Abstract
Introduction
Tuberculosis (TB) is a widespread infectious disease that represents a major health problem and a major cause of morbidity and mortality in the world.
Approximately one-third of the world’s population has latent infection with Mycobacterium Tuberculosis.1,2 Tunisia is the third endemic country in the world among countries with high TB burden countries. 3
In 1855, Rokitansky put forward the theory that TB and malignancy cannot exist within the same organ and highlighted a definite antagonism between the 2. 4 However, Whartin described for the first time in 1899, the coexistence of cancer and TB within the same lymphadenopathy.
This condition is very rare, and only a few cases have been reported in the literature,3,5 especially in countries with a high incidence of TB. 6
The purpose of this article was to discuss, through our cases, the clinical characteristics and treatment options of concomitant TB and malignancy within the same cervical lymph node.
Methods
The study was conducted from 2015 to 2023. It included 10 patients treated for concomitant TB and malignancy within the same cervical lymph node at our ENT department. The inclusion criteria comprised patients with confirmed histological evidence of both malignancy and TB within the same cervical lymph node. Ineligible or incomplete records were excluded.
Cases were retrospectively reviewed. We collected, through medical files, demographic data (age, gender, history), clinical features, laboratory tests, bacteria culture tests, imaging, treatment, and outcomes. Data collection was performed using SPSS software.
Ethical approval for the study was obtained from the ethics committee with the approval number : 71/2024/CLPP
Results
The study included 10 patients (5 men and 5 women) (Table 1). Their mean age was 47 years. All patients have been BCG-vaccinated.
Characteristics of the Reported Cases.
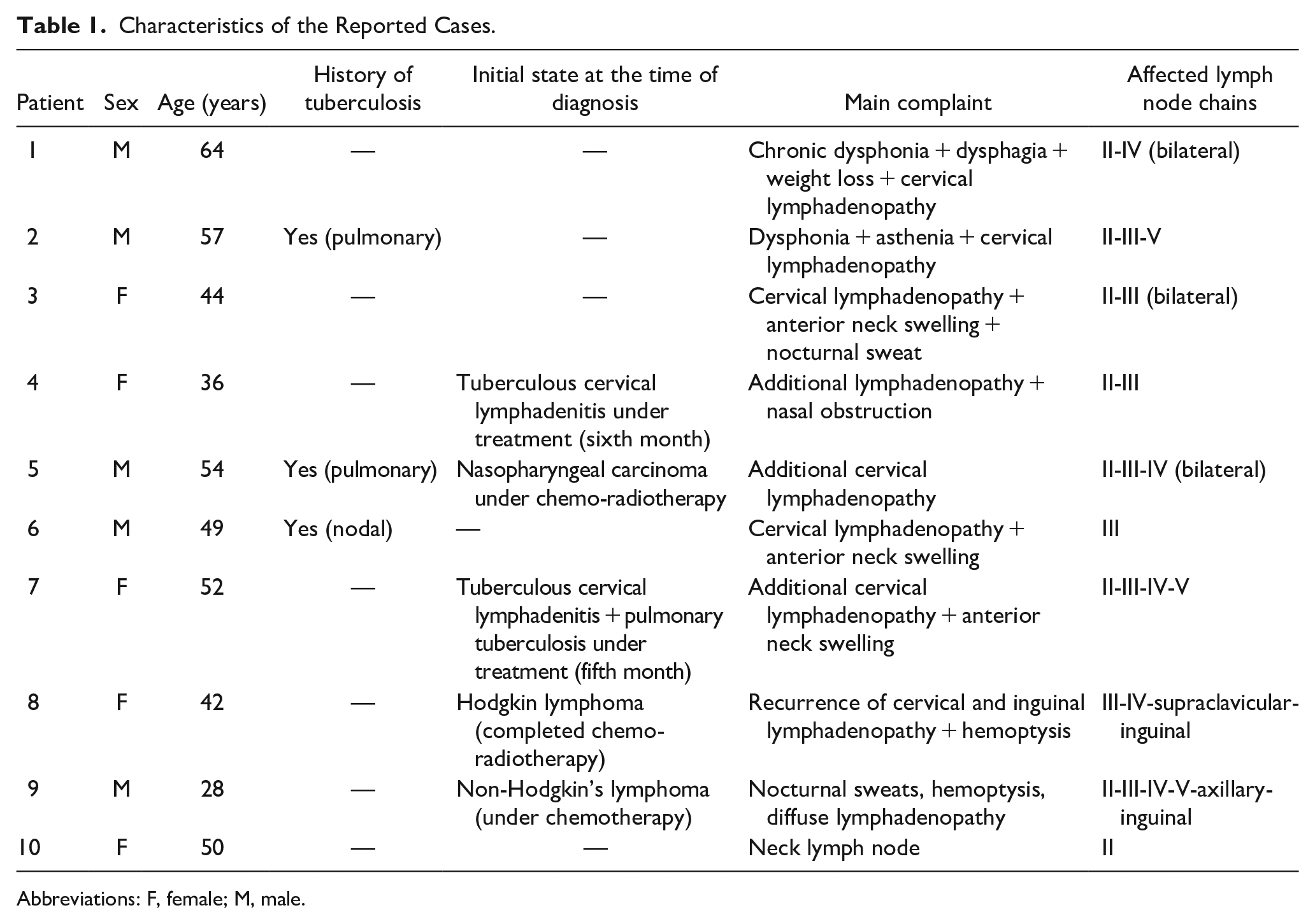
Abbreviations: F, female; M, male.
Two patients have first been diagnosed with cancer and were receiving (1 case) or receiving (1 case) treatment. Four patients received (2 cases) or received (2 cases) treatment for TB. One patient had a history of cured TB and was being treated for nasopharyngeal carcinoma. The other 3 cases were not diagnosed with cancer or TB.
