Abstract
Rosai–Dorfman disease is a very rare disease characterized by histiocytic accumulation in the head and neck region and lymph node enlargement. We report a rare pseudo-malignant paranasal extranodal Rosai–Dorfman disease. A 69-year-old-man presented nasal bleeding and nasal obstruction. Paranasal mass was detected in the left nasal cavity and computed tomography (CT) findings are the sphenoid sinus, maxillary sinus, and ethmoid sinus were involved with inconstant bone thickening, however, no bone destruction was detected. Magnetic resonance imaging scans show iso-intensity signal in T1-weighed image and T2-weighed image. Positron emission tomography/CT fluorodeoxyglucose (FDG) uptake in posterior ethmoid sinus and sphenoid sinus, bilateral cervical lymph node, clavicle, and sternum. Based on the above results, we considered malignant lymphoma and performed a biopsy. After pathological examination, a diagnosis of Rosai–Dorfman disease was established.
Introduction
Various mass lesions may form within the nose, and there are scattered cases in which patients are unaware of the disease until it begins to affect nasal ventilation. Inflammatory diseases such as nasal polyps associated with eosinophilic chronic rhinosinusitis, benign tumors such as inverted papilloma, and malignant tumors such as squamous cell carcinoma are examples of diseases that form masses in the nose. In particular, inverted papilloma often shows convoluted cerebriform pattern (CCP) on magnetic resonance imaging (MRI) T2-weighed images.1,2 In the case of malignant tumors, significant bone destruction on computed tomography (CT) and 18F-FDG accumulation on positron emission tomography (PET)/CT are commonly detected, and imaging examinations are very useful in the diagnosis of tumor diseases. However, it is difficult to make a complete qualitative diagnosis based on imaging findings alone, not only for nasal sinus disease, and even when malignant disease is considered, the possibility of an atypical, very rare inflammatory disease cannot be excluded.
We report the rare case of a 69-year-old man who diagnosed pseudo-malignant paranasal rare extranodal Rosai–Dorfman disease and review the literature.
Case Report
A 69-year-old man was referred to our department because he exhibited symptoms of nasal obstruction and nasal bleeding. The patient’s general condition was good and smooth surface mass derived from the left middle meatus was observed during nasal endoscopy (Figure 1A). Heterogeneous contrasting shadows were observed in both paranasal sinuses, besides irregular bone augmentation in the medial wall of the right maxillary sinus by paranasal contrast-enhanced CT (Figure 1B). T1-weighted iso intensity and T2-weighted low intensity signal areas are observed in the medial regions of bilateral maxillary sinuses (Figure 1C). PET/CT demonstrated bilateral cervical, axillary, and supraclavicular lymph node accumulations, as well as bony lesions in the clavicle and sternum, in addition to paranasal sinuses (Figure 1D). Resultantly, we considered the patient with malignant lymphoma and decided to perform biopsy of paranasal tumor. As a result, histiocyte-like cells were observed under the nasal epithelium, showing emperipolesis. Immunostaining demonstrated that histiocyte-like cells were CD68 (+), CD163 (+), S100 (+), cyclinD1 (+), oct-2 (+), CD1a (−), BRAFV600E (−), and CK(AE1/AE3) (−), suggesting Rosai–Dorfman disease (Figure 2A, B, C, and D). However, considering it difficult to make a diagnosis based on only extranodal lesions, a left cervical lymph node biopsy was also performed, which showed similar findings (Figure 2E and F) and confirmed the diagnosis of the Rosai–Dorfman disease. The patient was referred to our hematology department for treatment.
