Abstract
Introduction
Epistaxis is a common medical emergency that may require admission to the emergency department (ED) and treatment by an otolaryngologist. 1 Over 60 years ago, epistaxis was already known to require careful management, and the recommendations were quite similar to those used today. 2
Sixty percent of the general population will suffer from a nosebleed. Approximately 6% to 10% will need medical treatment in the ED, while only a minority will be hospitalized (0.2%–2.6%).3–6 Ten percent of those treated in the ED may experience severe life-threatening bleeding due to hemodynamic or respiratory compromise, 7 and 10% will have recurrent epistaxis. 8 In most cases, the source of the bleeding is the anterior part of the nasal cavity, defined as anterior to the natural maxillary sinus ostium. 9 A less common site is the posterior part, which is harder to locate and treat.8,9 The conventional methods of treating epistaxis are based on localizing the source of bleeding and chemical/electrical cauterization or packing, creating solid pressure.10-13 Intravenous or topical tranexamic acid (e.g., Hexakapron®) is administered to patients with no contraindications. 14 In more severe or difficult to control cases, mainly posterior bleeding unresponsive to posterior balloon packing, surgical intervention may be required, either ligation or embolization of specific vessels, as was described by Sokoloff in 1974. 15 Some medical conditions are associated with nosebleeds, for example, hypertension, anticoagulation, or non-steroidal anti-inflammatory drug therapy.1,8,16-18
Following a systematic review of the literature and consensus panel discussion, efforts were made to establish evidence-based recommendations regarding the management of epistaxis in hospitals.19,20 These recommendations included many aspects of epistaxis but did not establish specific indications for hospitalization. Nevertheless, it was stated that patients treated with absorbable packs do not have to be routinely admitted unless there are concerns regarding discharge safety, without specifying which concerns or therapies may indicate admission.
Currently, the common indications for hospitalization include a presentation with massive epistaxis, a large amount of blood loss, general weakness, or the need for treatment with posterior packing while pending a decision on whether further surgical intervention is needed.8,9,15,21 However, in some cases, deciding whether to hospitalize a patient is flexible. It depends on the subjective opinion of the otolaryngologist (e.g., a patient with recurrent epistaxis even in the absence of active bleeding).
Thus, due to the high incidence and potential hemodynamic compromise in epistaxis, there is a need to identify, at an early stage, possible hemodynamic compromise, major risk factors, and other negative prognostic factors.
This study aims to create objective parameters that can serve as indications for hospitalization in borderline cases without definitive indications due to their marked influence on the clinical course.
Patients and Methods
This is a retrospective study of 1171 medical records of epistaxis patients treated at our tertiary medical center ED between 2013 and 2018 with no age limitation. All 1171 files were reviewed thoroughly from the time of admission until discharge. The following data were collected on each patient: demographic details (age, sex, month of admission), primary medical history of hypertension (confirmed by medical records or by antihypertensive medication) or anticoagulation therapy, method of treatment (cauterization, packing, etc.), and blood tests: complete blood count and coagulation factor levels. The primary outcome was the severity of the bleeding, defined as severe by the presence of 1 or more of the following factors: recurrent epistaxis, a posterior source of bleeding, the need for hospitalization, the need for blood transfusion, or surgical intervention. Statistical analysis was conducted using SPSS software (IBM® SPSS® Statistics, version 23, Chicago, IL, USA). Data were presented as number of patients and percentage. Fisher’s exact test was used to test the association between 2 categorical variables. Logistic and multinomial regression models were used when the dependent variable had 2 or more categories, respectively. All tests applied were 2-tailed, and P ≤ .05 was considered statistically significant.
