Abstract
Introduction
Chemo-irradiation to the head and neck region causes nasal dryness, nasal crusting, and olfactory dysfunction. These can significantly impair sino-nasal quality of life (QoL). Nasopharynx cancer (NPC) is an endemic disease in southern China. Treatment of NPC depends on the clinical staging; in our locality, for stage I and stage II disease, radial radiotherapy will be given. For stage II disease with high risk (EBV-DNA > 1500 copies/mL or lymph node >3 cm), concurrent chemoradiotherapy will be offered. For stage III (T3N0) disease, concurrent chemoradiotherapy will be offered if age ≤75 years and performance stage ≤2. For stage III-IV (except T3N0), induction chemotherapy followed by concurrent chemoradiotherapy will be suggested if age ≤75 years and performance stage ≤2. Induction chemotherapy regime will be subjected to age, performance stage, and renal function. Radial radiotherapy will be offered either if age >75 years, performance stage >2, or in poor premorbid status.1-3 Although in those non-endemic areas, the treatment regime may be different. 4 As the main primary treatment of NPC is radiation therapy (RT), often combined with chemotherapy, deterioration of sino-nasal QoL after treatment is expected.5,6 Limited local study addressing sino-nasal QoL for NPC patients after receiving chemo-irradiation, especially on objective smell sensation, could be found on the literature. Effect of RT toward nasal soft tissue edema and nasal flow alternation were also seldom reported. In this study, we investigate the nasal QoL of NPC patients after primary treatment and compare the result with healthy control group.
Method
Study Design
It is a case-control study. The primary objective is to measure the objective smell sensation for NPC patients after receiving primary chemo-irradiation treatment, the result was compared with the healthy control group. The secondary objective is to obtain the subjective data of sino-nasal lifestyle impact for NPC patients after receiving primary treatment using questionnaire, and also to measure the nasal obstruction, nasal cross-sectional area, and nasal flow after treatment. All the results were compared with the healthy control group.
Case Recruitment
Cases were recruited from January 1, 2021 to December 31, 2021. The NPC group and control group were recruited into 1:1 ratio. The inclusion criteria for the study group were those patients with a history of nasopharyngeal cancer who had completed primary treatment. For those patients with a history of nasal or brain surgery, history of head and neck chemo-irradiation except current NPC primary treatment, residual NPC disease, other nasal diseases like nasal polyps, preexisting smell disorders before suffering from NPC, poor premorbid status, mentally incompetent patients, less than 18 years old, and pregnant, all were excluded. Voluntary healthy control participants were recruited in the control group who were mainly our medical and nursing staff who enjoyed good past health and denied of smell and nasal problem.
Assessment
Patients were referred to a designated rhinology clinic. A completed ear, nose, and throat physical examination and naso-endoscope examination were performed to rule out any nasal pathology. Suitable patients were recruited and signed consent form. Objective smell assessment was done by using Butanol Threshold Test (BTT) and the traditional Chinese version of University of Pennsylvania Smell Identification Test [(UPSIT), Sensonics International, Haddon Heights, NJ]. Subjective nasal QoL was obtained by sino-nasal outcome test (SNOT-22) questionnaire. Nasal obstruction, nasal flow, and nasal cross-sectional area were measured using acoustic rhinometry and rhinomanometry (Otopront, RHINO-SYS). Results of the study group and the control group were compared.
Outcome Measurement
Butanol Threshold Test
The BTT was used for the quantitation of olfactory threshold in this study. The BTT was performed as previously described in literature with modifications. 7 99.9% 1-butanol was diluted in distilled water to produce a range of 10 serial dilutions (Appendix 1), from 0.00002% (step 10) to 4% (step 1). Identical bottles containing distilled water were used as controls. The participants were blindfolded throughout the assessment. Each nose was tested separately starting from the lowest concentration [0.00002% 1-butanol (step 10)]. Each concentration was tested along with a control in a forced-choice paradigm, in which the participant was required to differentiate between the test solution and control solution. The concentration step at which the participant was able to correctly differentiate between the test solutions and controls over 4 consecutive trials was defined as the BTT score. The average score of both nostrils was the participant’s final BTT score. Final BTT scores of the NPC groups and the control groups were compared.
University of Pennsylvania Smell Identification Test
UPSIT was performed as an identification test of olfactory function. 8 UPSIT consisted of 40 test odorants. The test score was the total of all correct answers. The UPSIT results were compared against an age-matched and gender-matched normative UPSIT score, which were further categorized into normal, mild hyposmia, moderate hyposmia, severe hyposmia, and anosmia accordingly.
Sino-Nasal Outcome Test
The SNOT-22 questionnaire consists of 22 symptoms with responses recorded on a 6-point scale ranging from 0 to 5 points for each item. Total scores range from 0 to 110. Lower score indicates a better nasal QoL. The 5 most severe symptoms were also selected by participants at the end of the questionnaire.