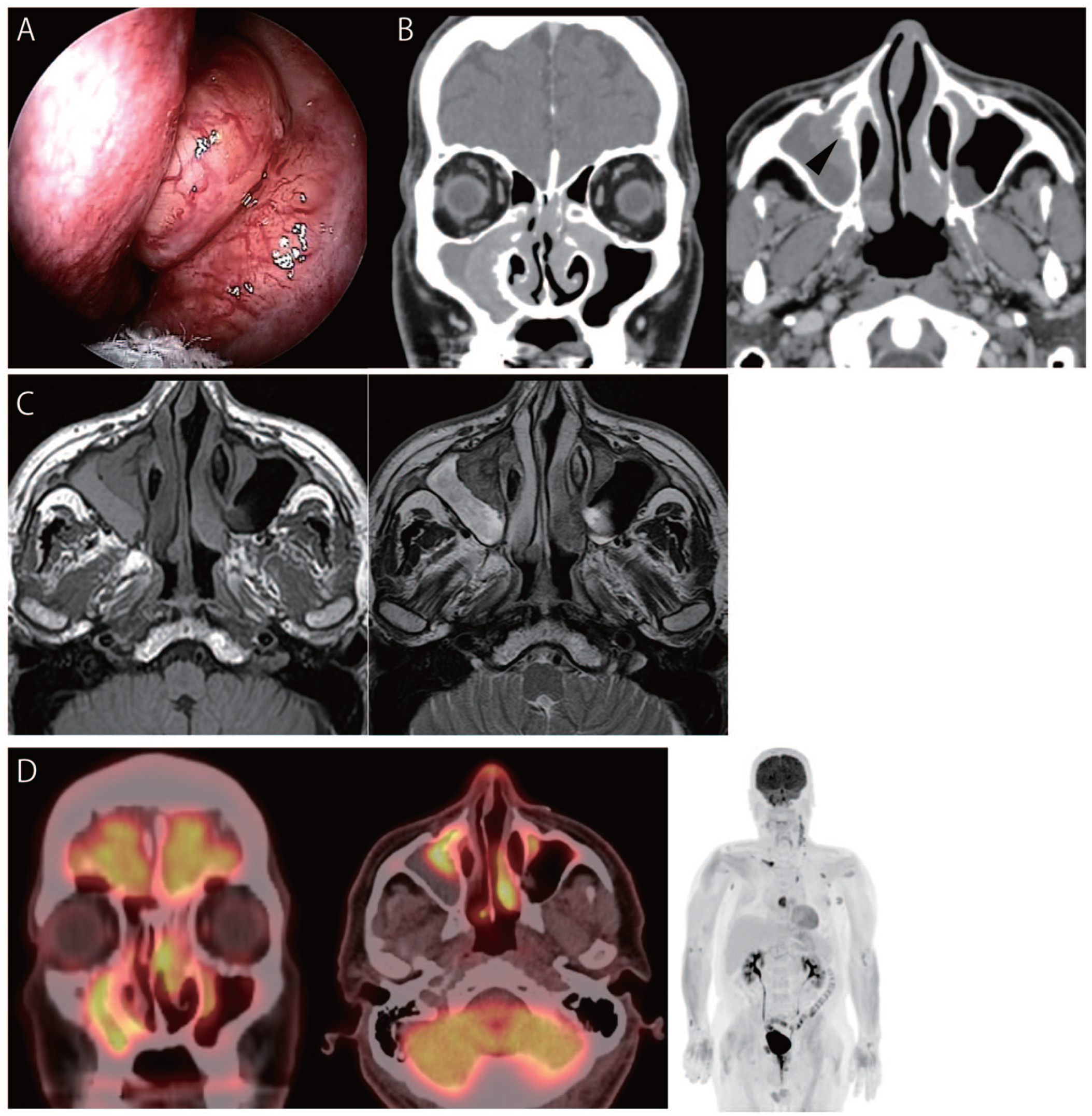
(A) Transnasal endoscopic view. Surface smooth mass is in the left middle nasal meatus. (B) Paranasal CT findings. Axial CT scan showed a bone thickening of interior wall of right maxillary sinus. On the other hand, bone destruction was not observed in the paranasal sinus (black arrowheads). The sinus mass is contrasted heterogeneously. (C) Paranasal MRI findings. T1-weighted iso intensity and T2-weighted low intensity signal areas in the medial regions of bilateral maxillary sinuses and the posterior part of left turbinate in MRI scans. (D) FDG PET/CT findings. FDG PET/CT findings. FDG uptake (SUVmax, 7.3) was detected in the left posterior ethmoidal sinus and sphenoid sinus. Accumulation was detected in bilateral cervical, supraclavicular, and axillary lymph nodes (SUVmax, 4.6), as well as in the right clavicle and sternum (SUVmax, 8.2, 6.9). S, septum; IT, inferior turbinate; T, tumor; CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography.
