Abstract
Introduction
Oral cancer refers to the malignancies that occur in the oral cavity, including the lips, buccal mucosa, anterior tongue, floor of the mouth, hard palate, upper and lower gingiva, and retromolar trigone. Oral cancer holds the 16th position in malignancy in 2020 all over the world (177,757 deaths and 377,713 new cases), of which more than 95% are squamous cell carcinoma, with unacceptably high mortality rates.1,2 The treatment for oral squamous cell carcinoma (OSCC) is dominated by surgery and radiochemotherapy, which is in part associated with high morbidity.3,4 OSCC is characterized by its susceptibility to local recurrence and regional and distant metastasis, and the 5-year survival rate is only 50% to 60%. 5
OSCC is predominantly seen in elderly adults, following a long history of tobacco, alcohol, and betel nut exposure; in addition, accumulated evidences indicate a close relationship between oral microbiome (especially Porphyromonas gingivalis) and OSCC oncogenesis.6,7 While, among young individuals, the incidence of OSCC is relatively rare, representing generally less than 5% of all cases. 8 Furthermore, young adults with OSCC have specific multi-omic traits in molecular and immune landscape, for example, high detection rate of MMP-1 2 G allele and KIR2DL1+-HLA-C2+ genotype. 9 Approximately over six-tenths of OSCC cases are diagnosed at stages III–IV, which result in a poor prognosis and worse quality of life.5,10
Surgery plus postoperative radiation therapy is commonly employed in the treatment of OSCC patients, particularly for advanced cases. Some studies have confirmed the efficacy of this approach in optimizing locoregional control in this group of patients.11,12 However, few series report their reports on the long-term effects of postoperative radiotherapy in patients with OSCC. For this reason, we performed this present retrospective case-control study to evaluate the possible influence of postoperative radiotherapy on the prognostic outcome of OSCC in the US population.
Materials and Methods
Ethics
The Surveillance, Epidemiology, and End Results (SEER) cancer registries, as an open-access database, only contains de-identified data and has been deemed exempt from the Western Institutional Review Board (IRB) by a qualified expert as defined in Section §164.514(b)(1) of the Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule.
Study Design
To address the research purpose, the authors implemented a retrospective case-control investigation. The study population was composed of all patients admitting for management of OSCC registering in the SEER database (https://seer.cancer.gov/), screening occurred over a 6-year timespan (between 2010 and 2015) by using SEER*Stat software (v8.3.9) (National Cancer Institute, Bethesda, Maryland, USA).
Inclusion criteria were as follows: (i) pathologically confirmed OSCC (anatomical sites of oral cavity included the lips, buccal mucosa, anterior tongue, floor of the mouth, hard palate, upper and lower gingiva, and retromolar trigone); (ii) OSCC in stages I to IV (T1-4 and/or N+); (iii) diagnosis time from 2010 to 2015; (iv) treated with at least standard radical resection. Patients who met any of the following criteria were excluded: (i) any reasons for refusing radical surgical intervention and (ii) loss of follow-up after treatment.
The tumor-node-metastasis (TNM) cancer staging system and clinicopathological classification of eligible OSCC patients were compiled jointly using the American Joint Commission on Cancer and the Union for In-ternational Cancer Control guidelines. 13 Demographics and clinical materials encompassing age, gender, race, primary site, tissue differentiation, clinical stage, and TNM stage were collected. Overall survival (OS) rate and cancer-specific survival (CSS) rate were used to assess the prognosis of patients with OSCC. According to the treatment modalities, the enrolled subjects were divided into 2 groups: (i) Group A—only underwent radical surgery; (ii) Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
Statistical Analysis
The data were entered into an Excel spreadsheet. Data analysis was performed using R-based programming package version 4.3.0 (R Foundation for Statistical Computing, Vienna, Austria). Categorical variables were presented as frequencies and percentages, and comparisons between the 2 groups were analyzed using the Chi-square test. Kaplan–Meier method was used to plot the survival curves, calculating survival time and rate. A weighted log-rank test was proposed for comparing group differences of survival functions. The primary and secondary endpoint were set as OS and CSS rate, respectively. All Group A cases were matched one-to-one with Group B cases using the propensity score matching (PSM) approach. Cox proportional hazards regression analysis was used to identify multivariate risk factors of survival outcomes of patients with OSCC. P < .05 was considered to indicate statistically significant differences.
Results
Demographics and Clinicopathological Characteristics
A total of 7231 patients were included, of which 4278 were males (59.2%) and 2953 were females (40.8%). Group A was composed of 4167 cases, accounting for 57.6%. Group B comprised 3064 cases taking up 42.4%. Only M stage was not statistically significant (P = .419); other variables including age, gender, race, site, differentiation, clinical stage, T stage, N stage, and chemotherapy showed significant differences between the 2 groups (all P < .001; Table 1). The majority of patients in Group A were clinical stage I (n = 2569, 61.7%), whereas most patients in Group B were clinical stages III to IV (n = 2360, 77.1%).
Comparison of Clinical Pathologic Variables Between the 2 Groups [n (%)].
