Abstract
Neck hybrid schwannoma/neurofibromas are uncommon and classified as peripheral nerve sheath tumors (PNSTs). PNSTs develop from soft tissues, of which schwannoma, perineurioma, and neurofibroma are the most common. Hybrid PNSTs consist of more than 1 type of PNST, such as hybrid schwannoma/neurofibromas. The exact epidemiology and pathogenesis of these tumors are still largely unknown because of the limited studies on this topic. Such tumors can spread over the soft tissues, although most cases reported involve the subcutaneous layer or dermis. Some studies have suggested that hybrid schwannoma/neurofibromas may be associated with neurofibromatosis. We present a case of a 51-year-old female patient with a neck hybrid schwannoma/neurofibroma diagnosed by histopathology and immunohistochemistry. The patient was referred to the neurology department for neurofibromatosis screening, which reported negative results. After 12 months, the patient showed no evidence of tumor recurrence.
Keywords
Introduction
Hybrid schwannoma/neurofibromas are uncommon, and they are a type of peripheral nerve sheath tumor (PNST) that can arise from any soft tissue in the body. PNSTs include schwannomas, perineuriomas, and neurofibromas. Hybrid peripheral nerve sheath tumors (HPNSTs) consist of 2 or more types of PNSTs. The most common combination is hybrid schwannoma/perineurioma.1,2 The second most common type is hybrid neurofibroma/schwannoma, which is associated with neurofibromatosis type 1 (NF1), neurofibromatosis type 2 (NF2), or schwannomatosis. 3 Neurofibroma/perineurioma combinations are infrequently seen and often associated with NF1 disease. 1 The World Health Organization officially classified these tumors as “tumors of soft tissue and bone” in the fourth edition guideline 2 published in 2013 and “tumors of the central nervous system” in the updated fourth edition guideline 4 published in 2016.
This article presents a case of a hybrid schwannoma/neurofibroma of the neck, where the patient was referred to the neurology department for further assessment.
Case Report
A 51-year-old female patient presented to our clinic with a 2-week history of a right neck mass. Physical examination showed a 2-cm-wide right neck level II mass. On fiberoptic endoscopy, no obvious masses were observed at the nasopharynx, oropharynx, or hypopharynx. The results of baseline blood tests were within the normal range, including those for Epstein–Barr Virus Capsid Antigen IgA and lactate dehydrogenase. Computed tomography with contrast showed the presence of a teardrop-shaped cystic mass at the right level II jugular chain that appeared subtly high density after injection of the contrast medium. The mass had a clear margin, no surrounding fat stranding, and no lymphadenopathy (Figure 1(A-C)). Focal neurogenic tumor formation was suspected; therefore, the patient underwent magnetic resonance imaging (MRI), which showed a focal soft tissue-like lesion around the right level II area (Figure 1-D).