Acoustic Rhinometry and Rhinomanometry
Acoustic rhinometry is an instrument to measure the nasal cavity cross-sectional area. The method is based on the computer-assisted analysis of reflections, a nasally applied sound signal. This method allows the calculation of the cross-sectional area in the nose starting from the nostril and beyond. Acoustic rhinometry can pick up narrowing points as a cause of increased nasal resistance as well as deformations.
In acoustic rhinometry, the first and second narrowing point were labeled as minimal cross-section area (MCA)1 (in cm2) and MCA2 (in cm2), which is representing the cross-sectional area of interval nasal valve region and the cross-sectional area of inferior turbinate head, respectively. Both sides of the nose were measured. To minimize the effect of a deviated nasal septum causing a significant discrepancy in MCA results on each side of the nose, we take the average score of MCA1 and MCA2 on both sides of the nose as the final MCA1 and MCA2 results. Total volume (TV) (in cm3) is the nasal cavity volume measuring from nostril to 5 cm distal to nostril. Again, the average TV score of both sides of the nose was calculated to establish the final TV score to minimize the effect caused by the nasal septum deviation. Rhinomanometry and rhinoresistometry provide objective data of nasal flow (in ml/s) and nasal resistance (in sPa/ml). The average value of nasal flow and nasal resistance of both sides of the nose were used as the final score again.
Rhinomanometry is used to detect the nasal flow rate via a breathing mask with a special measuring nozzle. The choanal pressure is measured by means of a stick-on, nondeforming nose adapter. The direct coupling of the micro-pressure sensors to the respiratory mask results in a better handling and higher measurement accuracy. A bacteria filter ensures good hygiene of the measuring instrument.
The rhinoresistometry is a further measurement of the rhinomanometry based on flow laws. It can differentiate the cause of the flow dynamics, for example, narrowing by swelling, skeletal deformation, and pathological inspiratory nasal flap collapse as well as pathological turbulence behavior. Rhinomanometry serves as a measurement method for rhinoresistometry.