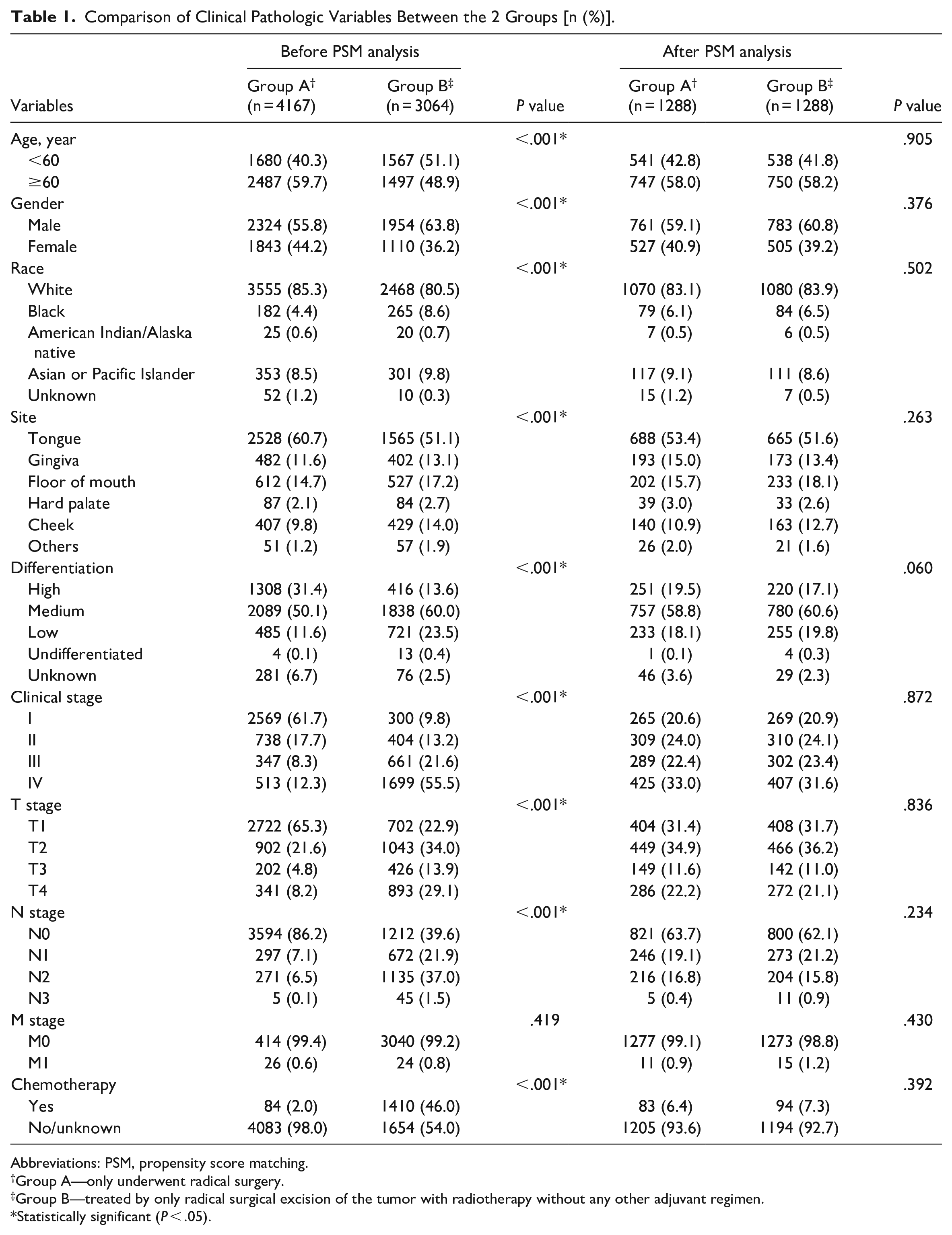
Abbreviations: PSM, propensity score matching.
Group A—only underwent radical surgery.
Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
Statistically significant (P < .05).
To eliminate the interference of confounding factors and determine the effects of treatments in this observational study, we performed PSM analysis. A total of 2576 patients were one-to-one matched successfully. After matching, each variable had similar distributions between 2 groups (all P > .05, Table 1). As for follow-up period, the median was 44 months (lower quartile (QL) = 26, upper quartile (QU) = 62) in Group A, and 43 months (lower quartile (QL) = 26, upper quartile (QU) = 63) in Group B, respectively.
Survival Outcomes
The median survival times of Group A was 24 months and that of Group B was 31.5 months. Group B had significantly higher 1-, 3-, and 5-year OS rates (88.1%, 76%, and 69.7%, respectively), compared with those of Group A (81%, 58.3%, and 51.3%, respectively; P < .0001, Figure 1A and Table 2).
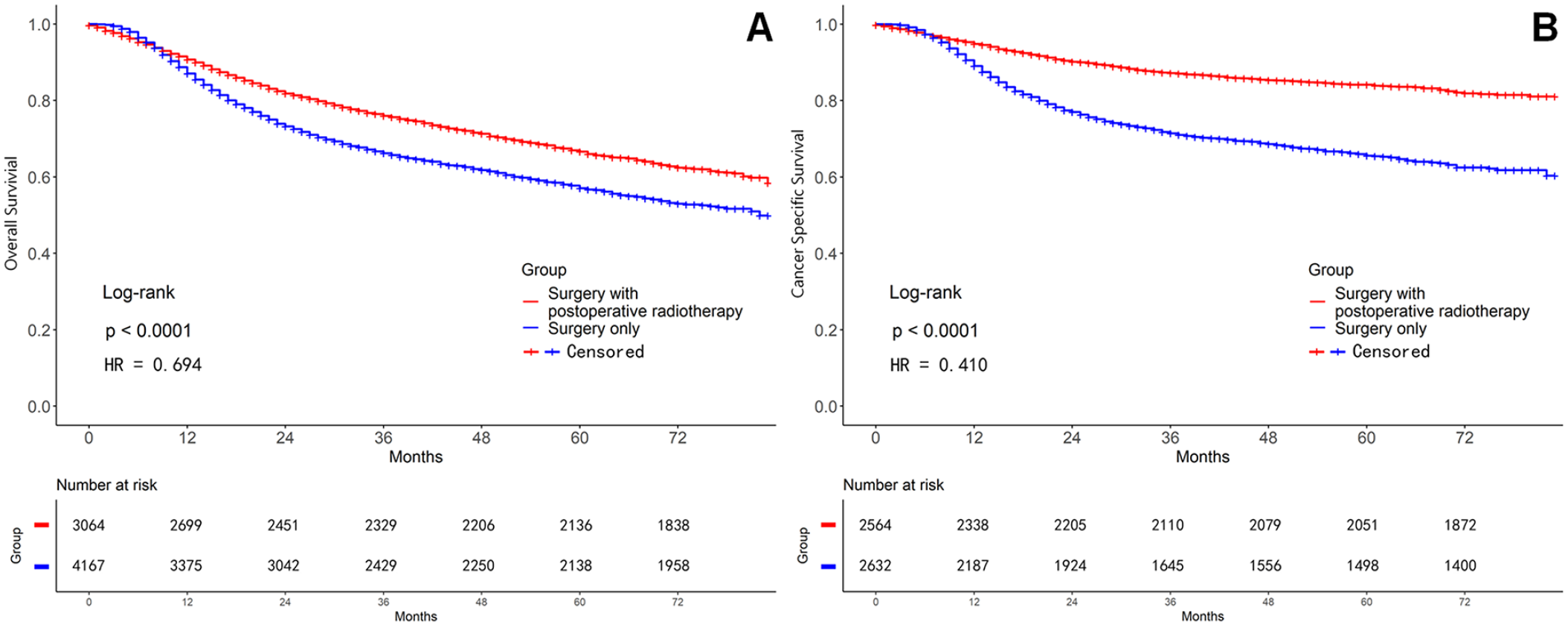
Comparison of survival curves before propensity score matching (PSM) analysis between the 2 groups. (A) Overall survival (OS) curve. (B) Cancer-specific survival (CSS) curve.
Survival Outcomes of 1, 3, and 5 Years Between the 2 Groups.