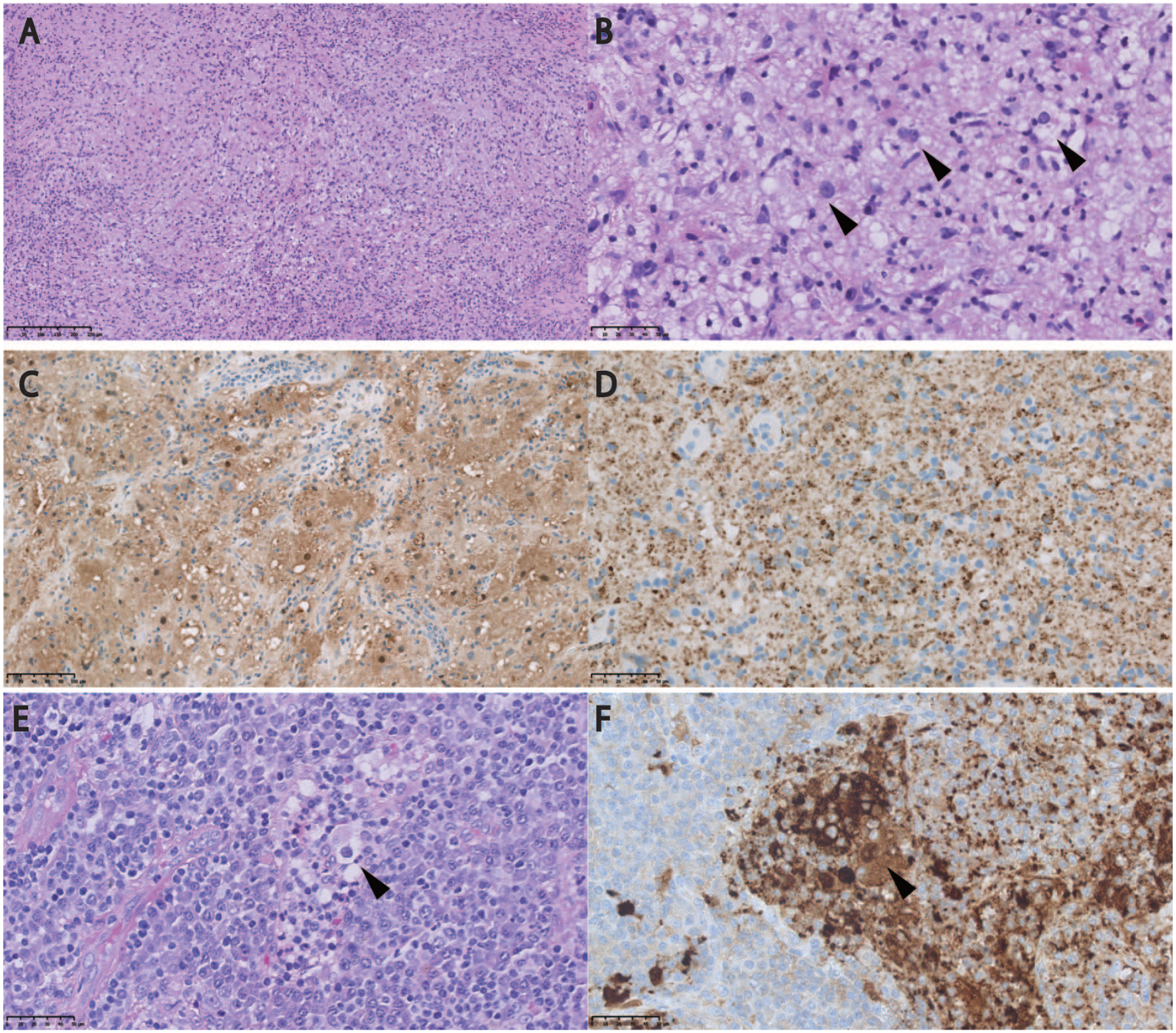
(A) Pathological observations of nasal lesions. Pale histiocyte-like cells with a rounded, enlarged nucleus diffusely infiltrated with lymphocytes, plasma cells, and neutrophils. (B) Lymphocytyes can be identified (black arrowhead). (C) The infiltrated histiocytes were positive for immunostaining of S-100 protein. (D) The infiltrated histiocytes were positive for immunostaining of CD68. (E) Pathological observations of left cervical lymph node. Infiltration of plasma cells and histiocytes is observed, and emperipolesis can be identified (black arrowhead) same as in nasal lesions. (F) Infiltrated histiocytes are immunostained by S-100 protein.
Discussion
In otorhinolaryngology, nasal and paranasal mass disease are very common. Inflammatory diseases include nasal polyps often associated with rhinosinusitis, cholesterol granuloma, and mycetoma.3,4 Benign tumors include inverted papilloma and pleomorphic adenomas. In contrast, there are cases involving various primary malignancies such as squamous cell carcinoma, adenocarcinoma, and malignant lymphoma, and metastasis from the lungs, kidneys, and prostate.5 –7 In the present case, the patient had nasal obstruction and nasal bleeding and irregularly shaped shadows were seen in the bilateral paranasal sinuses. Since there was little bone destruction on CT and no typical CCP on MRI, and the mass was smooth on the surface, the possibility of squamous cell carcinoma was low and biopsy was performed with malignant lymphoma as the main concern. Interestingly, the diagnosis of Rosai–Dorfman disease in extranodal lesion was made.
Rosai–Dorfman disease is a very rare subtype of non-Langerhans cell histiocytosis.8,9 Although nodal involvement of the skin, subcutaneous tissue, and lymph nodes is common, some cases are associated with an extranodal type. In the head and neck region, nasal cavity and paranasal sinuses and/or cervical lymph nodes are often involved.10,11 Differentiation from