Abstract
Myiasis is the invasion of mammalian tissues by dipterous larvae. Cutaneous myiasis is the commonest type, and less commonly, internal tissues and organs are affected. Here, we report the first case of oro-nasopharyngeal myiasis caused by the third-instar larvae of Chrysomya bezziana (Old-World screwworm) in Sri Lanka. A retired 71-year-old female tea-plucker presented to the hospital with a 4-day history of nasal bleeding and neck discomfort. Except for well-controlled hypertension, she had no significant medical history. On examination, there was an ulcerated area in the posterior oro-nasopharynx filled with maggots. Hematological analysis showed a high C-reactive protein level (24 mg/dL) and white blood cell count (17.5 × 109/L) with 80% neutrophils. Computed tomography showed severe inflammation of the pharynx with no features of a neoplasm or parapharyngeal abscess. An examination was performed under anesthesia followed by manual removal of larvae, surgical debridement with biopsy, and treatment with antiparasitic medicines and antibiotics. Histopathology excluded neoplasm and only showed inflammatory changes. All extracted larvae were morphologically similar and identified as third-instar larvae of C. bezziana using a trinocular zoom stereomicroscope. The patient recovered and was discharged from the hospital on the 15th day and was well at 2 months follow-up.
Introduction
Myiasis is the invasion of mammalian tissues by dipterous larvae. For some species of flies, this is an essential part of their life cycle (obligatory myiasis), while for carrion breeding larvae it is an optional host–parasite relationship (facultative myiasis). Meanwhile, myiasis also occurs when the eggs or larvae of the fly are ingested inadvertently and not killed (accidental myiasis). In humans, myiasis can affect the skin, body cavities, and organs of the body (intestinal, urogenital). Predisposing factors for myiasis are overcrowded conditions, low socioeconomic backgrounds, rural settings, poor sanitary conditions, poor personal hygiene, immunosuppressed status, chronic diseases such as diabetes mellitus, open wounds, infections, debility, tube feeding, cancer, use of immunosuppressive agents, psychiatric illness, intellectual disability, and hemiplegia. 1
Case Report
A 71-year-old woman from a rural suburb of Nuwara Eliya, central province in Sri Lanka presented with a 4-day history of nasal bleeding and neck discomfort. She was a diagnosed patient with hypertension for more than 15 years with good control. The patient had no history of diabetes mellitus or malignancy and denied any clinical symptoms of immune deficiency. There was no history of craniofacial trauma, head and neck surgery, psychiatric illness, or mental subnormality. She had been a retired tea-plucker from a low-middle socioeconomic background.
