analyzed during the current study available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Inha University Hospital Research grant.
Ethical Approval
Inha University Hospital’s institutional review board (IRB) authorized this study (Investigation No.: 2022-08-050).
Patient Consent
The IRB waived the patient’s informed consent requirement because this research did not involve any personally identifiable information.
Permission to Reproduce Material From Other Sources
N/A.