All patients presented with recurrent or new lateral neck swelling. The average size was 3.5 cm. It was firm in 7 cases, hard in 2 cases, and fluctuant in 1 case. It was mobile in 5 cases and fixed elsewhere. The overlying skin was healthy in all cases. Other symptoms included hoarseness, dysphagia, nosebleed, nasal obstruction, hemoptysis, weight loss, and nocturnal sweat. On physical examination, the lymph nodes were located in levels II and III of the neck. They were bilateral in 3 cases.
Neck ultrasound showed well-defined hypoechogenic lymph nodes bilaterally, with preserved echostructure in 2 cases. Lymph nodes had a thyroid-like appearance in 1 case. They had necrosis areas in 3 cases.
Computed tomography (CT scan) was performed in 7 cases. It showed necrotic lymph nodes (in 5 cases) and mediastinal nodes in 2 cases (cases 7 and 8). One patient had miliary TB on CT scan.
Lymph node fine needle aspiration cytology (FNAC) was performed in 5 cases. It showed lymphoma cells in 2 cases and caseous necrosis in 1 case. It was negative 2 cases. Mycobacterium Bovis was identified in 1 case by polymerase chain reaction (PCR) exam.
In all cases, a histopathological examination of the swollen lymph node was performed. Four patients underwent cervical lymph node dissection due to the diagnosis of cancer: thyroid cancer (patients 3, 6, 7) and laryngeal cancer (patient 1). For patient 2, who had T1 laryngeal cancer, considering the history of TB, cervical lymph node TB was suspected rather than lymph node metastasis of laryngeal cancer. Therefore, a cervicotomy with lymph node excision and frozen section examination was performed. Patient 4 was initially diagnosed with lymph node TB. However, after 6 months of antituberculous treatment, physical exam revealed a nasopharyngeal tumor and new lymphadenopathy. Given that, fine needle aspiration (FNA) did not aid in the diagnosis, we proceeded with an excisional biopsy to determine whether the lymphadenopathy was due to the tumor or TB.
For Case 5, the patient had a history of treated pulmonary TB and was being monitored for nasopharyngeal cancer. The emergence of cervical lymphadenopathy during cancer treatment raised the suspicion of metastatic disease. Although FNA was performed, the cytological results were more consistent with lymph node TB (showing caseous necrosis without epithelioid granulomas and giant cells). The presence of caseous necrosis alone, without granulomas or giant cells, was not considered sufficient for a TB diagnosis and treatment. Therefore, we opted for an excisional biopsy to confirm the diagnosis.