Statistical Analysis
The numerical results were compared using unpaired-t test. Categorical variables were compared using the Fisher’s exact test. SPSS 26.0 for Windows (IBM Corp. Released 2019. IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp) was used for statistical computation. P-value < .05 represents a significant difference.
Result
There were 14 NPC patients and 14 control participants in this study. The demographic data are summarized in Table 1. The age of the control group ranged from 28 years to 63 years (mean ± standard deviation, 43.1 ± 14.8) and the age of the NPC group ranged from 36 years to 72 years (mean ± standard deviation, 50.6 ± 10.7). Statistically there is no difference in the mean age of both group with P = .065. Among the 14 NPC patients, 1 patient suffered from stage II disease (2/14, 14%), 7 patients suffered from stage III disease (7/14, 50%), and 5 patients suffered from stage IV disease (5/14, 36%), respectively. The posttreatment duration of the NPC group ranged from 6 months to 183 months (mean ± standard deviation, 79.4 ± 58.0).
Demographic and Clinical Characteristics of NPC Patients and Controls.
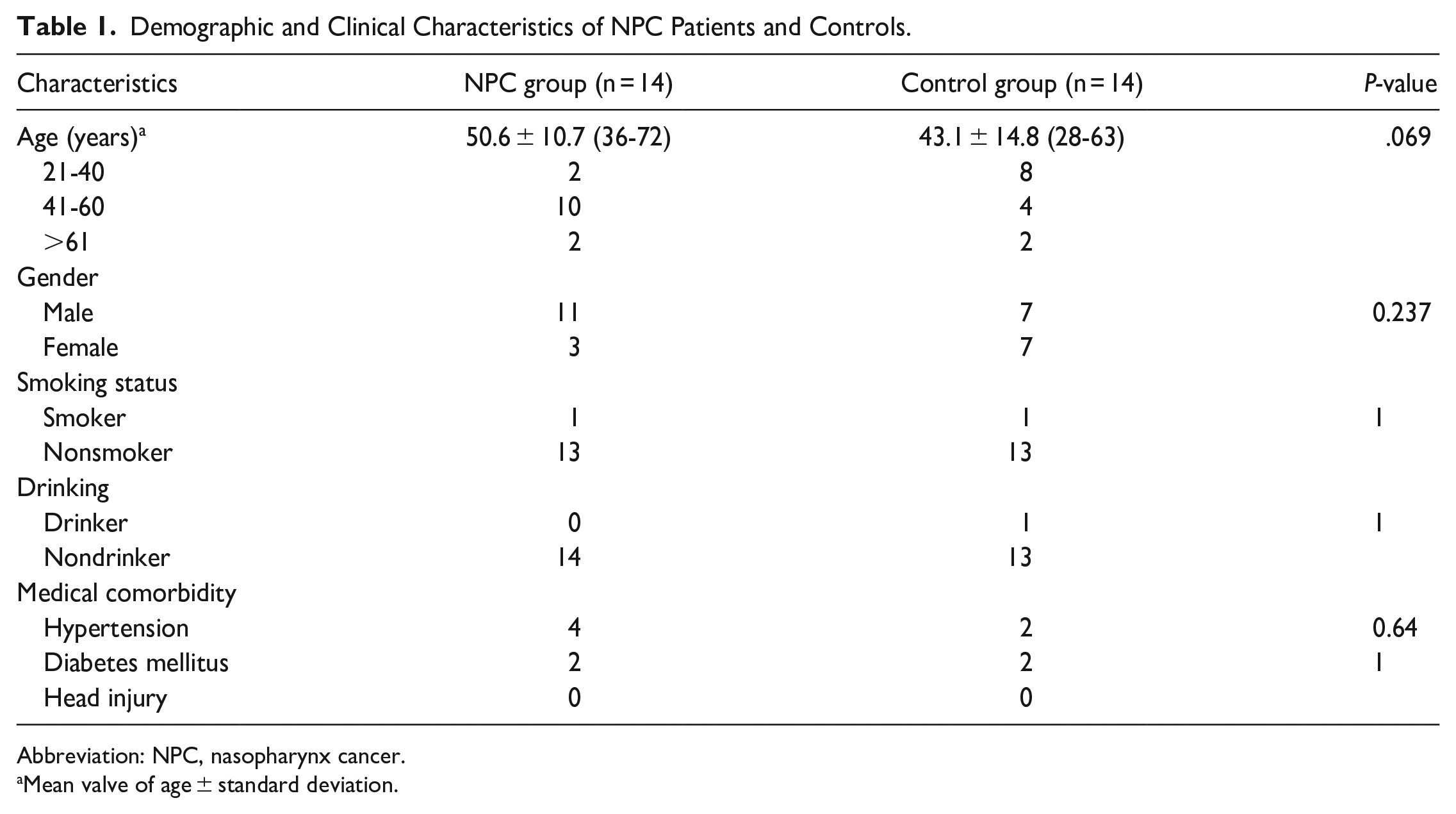
Abbreviation: NPC, nasopharynx cancer.
Mean valve of age ± standard deviation.
The mean BTT score of the control group was 5.17 (standard deviation = 1.49), whereas the BTT score of the NPC group was 2.71 (standard deviation = 1.37) (Figure 1). The BTT score of the NPC group was significantly lower than the BTT score of the control group indicating an impaired olfactory threshold in the former group (P < .05, difference in mean = 2.46 with 95% confidence interval: 1.35-3.57).
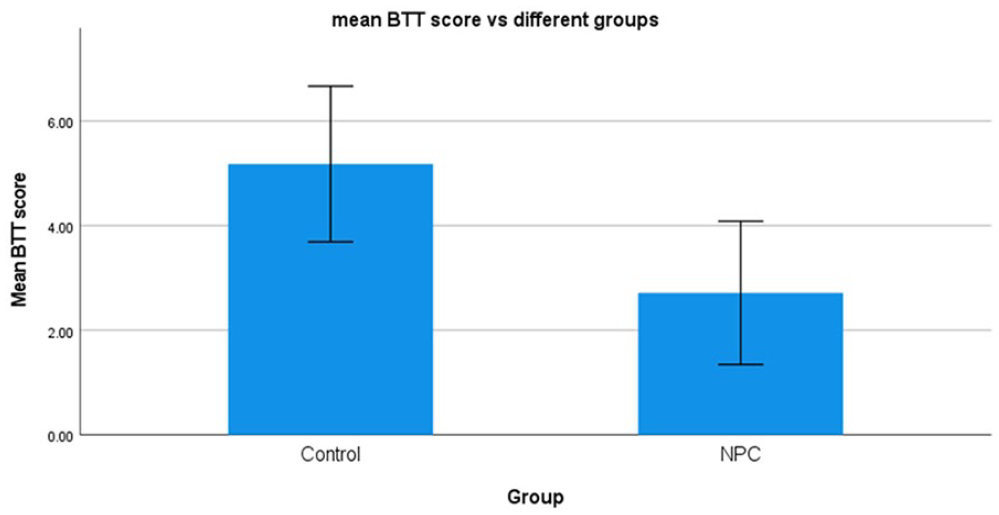
Comparison of mean Butanol Threshold Test scores between the NPC patient group and the healthy control groups. Bars represent standard deviation of mean. P < .05.
The UPSIT score of the control group was 31.93 (standard deviation = 2.80) and the UPSIT score of the NPC group was 25.14 (standard deviation = 8.21) (Figure 2). The mean difference was statistically significant (P < .05, difference in mean = 6.79 with 95% confidence interval: 2.02-11.55). The results revealed that the NPC group had a lower olfactory identification level.