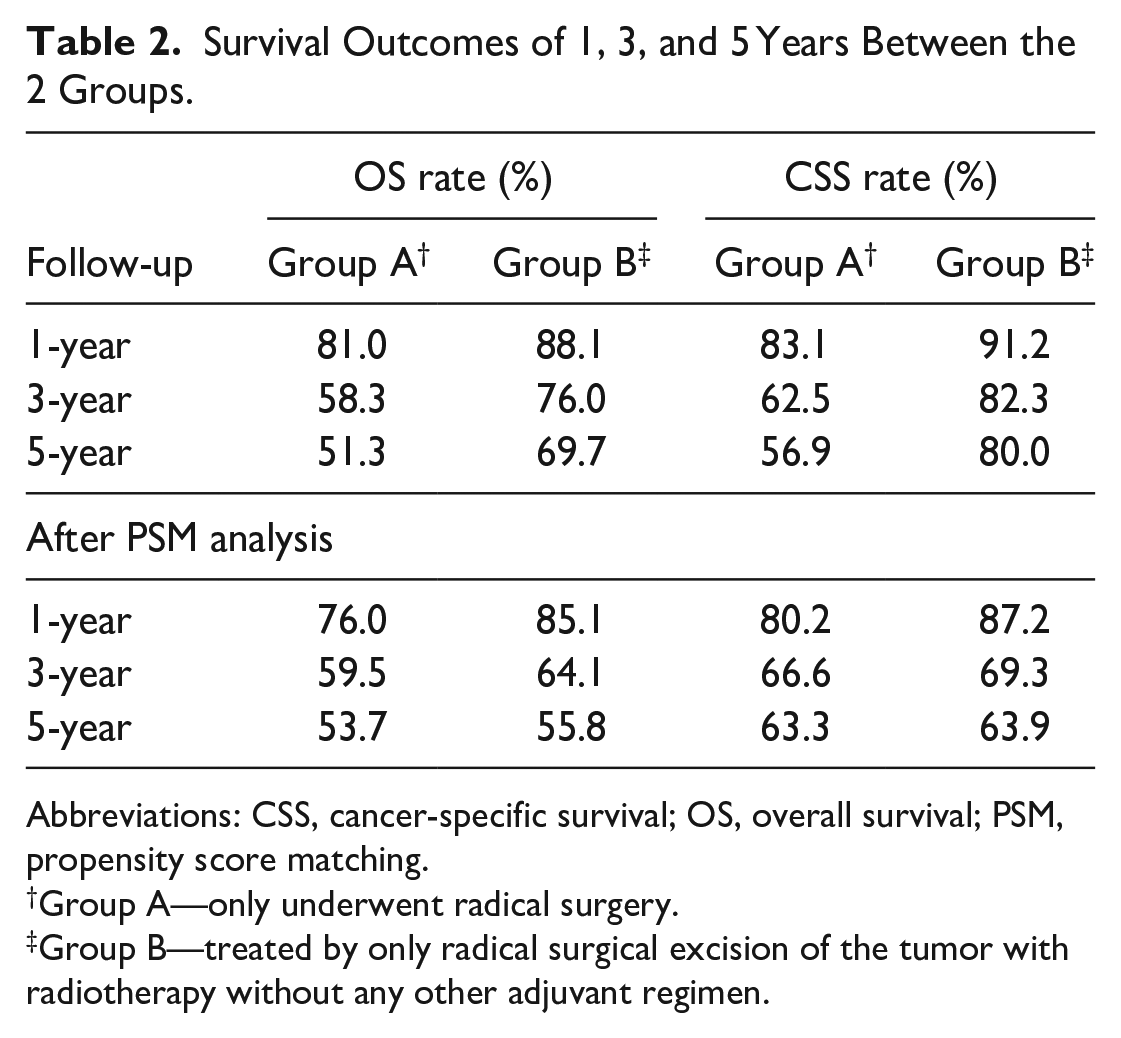
Abbreviations: CSS, cancer-specific survival; OS, overall survival; PSM, propensity score matching.
Group A—only underwent radical surgery.
Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
The 1-, 3-, and 5-year CSS rates of Group B (91.2%, 82.3%, and 80%, respectively) were significantly higher than those of Group A (83.1%, 62.5%, and 56.9%, respectively; P < .0001, Figure 1B and Table 2).
After PSM adjustment, in Group A, the 1-, 3-, and 5-year OS rates (76%, 59.5%, and 53.7%, respectively) were significantly lower than those of Group B (85.1%, 64.1%, and 55.8%, respectively; P < .0001, Figure 2A and Table 2). Similarly, the 1-, 3-, and 5-year CSS rates of Group A (80.2%, 66.6%, and 63.3%, respectively) were significantly lower than those of Group B (87.2%, 69.3%, and 63.9%, respectively; P < .0001, Figure 2B and Table 2). The survival analysis on the basis of different clinical stages had the results as following. In general, the 1-, 3-, and 5-year OS rates of Group A were lower than those of Group B, respectively. The difference was statistically significant in clinical stages I, III, and IV (all P < .0001, Figure 3 and Table 3); but no statistical significance existed in stage II (P = .269). Group B had significantly higher 1-, 3-, and 5-year CSS rates than those in Group A. Except for stage III (P = .971), stages I, II, and IV all had statistically significant difference (all P < .0001, Figure 4 and Table 3). As the clinical stage evolved, the harm of the tumor increased, and both OS and CSS rates decreased.
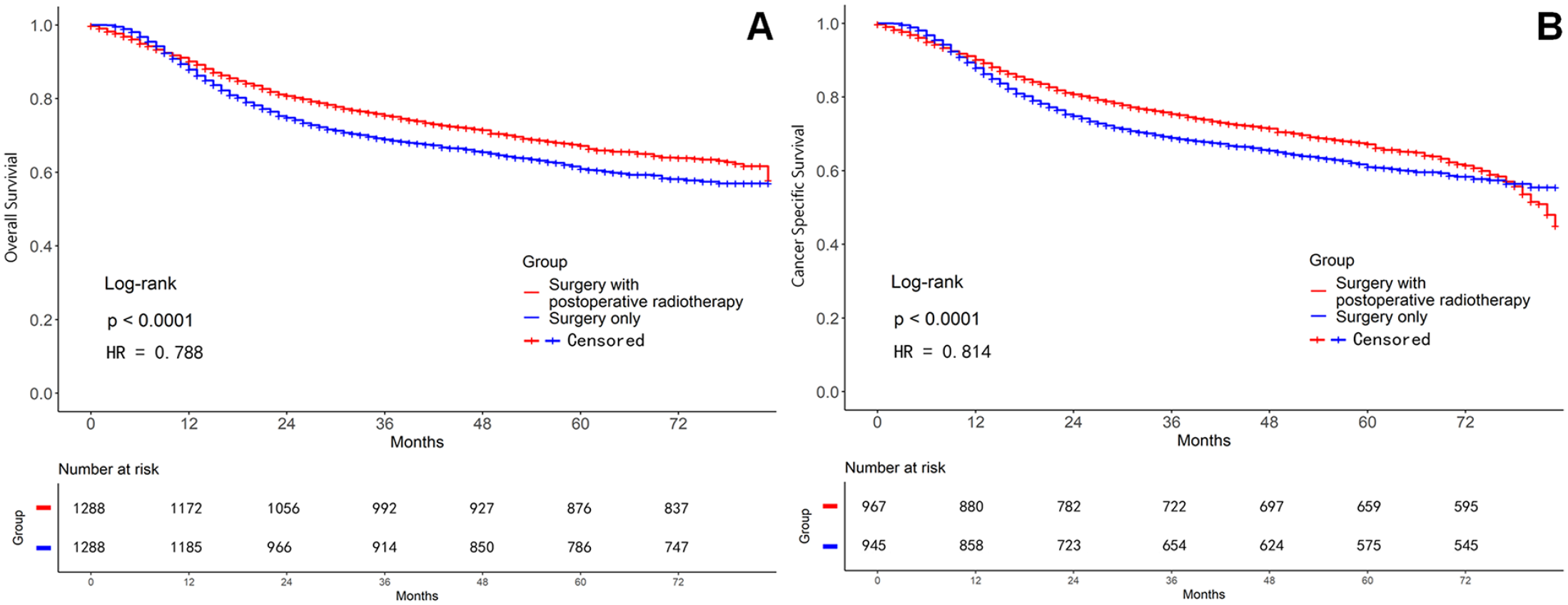
Comparison of survival curves after PSM analysis between the 2 groups. (A) OS curve. (B) CSS curve.