On examination, the patient had a temperature of 37.4°C, a heart rate of 100 beats per minute, and a blood pressure of 140/90 mmHg. Her respiratory parameters were normal. There was an unhealthy ulcerated area of 3 cm × 3 cm in the posterior oro-nasopharynx filled with maggots (Figure 1A). As there was persistent epistaxis, an anterior nasal packing was performed at the emergency treatment unit.
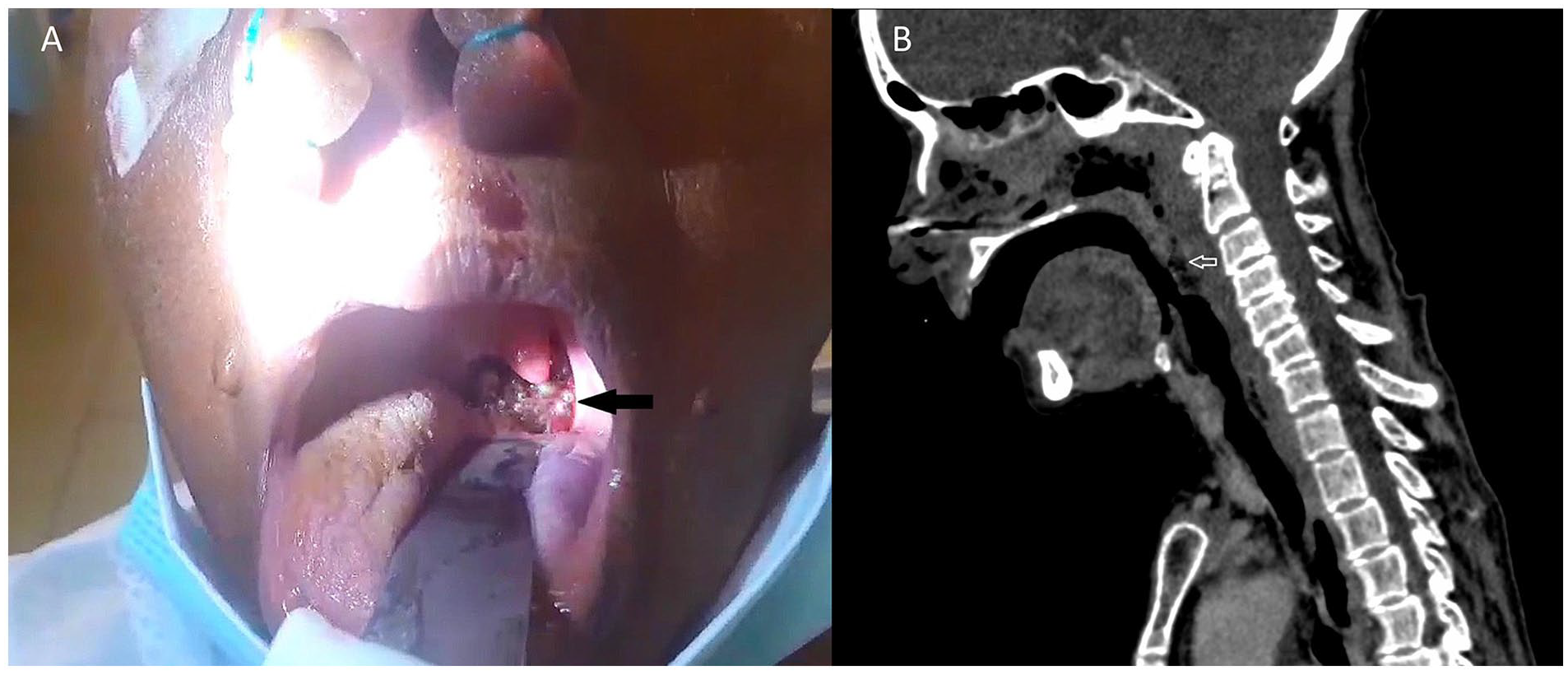
(A) Ulcerated area in the posterior oro-nasopharynx with maggots. (B) Computed tomography (CT) of the neck shows inflammation of the pharynx. There are no features of a neoplasm or parapharyngeal abscess.
The patient had a white blood cell count of 17.5 × 109/L with 80% neutrophils, 16% lymphocytes, 4% eosinophils, hemoglobin of 11 g/dL, and platelet count of 275 × 109/L. Her C-reactive protein (CPR) level was 24 mg/dL with normal renal and liver biochemistry. She was empirically started on an intravenous co-amoxiclav of 1.2 g 8 hourly. The patient was subjected to computed tomography (CT) of the neck, which showed severe inflammation of the pharynx with no definitive neoplasm or a parapharyngeal abscess formation (Figure 1B). Her HIV screening was negative.
During the next 72 hours, the patient underwent 2 examination sessions of the pharynx under general anesthesia. It was a predicted difficult intubation with a considerable risk of maggot aspiration into the trachea. All precautions were taken to prepare for difficult intubation and ventilation. The patient was induced with rapid sequence induction with cricoid pressure to prevent aspiration. More than 50 maggots were removed manually. Multiple biopsies were taken from the edge and base of the ulcer for histological evaluation. Fibro-optic nasal endoscopy showed maggot infestation in the paranasal sinuses too. In addition to the initial antibiotics, the patient was treated with a stat dose of ivermectin 12 mg followed by albendazole 400 mg daily for 5 days. Due to severe pharyngeal edema, the patient was ventilated for 4 days. She made a slow but steady recovery over the next 2 weeks, leaving her to be discharged on the 15th day after admission. Two months after discharge, the pharyngeal ulcer had completely healed, and she was back to her normal routine. The histology of the ulcer biopsy only showed inflammatory changes.