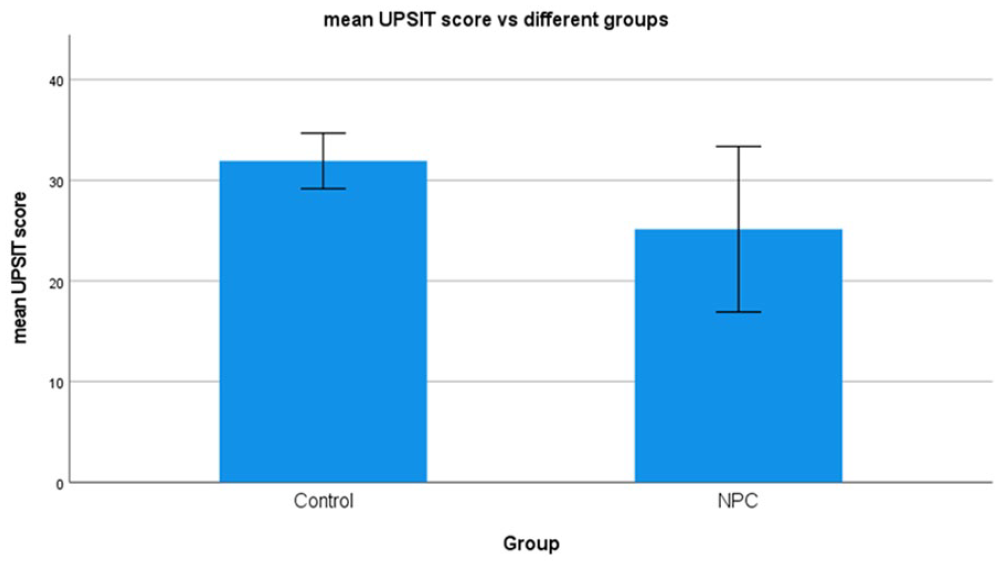
Comparison of mean UPSIT scores between the NPC patient group and the healthy control groups. Bars represent standard deviation of mean. P < .05.
The mean SNOT-22 score of control group was 16.71 (standard deviation = 8.02), which is statistically lower (P < .05, difference in mean = 21.00 with 95% confidence interval: 8.23-33.77) than the mean SNOT-22 score NPC group of 37.71 (standard deviation = 21.82). The higher score of the SNOT-22 questionnaire reflected worsening nasal QoL in the NPC group (Figure 3).
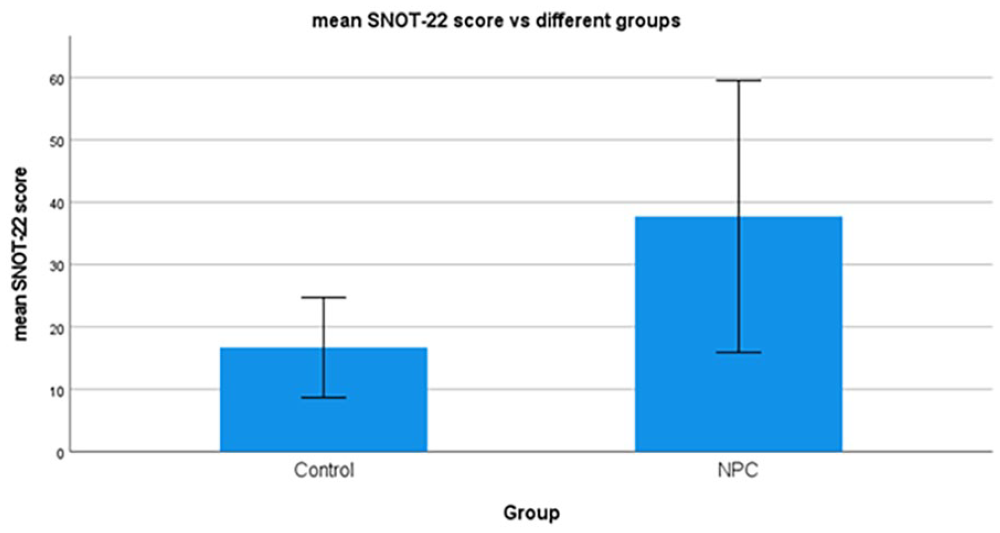
Comparison of mean SNOT-22 questionnaire scores between the NPC patient group and the healthy control groups. Bars represent standard deviation of mean. P < .05.
The mean score and the standard deviation of each question in SNOT-22 were listed (Table 2). We further categorized the SNOT-22 questions into 5 subdomains. Questions 1, 2, 3, 5, 6, 21, and 22 were grouped as rhinologic symptoms; Questions 4 and 8 were grouped as extra-nasal rhinologic symptoms; Questions 7, 9, and 10 were grouped as ear/facial symptoms; Questions 15 to 20 were grouped as psychological dysfunction; and Questions 11 to 14 were grouped as sleep dysfunction. The mean scores and the standard deviation of each subdomain were showed (Table 3). The result showed that the treatment group experienced worsening ear/facial symptoms and psychological dysfunction, which were statistically significant.