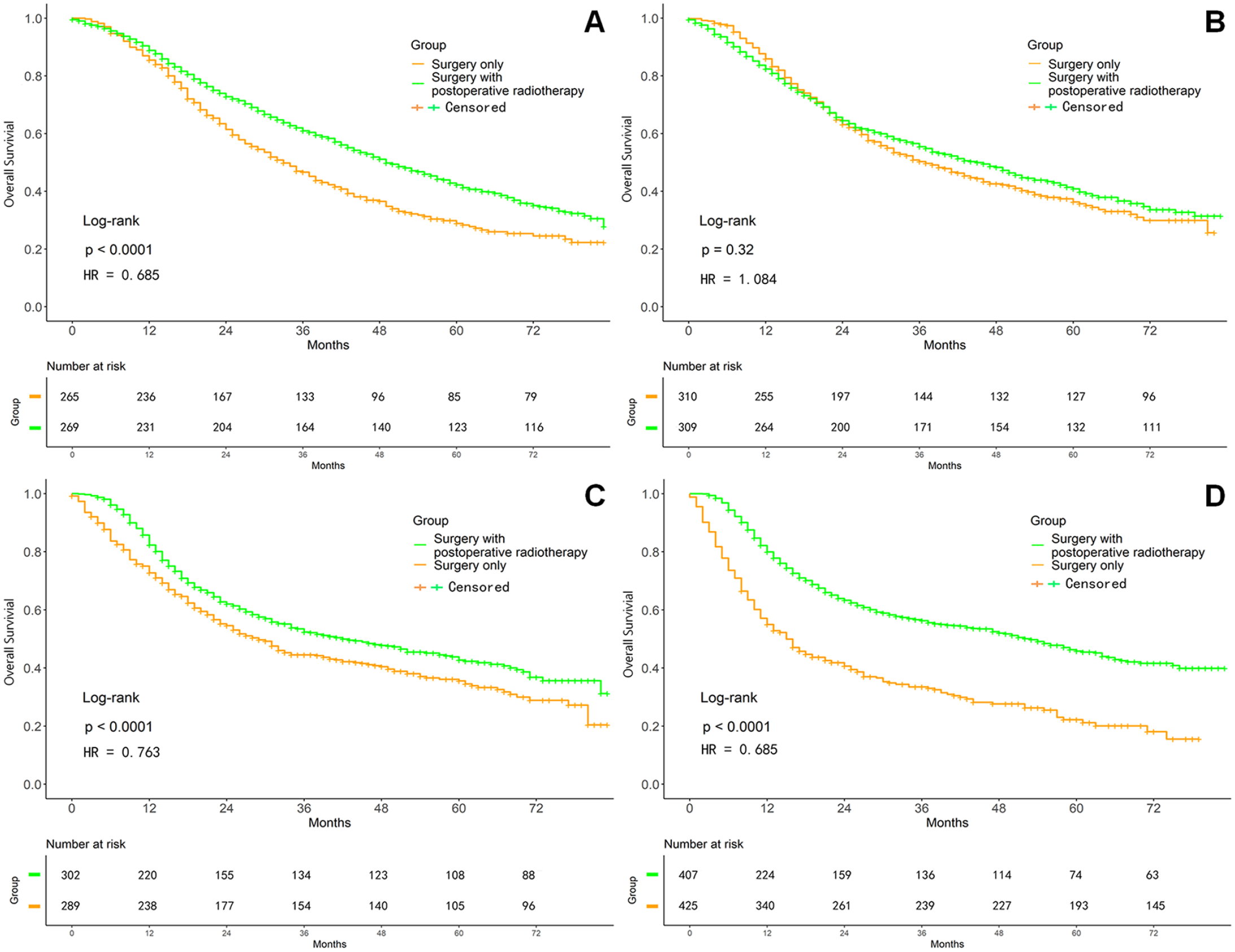
Survival curves on OS after PSM adjustment in different clinical stages. (A) Stage I. (B) Stage II. (C) Stage III. (D) Stage IV.
After PSM Adjustment, One-, Three-, and Five-Year Survival Outcomes Regarding Different Clinical Stages Between the 2 Groups.