Results
Demographic and Medical History
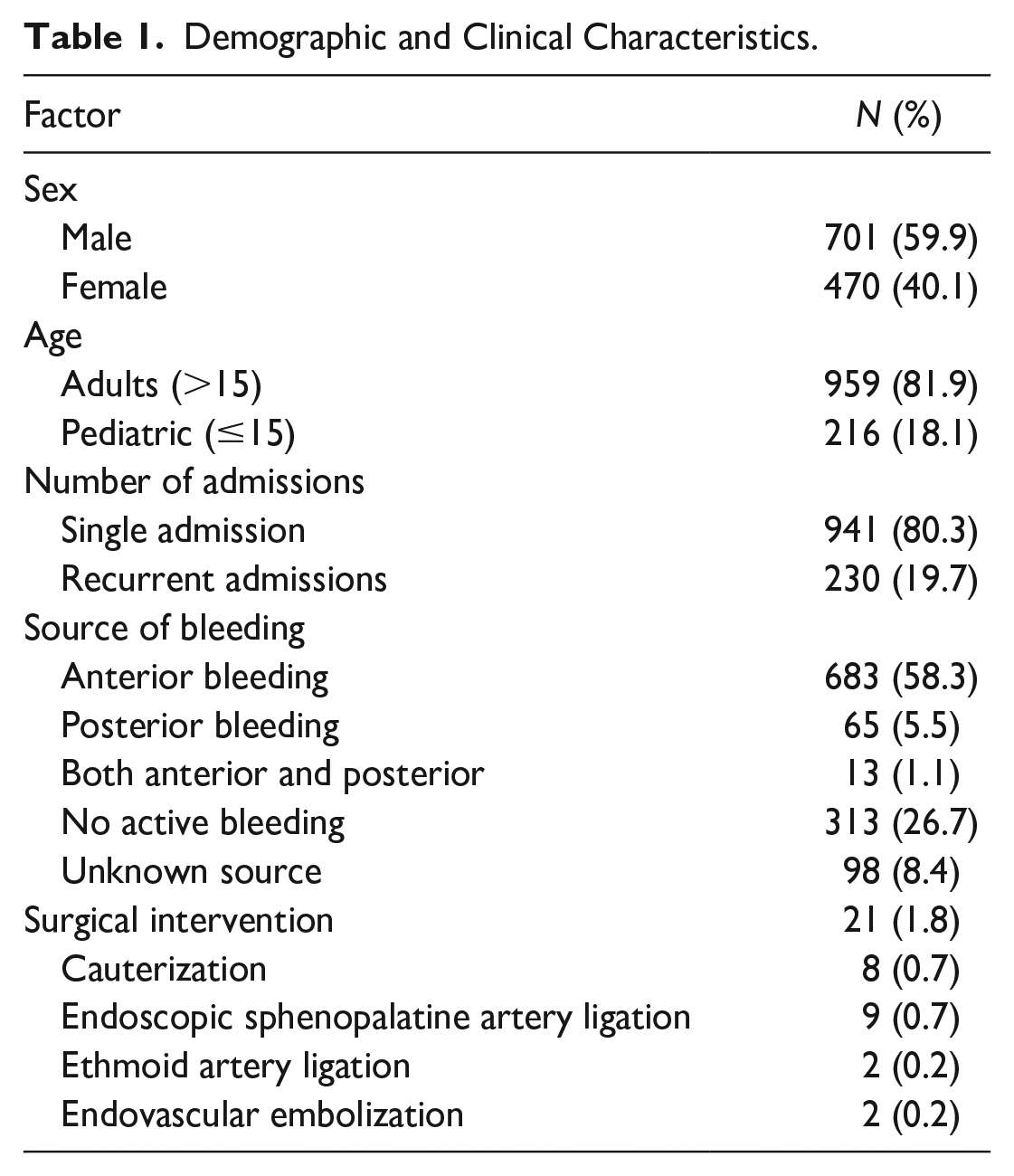
Between 2013 and 2018, there were 1171 admissions to the ED in our tertiary medical center, due to epistaxis, with 230 (19.7%) recurrent admissions for a total of 941 patients; 131 patients were hospitalized (11%). The number of admissions ranged from 1 to 7 for a single patient. The majority of patients were male (Table 1). The admission age ranged from 4 months to 104 years, with an average age of 6.6 years. The distribution of the patients’ ages was not homogeneous and showed 2 peaks of incidence: 255 patients (27%) aged 0 to 20 years and 407 patients (43%) aged 60 to 90 years. In 523 cases, the patient had a primary diagnosis of hypertension (44.7%). In 499 cases (42.6%), the patient was treated with antiplatelet/anticoagulation therapy, whereas 59 cases (5%) involved treatment with combined antiplatelet + anticoagulation medicines (Table 2). Most patients (63%) came during the winter months—November to April—with 422 admissions alone (36%) in February–April.
Demographic and Clinical Characteristics.
Correlations Between Various Risk Factors and Severe Clinical Course.