Descriptive Statistics for SNOT-22 Items Before and After Treatment.
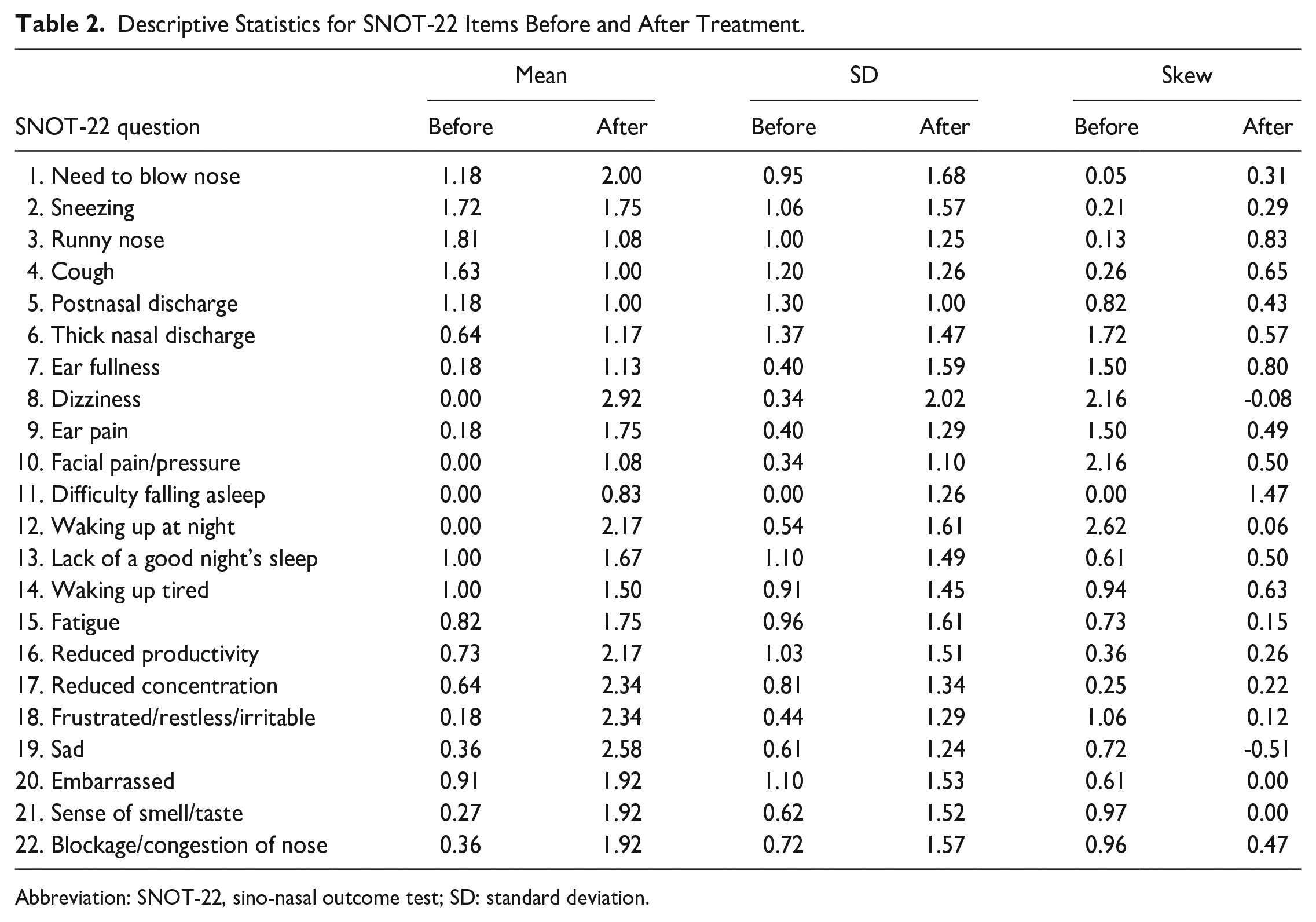
Abbreviation: SNOT-22, sino-nasal outcome test; SD: standard deviation.
Subdomain of SNOT-22 Items Before and After Treatment.