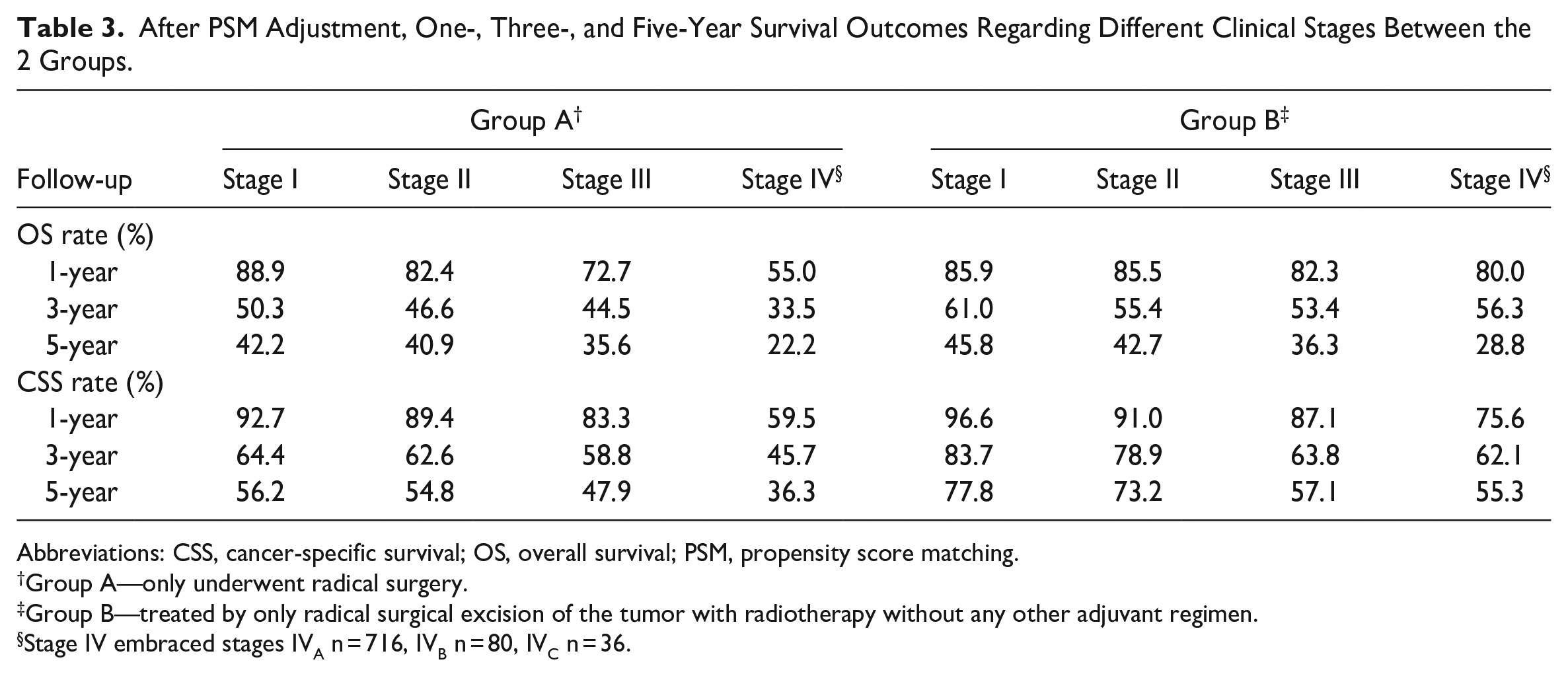
Abbreviations: CSS, cancer-specific survival; OS, overall survival; PSM, propensity score matching.
Group A—only underwent radical surgery.
Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
Stage IV embraced stages IVA n = 716, IVB n = 80, IVC n = 36.
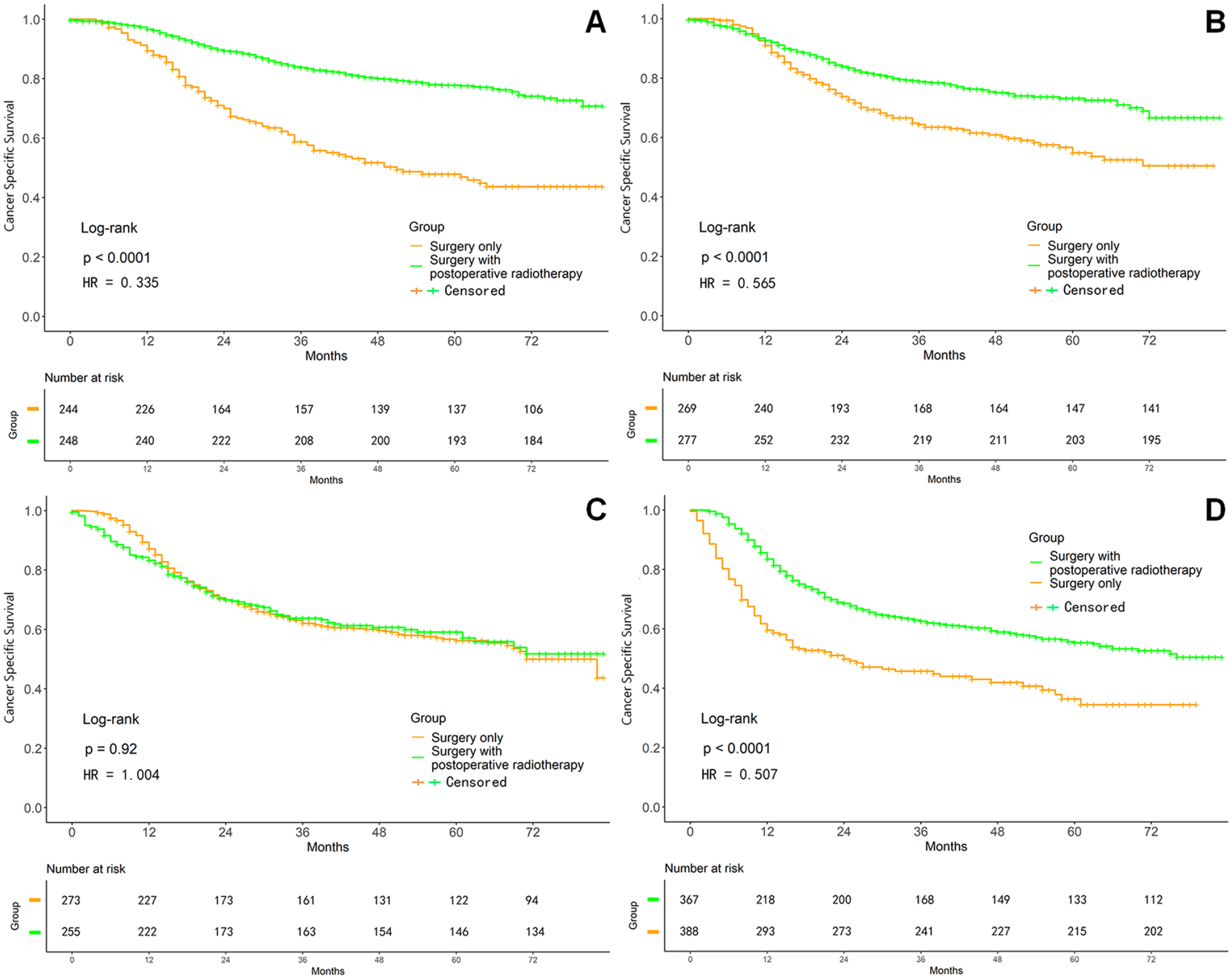
Survival curves on CSS after PSM adjustment in different clinical stages. (A) Stage I. (B) Stage II. (C) Stage III. (D) Stage IV.
Univariate Analysis of Clinicopathological Factors Influencing Survival Following OSCC Treatment
After PSM analysis, the Cox univariate proportional hazards regression showed that age, differentiation, clinical stage, T stage, N stage, M stage, and chemotherapy had significant difference (all P < .05; Table 4). Consequently, these variables were chosen as covariants into the next multivariate Cox analysis.
Univariate Cox Proportional Hazards Regression Analysis for Screening Predictive Factors.