For patients 8 and 9, they experienced a relapse during treatment for lymphoma. A FNAC was performed, showing the presence of lymphoma cells. Lymph node excision was carried out for immunohistochemical study. For patient 10, the PCR test on the fine needle aspiration fluid was positive. However, the diagnosis of lymphoma was strongly suspected based on clinical, biological, and radiological data. Lymph node excision was performed.
The coexistence of TB and malignancy was confirmed histologically for all patients (Figure 1A and B). The histological types of the diagnosed cancers were dominated by papillary thyroid carcinoma and undifferentiated nasopharyngeal carcinoma (Table 2).
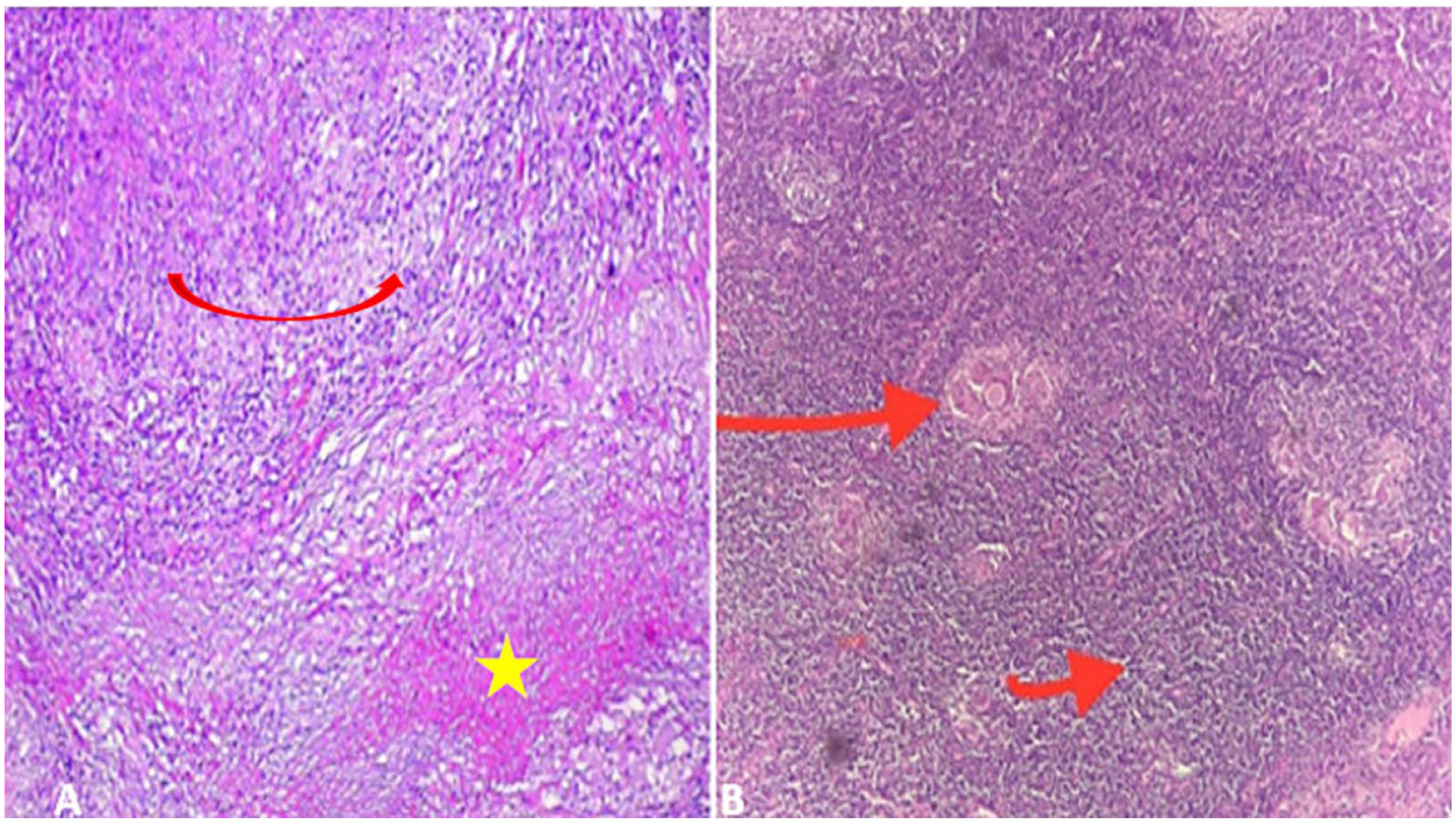
HE*250: (A) proliferation of undifferentiated carcinoma cells (yellow star) with an epithelioid granuloma centered by caseating necrosis (big red arrow); (B) Reed-Sternberg cells (little red arrow) and epithelioid and gigantocellular granulomas (big red arrow).