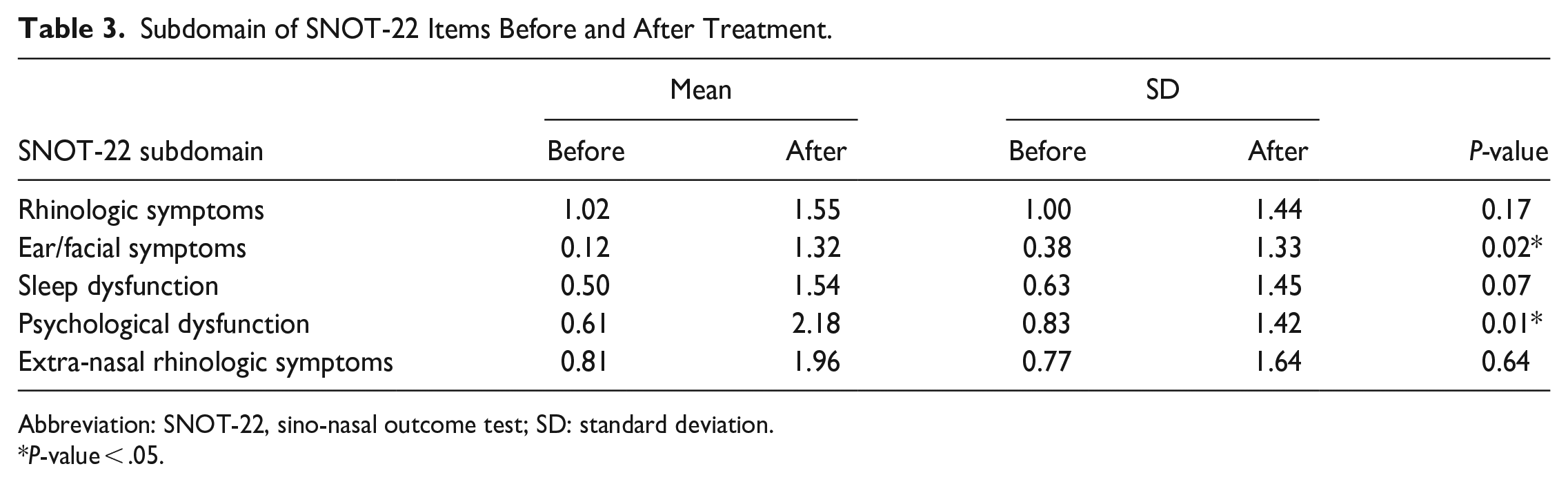
Abbreviation: SNOT-22, sino-nasal outcome test; SD: standard deviation.
P-value < .05.
The distribution of 5 most severe problems in SNOT-22 questions was different from those in NPC patients. In control group, the 5 most troublesome nasal problems were nasal blockage, need to blow nose, sneezing, runny nose, and postnasal discharge, whereas the most disturbing symptoms for NPC group were ear fullness, thick nasal discharge, decreased sense of smell/taste, fatigue, and reduced productivity (Table 4). For the control group, those symptoms were mainly rhinitis-related problems. However, for the NPC group, the side effects of radiotherapy distress them to a certain extent even after months of chemo-irradiation. It can be explained that those control groups of patients may have undiagnosed mild rhinitis symptoms that may slightly affect their daily lives; even though they denied having rhinitis symptoms during case recruitment, they revealed them when specifically asked in the SNOT-22 questionnaire. This part of results just indicated the existence of symptoms without reflecting the severity of the symptoms.
Distribution of 5 Most Severe Symptoms of SNOT-22 Questionnaire in Both Control Group and NPC Group.
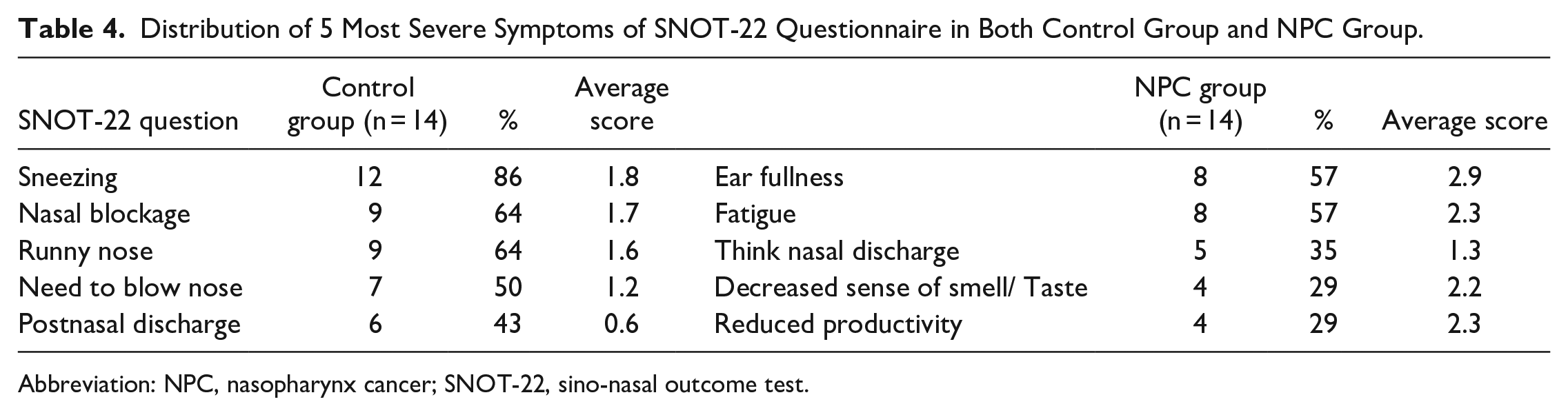
Abbreviation: NPC, nasopharynx cancer; SNOT-22, sino-nasal outcome test.