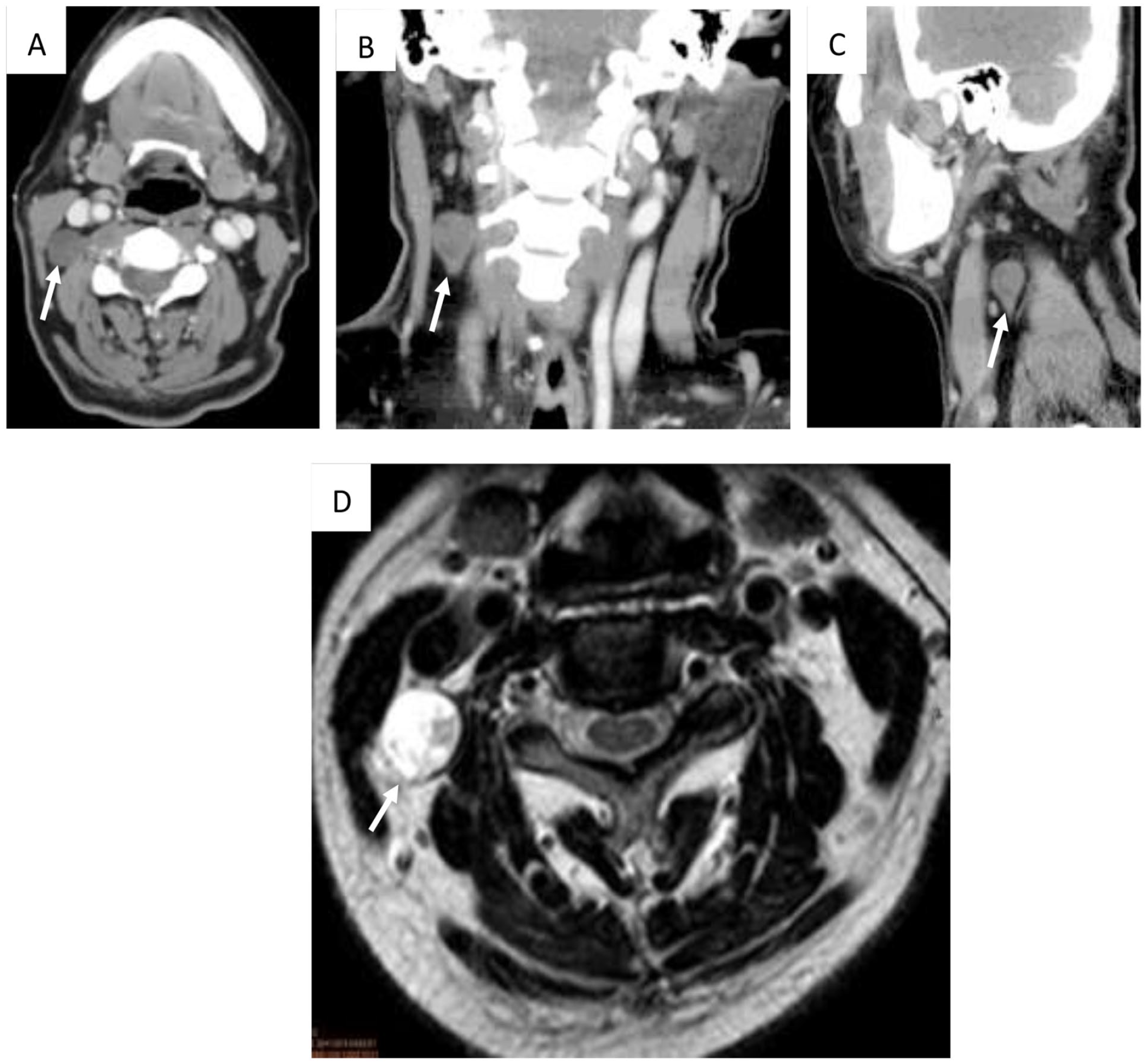
Preoperative images of the patient’s neck, including contrast-enhanced computed tomography scan (A, B, C) and magnetic resonance imaging scan (D). (A) The transverse view shows a cystic mass (white arrow) with a smooth margin and subtle heterogeneity at the right level II jugular chain. (B, C) The coronal and sagittal views show that the cystic mass is teardrop shaped (white arrow). (D) The transverse view shows a focal soft tissue mass (white arrow) with a smooth margin and subtle heterogeneity at the right level II jugular chain.
The neck mass was surgically excised, and histopathology showed an HPNST composed of solid schwannomatous nodules and a loose area of neurofibroma. Microscopically, immunostaining demonstrated a mixed population of cells in the neurofibromatous component, including CD34-positive fibroblasts and epithelial membrane antigen (EMA)-positive perineurial cells. The solid nodules of Schwann cells were diffusely positive after S100 and Sry-related HMg-Box gene 10 (SOX10) immunostaining (Figure 2).