The maggots extracted were sent in normal saline and 70% ethanol to the Department of Parasitology of the Faculty of Medicine of the University of Peradeniya for identification. Larvae were examined using a trinocular zoom stereomicroscope (Model-SZN-6, Optika, Italy). The thoracic and abdominal segments and the anterior and posterior ends of larvae were photographed for identification (Figure 2). Based on the morphological characteristics observed during the macroscopic examination, the larvae were identified as third instars. They had non-hairy whitish-to-cream-colored bodies. Each third-instar larva was approximately 12 mm in length (Figure 2A). It had a longitudinal worm-shaped body with 12 visible segments, including the head (Figure 2B and C), thoracic (Figure 2E), and abdominal segments (Figure 2F). There were prominent bands of sclerotized thorn-like spines between the segments (Figure 2E and F). The cephalopharyngeal skeleton had large, sclerotized mouth hooks, curved downward (Figure 2B and C). The anterior spiracle consisted of 6 lobes arranged in a single row (Figure 2D). There were prominent bands of sclerotized thorn-shaped spines between the segments at the posterior end (Figure 3A). The posterior spiracles consisted of ventrally incomplete, deep brown-to-blackish posterior peritremes that enclosed 3 distinct spiracular slits (Figure 3B-D).

Anterior end of the third-instar larva of Chrysomya bezziana. (A) The third-instar larva is approximately 14 mm in length. (B) Large and sclerotized mouth hooks (mh), which are curved downward. (C) Showing antenna (a), maxillary pulp (mp), oral groove (og), and mouth hooks. (D) Showing anterior spiracle with 6 lobes arranged in a single row (as). (E) Thoracic segments show rows of thorn-shaped spines (sp). (F) Abdominal segments showing rows of thorn-like spines (sp).
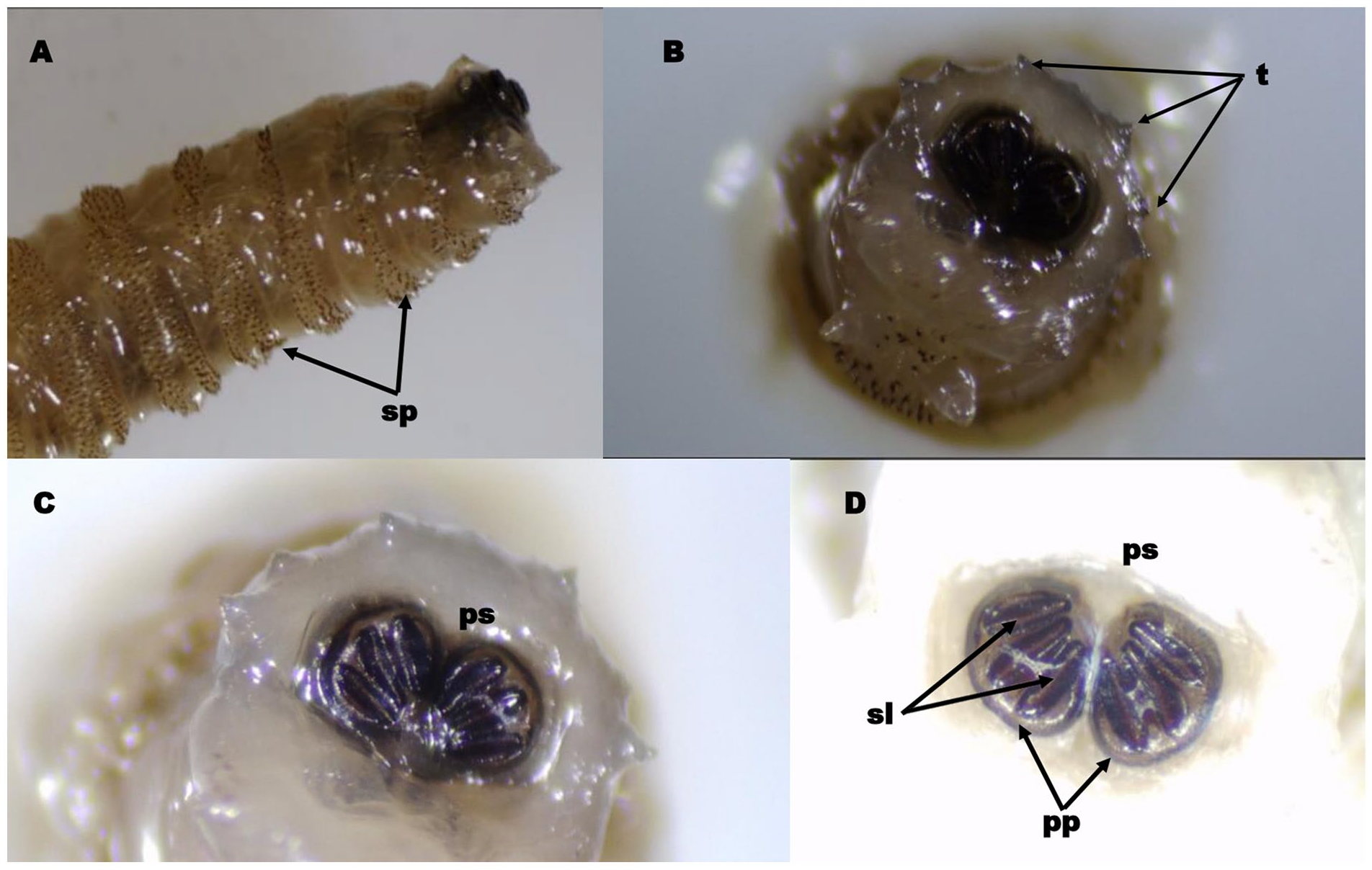
Posterior end of the third-instar larva of Chrysomya bezziana. (A) The posterior end of the third-instar larva shows sclerotized thorn-like spines (sp). (B) Posterior view of the last abdominal segment showing prominent tubercles (t) along the peripheral rim. (C, D) Showing posterior spiracles (ps) consist of ventrally incomplete deep brown-to-blackish posterior peritremes (pp) enclosing 3 distinct spiracular slits (sl).
Based on morphological characteristics, the larvae were identified as species of Chrysomya bezziana. Microscopic images were sent to the Parasitic Disease Branch, Diagnostics and Biology Team, Division of Parasitic Diseases and Malaria, Centers for Disease Control and Prevention, USA (http://www.cdc.gov) who confirmed species identification.
Discussion
The first case of Chrysomya bezziana myiasis in humans in the world was reported in India in 1909. 2 The first report of myiasis from Sri Lanka was a case of nasal myiasis by C. bezziana in 1954. 3 In a review of cutaneous myiasis cases (n = 18) from the Colombo district in 2000, reported that 16 cases were due to C. bezziana and 2 due to C. megacephala. 4 In a review of cases (n = 28) from the Central Province of Sri Lanka (same area as this case) in 2019, the authors reported that the majority (n = 26) were cutaneous and the rest (n = 2) were urogenital myiasis. 5 Of the total of 28 cases, 27 were caused by the Old-World screwworm, C. bezziana Villeneuve (Diptera: Calliphoridae), and the remaining 1 by the blowfly, C. megacephala Fabricius (Diptera: Calliphoridae). 5 In a study among animals in Sri Lanka in 2016, C. bezziana was identified as the causative agent in all 299 cases. 6