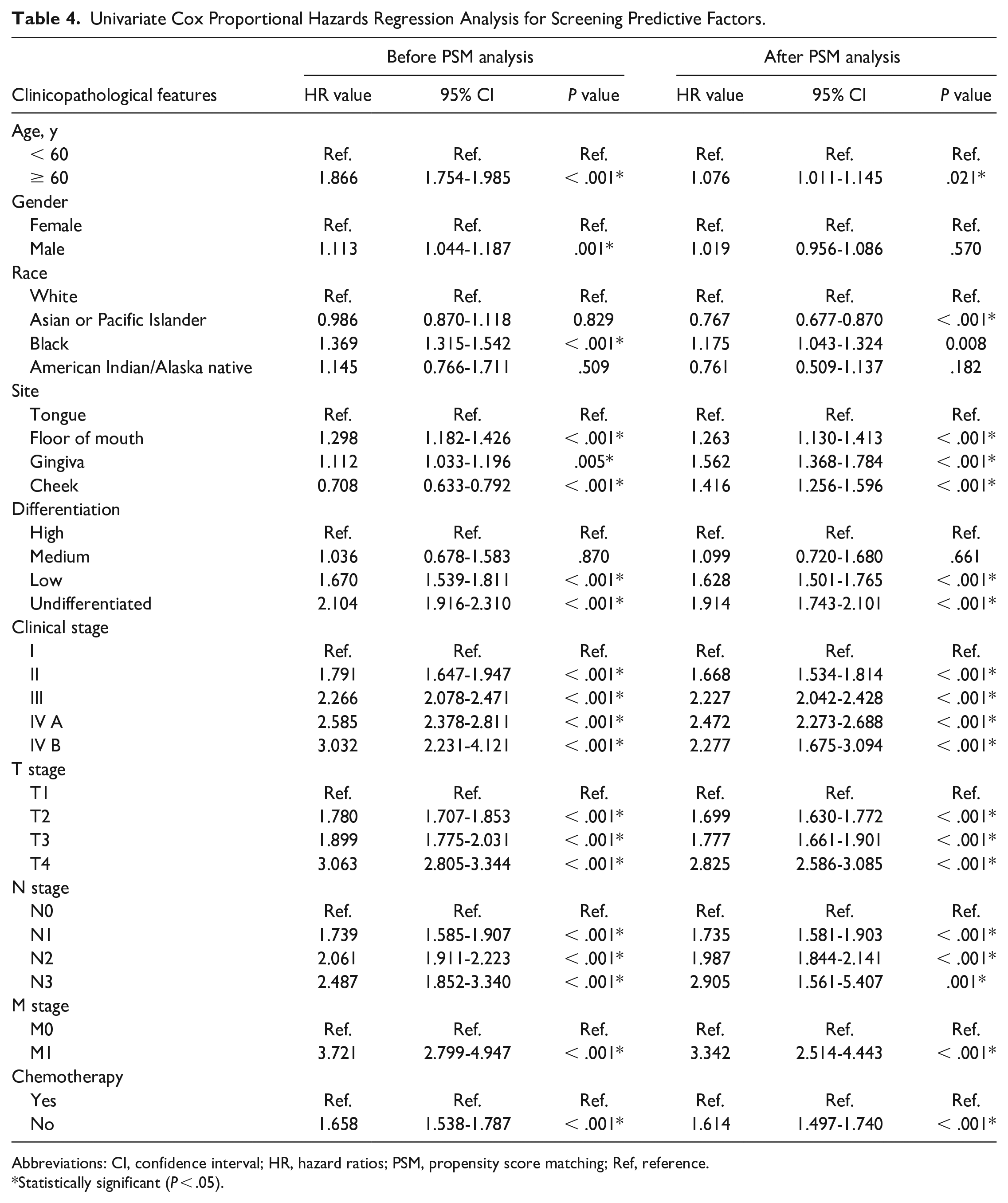
Abbreviations: CI, confidence interval; HR, hazard ratios; PSM, propensity score matching; Ref, reference.
Statistically significant (P < .05).
Multivariate Analysis of Clinicopathological Factors Influencing Survival Following OSCC Treatment
The Cox multivariate analysis showed that age, differentiation, clinical stage, T stage, N stage, and M stage affected the prognosis of patients with OSCC, while postoperative radiotherapy was a protective factor [OS: hazard ratios (HR) = 0.649 (95% confidence interval (CI) 0.580-0.727), P < .001; CSS: HR = 0.702 (95% CI 0.618-0.799), P < .001] (Table 5).
Multivariate Cox Proportional Hazards Regression Analysis for Screening Predictive Factors.
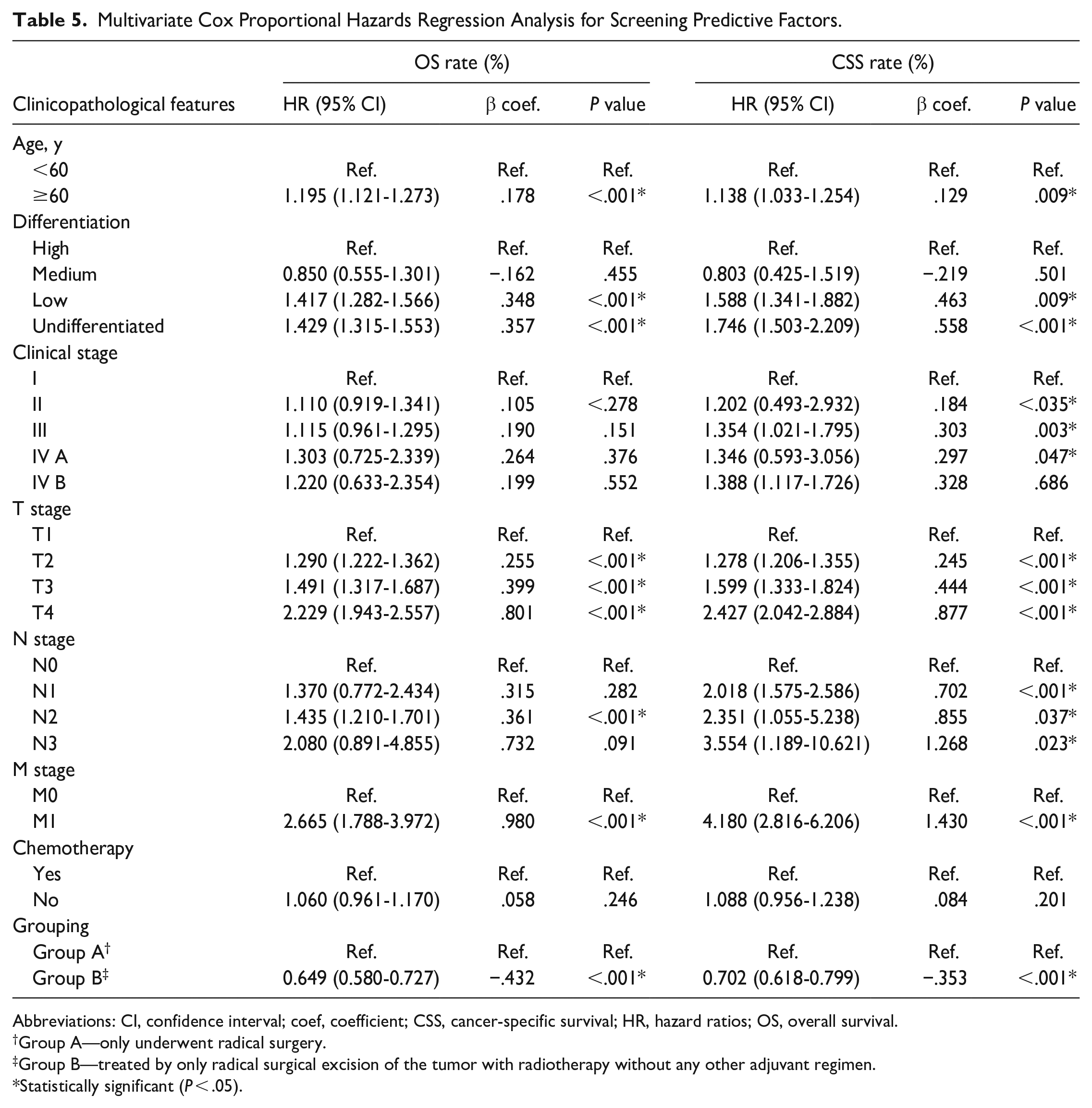
Abbreviations: CI, confidence interval; coef, coefficient; CSS, cancer-specific survival; HR, hazard ratios; OS, overall survival.