The results of acoustic rhinometry and rhinomanometry are summarized in Table 5. The MCA1, MCA2, and TV scores for the control group detected by acoustic rhinometry were 0.56, 1.71, and 6.35 whereas the scores in the NPC group were 0.65, 1.11, and 6.58, respectively. There was no statistical difference between these 2 groups. The nasal flow of the control group and NPC group were 553 and 546, respectively and the nasal resistance of the control group and NPC group were 0.35 and 0.53 separately. Both nasal flow and nasal resistance among these 2 groups did not show statistically significant results.
Result of Rhinomanometry, Rhinoresistometry, Acoustic Rhinometry in Both Control Group and NPC Group.
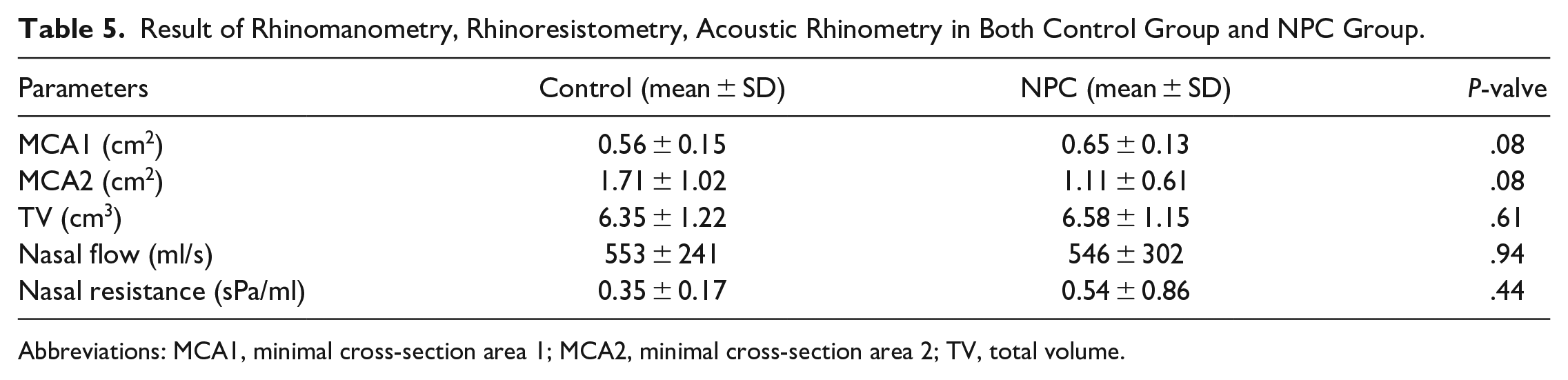
Abbreviations: MCA1, minimal cross-section area 1; MCA2, minimal cross-section area 2; TV, total volume.
Discussion
Olfaction Alteration During RT
Intensity-modulated radiotherapy (IMRT) is currently the standard of care for management of nasopharyngeal carcinoma. IMRT allows better conformation of radiation dose to the tumor, while sparing the surrounding organ at risk (OAR). IMRT has demonstrated better long-term disease control, 9 less toxicity 10 and fewer serious complications, when compared with conventional techniques.
Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, the toxicity criteria of the Radiation Therapy Oncology Group, and the European Organization for Research and Treatment of Cancer toxicity grading are the most commonly used systems in grading and reporting of toxicities associated with radiotherapy. Though included in the CTCAE, nasal toxicities are less well recognized and are not commonly reported in the literature. There is a lack of understanding of the relationship between radiation dosimetry and nasal toxicities endpoints. As a result, the nasal passage and the olfactory neural pathway are usually not considered as OARs in the RT planning process.
Olfactory cleft may be exposed to the radiation field leading to its deleterious effects. Patients will have deterioration of smell perception during the course of radiotherapy which may lead to domestic hazard. In a study by Jalali et al., 11 the olfactory threshold was reduced 2 weeks after initiation of radiotherapy and those patients showed no olfactory recovery up to 6 months follow-up. In the study by Gurushekar et al., 12 both olfactory threshold level and identification level will start to decrease after 3 weeks of RT and maintain throughout the course of RT. However, 24% of patients showed complete olfaction recovery after 3 months follow-up while the remaining 76% showed incomplete recovery. Veysellar et al. 13 investigated nasopharyngeal cancer patients who were treated with radiotherapy and found out that the olfactory bulb volumes on MRI scan and olfactory threshold were decreased in those patients when compared to the healthy controls group. This finding can be explained by the direct radiation damage to the olfactory bulb and mucosal injury to olfactory epithelium.