Histological Type of Malignancy and Its Treatment.
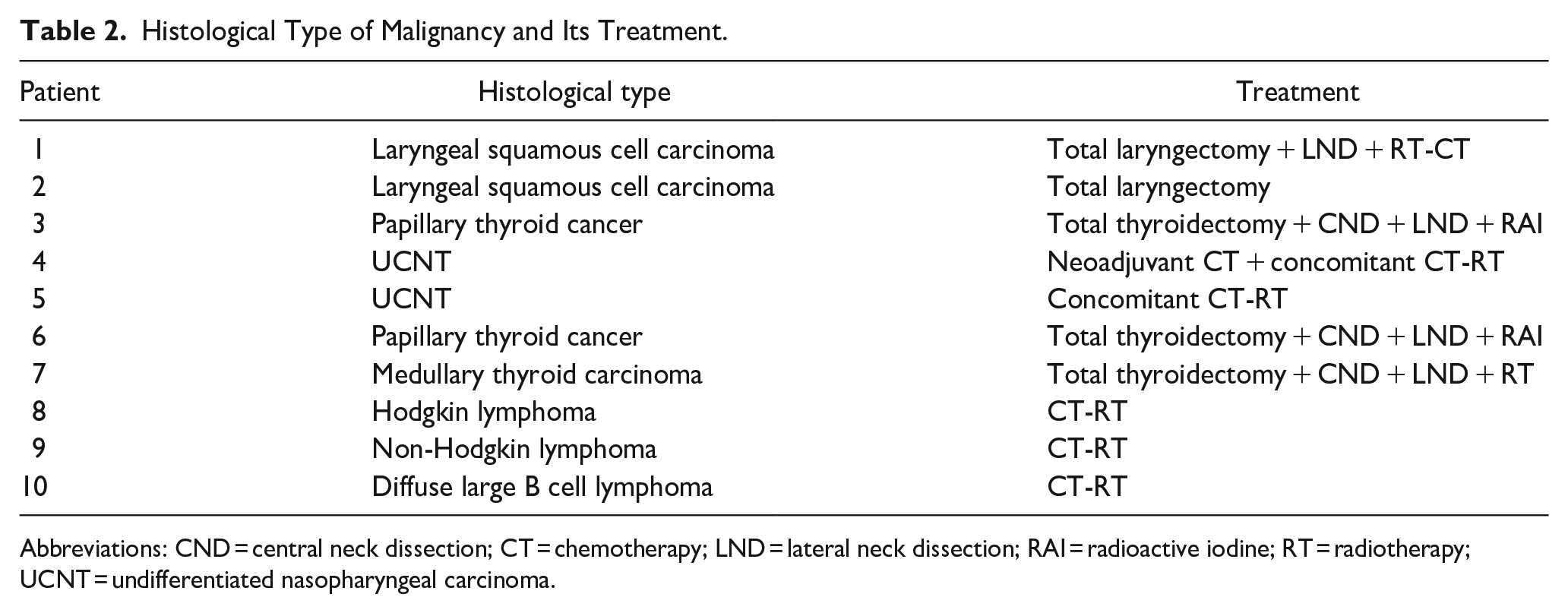
Abbreviations: CND = central neck dissection; CT = chemotherapy; LND = lateral neck dissection; RAI = radioactive iodine; RT = radiotherapy; UCNT = undifferentiated nasopharyngeal carcinoma.