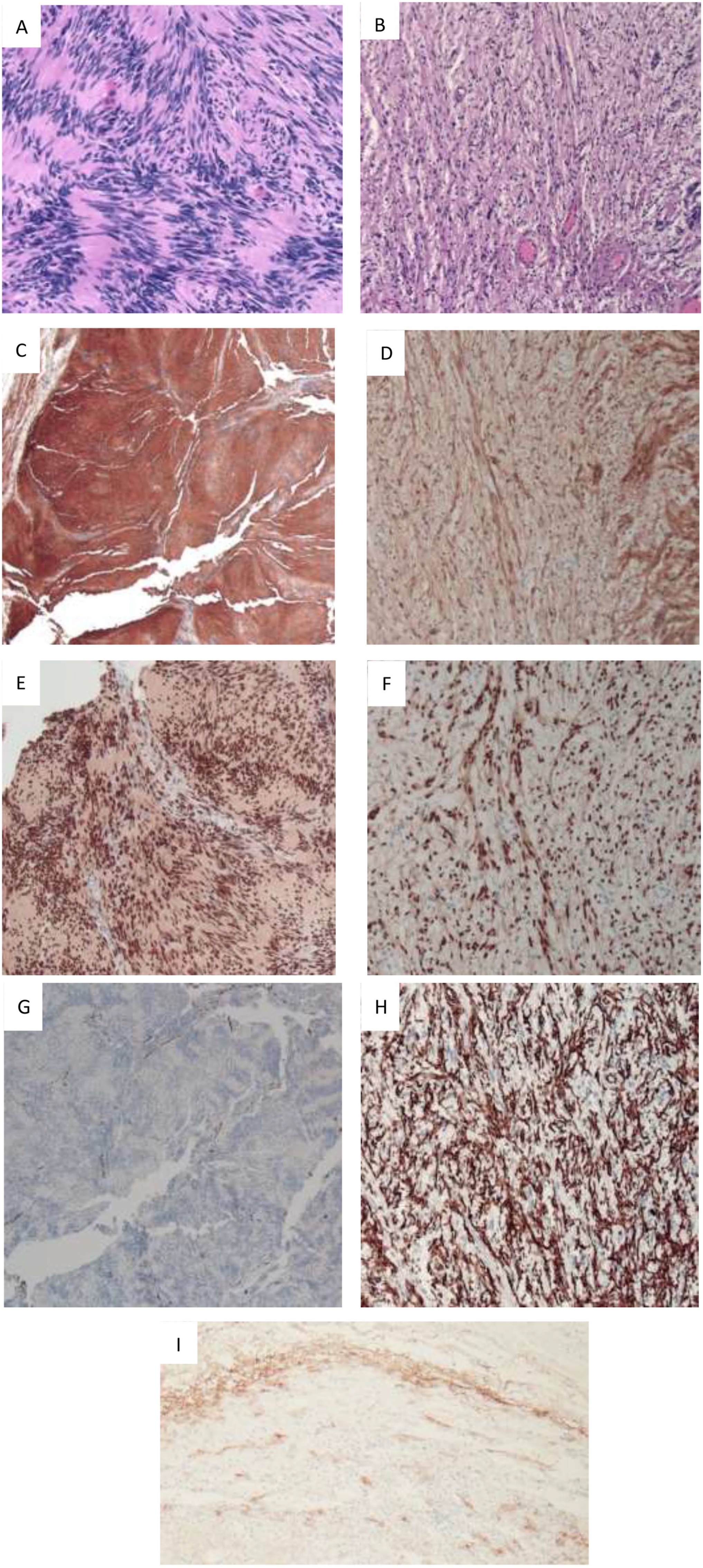
Micrographs with pathological stains of the tumor.
We referred the patient to neurology for NF screening based on the association between the hybrid neurofibroma/schwannoma and NF. She had no known family history of NF or central nervous system tumors. She also had pure-tone audiometry and auditory brainstem response, showing no obvious abnormality. After 12 months, a follow-up MRI showed no evidence of tumor recurrence. The patient is still followed up at the otolaryngology and the neurology department.