Effect of Radiation on Nasal QoL
RT can cause direct injury to nasal respiratory mucosa and cause various physiological changes such as nuclear pyknosis, vacuolation of the ciliary cells, decreased ciliary cells, and sloughing. Majority of patients undergoing head and neck irradiation complained of nasal congestion, nasal crusting, facial pain, and foul smell during the treatment. This was caused by the impaired ciliary function thus inadequate drainage of secretions. Nasal douching was recommended to those patients suffering from such symptoms. It aimed to restore the nasal microenvironment as much as possible. There is no definition of nasal QoL and there is no standard guideline to measure it either. In our study, we measured it via the objective olfactory assessment, subjective questionnaire score. and objective nasal flow measurement, in which the former 2 parameters were statistically diminished in the post-RT group.
Alteration of Nasal Cavity Volume After Radiotherapy
Radiation causes nasal mucosa change like mucosal edema 14 and thus affecting the nasal flow and nasal resistance. However, there is no literature investigating the objective changes of nasal cavity volume after radiotherapy. We used acoustic rhinometry to measure the overall cross-sectional area of the nasal cavity in both the NPC group and control group. Also, rhinomanometry and rhinoresistometry were used to give the objective data of nasal flow and nasal resistance. In our study, there was no statistical difference between these groups in all 3 parameters. It may be due to our small sample volume. A larger sample size study is suggested to investigate the sino-nasal structure changes after radiotherapy.
Smell Rehabitation on NPC Post-RT Patients
Olfactory dysfunction can be classified into conductive and sensorineural causes. Conductive olfactory loss is mainly due to structural nasal problems. Sensorineural loss is mainly caused by postinfectious, post-head injury, post-head and neck irradiation, and other central nervous system diseases. 15 For those locally advanced stages NPC, the olfactory pathway is blocked by the tumor which contributes to the conductive olfactory dysfunction. After completion of the NPC primary treatment, the tumor was resolved and there is no conductive element. It is supposed that the olfactory dysfunction in those post-RT patients is purely due to a sensorineural cause if endoscopically excluded nasal adhesion and scarring. Smell rehabilitation is a well-established treatment for sensorineural olfactory loss in foreign countries. 16 Smell rehabilitation is proved to be effective treatment in COVID-19-related olfactory dysfunction in a recent Hong Kong study. 17 However, there is no current literature worldwide about the effect of smell rehabilitation on those olfactory loss patients after RT.
Limitation and Future Study
In Hong Kong, the incidence rate of NPC is around 10 in 100,000. 18 Due to the small sample size of our study, it might cause sampling error in which the observed sample might not represent the whole population.
Also, the traditional Chinese version is not validated in Hong Kong population. It may cause measurement bias of the identification score in both NPC group and control group. However, when we compared the mean UPSIT score among both groups, the measurement bias can be eliminated.
The staging of NPC, different chemotherapy regimes and dosage of RT may contribute to different degrees of olfactory loss, but in this study, it is not limited to the small sample size. A larger sample size and multicenter study is suggested to reduce the sample bias and analyzes the correlation between olfactory impairment with RT dosage and disease staging.
To investigate the true effect of radiation on sensorineural olfaction loss, a prospective cohort is advocated to compare the smell alteration before and after irradiation. However, careful selection of suitable candidates is required to make sure that only those early-stage NPC without gross structural blockage to the olfactory pathway causing conductive olfactory loss are eligible to the study.
Conclusion
This study provides a quantitative olfactory threshold and identification changes after primary treatment for NPC patient. The olfactory function and QoL for those patients receiving chemo-irradiation is statistically significantly impaired when compared to the healthy control group. Otolaryngologists should pay more attention to olfaction and nasal QoL to those NPC patients as it is commonly neglected in our daily practice.
Supplemental Material
sj-docx-1-ear-10.1177_01455613231204210 – Supplemental material for Effect of Radiotherapy on Sino-nasal Function in Nasopharynx Cancer Patients
Supplemental material, sj-docx-1-ear-10.1177_01455613231204210 for Effect of Radiotherapy on Sino-nasal Function in Nasopharynx Cancer Patients by Fergus Kai-Chuen Wong, Edwin Chun-Yin Wong, Alvin Po-Ngai Chu, Peter Ka-Chung Kwan and Fu-Man Tong in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
FK-CW and F-MT contributed to the conception and design of the study. FK-CW contributed to the planning and acquisition of data. FK-CW contributed to analysis of data. FK-CW and EC-YW wrote the first article draft. FK-CW, PK-KC and AP-NC contributed to the interpretation of results. All authors contributed to the critical review and revision of the article. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Availability of data and materials
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Informed consent was obtained from all subjects involved in the study. This study was approved by Hong Kong East Cluster Research Ethics Committee (HKECREC-2020-096).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