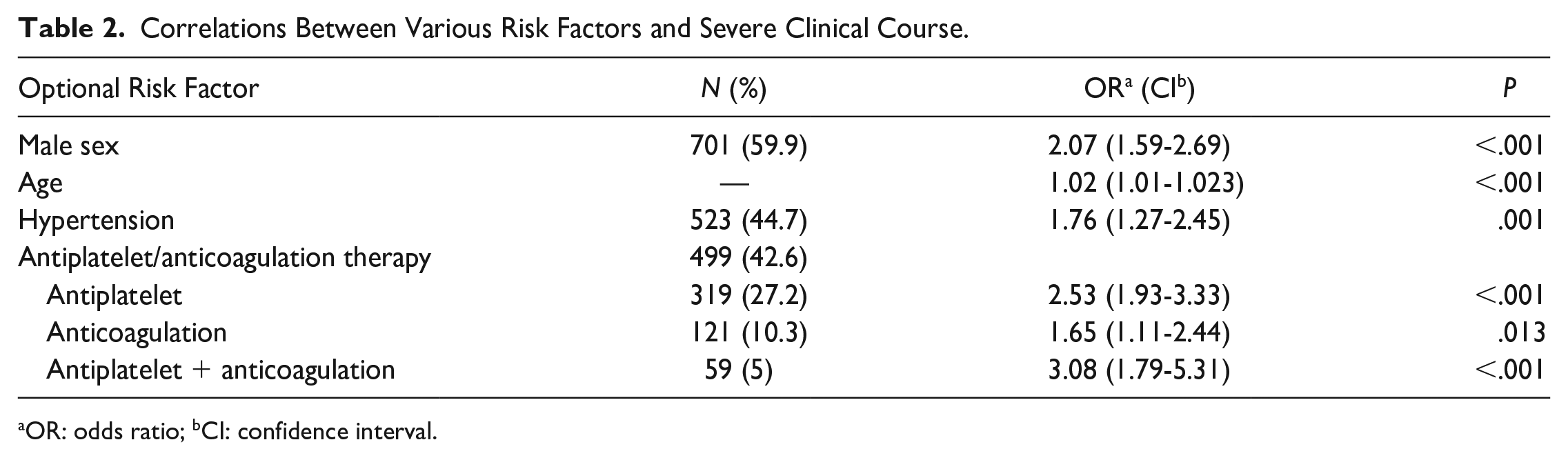
OR: odds ratio; bCI: confidence interval.
Bleeding Localization
In 761 patients (65%), the bleeding source was identified. In 411 cases (35%), it was not possible to locate the specific site: in 313 cases (26.6%), because of an absence of active bleeding and in 98 cases (8.4%) due to profuse active bleeding denying identification of its source (Table 1). One patient with hereditary hemorrhagic telangiectasia (Osler–Weber–Rendu syndrome) presented with anterior bleeding controlled by chemical cauterization. Eighty admissions (6.8%) to the ED followed various nasal surgical interventions, mostly septoplasty/turbinate reduction (87%).
Blood Test Results
Coagulation tests were taken in most cases (929 admissions) and prolonged in 158 cases (17%). An international normalized ratio (INR) >2 was detected in 85 patients, while only 14 patients (1.2%) had INR > 3.5, most of whom (70%) were being treated with warfarin. In 34 patients (2.9%), hemoglobin levels decreased by >1gr% following the first complete blood count in the ED; of these, 7 (0.6%) were given a blood transfusion due to hemodynamic compromise or low hemoglobin levels (<8gr%).
Medical and Surgical Intervention
An otolaryngologist treated all the patients, and the treatment modality was divided as follows: chemical cauterization with silver nitrate (36.7%), electrical cauterization (2.5%), anterior nasal pack (25.9%) using materials such as polyvinyl acetate polymer sponges (eg, Merocel®; Medtronic Inc., Jacksonville, FL, USA), or absorbable hemostat alone (7.1%) using products like oxidized regenerated cellulose (eg, Surgicel®; Ethicon Inc., Somerville, NJ, USA). A Foley catheter was used as a posterior pack in 7 cases (0.7%). In 7% of cases with chemical cauterization, the bleeding was not controlled, and additional anterior packing was required.
Surgical bleeding control was required for 21 patients (1.8%). In 8 cases, the source of the bleeding was localized and cauterized. In 9 cases, endoscopic ligation of the sphenopalatine artery was conducted, and in 2 cases, there was a need for unilateral endoscopic ligation of the anterior or posterior ethmoid artery. Endovascular embolization was performed after sphenopalatine artery ligation failed in only 2 cases. Of all 80 cases of postoperative bleeding mentioned above, 32 (40%) were hospitalized. However, only 2 required surgical control of the bleeding with cauterization.
According to these findings, logistic and multinomial regression models were used (according to the number of categories in the dependent variables) for the following independent variables: age, sex, presence of a former diagnosis of hypertension, antiplatelet or anticoagulation therapy, month of admission and treatment modality in the ED. The dependent variable was the clinical course, defined as more severe based on the number of admissions, the source of bleeding (anterior vs posterior), and the need for hospitalization, blood transfusion, and/or surgical intervention.
After adjusting for confounding factors, the following factors were found to be significantly associated with a more severe clinical course (odds ratio and confidence interval): male sex (OR 2.07; CI 1.59-2.69), age (OR 1.02; CI 1.01-1.023), and former diagnosis of hypertension (OR 1.76; CI 1.27-2.45).
Although age was significantly associated with a more severe course, the effect size was small (Table 2). Antiplatelet/anticoagulation therapy was a significant risk factor (OR 2.53; CI 1.93–3.33, OR 1.65; CI 1.11–2.44, respectively), with the highest rate in patients who were treated with combined antiplatelet + anticoagulation therapy (OR 3.08; CI 1.79–5.31).