malignant disease is important because PET/CT shows nodular lesions with aggregation in multiple locations throughout the body.12,13 There are no established treatment guidelines; however, surgical resection or systemic corticosteroids are often used as initial treatment. There are often cases in which multiple lesions are difficult to resect or steroid-refractory, and chemotherapy may be administered in these cases. On the other hand, the course of the disease varies widely, including spontaneous resolution in some cases. 14 Due to the multiple lesions throughout the body, surgical resection was not indicated in this case, and the patient was referred to the Department of Hematology for systemic corticosteroids because of the increasing disease lesions. Since the disease is pathologically benign but can progress dramatically in some cases, proper diagnosis and tailored treatment are considered important.
In conclusion, we encountered a patient with pseudo-malignant paranasal extranodal Rosai–Dorfman disease. Though it is rare, to distinguish Rosai–Dorfman disease is very important if a mass is found in the nasal cavity.
Footnotes
Acknowledgements
We thank Rintaro Ohe of the Department of Pathology, Faculty of Medicine, Yamagata University, for his invaluable advice on pathological diagnosis.
Data Availability
The data that support the findings of this study are available on request from the corresponding author, TT. The data are not publicly available due to restrictions, for example, their containing information that could compromise the privacy of research participants.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of Osaka University (No. 16329) and informed consent was obtained from the patient for the publication of their data.
Human and Animal Rights
This article does not contain any studies involving human or animal subjects.
Informed Consent
Informed consent was obtained from the patient for their anonymized information to be published in this article.