C. bezziana which causes nasopharyngeal myiasis has been reported in other countries. 7 However, no cases of pharyngeal myiasis from Sri Lanka have been published. This is the first case of oro-nasopharyngeal myiasis reported from Sri Lanka.
C. bezziana is present in 63 countries in the world. 8 Of all human myiasis cases caused by C. bezziana, Asia contributes 94.5% (China 36%, India 36%, Sri Lanka 15.6%). 1 The number of myiasis cases in Sri Lanka is increasing (02: before 1968; 14:1989-1998; 27:2009-2019; 0 reports in gap years) due to an increase in incidence, detection, or reporting or all 3 reasons. 1
Infection, open wounds, bedridden/debilitated/wheelchair-bound, multiple underlying diseases, tube feeding, diabetes, cancer, dementia, stroke, and so on, were considered as associated conditions in C. bezziana myiasis cases. 1 Advanced age, poverty, low socioeconomic background, poor oral hygiene, mouth breathing, severe halitosis, history of craniomaxillofacial trauma, cancer, neurological conditions, debilitating conditions, neglected fractures, living close to animals, alcoholism, and patients with mechanical ventilation were reported as risk factors associated with oral myiasis. 9 In a review of 10 years of experience with 252 cases of nasal myiasis in India, atrophic rhinitis was present in more than 97%. 10
In a systematic review of 199 global cases of C. bezziana myiasis, there was no gender preponderance; most were in the over-65 age group, and the mouth was the most common site among the over-65 age category, followed by the limbs. 1 Furthermore, the most common clinical signs and symptoms reported were visibility of the larvae, swelling, ulcer/perforations/tunnels, bleeding, pain, discharge, fever, necrosis, bone or tissue destruction, foul smell, pruritis, and inflammation. 1
In the case of myiasis in and around the oral, nasal, and pharynx area, oviposition is likely to occur while the patient is asleep. 11 In the world literature, among the cases of myiasis in the region of the ear, nose, and throat pharyngeal myiasis represents the least burden (2.5%). 12 Oestrus ovis (sheep botfly) is referred to as the most common fly that causes throat and nasomyiasis and is often seen among people in close contact with sheep and goats.11,13 In Sri Lanka, there is 1 report of a case of an external ophthalmomyiasis by O. ovis 14 . Furthermore, many other fly species also cause nasomyiasis such as Sarcophaga species, 15 Drosophila melanogaster (fruit fly), 16 C. albiceps and C. rufifacies, 11 Calliphora erythrocephala, 17 Cochliomyia hominivorax, C. bezziana, O. ovis, Wohlfahrtia magnifica, Lucilia sericata, Drosophila melanogaster, C. vicina, 11 and Eristalis tenax. 18
The genus Chrysomya in the order Diptera, family Calliphoridae, suborder Cyclorrhapha, consists of 12 species, including C. bezziana. 19 Chrysomya adult flies are metallic bluish green in color. Adult females lay about 150-250 eggs that hatch in 24 hours, releasing larvae. With their mouth hooks, Chrysomya larvae dig into living tissue in a screw-like manner (reason for the common name: screwworm fly), while attaching to the tissue with intersegmental spines. Within the next 5-7 days, the larvae go through 3 developmental changes (3 instar stages) and fall to the ground and pupate. Subsequently, the adult is released from the pupa. The number of days taken depends on the temperature of the environment. 1