Treatment is detailed in Table 2. It was provided in collaboration with infectious disease specialists and oncologists. All patients who did not have chemotherapy at the time of diagnosis received 15 to 30 days of TB treatment before starting chemotherapy. For patients diagnosed with cancer during TB treatment, TB treatment was continued, and cancer treatment was initiated immediately. The patients received combined TB treatment for an average duration of 8 months [2 months of isoniazid (H), rifampicin (R), pyrazinamide (Z), and ethambutol (E) (HRZE) quadritherapy and 4 to 8 months of bitherapy (HR)].
Follow-up was based on clinical, biologic, and radiologic exams (neck ultrasound and facial magnetic resonance imaging). The side effects of TB treatment were dominated by vomiting and/or hematologic disturbance in 3 cases. Chemotherapy was delayed until the biological balance was normalized. Two patients died during treatment, one of them due to miliary TB with pulmonary and liver complications, the second due to septicemia due to secondary aplasia to chemotherapy (case 9).
Discussion
A complex relationship between TB and malignancy has been highlighted, especially in countries with high endemicity for TB. 7 The mutual influence between TB and cancer is not fully understood. Some suggest that coexistence is purely coincidental. Others propose that cancer cachexia may offer a good nutritional basis for dormant bacteria. 4 Other authors suggest that chronic immunosuppression may be favorable for the development of the simultaneous or secondary existence of both disorders in the same patient.8,9 In our study, TB in patients diagnosed with lymphoma or/and treated with chemotherapy and allograft can be explained by the immunodeficiency induced by the tumor and its treatment. The tumor itself, particularly locally advanced or metastatic tumors, can also predispose patients to the reactivation of TB. 10 TB may play a major role in carcinogenesis due to chronic inflammation and resulting DNA alterations.9,11
Wu et al 8 reported an average delay of 4 and 11 months in cancer diagnosis in TB patients. Clinically, TB has similar symptoms to cancer, especially hematologic malignancies. 6 Lymph nodes, weight loss, and night sweats can be seen in these 2 illnesses. 6 In the reported cases of synchronous TB and head and neck cancer, failure in TB treatment was the most frequent circumstance of discovery of cancer.10,12,13 In our study, 4 patients received or were receiving treatment for TB. They presented with recurrent or new lymph nodes. Treatment failure was suspected.
To our knowledge, only 2 cases of coexisting tubercular lymphadenitis and metastatic nasopharyngeal cells within a cervical lymph node have been reported. 3 The coexistence of TB and squamous cell carcinoma (SCC) of the upper aerodigestive tract in the same lymph node was first described by Gheriani et al, in 2006. 14 To date, only a dozen cases have been reported in the literature. 14 In SCC, TB should always be considered, even in nonendemic areas, particularly in elderly patients, mainly due to cancer-induced immune suppression. 12 The coexistence of TB and metastasis of papillary thyroid carcinoma (PTC) in the same lymph node is also very uncommon. 15
On imaging, suspicion of nodal TB is usually difficult in patients diagnosed with cancer. In fact, TB and malignant tumors share common imaging characteristics such as necrosis and intranodal calcification.4,13,14,16
In our study, ultrasound and CT were not helpful to distinguish between TB and metastatic nodes. They showed calcification and necrosis, both of which can be found in the 2 situations.
The diagnostic efficiency of FNAC varies between 46% and 90%, and PCR has a sensitivity of 82% and a specificity of 100% for detecting TB. 16 Combined FNAC and PCR to detect Mycobacterium Tuberculosis should be used in cancer patients every time TB is suspected. 17 In our study, we performed 5 FNAC, 2 of which were not conclusive. It showed lymphoid cells in 2 cases and caseous necrosis in 1 case. PCR was positive in 1 case for detection of Bovis tuberculosis.