Group A—only underwent radical surgery.
Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
Statistically significant (P < .05).
Based on different clinical staging of OSCC, the subgroup analyses were accordingly conducted. In early-onset OSCC (stage I and II) patients, age, differentiation, clinical stage, T stage, and postoperative radiotherapy had significant impact on OS and CSS (all P < .05, Table 6). In advanced OSCC (stage III and IV) patients, age, differentiation, T stage, N stage, M stage, and postoperative radiotherapy had obvious influence on OS and CSS of OSCC patients (all P < .05, Table 6).
Subgroup Analysis on the Basis of Clinical Staging of OSCC.
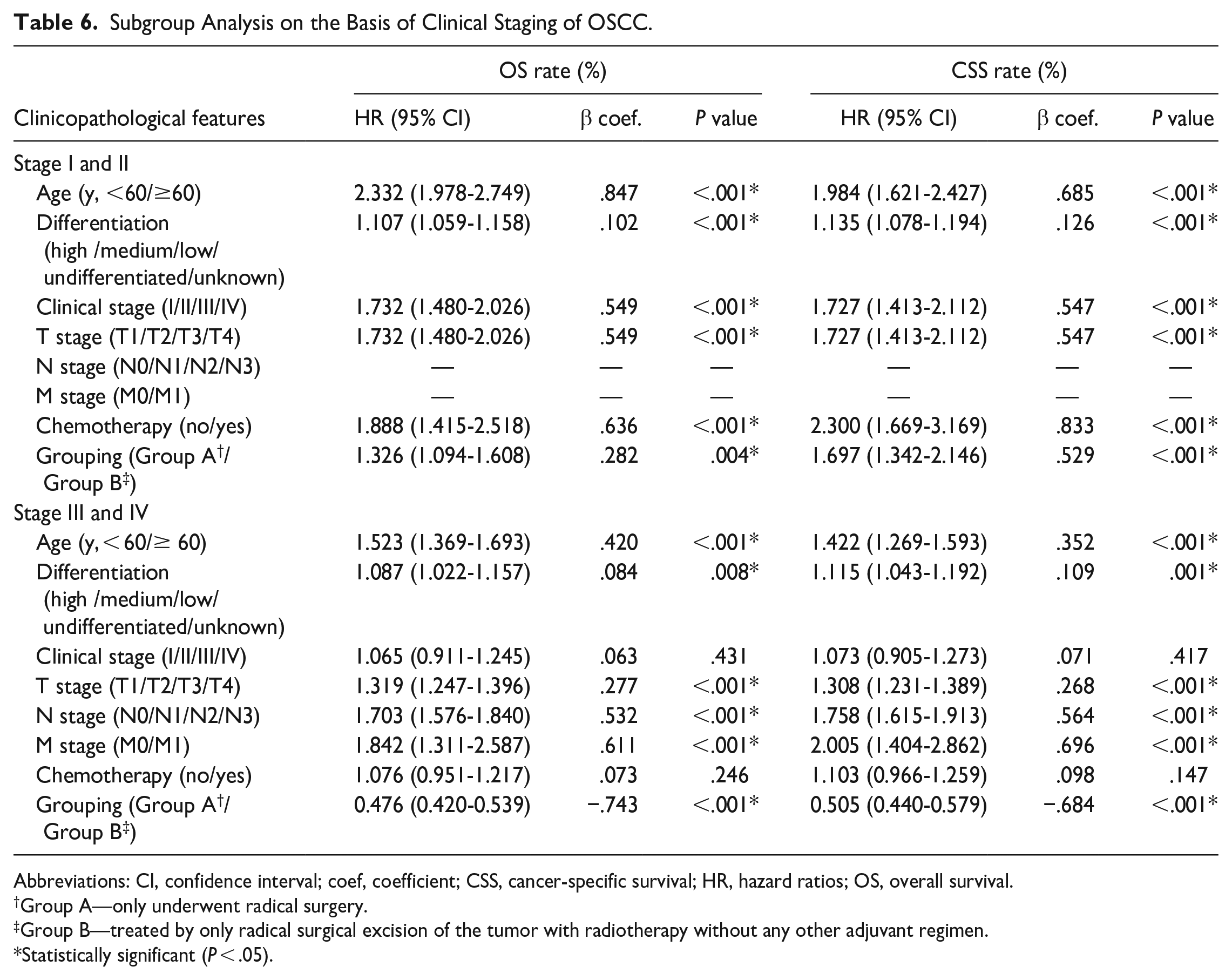
Abbreviations: CI, confidence interval; coef, coefficient; CSS, cancer-specific survival; HR, hazard ratios; OS, overall survival.
Group A—only underwent radical surgery.
Group B—treated by only radical surgical excision of the tumor with radiotherapy without any other adjuvant regimen.
Statistically significant (P < .05).