As mentioned above, only 21 patients required surgery, none of whom belonged to the pediatric population. Treatment modality did not correlate with a more severe clinical course. The number of admissions during the winter (January–March) was significantly higher (P < .001) than the number of admissions throughout the rest of the year. However, there was no correlation between the season and the more severe clinical course.
Discussion
In the current study, we tried to identify risk factors and negative prognostic factors for severe nosebleeds to create accepted indications for hospitalization. A thorough review was conducted for all 1171 patients admitted to a tertiary ED over 5 years.
The sex distribution in our study was similar to that of former studies,2,16,22 with a higher proportion of males. A significant correlation was found between male sex and a more severe clinical course; thus, we think it should be considered a risk factor.
Regarding the age distribution, it seems that in our study, the average age was lower than in other studies.1,22,23 This can be attributed to our region’s high proportion of the younger population sector. The high prevalence of severe nosebleeds in the younger and older age groups matches data described decades ago. 2 Some studies have noted that older age is correlated with a more severe clinical course presented as posterior bleeding, as well as recurrent admissions.24,25 We found a significant correlation in the current study, but the effect size was small. Therefore, we suggest age as a relative risk factor.
In our study, the prevalence of a primary hypertension diagnosis was relatively low compared to other studies.22,23 Currently, it is unclear whether hypertension causes an increased probability of bleeding.16,26 Some studies only present descriptive statistics of hypertension prevalence with no analysis of negative prognostic factors. However, despite the lack of consensus in the literature and the British Rhinological Society’s multidisciplinary consensus recommendations regarding the influence of primary hypertension on the clinical course of epistaxis and its severity, 19 we think hypertension should be considered a risk factor for severe clinical course. Hence, we suggest this parameter be considered a major risk factor. It should be mentioned that hypertension measured during an episode of epistaxis may not be reliable as it is a stressogenic event.
The percentage of patients treated with anticoagulation therapy differs among studies, ranging from 10% to 70%.1,9,23 Our study showed an average of 40% associated significantly with the more severe clinical course. The British Rhinological Society’s multidisciplinary consensus recommendations have already concluded that anticoagulation therapy carries a higher risk for a severe outcome 19 ; therefore, we suggest considering this parameter an indication for hospitalization, specifically in patients with warfarin treatment or combined antiplatelet + anticoagulation therapy who may require further investigation to rule out coagulopathy.
These findings suggest considering hospitalization in borderline cases without definitive indication but with more than one risk factor.
As in previous studies,27,28 we observed a significantly higher incidence of admission due to epistaxis during the winter, probably due to the decreased humidity and increased dryness, which may result in drier mucosa of the nasal cavity and increased proneness to bleeding. 29
Regarding the bleeding location, different rates have been described. Nevertheless, it is accepted that anterior bleeding accounts for 85% to 95% of cases, while posterior bleeding accounts for 5% to 15% of cases.2,26 Similarly, in this study, an anterior source was located in 85% to 90% of active bleeding cases and a posterior source in 10% to 15% of cases. The number of cases with non-active bleeding was similar to other studies. 8
In 46 cases (4%), admission to the ED followed failure to control the bleeding by chemical cauterization. The number of actual failures may be higher than observed due to a lack of documentation in the medical record; nevertheless, there is not enough information regarding chemical cauterization to establish definitive conclusions regarding this method. 9
As previously described, coagulation tests were routinely taken for every patient with epistaxis even though only 158 cases (17%) had prolonged coagulation tests, and it seems that it should be reserved for patients treated with warfarin or suffering recurrent bleeding, or cases where coagulation disorder is suspected.30,31 A complete blood count is indicated for patients with a history of substantial pre-hospital bleeding and hemodynamic instability.
The limitation of our study is its retrospective nature and the fact that some of the patients with epistaxis were treated at pre-hospital clinics, which may have resulted in incomplete data collection.
Conclusion
Our findings suggest a few negative prognostic factors, including male sex, age, previous diagnosis of hypertension, and antiplatelet/anticoagulation therapy, which may increase the risk for a more severe clinical course of epistaxis. Therefore, we suggest that these factors be defined as indicators for hospitalization and close monitoring in borderline cases with no definitive indication for hospitalization, especially for patients with more than one risk factor. The current treatment modalities in the ED appear to be sufficient and allow for proper bleeding control, usually without posterior packing or surgical intervention. Similar to previous recommendations, we agree that coagulation tests may only be required for patients treated with warfarin or combined antiplatelet + anticoagulation therapy.
Footnotes
Authors’ Contribution Statement
All authors contributed to the study conception and design and all authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The institutional review board of Shaare-Zedek Medical Center approved the study (approval #0326-21-SZMC).
Informed Consent
Due to the retrospective nature of the study, the need for informed consent was waived.