Discussion
The exact incidence rates of HPNSTs remain largely unknown owing to their rarity. All age groups of patients with HPNSTs have been described, but young adult patients are relatively common. No obvious sex predilection has been noted for these tumors. 1 Most tumors are in the subcutaneous or dermal layers, but they also demonstrate a wide anatomic distribution and can occur anywhere in the somatic soft tissues, such as the orbit, spinal foraminal regions, or femur.5 -8
The pathogenesis of HPNSTs is poorly understood. Previous studies consider localized changes in the microenvironment to be a possible cause, but it remains unclear. Most patients with HPNSTs show painless nodules in the dermis or subcutaneous tissue. In some cases, symptoms such as pain or paresthesia may occur when tumors demonstrate a mass effect on adjacent areas.1,9,10
To date, no consensus about the diagnostic criteria for hybrid neurofibroma/schwannoma tumors has been achieved. Histology and immunohistochemistry morphology may aid in diagnosis.3,11 Schwannomas consist of Schwann cells, with spindle cells having relatively large nuclei. In contrast, neurofibromas have Schwann cells with smaller nuclei having lengthened and curled figures.2,3,11,12 Histologically, interspersed neurofibroma regions and nests of schwannoma may be seen within the same lesion in varying proportions. The neurofibroma component should be distinctly separated from the Antoni B areas of schwannoma. Immunohistochemistry was performed for neurofilaments stained with S100, SOX10, CD34, and EMA. S100 and SOX10 staining were positive for schwannomas and neurofibromas to different degrees. The schwannoma showed diffusely and strongly positive S100 and SOX10 staining, whereas neurofibromas demonstrated limited S100 and SOX10 staining. 11 CD34 staining was highlighted in reticular components such as fibroblastic parts in neurofibromas and Antoni B regions of schwannomas, while the staining was negative in Antoni A areas of schwannomas. EMA staining was present in the perineural components of the neurofibromas and tumor capsule.3,11
No gold standard treatment for HPNST has been established. Based on a literature review, surgical excision is the most common treatment for HPNSTs. 13 The exact rates of recurrence remain largely unknown owing to the extreme rarity of these tumors. Cases of local recurrence are rare but do exist.9,14,15 However, no guidelines on the follow-up period have been suggested. Reported instances of neurofibroma/schwannoma showed no follow-up or a limited 6- to 118-month postoperative follow-up.10,12
According to a literature review of pathology, hybrid neurofibroma/schwannoma tumors are surprisingly associated with neurofibromatoses, 3 including NF1. NF1 has the risk of transforming into malignant PNSTs or possesses other coexisting malignancies, such as glioma, rhabdomyosarcoma, or gastrointestinal stromal tumors. 16 Therefore, examining patients with neurofibromas/schwannoma for possible underlying NF is essential. The diagnostic criteria for NF1, NF2, and schwannomatosis include multiorgan system involvement, including the ears, central nervous system, skin, bones, and genetic analysis. 16 Multidepartment collaboration is needed to confirm the diagnosis.
In conclusion, this case highlights that a patient with hybrid schwannoma/neurofibroma may have coexisting neurofibromatosis, including NF1, which has the risk of malignant transformation to an HPNST. Multidisciplinary teamwork is needed to help evaluate the patient, diagnose neurofibromatosis, and provide further care.
Footnotes
Data Availability Statement
Sharing does not apply to this article because no data sets were generated or analyzed during the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethics Committee of Mackay Memorial Hospital, Taitung, Taiwan (23MMHIS139e).
Statement of Informed Consent
Patient consent was waived as the waiver will not adversely affect the rights and welfare of the subjects. A waiver of informed consent from the patient was approved by the Institutional Review Board committee of Mackay Memorial Hospital, Taitung, Taiwan.