Entomological examination for characteristic features is the gold standard for identification. 1
In the management of invasive forms of myiasis, the manual removal of larvae is performed as an initial step. The larvae display negative phototaxis and thus try to move away from light by borrowing deeper into the tissue. Forceful removal may lead to the retention of larval particles in tissues, leading to granulomatous inflammation and calcification. 19 Therefore, some centers use various suffocating agents (e.g., liquid paraffin, petroleum jelly, beeswax, adhesive tape, glue, turpentine oil) or larvicidal agents such as hydrogen peroxide and isopropyl alcohol before mechanical removal that can be done using non-toothed forceps. 19 Surgical debridement is necessary in most cases, such as in this case report. Broad-spectrum antibiotics (e.g., amoxicillin with clavulanic acid, metronidazole, and cefazolin) are recommended to prevent secondary bacterial infections. Anthelminthic drugs (e.g., ivermectin) and benzimidazoles (e.g., albendazole) are prescribed as adjunctive therapy. Ivermectin is absorbed rapidly and reaches high tissue concentration, thus the larvae are quickly expelled. 20
Public awareness of the infestation, its consequences, and predisposing factors are important measures for prevention and reinfestation. Wounds exudate and their odor attracts flies; thus, wounds should be kept clean and properly dressed. 19 Environmental cleanliness and improved living conditions are also helpful.
In conclusion, from the available literature, it is evident that C. bezziana is the most common fly involved in cases of myiasis in Sri Lanka. C. bezziana is an aggressive, rapidly progressing, and destructive species. 1 Myiases in the head and neck regions lead to more severe complications, such as the destruction of tissues and organs, leading to loss of function, amputation, or even death. 1 Therefore, it is important to be vigilant about this cryptic form of myiasis. To our knowledge, this is the first report of oro-nasopharyngeal invasive cryptic myiasis caused by third-instar larvae of C. bezziana reported from Sri Lanka.
Footnotes
Acknowledgements
We would like to thank Nalin Nandasiri and other laboratory staff of the Department of Parasitology of the Faculty of Medicine at the University of Peradeniya in Sri Lanka for their assistance with species identification.
Data Availability Statement
Any further information or data will be provided on enquiry for all scientific purposes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case was granted by the Ethics Review Committee of Faculty of Medical Sciences of the University of Sri Jayewardenepura in Sri Lanka at the meeting held on August 24, 2023.
Statement of Human and Animal Rights
All procedures in this case study were conducted in accordance with the standard guidelines and practices approved by the Ethics Review Committee of Faculty of Medical Sciences of the University of Sri Jayewardenepura in Sri Lanka.
Statement of Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