Quantiferon TB-2G testing is relatively new and is often used for extrapulmonary TB. Sensitivity and specificity were shown to be 70% to 90% and 90% to 100%. 16 The diagnostic utility of the tuberculin skin test is very low in the background of a malignancy due to underlying immune suppression. 18 However, tuberculin skin test can be considered positive for patients with hematological cancer since they have the highest active TB rates, followed by patients with head and neck cancer. 19 In our study, the tuberculin skin test was performed on 5 patients and was positive in 3 cases.
To confirm the diagnosis of concomitant TB and malignancy, histology remains the most reliable exam; although in some cases, necrotizing or caseating granulomatous lesions typical of TB can also be found in other tumors, including Hodgkin Lymphoma and non-Hodgkin lymphoma. 20 Furthermore, a granulomatous reaction can be observed in SCC against keratin produced by tumor cells. 11 Additionally, the distinction between nasopharyngeal cancer and TB can be difficult, since nasopharyngeal cancer can cause a granulomatous response within the peritumoral tissue. 21
Treatment of TB in patients with cancer is not well codified. However, it must always be discussed in a multidisciplinary consultation meeting. 18 In practice, according to Reddy et al, 22 once a patient has developed TB, anti-TB treatment takes priority, although this depends on the severity of the infection. Cheng et al, 23 in their study, recommended starting anti-TB treatment at least 15 days before anticancer treatment. However, in some cases, anti-TB and anticancer treatment could be administered simultaneously. 23 Only poor performance status, extremely drug-resistant Mycobacterium Tuberculosis, and severe organ dysfunction were shown to be the contraindications for simultaneous treatment with concomitant chemotherapy. 24 Hirashima et al 25 suggested waiting up to 2 months before initiating anticancer treatment, while remaining cautious about the impact of this approach on the prognosis and progression of cancer. Ito et al 26 recommend surgery of the primary tumor and anti-TB treatment prior to lymph node dissection to avoid the spread of TB infection. Furthermore, cancer treatment options including chemotherapy, radiotherapy, immunotherapy, and targeted therapy could lead to drug interactions and worsen the side effects of anti-TB drugs, including skin rashes, hepatitis, kidney failure, and lymphopenia.23,25 In our study, for patients who had chemotherapy, a 15-day minimum deadline was respected before starting anticancer treatment.
The response to treatment also varied between the authors. In the series by Hedhli et al, 27 7/8 patients reported adverse events such as vomiting and/or disturbed liver and/or hematological balance, necessitating postponement or discontinuation of the treatment protocol, with 7 deaths. Adzic 28 reported a good response rate of 90%, with only 1 death due to miliary TB.
In our study, the side effects of TB treatment were dominated by vomiting and hematologic disorders in 3 cases. Chemotherapy was delayed until biological equilibrium was normalized. Two patients died during treatment, due to miliary TB in 1 case and sepsis due to aplasia secondary to chemotherapy.
Conclusions
The coexistence of neoplasia and TB infection within the same lymphadenopathy can mislead and delay the diagnosis and treatment of 1 disease. TB infection that occurs during or after chemotherapy can also make the differential diagnosis between resistant and recurrent malignancy difficult. This diagnosis must be particularly taken into account in countries where TB is highly endemic.
To avoid missing the correct diagnosis of concurrent TB with cancer when both coexist, it is imperative to adopt a systematic and comprehensive approach. This includes suspecting this condition based on the patient’s medical history, clinical symptoms, and complementary test results such as imaging, biopsy, and PCR.
Additionally, close collaboration among specialists involved in patient care, including ENT, oncologists, and infectious disease specialists, is essential for ensuring holistic assessment and optimal treatment.
Footnotes
Acknowledgements
None.
Data Availability Statement
All data generated or analyzed during this research are included in this published article. Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical/Consent Statement
Approval was obtained from the local hospital ethics committee before data were collected for the study. Informed written consent was obtained from surviving patients or from the descendants of deceased patients.
Grant Number
Not applicable.
Trial Registration
Not applicable.