Discussion
OSCC is the most common type of oral malignancy that usually originates from the precancerous lesion of oral mucosa, and metastasis accounts for its poor prognosis. It has a profound effect on human health and quality of life due to the high morbidity and mortality. Recent advances have brought substantial progress in prognostic outcomes, despite the fact that OSCC is associated with disfiguration, psychosocial distress, dysfunction, and death. Advances in standard therapy, such as organ-sparing, minimally invasive surgical techniques, improvements in radiation therapy, and curative multimodal approaches, have enhanced the long-term functional and structural preservation. The principal treatment for OSCC is generally determined based on tumor stage and pathological diagnosis, with surgical resection remaining the mainstay of comprehensive treatment. Treatment strategies must be chosen carefully as well as individually tailored to the patient’s needs to optimize the quality of life and survival of patient. 14 Our SEER-based study is the first to stratify the prognosis of newly diagnosed OSCC and screen the patients benefiting from postoperative radiotherapy. The findings of multivariate Cox proportional hazards regression and PSM analyses showed that radiotherapy remarkably ameliorated both OS and CSS in the entire group of patients [OS: HR = 0.649 (95% CI: 0.580-0.727), P < .001; CSS: HR = 0.702 (95% CI: 0.618-0.799), P < .001]. The Kaplan–Meier curves also evidently illustrated higher 1-, 3-, and 5-year OS rates as well as CSS rates in patients treated by surgery combined with postoperative radiotherapy than that in patients treated with surgery alone (all P < .0001).
Stage I to II OSCC is traditionally treated with surgery alone, especially since progress in surgical excision and microvascular reconstruction has evolved. 15 According to National Comprehensive Cancer Network and Chinese Society of Clinical Oncology guidelines, early-stage (T1-2, N0) can be treated effectively through radical resection or local radiation when surgery is not suitable. But in our investigation, the early-onset OSCC cases amounted in aggregate to 23% (stage I, 9.8%; stage II, 13.2%) who were treated with surgery plus postoperative radiotherapy. This “irregular” phenomenon might be inextricably linked with pathologic degree of tumor differentiation of OSCC. Low-grade or undifferentiated OSCC hints higher malignancy with a stronger proliferative ability showing significantly increased Ki-67 immunochemical staining. 16 These patients presenting rapid progression in a short-run may be better necessary to adjuvant postoperative radiation. The depth of tumor invasion, as one risk factor for early-stage OSCC, is considered to be related with increased risk of metastasis to the neck and decreased OS in previous studies.17-21 However, Rubin et al. 15 demonstrated that irrespective of the depth of tumor invasion, an OS raise was not found in T2N0 patients received regardless of surgery alone or postoperative radiation therapy. In this present study, we confirmed that age, tumor differentiation grade, clinical stage, T stage, and postoperative radiotherapy correlated significantly with the OS and CSS of stage I and II OSCC patients (all P < .05).
Clinically, a large number of OSCC patients are diagnosed at late stage. 22 The ideal treatment for patients with advanced-stage OSCC at diagnosis remains controversial. 23 Thorough and systemic therapy is of great importance because patients at advanced stage frequently experience higher recurrence rate and poor survival.24,25 Unfortunately, 20.6% patients with advanced OSCC (stage III, 8.3%; stage IV, 12.3%) received surgery only. The irregularity could depend on the socioeconomic status. These patients’ income level could be too low to afford the medical expenditure covering the comprehensive and sequential therapy. More importantly, some surgeons found that many patients with locally advanced OSCC without postoperative radiotherapy also have a good prognosis.26,27 In their opinions, postoperative radiotherapy may not be necessarily needed to achieve a good prognosis under the premise of high-quality surgical resection of the tumor. Alabi et al. 23 discovered that patients with surgery combined with postoperative radiotherapy showed better OS than that with postoperative chemoradiotherapy or surgical treatment alone. Our study revealed, in patients with stage III and IV OSCC, age, tumor differentiation grade, T stage, N stage, M stage, and postoperative radiotherapy had significant impact on their OS and CSS (all P < .05); but the role of adjuvant chemoradiation showed no statistically significant difference (Pos = .246; Pcss = .098).
Some limitations of this study should not be ignored. First, subjects in the study all come from the United States. It would be better to include as many research subjects as possible, such as the International Head and Neck Cancer Epidemiology Consortium database. Second, presence of genetic mutations and oral bacteria as potential risk factors associated with developing OSCC could not be avoided. Third, it is noted that analytical approach for young patients with OSCC were not performed due to too many censored data in follow-up. 28 Finally, prospective controlled clinical trials are needed to further confirm our findings and to assert a cause-effect relationship between different treatment modalities and survival outcomes based on a multicentric survey in the future.
In conclusion, both 1-, 3-, and 5-year OS and CSS rates among patients treated with radical excision of the tumor with adjuvant postoperative radiotherapy have significantly improved than that in patients treated with surgery alone. Our population-based study showed that the prognosis of patients with OSCC is affected by many factors, including age, tumor differentiation grade, clinical stage, T stage, N stage, and M stage, and postoperative radiotherapy is a particularly important protective factor for OSCC patients. Notably, the understanding of survival prognosis in OSCC is crucial for the planning of patient counseling and personalized care. The onus is on the clinicians to make insightful conclusions regarding the best treatment for the patients.
Footnotes
Acknowledgements
We would like to express our cordial appreciation to SEER databank for the permission to use its open-access data to conduct our clinical study. We also thank Prof. Hua-rong Zhao (Department of Radiotherapy and Oncology, Cancer Center, Xinjiang Medical University Affiliated First Hospital, Urumqi 830054, China), Dr. Jia-lin Sun (School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China), and Cheng Li (MSc, MPH. Statistical Analysis Center, the First Affiliated Hospital of Xinjiang Medical University. Urumqi 830054, China) for their professional assistance to our work.
Authors’ Contributions
C-X.L. conceptualized the study and guaranteed the integrity of the whole procedure. Z-Y.W. and Q-Y.T. were incharge of double-checking the original sheets and preparing tables and figures. C-X.L., M-Q.L. and W.W. organized the acquired data and performed the statistical analysis. Z-C.G. critically revised the article for important intellectual content. Z-C.G. and C-X. L. acquired financial support. C-X.L. was responsible for methodology and drafting, revising, and editing the manuscript. All authors approved the final article as submitted and agree to be accountable for all aspects of the work. The requirements for authorship as stated earlier in this document have been met, and each author believes that the article represents honest work.
Availability of Data and Materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All phases of this study was supported by National Natural Science Foundation of China (grant number: 82360481); Open Project of Hubei Province Key Laboratory of Oral and Maxillofacial De-velopment and Regeneration (grant number: 2022kqhm008); and Xinjiang Postgraduate Scientific Research Innovation Project (grant number: XJ2023G174).
Ethics Approval and Consent to Participate
The SEER cancer registries, as an open-access database, only contain de-identified data and have been deemed exempt from the Western Institutional Review Board (IRB) by a qualified expert as defined in Section §164.514(b)(1) of the HIPAA Privacy Rule.
Consent for Publication
Not applicable.
